Antibodies to Commonly Circulating Viral Pathogens Modulate Serological Response to Severe Acute Respiratory Syndrome Coronavirus 2 Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design, Setting, Selection of Households and Participants
2.2. Collection of Data
2.3. Collection, Processing and Storage of Specimen
2.4. Assessment of SARS-CoV-2 Specific Antibodies
2.5. Assessment of Antibodies to Other Viral Pathogens
2.6. Complete Blood Count (CBC)
2.7. Data Analysis Plan
3. Results
3.1. Demography
3.2. Seroprevalence of SARS-CoV-2 and Other Circulating Viruses
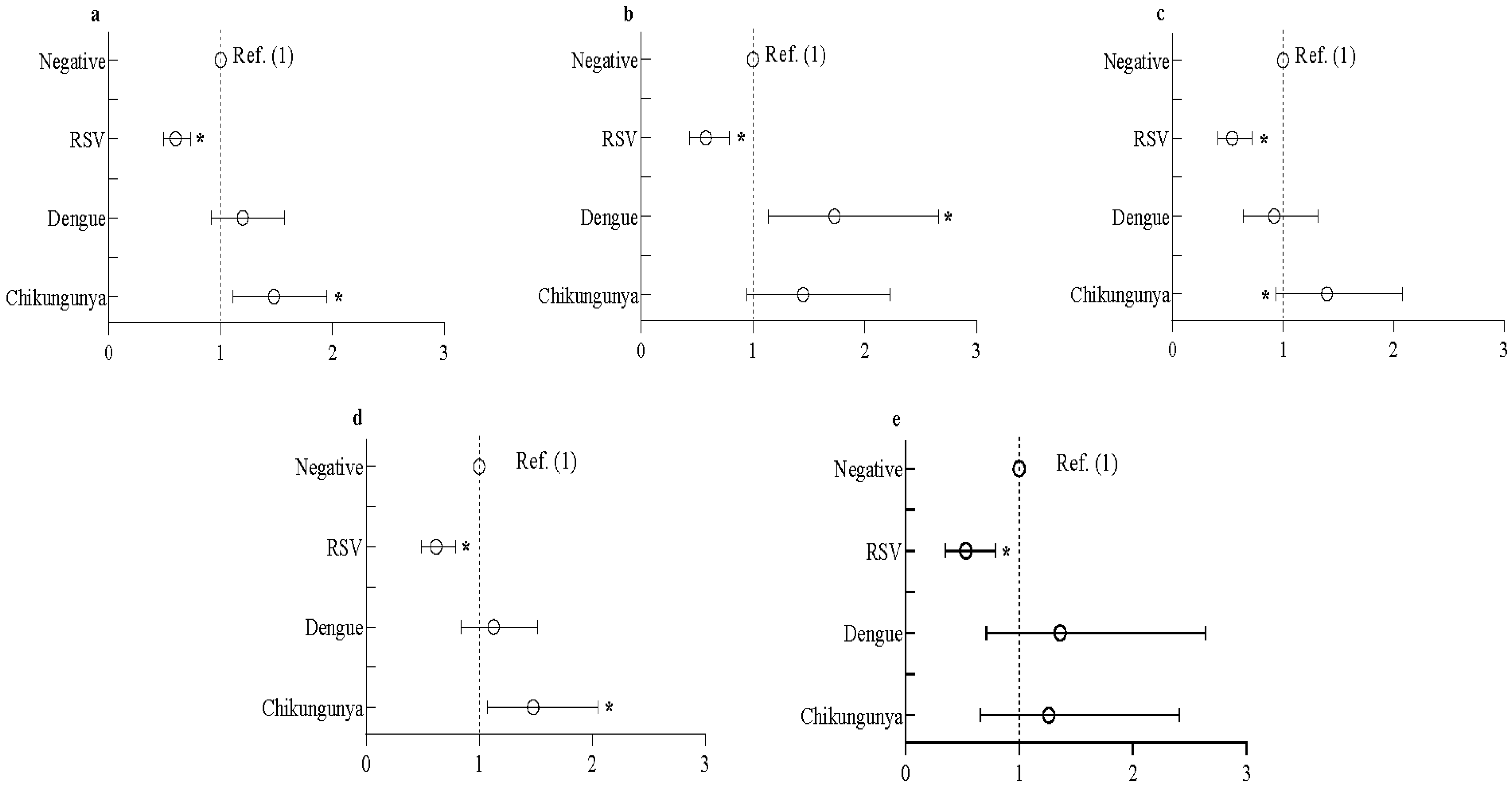
3.3. Association between Seropositivity of SARS-CoV-2 and RSV/Dengue/Chikungunya
3.4. Blood Cell Counts in Seropositive vs. Seronegative Groups of SARS-CoV-2/RSV/Dengue/Chikungunya
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kutter, J.S.; Spronken, M.I.; Fraaij, P.L.; Fouchier, R.A.M.; Herfst, S. Transmission routes of respiratory viruses among humans. Curr. Opin. Virol. 2018, 28, 142–151. [Google Scholar] [CrossRef] [PubMed]
- Nickbakhsh, S.; Thorburn, F.; Wissmann, B.V.; McMenamin, J.; Gunson, R.N.; Murcia, P.R. Extensive multiplex PCR diagnostics reveal new insights into the epidemiology of viral respiratory infections. Epidemiol. Infect. 2016, 144, 2064–2076. [Google Scholar] [CrossRef]
- Alpaydin, A.O.; Gezer, N.S.; Simsek, G.O.; Tertemiz, K.C.; Kutsoylu, O.O.E.; Zeka, A.N.; Guzel, I.; Soyturk, M.; Sayiner, A.A.; Oguz, V.A. Clinical and radiological diagnosis of non-SARS-CoV-2 viruses in the era of COVID-19 pandemic. J. Med. Virol. 2021, 93, 1119–1125. [Google Scholar] [CrossRef]
- Chen, X.; Liao, B.; Cheng, L.; Peng, X.; Xu, X.; Li, Y.; Hu, T.; Li, J.; Zhou, X.; Ren, B. The microbial coinfection in COVID-19. Appl. Microbiol. Biotechnol. 2020, 104, 7777–7785. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.; Quinn, J.; Pinsky, B.; Shah, N.H.; Brown, I. Rates of Co-infection Between SARS-CoV-2 and Other Respiratory Pathogens. JAMA 2020, 323, 2085–2086. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.W.; Deveson, I.W.; Pang, C.N.I.; Yeang, M.; Naing, Z.; Adikari, T.; Hammond, J.M.; Stevanovski, I.; Beukers, A.G.; Verich, A.; et al. Respiratory viral co-infections among SARS-CoV-2 cases confirmed by virome capture sequencing. Sci. Rep. 2021, 11, 3934. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.C.; Wang, C.Y.; Hsueh, P.R. Co-infections among patients with COVID-19: The need for combination therapy with non-anti-SARS-CoV-2 agents? J. Microbiol. Immunol. Infect. 2020, 53, 505–512. [Google Scholar] [CrossRef]
