Risk/Benefit Profiles of Currently Approved Oral Antivirals for Treatment of COVID-19: Similarities and Differences
Abstract
:1. Introduction
2. Methods
3. Regulatory and Clinical Trial Landscape
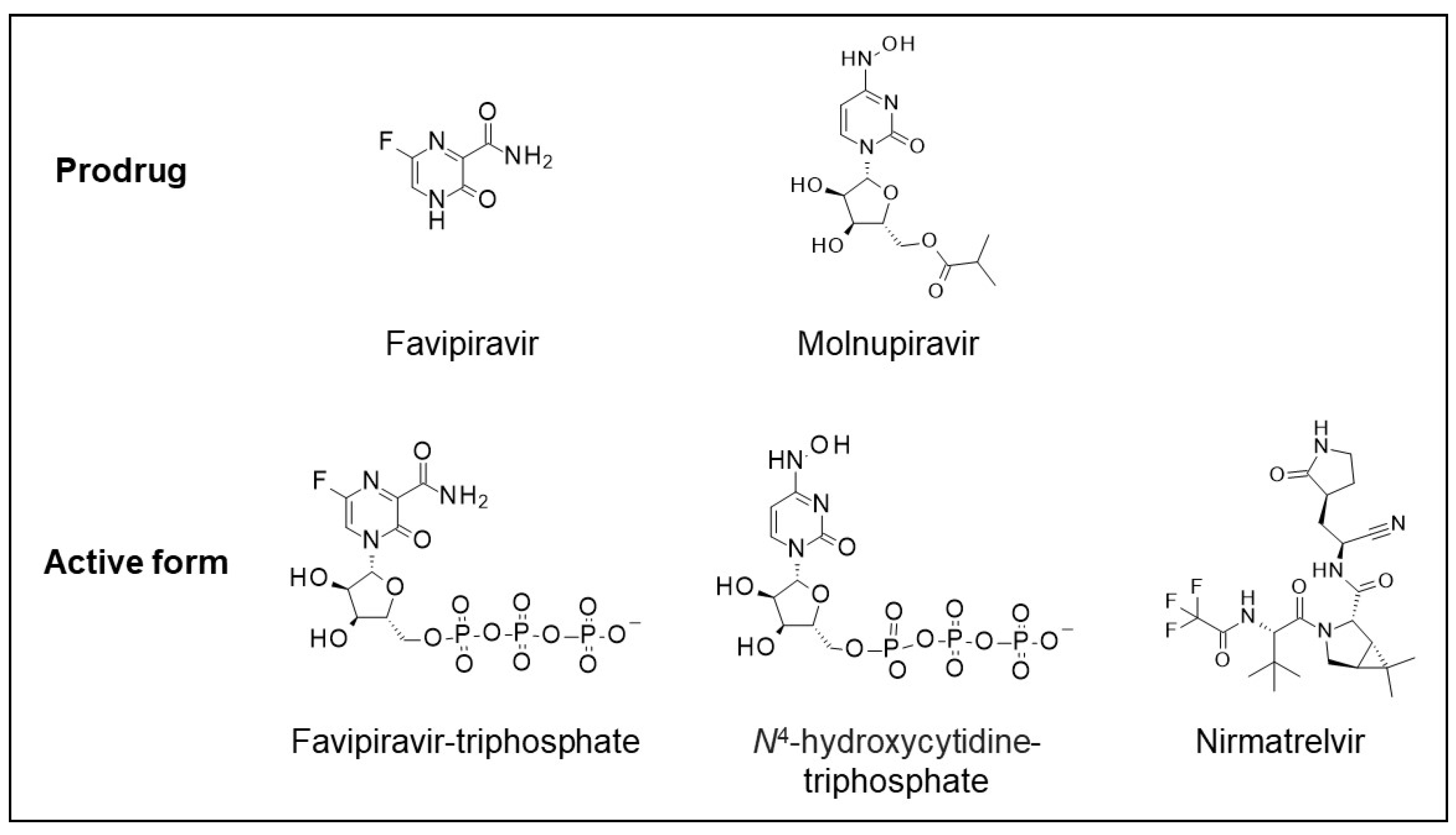
4. Drug Mechanisms of Action
5. Preclinical Potency against SARS-CoV-2 Virus
6. Pharmacokinetics
7. Clinical Pharmacology
8. Toxicology
9. Clinical Safety
10. Clinical Efficacy
11. Potential for Developing Antiviral Drug Resistance
12. Summary of the Clinical Risk/Benefit Profiles
13. Discussion
14. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- US Centers for Disease Control and Prevention. COVID Data Tracker. Available online: https://covid.cdc.gov/covid-data-tracker/#datatracker-home (accessed on 20 May 2022).
- World Health Organization. Available online: https://covid19.who.int/ (accessed on 8 March 2022).
- Paules, C.I.; Fauci, A.S. COVID-19: The therapeutic landscape. Med 2021, 2, 493–497. [Google Scholar] [CrossRef] [PubMed]
- Furuta, Y.; Komeno, T.; Nakamura, T. Favipiravir (T-705), a broad spectrum inhibitor of viral RNA polymerase. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2017, 93, 449–463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- FUJIFILM Toyama Chemical Co., Ltd. Avigan Tablets 200 mg. Available online: https://www.sukl.cz (accessed on 14 June 2022).