- Lehmann, C.J.; Pho, M.T.; Pitrak, D.; Ridgway, J.P.; Pettit, N.N. Community-acquired Coinfection in Coronavirus Disease 2019: A Retrospective Observational Experience. Clin. Infect. Dis. 2021, 72, 1450–1452. [Google Scholar] [CrossRef]
- Marriott, D.; Beresford, R.; Mirdad, F.; Stark, D.; Glanville, A.; Chapman, S.; Harkness, J.; Dore, G.J.; Andresen, D.; Matthews, G.V. Concomitant Marked Decline in Prevalence of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) and Other Respiratory Viruses Among Symptomatic Patients Following Public Health Interventions in Australia: Data from St Vincent’s Hospital and Associated Screening Clinics, Sydney, NSW. Clin. Infect. Dis. 2021, 72, e649–e651. [Google Scholar]
- Wee, L.E.; Ko, K.K.K.; Ho, W.Q.; Kwek, G.T.C.; Tan, T.T.; Wijaya, L. Community-acquired viral respiratory infections amongst hospitalized inpatients during a COVID-19 outbreak in Singapore: Co-infection and clinical outcomes. J. Clin. Virol. 2020, 128, 104436. [Google Scholar] [CrossRef] [PubMed]
- Biernat, M.M.; Zinczuk, A.; Biernat, P.; Bogucka-Fedorczuk, A.; Kwiatkowski, J.; Kalicinska, E.; Marciniak, D.; Simon, K.; Wrobel, T. Nosocomial outbreak of SARS-CoV-2 infection in a haematological unit—High mortality rate in infected patients with haematologic malignancies. J. Clin. Virol. 2020, 130, 104574. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, S.A.; Safamanesh, S.; Ghasemzadeh-Moghaddam, H.; Ghafouri, M.; Azimian, A. High prevalence of SARS-CoV-2 and influenza A virus (H1N1) coinfection in dead patients in Northeastern Iran. J. Med. Virol. 2021, 93, 1008–1012. [Google Scholar] [CrossRef] [PubMed]
- Calderaro, A.; De Conto, F.; Buttrini, M.; Piccolo, G.; Montecchini, S.; Maccari, C.; Martinelli, M.; Di Maio, A.; Ferraglia, F.; Pinardi, F.; et al. Human respiratory viruses, including SARS-CoV-2, circulating in the winter season 2019–2020 in Parma, Northern Italy. Int. J. Infect. Dis. 2021, 102, 79–84. [Google Scholar] [CrossRef]
- Galli, C.; Pellegrinelli, L.; Bubba, L.; Primache, V.; Anselmi, G.; Delbue, S.; Signorini, L.; Binda, S.; Cereda, D.; Gramegna, M.; et al. When the COVID-19 Pandemic Surges during Influenza Season: Lessons Learnt from the Sentinel Laboratory-Based Surveillance of Influenza-Like Illness in Lombardy during the 2019–2020 Season. Viruses 2021, 13, 695. [Google Scholar] [CrossRef]
- Liu, Y.; Lillepold, K.; Semenza, J.C.; Tozan, Y.; Quam, M.B.M.; Rocklov, J. Reviewing estimates of the basic reproduction number for dengue, Zika and chikungunya across global climate zones. Environ. Res. 2020, 182, 109114. [Google Scholar] [CrossRef]
- Tozan, Y.; Sjodin, H.; Munoz, A.G.; Rocklov, J. Transmission dynamics of dengue and chikungunya in a changing climate: Do we understand the eco-evolutionary response? Expert Rev. Anti-Infect. Ther. 2020, 18, 1187–1193. [Google Scholar] [CrossRef]
- Banu, S.; Hu, W.; Hurst, C.; Guo, Y.; Islam, M.Z.; Tong, S. Space-time clusters of dengue fever in Bangladesh. Trop. Med. Int. Health 2012, 17, 1086–1091. [Google Scholar] [CrossRef]
- Rahman, M.; Rahman, K.; Siddque, A.K.; Shoma, S.; Kamal, A.H.; Ali, K.S.; Nisaluk, A.; Breiman, R.F. First outbreak of dengue hemorrhagic fever, Bangladesh. Emerg. Infect. Dis. 2002, 8, 738–740. [Google Scholar] [CrossRef]
- Sharmin, S.; Viennet, E.; Glass, K.; Harley, D. The emergence of dengue in Bangladesh: Epidemiology, challenges and future disease risk. Trans R Soc. Trop. Med. Hyg. 2015, 109, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Yunus, E.B.; Bangali, M.A.; Mahmood, M.A.H.; Rahman, M.M.; Chowdhury, A.R.; Talukder, K.R. Dengue Outbreak 2000 in Bangladesh: From Speculation to Reality and Exercises. Dengue Bull. 2001, 25, 15–20. [Google Scholar]
- Karim, M.N.; Munshi, S.U.; Anwar, N.; Alam, M.S. Climatic factors influencing dengue cases in Dhaka city: A model for dengue prediction. Indian J. Med. Res. 2012, 136, 32–39. [Google Scholar] [PubMed]
- Mutsuddy, P.; Jhora, S.T.; Shamsuzzaman, A.K.M.; Kaisar, S.M.G.; Khan, M.N.A. Dengue Situation in Bangladesh: An Epidemiological Shift in terms of Morbidity and Mortality. Can. J. Infect. Dis. Med. Microbiol. 2019, 2019, 3516284. [Google Scholar] [CrossRef]
- Harapan, H.; Ryan, M.; Yohan, B.; Abidin, R.S.; Nainu, F.; Rakib, A.; Jahan, I.; Emran, T.B.; Ullah, I.; Panta, K.; et al. Covid-19 and dengue: Double punches for dengue-endemic countries in Asia. Rev. Med. Virol. 2021, 31, e2161. [Google Scholar] [CrossRef]
- Nicolelis, M.A.L.; Raimundo, R.L.G.; Peixoto, P.S.; de Andreazzi, C.S. How super-spreader cities, highways, hospital bed availability, and dengue fever influenced the covid-19 epidemic in Brazil. medRxiv 2020. [Google Scholar] [CrossRef]
- Raqib, R.; Sarker, P.; Akhtar, E.; Huda, T.M.N.; Haq, M.A.; Roy, A.K.; Hosen, M.B.; Haque, F.; Chowdhury, M.R.; Reidpath, D.D.; et al. Seroprevalence of SARS-CoV-2 infection and associated factors among Bangladeshi slum and non-slum dwellers in pre-COVID-19 vaccination era: October 2020 to February 2021. PLoS ONE 2022, 17, e0268093. [Google Scholar] [CrossRef] [PubMed]