- Mentre, F.; Taburet, A.M.; Guedj, J.; Anglaret, J.; Keita, S.; de Lamballerie, X. Dose regimen of favipiravir for Ebola virus disease. Lancet Infect. Diseases 2015, 15, 150–151. [Google Scholar] [CrossRef]
- Nagata, T.; Lefor, A.K.; Hasegawa, M.; Ishii, M. Favipiravir: A new medication for the Ebola virus disease pandemic. Disaster Med. Public Health Prep. 2015, 9, 79–81. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Jiang, X.M.; Cui, N.; Yuan, C.; Zhang, S.F.; Lu, Q.B.; Yang, Z.D.; Xin, Q.L.; Song, Y.B.; Zhang, X.A.; et al. Clinical effect and antiviral mechanism of T-705 in treating severe fever with thrombocytopenia syndrome. Signal Transduct. Target. Ther. 2021, 6, 145. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Lu, Q.B.; Yao, W.S.; Zhao, J.; Zhang, X.A.; Cui, N.; Yuan, C.; Yang, T.; Peng, X.F.; Lv, S.M.; et al. Clinical efficacy and safety evaluation of favipiravir in treating patients with severe fever with thrombocytopenia syndrome. EBioMedicine 2021, 72, 103591. [Google Scholar] [CrossRef]
- Shiraki, K.; Daikoku, T. Favipiravir, an anti-influenza drug against life-threatening RNA virus infections. Pharmacol. Ther. 2020, 209, 107512. [Google Scholar] [CrossRef]
- Jain, M.; Bahrate, S.D. Favipiravir has been investigated for teh treatment of life-threatening pathogens such as Ebola virus, Lassa virus, and now COVID-19: A Review. Asian J. Parmacy Res. 2021, 11, 39–42. [Google Scholar] [CrossRef]
- Yoon, J.J.; Toots, M.; Lee, S.; Lee, M.E.; Ludeke, B.; Luczo, J.M.; Ganti, K.; Cox, R.M.; Sticher, Z.M.; Edpuganti, V.; et al. Orally Efficacious Broad-Spectrum Ribonucleoside Analog Inhibitor of Influenza and Respiratory Syncytial Viruses. Antimicrob. Agents Chemother. 2018, 62, e00766-18. [Google Scholar] [CrossRef] [Green Version]
- Hoffman, R.L.; Kania, R.S.; Brothers, M.A.; Davies, J.F.; Ferre, R.A.; Gajiwala, K.S.; He, M.; Hogan, R.J.; Kozminski, K.; Li, L.Y.; et al. Discovery of Ketone-Based Covalent Inhibitors of Coronavirus 3CL Proteases for the Potential Therapeutic Treatment of COVID-19. J. Med. Chem. 2020, 63, 12725–12747. [Google Scholar] [CrossRef]
- Peng, Q.; Peng, R.; Yuan, B.; Wang, M.; Zhao, J.; Fu, L.; Qi, J.; Shi, Y. Structural Basis of SARS-CoV-2 Polymerase Inhibition by Favipiravir. Innovation 2021, 2, 100080. [Google Scholar] [CrossRef]
- Shannon, A.; Selisko, B.; Le, N.T.; Huchting, J.; Touret, F.; Piorkowski, G.; Fattorini, V.; Ferron, F.; Decroly, E.; Meier, C.; et al. Rapid incorporation of Favipiravir by the fast and permissive viral RNA polymerase complex results in SARS-CoV-2 lethal mutagenesis. Nat. Commun. 2020, 11, 4682. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.P.; Daifuku, R.; Loeb, L.A. Viral error catastrophe by mutagenic nucleosides. Annu. Rev. Microbiol. 2004, 58, 183–205. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kabinger, F.; Stiller, C.; Schmitzova, J.; Dienemann, C.; Kokic, G.; Hillen, H.S.; Hobartner, C.; Cramer, P. Mechanism of molnupiravir-induced SARS-CoV-2 mutagenesis. Nat. Struct. Mol. Biol. 2021, 28, 740–746. [Google Scholar] [CrossRef] [PubMed]
- Abdelnabi, R.; Foo, C.S.; Kaptein, S.J.F.; Zhang, X.; Do, T.N.D.; Langendries, L.; Vangeel, L.; Breuer, J.; Pang, J.; Williams, R.; et al. The combined treatment of Molnupiravir and Favipiravir results in a potentiation of antiviral efficacy in a SARS-CoV-2 hamster infection model. EBioMedicine 2021, 72, 103595. [Google Scholar] [CrossRef]
- Gordon, C.J.; Tchesnokov, E.P.; Schinazi, R.F.; Gotte, M. Molnupiravir promotes SARS-CoV-2 mutagenesis via the RNA template. J. Biol. Chem. 2021, 297, 100770. [Google Scholar] [CrossRef]
- Jin, Z.; Du, X.; Xu, Y.; Deng, Y.; Liu, M.; Zhao, Y.; Zhang, B.; Li, X.; Zhang, L.; Peng, C.; et al. Structure of M(pro) from SARS-CoV-2 and discovery of its inhibitors. Nature 2020, 582, 289–293. [Google Scholar] [CrossRef] [Green Version]
- Rut, W.; Groborz, K.; Zhang, L.; Sun, X.; Zmudzinski, M.; Pawlik, B.; Wang, X.; Jochmans, D.; Neyts, J.; Mlynarski, W.; et al. SARS-CoV-2 M(pro) inhibitors and activity-based probes for patient-sample imaging. Nat. Chem. Biol. 2021, 17, 222–228. [Google Scholar] [CrossRef]