- Galipeau, Y.; Greig, M.; Liu, G.; Driedger, M.; Langlois, M.A. Humoral Responses and Serological Assays in SARS-CoV-2 Infections. Front. Immunol. 2020, 11, 610688. [Google Scholar] [CrossRef]
- Falsey, A.R.; McElhaney, J.E.; Beran, J.; van Essen, G.A.; Duval, X.; Esen, M.; Galtier, F.; Gervais, P.; Hwang, S.J.; Kremsner, P.; et al. Respiratory syncytial virus and other respiratory viral infections in older adults with moderate to severe influenza-like illness. J. Infect. Dis. 2014, 209, 1873–1881. [Google Scholar] [CrossRef]
- Glezen, W.P.; Taber, L.H.; Frank, A.L.; Kasel, J.A. Risk of primary infection and reinfection with respiratory syncytial virus. Am. J. Dis. Child 1986, 140, 543–546. [Google Scholar] [CrossRef]
- Hall, H.C.B.; Long, C.E.; Schnabel, K.C. Respiratory syncytial virus infections in previously healthy working adults. Clin. Infect. Dis. 2001, 33, 792–796. [Google Scholar] [CrossRef]
- Bourgeois, F.T.; Valim, C.; McAdam, A.J.; Mandl, K.D. Relative impact of influenza and respiratory syncytial virus in young children. Pediatrics 2009, 124, e1072–e1080. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.B.; Weinberg, G.A.; Blumkin, A.K.; Edwards, K.M.; Staat, M.A.; Schultz, A.F.; Poehling, K.A.; Szilagyi, P.G.; Griffin, M.R.; Williams, J.V.; et al. Respiratory syncytial virus-associated hospitalizations among children less than 24 months of age. Pediatrics 2013, 132, e341–e348. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Thompson, W.W.; Viboud, C.G.; Ringholz, C.M.; Cheng, P.Y.; Steiner, C.; Abedi, G.R.; Anderson, L.J.; Brammer, L.; Shay, D.K. Hospitalizations associated with influenza and respiratory syncytial virus in the United States, 1993–2008. Clin. Infect. Dis. 2012, 54, 1427–1436. [Google Scholar] [CrossRef]
- Crowcroft, N.S.; Zambon, M.; Harrison, T.G.; Mok, Q.; Heath, P.; Miller, E. Respiratory syncytial virus infection in infants admitted to paediatric intensive care units in London, and in their families. Eur. J. Pediatr. 2008, 167, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Hall, C.B.; Geiman, J.M.; Biggar, R.; Kotok, D.I.; Hogan, P.M.; Douglas, G.R., Jr. Respiratory syncytial virus infections within families. N. Engl. J. Med. 1976, 294, 414–419. [Google Scholar] [CrossRef]
- Heikkinen, T.; Valkonen, H.; Waris, M.; Ruuskanen, O. Transmission of respiratory syncytial virus infection within families. Open Forum Infect. Dis. 2015, 2, ofu118. [Google Scholar] [CrossRef] [PubMed]