- Pillaiyar, T.; Manickam, M.; Namasivayam, V.; Hayashi, Y.; Jung, S.H. An Overview of Severe Acute Respiratory Syndrome-Coronavirus (SARS-CoV) 3CL Protease Inhibitors: Peptidomimetics and Small Molecule Chemotherapy. J. Med. Chem. 2016, 59, 6595–6628. [Google Scholar] [CrossRef]
- Waters, L.; Marra, F.; Pozniak, A.; Cockburn, J.; Boffito, M. Ritonavir and COVID-19: Pragmatic guidance is important. Lancet 2022, 399, 1464–1465. [Google Scholar] [CrossRef]
- Heskin, J.; Pallett, S.J.C.; Mughal, N.; Davies, G.W.; Moore, L.S.P.; Rayment, M.; Jones, R. Caution required with use of ritonavir-boosted PF-07321332 in COVID-19 management. Lancet 2022, 399, 21–22. [Google Scholar] [CrossRef]
- Wang, Y.; Yuan, C.; Xu, X.; Chong, T.H.; Zhang, L.; Cheung, P.P.; Huang, X. The mechanism of action of T-705 as a unique delayed chain terminator on influenza viral polymerase transcription. Biophys. Chem. 2021, 277, 106652. [Google Scholar] [CrossRef] [PubMed]
- Cox, R.M.; Wolf, J.D.; Plemper, R.K. Therapeutically administered ribonucleoside analogue MK-4482/EIDD-2801 blocks SARS-CoV-2 transmission in ferrets. Nat. Microbiol. 2021, 6, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Owen, D.R.; Allerton, C.M.N.; Anderson, A.S.; Aschenbrenner, L.; Avery, M.; Berritt, S.; Boras, B.; Cardin, R.D.; Carlo, A.; Coffman, K.J.; et al. An oral SARS-CoV-2 M(pro) inhibitor clinical candidate for the treatment of COVID-19. Science 2021, 374, 1586–1593. [Google Scholar] [CrossRef] [PubMed]
- Driouich, J.S.; Cochin, M.; Lingas, G.; Moureau, G.; Touret, F.; Petit, P.R.; Piorkowski, G.; Barthelemy, K.; Laprie, C.; Coutard, B.; et al. Favipiravir antiviral efficacy against SARS-CoV-2 in a hamster model. Nat. Commun. 2021, 12, 1735. [Google Scholar] [CrossRef]
- Kaptein, S.J.F.; Jacobs, S.; Langendries, L.; Seldeslachts, L.; Ter Horst, S.; Liesenborghs, L.; Hens, B.; Vergote, V.; Heylen, E.; Barthelemy, K.; et al. Favipiravir at high doses has potent antiviral activity in SARS-CoV-2-infected hamsters, whereas hydroxychloroquine lacks activity. Proc. Natl. Acad. Sci. USA 2020, 117, 26955–26965. [Google Scholar] [CrossRef] [PubMed]
- Greasley, S.E.; Noell, S.; Plotnikova, O.; Ferre, R.; Liu, W.; Bolanos, B.; Fennell, K.; Nicki, J.; Craig, T.; Zhu, Y.; et al. Structural basis for the in vitro efficacy of nirmatrelvir against SARS-CoV-2 variants. J. Biol. Chem. 2022, 298, 101972. [Google Scholar] [CrossRef]
- Li, P.; Wang, Y.; Lavrijsen, M.; Lamers, M.M.; de Vries, A.C.; Rottier, R.J.; Bruno, M.J.; Peppelenbosch, M.P.; Haagmans, B.L.; Pan, Q. SARS-CoV-2 Omicron variant is highly sensitive to molnupiravir, nirmatrelvir, and the combination. Cell Res. 2022, 32, 322–324. [Google Scholar] [CrossRef]
- Standard, B. COVID-19: Russia’s Avifavir Pill Shows Promise against Omicron, Delta. Available online: https://www.business-standard.com/article/current-affairs/COVID-19-russia-s-avifavir-pill-shows-promise-against-omicron-delta-121122800498_1.html (accessed on 28 December 2021).
- Japanese Pharmaceuticals and Medical Devices Agency. Avigan, Japanese Pharmaceuticals and Medical Devices Agency (PMDA) Report on the Deliberation Results. Available online: https://www.pmda.go.jp/files/000210319.pdf (accessed on 7 March 2022).
- European Medicines Agency. Molnupiravir Article 5(3) Opinions. Available online: https://www.ema.europa.eu/en/human-regulatory/post-authorisation/referral-procedures/article-53-opinions (accessed on 19 November 2021).
- Foisy, M.M.; Yakiwchuk, E.M.; Hughes, C.A. Induction effects of ritonavir: Implications for drug interactions. Ann. Pharmacother. 2008, 42, 1048–1059. [Google Scholar] [CrossRef]
- European Medicines Agency. Paxlovid for the Treatment of COVID-19—EMEA/H/A-5(3)/1513. Available online: https://www.ema.europa.eu/en/documents/referral/paxlovid-pf-07321332-ritonavir-COVID-19-article-53-procedure-conditions-use-conditions-distribution_en.pdf (accessed on 16 December 2021).
- Koshi, E.; Saito, S.; Okazaki, M.; Toyama, Y.; Ishimoto, T.; Kosugi, T.; Hiraiwa, H.; Jingushi, N.; Yamamoto, T.; Ozaki, M.; et al. Efficacy of favipiravir for an end stage renal disease patient on maintenance hemodialysis infected with novel coronavirus disease 2019. CEN Case Rep. 2021, 10, 126–131. [Google Scholar] [CrossRef]
- Telbisz, A.; Ambrus, C.; Mozner, O.; Szabo, E.; Varady, G.; Bakos, E.; Sarkadi, B.; Ozvegy-Laczka, C. Interactions of Potential Anti-COVID-19 Compounds with Multispecific ABC and OATP Drug Transporters. Pharmaceutics 2021, 13, 81. [Google Scholar] [CrossRef] [PubMed]
- Painter, W.P.; Holman, W.; Bush, J.A.; Almazedi, F.; Malik, H.; Eraut, N.; Morin, M.J.; Szewczyk, L.J.; Painter, G.R. Human Safety, Tolerability, and Pharmacokinetics of Molnupiravir, a Novel Broad-Spectrum Oral Antiviral Agent with Activity Against SARS-CoV-2. Antimicrob. Agents Chemother. 2021, 65, e02428-20. [Google Scholar] [CrossRef]
- Ivashchenko, A.A.; Dmitriev, K.A.; Vostokova, N.V.; Azarova, V.N.; Blinow, A.A.; Egorova, A.N.; Gordeev, I.G.; Ilin, A.P.; Karapetian, R.N.; Kravchenko, D.V.; et al. AVIFAVIR for Treatment of Patients With Moderate Coronavirus Disease 2019 (COVID-19): Interim Results of a Phase II/III Multicenter Randomized Clinical Trial. Clin. Infect. Dis. 2021, 73, 531–534. [Google Scholar] [CrossRef] [PubMed]
- Madelain, V.; Mentre, F.; Baize, S.; Anglaret, X.; Laouenan, C.; Oestereich, L.; Nguyen, T.H.T.; Malvy, D.; Piorkowski, G.; Graw, F.; et al. Modeling Favipiravir Antiviral Efficacy Against Emerging Viruses: From Animal Studies to Clinical Trials. CPT Pharmacomet. Syst. Pharmacol. 2020, 9, 258–271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mendenhall, M.; Russell, A.; Smee, D.F.; Hall, J.O.; Skirpstunas, R.; Furuta, Y.; Gowen, B.B. Effective oral favipiravir (T-705) therapy initiated after the onset of clinical disease in a model of arenavirus hemorrhagic Fever. PloS Negl. Trop. Dis. 2011, 5, e1342. [Google Scholar] [CrossRef] [PubMed]
- Irie, K.; Nakagawa, A.; Fujita, H.; Tamura, R.; Eto, M.; Ikesue, H.; Muroi, N.; Tomii, K.; Hashida, T. Pharmacokinetics of Favipiravir in Critically Ill Patients With COVID-19. Clin. Transl. Sci. 2020, 13, 880–885. [Google Scholar] [CrossRef] [PubMed]
- Sissoko, D.; Laouenan, C.; Folkesson, E.; M’Lebing, A.B.; Beavogui, A.H.; Baize, S.; Camara, A.M.; Maes, P.; Shepherd, S.; Danel, C.; et al. Experimental Treatment with Favipiravir for Ebola Virus Disease (the JIKI Trial): A Historically Controlled, Single-Arm Proof-of-Concept Trial in Guinea. PloS Med. 2016, 13, e1001967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mishima, E.; Anzai, N.; Miyazaki, M.; Abe, T. Uric Acid Elevation by Favipiravir, an Antiviral Drug. Tohoku J. Exp. Med. 2020, 251, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Pfizer Inc. Paxlovid Fact Sheet For Healthcare Providers. Available online: https://www.covid19oralrx-hcp.com/?source=google&HBX_PK=s_paxlovid+healthcare&skwid=43700068281648831&gclid=Cj0KCQjw-JyUBhCuARIsANUqQ_LsoVyWK8Yyq8Ox7nmhOZV6BhZafwaQJkhAuN92ClzxO1Je9mAEz8IaAr8yEALw_wcB&gclsrc=aw.ds (accessed on 22 December 2021).
- Marzolini, C.; Kuritzkes, D.R.; Marra, F.; Boyle, A.; Gibbons, S.; Flexner, C.; Pozniak, A.; Boffito, M.; Waters, L.; Burger, D.; et al. Prescribing Nirmatrelvir-Ritonavir: How to Recognize and Manage Drug-Drug Interactions. Ann. Intern. Med. 2022, 175, 744–746. [Google Scholar] [CrossRef]
- Marzolini, C.; Kuritzkes, D.R.; Marra, F.; Boyle, A.; Gibbons, S.; Flexner, C.; Pozniak, A.; Boffito, M.; Waters, L.; Burger, D.; et al. Recommendations for the Management of Drug-Drug Interactions Between the COVID-19 Antiviral Nirmatrelvir/Ritonavir (Paxlovid) and Comedications. Clin. Pharmacol. Ther. 2022. [Google Scholar] [CrossRef]
- Outpatient Guidance for Treatment of COVID-19 in Adults and Children. Available online: https://www.med.umich.edu/asp/pdf/outpatient_guidelines/COVID-19-amb-treatment.pdf (accessed on 14 June 2022).
- COVID-19 Drug Interactions. Available online: https://www.covid19-druginteractions.org/ (accessed on 14 June 2022).
- Drug-Drug Interactions between Ritonavir-Boosted Nirmatrelvir (Paxlovid) and Concomitant Medications. Available online: https://www.covid19treatmentguidelines.nih.gov/therapies/antiviral-therapy/ritonavir-boosted-nirmatrelvir—paxlovid-/paxlovid-drug-drug-interactions/ (accessed on 14 June 2022).
- Jothimani, D.; Venugopal, R.; Abedin, M.F.; Kaliamoorthy, I.; Rela, M. COVID-19 and the liver. J. Hepatol. 2020, 73, 1231–1240. [Google Scholar] [CrossRef] [PubMed]
- Abbott Inc. Norvir Prescribing Information. 2012. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/020945s034lbl.pdf (accessed on 7 March 2022).