- Munywoki, P.K.; Koech, D.C.; Agoti, C.N.; Lewa, C.; Cane, P.A.; Medley, G.F.; Nokes, D.J. The source of respiratory syncytial virus infection in infants: A household cohort study in rural Kenya. J. Infect. Dis. 2014, 209, 1685–1692. [Google Scholar] [CrossRef]
- Nickbakhsh, S.; Mair, C.; Matthews, L.; Reeve, R.; Johnson, P.C.D.; Thorburn, F.; von Wissmann, B.; Reynolds, A.; McMenamin, J.; Gunson, R.N.; et al. Virus-virus interactions impact the population dynamics of influenza and the common cold. Proc. Natl. Acad. Sci. USA 2019, 116, 27142–27150. [Google Scholar] [CrossRef] [PubMed]
- Loos, C.; Atyeo, C.; Fischinger, S.; Burke, J.; Slein, M.D.; Streeck, H.; Lauffenburger, D.; Ryan, E.T.; Charles, R.C.; Alter, G. Evolution of Early SARS-CoV-2 and Cross-Coronavirus Immunity. mSphere 2020, 5, e00622-20. [Google Scholar] [CrossRef]
- Masyeni, S.; Santoso, M.S.; Widyaningsih, P.D.; Asmara, D.W.; Nainu, F.; Harapan, H.; Sasmono, R.T. Serological cross-reaction and coinfection of dengue and COVID-19 in Asia: Experience from Indonesia. Int. J. Infect. Dis. 2021, 102, 152–154. [Google Scholar] [CrossRef]
- Khairunisa, S.Q.; Amarullah, I.H.; Churrotin, S.; Fitria, A.L.; Amin, M.; Lusida, M.I.; Soegijanto, S. Potential Misdiagnosis between COVID-19 and Dengue Infection Using Rapid Serological Test. Infect. Dis. Rep. 2021, 13, 50. [Google Scholar] [CrossRef]
- Vanroye, F.; den Bossche, D.V.; Brosius, I.; Tack, B.; Esbroeck, M.V.; Jacobs, J. COVID-19 Antibody Detecting Rapid Diagnostic Tests Show High Cross-Reactivity When Challenged with Pre-Pandemic Malaria, Schistosomiasis and Dengue Samples. Diagnostics 2021, 11, 1163. [Google Scholar] [CrossRef]
- Lippi, G.; Plebani, M.; Henry, B.M. Thrombocytopenia is associated with severe coronavirus disease 2019 (COVID-19) infections: A meta-analysis. Clin. Chim. Acta 2020, 506, 145–148. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.; Niloofa, R.; Jayarajah, U.; De Mel, S.; Abeysuriya, V.; Seneviratne, S.L. Hematological Abnormalities in COVID-19: A Narrative Review. Am. J. Trop. Med. Hyg. 2021, 104, 1188. [Google Scholar] [CrossRef] [PubMed]
| Variables | Overall (n = 2012) | Slum (n = 1099) | Non-Slum (n = 913) | p Value | |
|---|---|---|---|---|---|
| Age, years | 31.48 ± 16.23 | 29.65 ± 15.58 | 33.67 ± 16.72 | <0.001 | |
| Sex | Male | 850 (42.2%) | 487 (44.3%) | 363 (39.8%) | 0.040 |
| Female | 1162 (57.8%) | 612 (55.7%) | 550 (60.2%) | ||
| Education | No education | 378 (18.8%) | 336 (30.6%) | 42 (4.60%) | <0.001 |
| 1–5 years | 564 (28.0%) | 445 (40.5%) | 119 (13.0%) | <0.001 | |
| 6–10 years | 665 (33.1%) | 281 (25.6%) | 384 (42.1%) | <0.001 | |