- Kaur, R.J.; Charan, J.; Dutta, S.; Sharma, P.; Bhardwaj, P.; Sharma, P.; Lugova, H.; Krishnapillai, A.; Islam, S.; Haque, M.; et al. Favipiravir Use in COVID-19: Analysis of Suspected Adverse Drug Events Reported in the WHO Database. Infect. Drug Resist. 2020, 13, 4427–4438. [Google Scholar] [CrossRef]
- Pilkington, V.; Pepperrell, T.; Hill, A. A review of the safety of favipiravir—A potential treatment in the COVID-19 pandemic? J. Virus Erad. 2020, 6, 45–51. [Google Scholar] [CrossRef]
- Catlin, N.R.; Bowman, C.J.; Campion, S.N.; Cheung, J.R.; Nowland, W.S.; Sathish, J.G.; Stethem, C.M.; Updyke, L.; Cappon, G.D. Reproductive and developmental safety of nirmatrelvir (PF-07321332), an oral SARS-CoV-2 M(pro) inhibitor in animal models. Reprod. Toxicol. 2022, 108, 56–61. [Google Scholar] [CrossRef]
- NORVIR Product Label. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/209512lbl.pdf (accessed on 13 June 2022).
- Ruzhentsova, T.A.; Oseshnyuk, R.A.; Soluyanova, T.N.; Dmitrikova, E.P.; Mustafaev, D.M.; Pokrovskiy, K.A.; Markova, T.N.; Rusanova, M.G.; Kostina, N.E.; Agafina, A.S.; et al. Phase 3 trial of coronavir (favipiravir) in patients with mild to moderate COVID-19. Am. J. Transl. Res. 2021, 13, 12575–12587. [Google Scholar] [CrossRef] [PubMed]
- Jayk Bernal, A.; Gomes da Silva, M.M.; Musungaie, D.B.; Kovalchuk, E.; Gonzalez, A.; Delos Reyes, V.; Martin-Quiros, A.; Caraco, Y.; Williams-Diaz, A.; Brown, M.L.; et al. Molnupiravir for Oral Treatment of COVID-19 in Nonhospitalized Patients. N. Engl. J. Med. 2022, 386, 509–520. [Google Scholar] [CrossRef] [PubMed]
- Cai, Q.; Yang, M.; Liu, D.; Chen, J.; Shu, D.; Xia, J.; Liao, X.; Gu, Y.; Cai, Q.; Yang, Y.; et al. Experimental Treatment with Favipiravir for COVID-19: An Open-Label Control Study. Engineering 2020, 6, 1192–1198. [Google Scholar] [CrossRef]
- Hammond, J.; Leister-Tebbe, H.; Gardner, A.; Abreu, P.; Bao, W.; Wisemandle, W.; Baniecki, M.; Hendrick, V.M.; Damle, B.; Simon-Campos, A.; et al. Oral Nirmatrelvir for High-Risk, Nonhospitalized Adults with COVID-19. N. Engl. J. Med. 2022, 386, 1397–1408. [Google Scholar] [CrossRef]
- Pfizer Inc. General Investigation for PAXLOVID PACK NCT05263908. Available online: https://clinicaltrials.gov/ct2/show/NCT05263908 (accessed on 23 April 2022).
- Ivashchenko, A.A.; Azarova, V.N.; Egorova, A.N.; Karapetian, R.N.; Kravchenko, D.V.; Krivonos, N.V.; Loginov, V.G.; Poyarkov, S.V.; Merkulova, E.A.; Rosinkova, O.S.; et al. Effect of Aprotinin and Avifavir((R)) Combination Therapy for Moderate COVID-19 Patients. Viruses 2021, 13, 1253. [Google Scholar] [CrossRef]
- Gunaratne, S.H.; Tieu, H.V.; Wilkin, T.J.; Taylor, B.S. CROI 2021: Advances in Antiretroviral Therapy for HIV and Antiviral Therapy for COVID-19. Top. Antivir. Med. 2021, 29, 361–378. [Google Scholar] [PubMed]
- Inoue, H.; Jinno, M.; Ohta, S.; Kishino, Y.; Kawahara, T.; Mikuni, H.; Sato, H.; Yamamoto, M.; Sato, Y.; Onitsuka, C.; et al. Combination treatment of short-course systemic corticosteroid and favipiravir in a successfully treated case of critically ill COVID-19 pneumonia with COPD. Respir. Med. Case Rep. 2020, 31, 101200. [Google Scholar] [CrossRef]
- Murohashi, K.; Hagiwara, E.; Kitayama, T.; Yamaya, T.; Higa, K.; Sato, Y.; Otoshi, R.; Shintani, R.; Okabayashi, H.; Ikeda, S.; et al. Outcome of early-stage combination treatment with favipiravir and methylprednisolone for severe COVID-19 pneumonia: A report of 11 cases. Respir. Investig. 2020, 58, 430–434. [Google Scholar] [CrossRef] [PubMed]
- Shindo, Y.; Kondoh, Y.; Kada, A.; Doi, Y.; Tomii, K.; Mukae, H.; Murata, N.; Imai, R.; Okamoto, M.; Yamano, Y.; et al. Phase II Clinical Trial of Combination Therapy with Favipiravir and Methylprednisolone for COVID-19 with Non-Critical Respiratory Failure. Infect. Dis. Ther. 2021, 10, 2353–2369. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhu, Q.; Zhang, C.; Li, J.; Wei, M.; Qin, Y.; Chen, G.; Wang, K.; Yu, J.; Wu, Z.; et al. Tocilizumab combined with favipiravir in the treatment of COVID-19: A multicenter trial in a small sample size. Biomed. Pharmacother. 2021, 133, 110825. [Google Scholar] [CrossRef] [PubMed]
- Doi, Y.; Hibino, M.; Hase, R.; Yamamoto, M.; Kasamatsu, Y.; Hirose, M.; Mutoh, Y.; Homma, Y.; Terada, M.; Ogawa, T.; et al. A Prospective, Randomized, Open-Label Trial of Early versus Late Favipiravir Therapy in Hospitalized Patients with COVID-19. Antimicrob. Agents Chemother. 2020, 64, e01897-20. [Google Scholar] [CrossRef]
- Schultz, D.C.; Johnson, R.M.; Ayyanathan, K.; Miller, J.; Whig, K.; Kamalia, B.; Dittmar, M.; Weston, S.; Hammond, H.L.; Dillen, C.; et al. Pyrimidine biosynthesis inhibitors synergize with nucleoside analogs to block SARS-CoV-2 infection. bioRxiv 2021. [Google Scholar] [CrossRef]
- Schultz, D.C.; Johnson, R.M.; Ayyanathan, K.; Miller, J.; Whig, K.; Kamalia, B.; Dittmar, M.; Weston, S.; Hammond, H.L.; Dillen, C.; et al. Pyrimidine inhibitors synergize with nucleoside analogues to block SARS-CoV-2. Nature 2022, 604, 134–140. [Google Scholar] [CrossRef]
- Padhi, A.K.; Shukla, R.; Saudagar, P.; Tripathi, T. High-throughput rational design of the remdesivir binding site in the RdRp of SARS-CoV-2: Implications for potential resistance. iScience 2021, 24, 101992. [Google Scholar] [CrossRef]
- Baranovich, T.; Wong, S.S.; Armstrong, J.; Marjuki, H.; Webby, R.J.; Webster, R.G.; Govorkova, E.A. T-705 (favipiravir) induces lethal mutagenesis in influenza A H1N1 viruses in vitro. J. Virol. 2013, 87, 3741–3751. [Google Scholar] [CrossRef] [Green Version]
- Perales, C.; Domingo, E. Antiviral Strategies Based on Lethal Mutagenesis and Error Threshold. Curr. Top. Microbiol. Immunol. 2016, 392, 323–339. [Google Scholar] [CrossRef] [PubMed]
- Pachetti, M.; Marini, B.; Benedetti, F.; Giudici, F.; Mauro, E.; Storici, P.; Masciovecchio, C.; Angeletti, S.; Ciccozzi, M.; Gallo, R.C.; et al. Emerging SARS-CoV-2 mutation hot spots include a novel RNA-dependent-RNA polymerase variant. J. Transl. Med. 2020, 18, 179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ullrich, S.; Ekanayake, K.B.; Otting, G.; Nitsche, C. Main protease mutants of SARS-CoV-2 variants remain susceptible to nirmatrelvir. Bioorg. Med. Chem. Lett. 2022, 62, 128629. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Hill, C.S.; Sarkar, S.; Tse, L.V.; Woodburn, B.M.D.; Schinazi, R.F.; Sheahan, T.P.; Baric, R.S.; Heise, M.T.; Swanstrom, R. β-d-N4-hydroxycytidine Inhibits SARS-CoV-2 through Lethal Mutagenesis but Is Also Mutagenic to Mammalian Cells. J. Infect. Dis. 2021, 224, 415–419. [Google Scholar] [CrossRef] [PubMed]
- Swanstrom, R.; Schinazi, R.F. Lethal mutagenesis as an antiviral strategy. Science 2022, 375, 497–498. [Google Scholar] [CrossRef] [PubMed]
- Sheahan, T.P.; Sims, A.C.; Zhou, S.; Graham, R.L.; Pruijssers, A.J.; Agostini, M.L.; Leist, S.R.; Schäfer, A.; Dinnon, K.H., 3rd; Stevens, L.J.; et al. An orally bioavailable broad-spectrum antiviral inhibits SARS-CoV-2 in human airway epithelial cell cultures and multiple coronaviruses in mice. Sci. Transl. Med. 2020, 12, eabb5883. [Google Scholar] [CrossRef] [Green Version]
- Kiso, M.; Takahashi, K.; Sakai-Tagawa, Y.; Shinya, K.; Sakabe, S.; Le, Q.M.; Ozawa, M.; Furuta, Y.; Kawaoka, Y. T-705 (favipiravir) activity against lethal H5N1 influenza A viruses. Proc. Natl. Acad. Sci. USA 2010, 107, 882–887. [Google Scholar] [CrossRef] [Green Version]
- Wang, M.; Cao, R.; Zhang, L.; Yang, X.; Liu, J.; Xu, M.; Shi, Z.; Hu, Z.; Zhong, W.; Xiao, G. Remdesivir and chloroquine effectively inhibit the recently emerged novel coronavirus (2019-nCoV) in vitro. Cell Res. 2020, 30, 269–271. [Google Scholar] [CrossRef]
- Du, Y.X.; Chen, X.P. Favipiravir: Pharmacokinetics and Concerns About Clinical Trials for 2019-nCoV Infection. Clin. Pharmacol. Ther. 2020, 108, 242–247. [Google Scholar] [CrossRef] [Green Version]
- Jia, X.; Ganter, B.; Meier, C. Improving properties of the nucleobase analogs T-705/T-1105 as potential antiviral. Annu. Rep. Med. Chem. 2021, 57, 1–47. [Google Scholar] [CrossRef]
- Cully, M. A tale of two antiviral targets—And the COVID-19 drugs that bind them. Nat. Rev. Drug Discov. 2022, 21, 3–5. [Google Scholar] [CrossRef]
| Therapeutic Class | Representative Drugs |
|---|---|
| Antimicrobials (antibiotics, antifungal agents, anti-tuberculotics) | Rifampin, Rifadin (rifampin, isoniazid, and pyrazinamide), erythromycin, clarithromycin, rifapentine, ketoconazole |
| Analgesics (mostly opioids) | Oxycodone, hydrocodone, fentanyl, tramadol |
| Antiarrhythmics | Amiodarone, disopyramide, dofetilide |
| Anticoagulants/Antiplatelets (including direct-acting oral anticoagulants) | Warfarin, rivaroxaban, apixaban, dabigatran, edoxaban, clopidrogel |
| Antihypertensives | Amlodipine (calcium channel inhibitor), |
| Antidiabetics | Saxagliptin, |
| Lipid-lowering agents | Statins, i.e., lovastatin, simvastatin |
| Anticonvulsants | Carbamazepine, phenytoin, phenobarbital, primidone |
| Antidepressants | Trazodone, St John’s Wort |
| Antipsychotics | Quetiapine, clozapine, aripiprazole, sertindole, pimavanserine, pimozide |
| Anxiolytics | Midazolam, diazepam, alprazolam, triazolam |
| Bronchodilators | Salmeterol |
| Anti-cancer drugs | Kinase inhibitors, cyclin-dependent kinase inhibitors, poly (ADP-ribose) polymerase inhibitors, apalutamide, enzalutamide, ibrutinib |