| 11–15 years | 405 (20.1%) | 37 (3.37%) | 368 (40.3%) | <0.001 | |
| Occupation | Service | 318 (15.8%) | 189 (17.2%) | 129 (14.1%) | 0.342 |
| Self-employed | 199 (9.89%) | 168 (15.3%) | 31 (3.40%) | 0.001 | |
| Business | 163 (8.10%) | 73 (6.64%) | 90 (9.86%) | 0.214 | |
| Housewife | 545 (27.1%) | 233 (21.2%) | 312 (34.2%) | 0.002 | |
| Unemployed | 236 (11.7%) | 157 (14.3%) | 79 (8.65%) | 0.110 | |
| Student | 551 (27.4%) | 279 (25.4%) | 272 (29.8%) | 0.865 | |
| Monthly income, taka | <20,000 | 360 (17.9%) | 340 (30.9%) | 20 (2.19%) | <0.001 |
| 20,000–40,000 | 798 (39.7%) | 624 (56.8%) | 174 (19.1%) | <0.001 | |
| 40,000–70,000 | 528 (26.2%) | 135 (12.3%) | 393 (43.0%) | <0.001 | |
| >70,000 | 326 (16.2%) | 0 | 326 (35.7%) | <0.001 | |
| BMI | Underweight | 374 (18.6%) | 281 (25.6%) | 93 (10.2%) | <0.001 |
| Normal | 837 (38.8%) | 483 (44.0%) | 354 (38.8%) | 0.120 | |
| Overweight | 801 (39.8%) | 335 (30.5%) | 466 (51.0%) | <0.001 | |
| COVID-19 like symptoms | 810 (40.3%) | 392 (35.7%) | 418 (45.8%) | <0.001 |
| Overall (n = 2012) | Slum vs. Non-Slum | Adult vs. Adolescent | |||||
|---|---|---|---|---|---|---|---|
| Slum (n = 1099) | Non-Slum (n = 913) | p Value | Adult (n = 1515) | Adolescent (n = 497) | p Value | ||
| SARS-CoV-2 | 1375 (68.3%) | 807 (73.4%) | 568 (62.2%) | <0.001 | 1064 (70.2%) | 311 (62.6%) | 0.001 |
| RSV | 1005 (50.0%) | 662 (60.2%) | 343 (37.6%) | <0.001 | 750 (49.5%) | 255 (51.3%) | 0.485 |
| HCoV-HKU1 | 67 (3.36%) | 42 (4.0%) | 25 (2.68%) | 0.116 | 44 (3.12%) | 23 (3.92%) | 0.009 |
| Dengue | 332 (16.5%) | 171 (15.6%) | 161 (17.6%) | 0.212 | 183 (18.7%) | 49 (9.86%) | <0.001 |
| Chikungunya | 312 (15.5%) | 169 (15.6%) | 143 (15.4%) | 0.927 | 260 (17.1%) | 52 (10.6%) | 0.007 |
| Overall (n = 2012) | Slum (n = 1099) | Non-Slum (n = 913) | Adult (n = 1515) | Adolescent (n = 497) | ||
|---|---|---|---|---|---|---|
| RSV | Seropositive | 61.3 (57.9, 64.6) | 65.4 (61.2, 69.4) | 53.9 (48.0, 59.7) | 64.2 (60.3, 67.9) | 53.6 (46.9, 60.1) |
| Seronegative | 72.4 (68.5, 75.9) | 76.9 (71.7, 81.4) | 69.6 (64.2, 74.5) | 74.0 (69.7, 78.0) | 67.2 (58.9, 74.6) | |
| Dengue | Seropositive | 73.2 (67.1, 78.5) | 80.1 (72.8, 85.7) | 66.6 (56.7, 75.1) | 74.2 (67.7, 79.9) | 67.9 (51.7, 80.6) |
| Seronegative | 65.1 (62.2, 67.8) | 67.2 (63.6, 70.7) | 62.7 (58.2, 66.9) | 67.6 (64.3, 70.7) | 58.6 (53.0, 63.9) | |
| Chikungunya | Seropositive | 75.3 (68.8, 80.8) | 79.4 (71.7, 85.4) | 71.7 (61.3, 80.2) | 76.4 (69.3, 82.2) | 68.9 (51.9, 82.0) |