| Gastrointestinal drugs | Gastroprokinetic cisapride and domperidone |
| HCV antivirals | Elbasvir, glecaprevir, grazoprevir, velpatasvir, voxilaprevir |
| Immunosuppressants | Everolimus, sirolimus, cyclosporin, tacrolimus |
| Steroids | Oral steroids |
| Ergot alkaloids | Dihydroergotamine, ergotamine, ergonovine |
| NCT Identifier | Study Title | Study Phase | Intervention | Study Design | Location |
|---|---|---|---|---|---|
| Favipiravir | |||||
| NCT04464408 | Favipiravir therapy in adults with mild COVID-19 | Phase 3 | Favipiravir: 1800 mg twice daily for one day, followed by 800 mg (4 tablets) twice daily, for 7 days Placebo: matching number of tablets and dosing regimen | Enrollment: 231 subjects, male and female, ≥18 yrs of age and older Intervention model: parallel, double-blind Primary endpoint: time from randomization to negativity in RT-PCR nucleic acid test for COVID-19 within 15 days of randomization | Saudi Arabia |
| NCT04346628 | Oral favipiravir compared to placebo in subjects with mild COVID-19 | Phase 2 | Favipiravir: 1800 mg on the first dose (day 1) followed by 800 mg twice daily for the next 9 days (days 2–10). Placebo: matching number of tablets and dosing regimen | Enrollment: 149 subjects, male and female, ≥18 yrs of age and older Intervention model: parallel, double-blind, placebo-controlled Primary endpoint: time until cessation of oral shedding of SARS-CoV-2 virus within 28 days of randomization | United States (Stanford University) |
| NCT04349241 | Efficacy and safety of favipiravir in management of COVID-19 (FAV-001) | Phase 3 | Favipiravir: 3200 mg (1600 mg 12 hourly) loading dose on day 1 followed by 1200 mg maintenance dose (600 mg 12 hourly daily) on day 2 to day 10 Active comparator: oseltamivir 75 mg 12 hourly for 5–10 days and hydroxychloroquine 400 mg 12 hourly day 1 followed by 200 mg 12 hourly daily on day 2 to day 5–10 | Enrolment: 100 subjects, male and female, 18–80 years of age Intervention model: parallel, open-label, active comparator-controlled Primary endpoint: time to viral clearance within 14 days of randomization | Egypt |
| NCT04600895 | The Prevent severe COVID-19 (PRESECO) | Phase 3 | Dosing regimen of favipiravir not specified | Enrolment: 1231 male and female subjects, ≥18 yrs of age and older Intervention model: parallel, double-blind, placebo-controlled Primary endpoint: time to sustained clinical recovery | United States Mexico Brazil |
| NCT04981379 | Efficacy and Safety of the Use of Hydroxychloroquine, Favipiravir or Hydroxychloroquine + Favipiravir in Early SARS-CoV-2 (COVID-19) Treatment | Phase 3 | Favipiravir + placebo (hydroxychloroquine): Favipiravir (1600 mg), as two tablets per day at the first day and then Favipiravir (600 mg) as two tablets per day for the remaining 4-day interval + placebo (Hydroxychloroquine (200 mg)), as two tablets per day for 5-day interval Hydroxychloroquine + placebo (favipiravir): Hydroxychloroquine (200 mg), as two tablets per day for 5-day interval + placebo (Favipiravir (1600 mg)), as two tablets per day at the first day and then Favipiravir (600 mg) as two tablet per day for the remaining 4-day interval Favipiravir + Hydroxychloroquine: Favipiravir (1600 mg), as two tablet per day at the first day and then Favipiravir (600 mg) as two tablet per day for the remaining 4-day interval + Hydroxychloroquine (200 mg), as two tablets per day for 5-day interval Placebo + active comparator: Placebo (favipiravir) 2 × 1600 mg loading, then 4 days 2 × 600 mg maintenance (5 days) + placebo (Hydroxychloroquine) 2 × 200 mg (5 days) | Enrolment: 1120 subjects, male and female, 18 to 59 years old Intervention model: parallel, double-blind, placebo- and active comparator-controlled Primary endpoint: clinical worsening | Turkey |
| NCT04499677 | FLARE: favipiravir +/− lopinavir | Phase 2 | Favipiravir + Lopinavir/ritonavir (LPV/r) Oral favipiravir at 1800 mg twice daily on Day 1, followed by 400 mg four (4) times daily from day 2 to day 7 PLUS Lopinavir/ritonavir (LPV/r) at 400 mg/100 mg twice daily on day 1, followed by 200 mg/50 mg four (4) times daily from day 2 to day 7 Favipiravir + Lopinavir/ritonavir (LPV/r) placebo Oral favipiravir at 1800 mg twice daily on day 1, followed by 400 mg four (4) times daily from day 2 to day 7 PLUS Lopinavir/ritonavir (LPV/r) matched placebo at 400 mg/100 mg twice daily on day 1, followed by 200 mg/50 mg four (4) times daily from day 2 to day 7 Favipiravir placebo + Lopinavir/ritonavir (LPV/r) Oral favipiravir matched placebo at 1800 mg twice daily on day 1, by 400 mg four (4) times daily from day 2 to day 7 PLUS Lopinavir/ritonavir (LPV/r) at 400 mg/100 mg twice daily on day 1, followed by 200 mg/50 mg four (4) times daily from day 2 to day 7 Favipiravir placebo + Lopinavir/ritonavir (LPV/r) placebo Oral favipiravir matched placebo at 1800 mg twice daily on day 1, by 400 mg four (4) times daily from day 2 to day 7 PLUS Lopinavir/ritonavir (LPV/r) matched placebo at 400 mg/100 mg twice daily on day 1, followed by 200 mg/50 mg four (4) times daily from day 2 to day 7 | Enrolment: 240 subjects, male and female, aged 17–70 years Intervention model: randomized, double-blind, 2 × 2 factorial placebo-controlled Primary endpoint: upper respiratory tract viral load at Day 5 | United Kingdom |
| NCT04373733 | Early intervention in COVID-19: favipiravir vs. standard care (PIONEER) | Phase 3 | Favipiravir and standard of care: Favipiravir—day 1, 1800 mg twice per day; days 2–10, 800 mg twice per day Standard care: No trial intervention | Enrolment: 502 adult subjects Intervention model: open-label parallel group randomized control trial. One trial treatment arm and one standard of care comparator arm Primary study endpoint: time from randomization to a sustained clinical improvement (maintained for 24 h) by two points on a seven-category ordinal scale or to discharge, whichever occurs first | UK Brazil |
| Molnupiravir | |||||
| NCT04575597 | A Phase 2/3, Randomized, Placebo-Controlled, Double-Blind Clinical Study to Evaluate the Efficacy, Safety, and Pharmacokinetics of MK-4482 in Non-Hospitalized Adults With COVID-19 | Phase 2/3 | Part I (Phase 2) Molnupiravir 200 mg 200 mg Molnupiravir administered orally every 12 h for 5 days (10 doses total) Molnupiravir 400 mg 400 mg Molnupiravir administered orally every 12 h for 5 days (10 doses total) Molnupiravir 800 mg 800 mg Molnupiravir administered orally every 12 h for 5 days (10 doses total) Placebo Placebo matching Molnupiravir administered orally every 12 h for 5 days (10 doses total) Part 2 (Phase 3) Molnupiravir 800 mg 800 mg Molnupiravir administered orally every 12 h for 5 days (10 doses total) Placebo Placebo matching Molnupiravir administered orally every 12 h for 5 days (10 doses total) | Enrolment: 1734 subjects ≥18 yrs of age and older Intervention model: parallel, double-blind, placebo-controlled Primary endpoint: percentage of participants who are hospitalized and/or die | United States |
| NCT04405570 | A Phase IIa Randomized, Double-Blind, Placebo-Controlled Trial to Evaluate the Safety, Tolerability and Efficacy of EIDD-2801 to Eliminate SARS-CoV-2RNA Detection in Persons with COVID-19 | Phase 2 | Molnupiravir: 200, 400, and 800 mg twice daily (BID) for 5 days Placebo: twice daily (BID) for 5 days | Enrolment: 204 subjects, ≥18 yrs of age and older Intervention model: parallel, double-blind, placebo-controlled Primary endpoint: number of participants until first non-detectable SARS-CoV-2 in nasopharyngeal (NP) swabs | United States |
| NCT04939428 | A Phase 3, Multicenter, Randomized, Double-blind, Placebo-controlled Study to Evaluate the Efficacy and Safety of MK-4482 for the Prevention of COVID-19 (Laboratory-confirmed SARS-CoV-2 Infection with Symptoms) in Adults Residing with a Person With COVID-19 (MOVe AHEAD) | Phase 3 | Molnupiravir: 800 mg every 12 h (Q12H) on Days 1 to 5 Placebo: Q12H on Days 1 to 5 | Enrolment: 1376 subjects, ≥18 yrs of age and older Intervention model: parallel, double-blind, placebo-controlled Primary endpoint: percentage of participants who have undetectable SARS-CoV-2 in baseline nasopharyngeal (NP) swabs and develop COVID-19 | United States |
| Paxlovid No completed Phase 3 trials in outpatients with mild to moderate COVID-19 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corritori, S.; Savchuk, N.; Pauza, C.D. Risk/Benefit Profiles of Currently Approved Oral Antivirals for Treatment of COVID-19: Similarities and Differences. COVID 2022, 2, 1057-1076. https://doi.org/10.3390/covid2080078
Corritori S, Savchuk N, Pauza CD. Risk/Benefit Profiles of Currently Approved Oral Antivirals for Treatment of COVID-19: Similarities and Differences. COVID. 2022; 2(8):1057-1076. https://doi.org/10.3390/covid2080078
Chicago/Turabian StyleCorritori, Suzana, Nikolay Savchuk, and C. David Pauza. 2022. "Risk/Benefit Profiles of Currently Approved Oral Antivirals for Treatment of COVID-19: Similarities and Differences" COVID 2, no. 8: 1057-1076. https://doi.org/10.3390/covid2080078
APA StyleCorritori, S., Savchuk, N., & Pauza, C. D. (2022). Risk/Benefit Profiles of Currently Approved Oral Antivirals for Treatment of COVID-19: Similarities and Differences. COVID, 2(8), 1057-1076. https://doi.org/10.3390/covid2080078