| Seronegative | 67.1 (64.3, 69.7) | 70.9 (67.4, 74.1) | 62.3 (57.8, 66.6) | 69.0 (65.9, 72.0) | 61.9 (56.4, 67.2) |
| Blood Cells | SARS-CoV-2 | RSV | Dengue | Chikungunya | ||||
|---|---|---|---|---|---|---|---|---|
| β (95% CI) | p Value | β (95% CI) | p Value | β (95% CI) | p Value | β (95% CI) | p Value | |
| Neutrophils | 0.06 (−0.80, 0.92) | 0.892 | −0.10 (−0.91, 0.71) | 0.802 | −0.31 (−1.38, 0.76) | 0.570 | 1.24 (0.15, 2.34) | 0.026 |
| Lymphocytes | 0.02 (−0.74, 0.79) | 0.955 | −0.45 (−1.15, 0.25) | 0.206 | 0.54 (−0.41, 1.49) | 0.265 | −0.29 (−1.26, 0.69) | 0.564 |
| Monocytes | 0.43 (0.30, 0.55) | <0.001 | −1.23 (−1.33, −1.12) | <0.001 | −0.06 (−0.22, 0.10) | 0.486 | 0.27 (0.12, 0.43) | 0.001 |
| Eosinophils | 0.70 (0.30, 1.09) | <0.001 | −1.56 (−1.93, −1.19) | <0.001 | −0.59 (−1.09, −0.09) | 0.020 | −0.08 (−0.57, 0.41) | 0.751 |
| Basophils | 0.02 (0.002, 0.03) | 0.024 | 0.01 (−0.001, 0.03) | 0.060 | 0.001 (−0.02, 0.02) | 0.891 | 0.01 (−0.01, 0.02) | 0.290 |
| Platelet | −18.6 (−27.7, −9.49) | <0.001 | 5.42 (−3.22, 14.1) | 0.219 | 9.68 (−1.73, 21.1) | 0.096 | −41.7 (−53.1, −30.2) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarker, P.; Akhtar, E.; Akter, S.; Rajia, S.; Kuddusi, R.U.; Ahmed, R.; Jakarea, M.; Islam, M.Z.; Hoque, D.M.E.; Ahmed, S.; et al. Antibodies to Commonly Circulating Viral Pathogens Modulate Serological Response to Severe Acute Respiratory Syndrome Coronavirus 2 Infection. COVID 2022, 2, 1625-1634. https://doi.org/10.3390/covid2120117
Sarker P, Akhtar E, Akter S, Rajia S, Kuddusi RU, Ahmed R, Jakarea M, Islam MZ, Hoque DME, Ahmed S, et al. Antibodies to Commonly Circulating Viral Pathogens Modulate Serological Response to Severe Acute Respiratory Syndrome Coronavirus 2 Infection. COVID. 2022; 2(12):1625-1634. https://doi.org/10.3390/covid2120117
Chicago/Turabian StyleSarker, Protim, Evana Akhtar, Sharmin Akter, Sultana Rajia, Rakib Ullah Kuddusi, Razu Ahmed, Md. Jakarea, Mohammad Zahirul Islam, Dewan Md Emdadul Hoque, Shehlina Ahmed, and et al. 2022. "Antibodies to Commonly Circulating Viral Pathogens Modulate Serological Response to Severe Acute Respiratory Syndrome Coronavirus 2 Infection" COVID 2, no. 12: 1625-1634. https://doi.org/10.3390/covid2120117
APA StyleSarker, P., Akhtar, E., Akter, S., Rajia, S., Kuddusi, R. U., Ahmed, R., Jakarea, M., Islam, M. Z., Hoque, D. M. E., Ahmed, S., & Raqib, R. (2022). Antibodies to Commonly Circulating Viral Pathogens Modulate Serological Response to Severe Acute Respiratory Syndrome Coronavirus 2 Infection. COVID, 2(12), 1625-1634. https://doi.org/10.3390/covid2120117







