Characterization and Functionalization Approaches for the Study of Polymeric Nanoparticles: The State of the Art in Italian Research
Abstract
1. Introduction
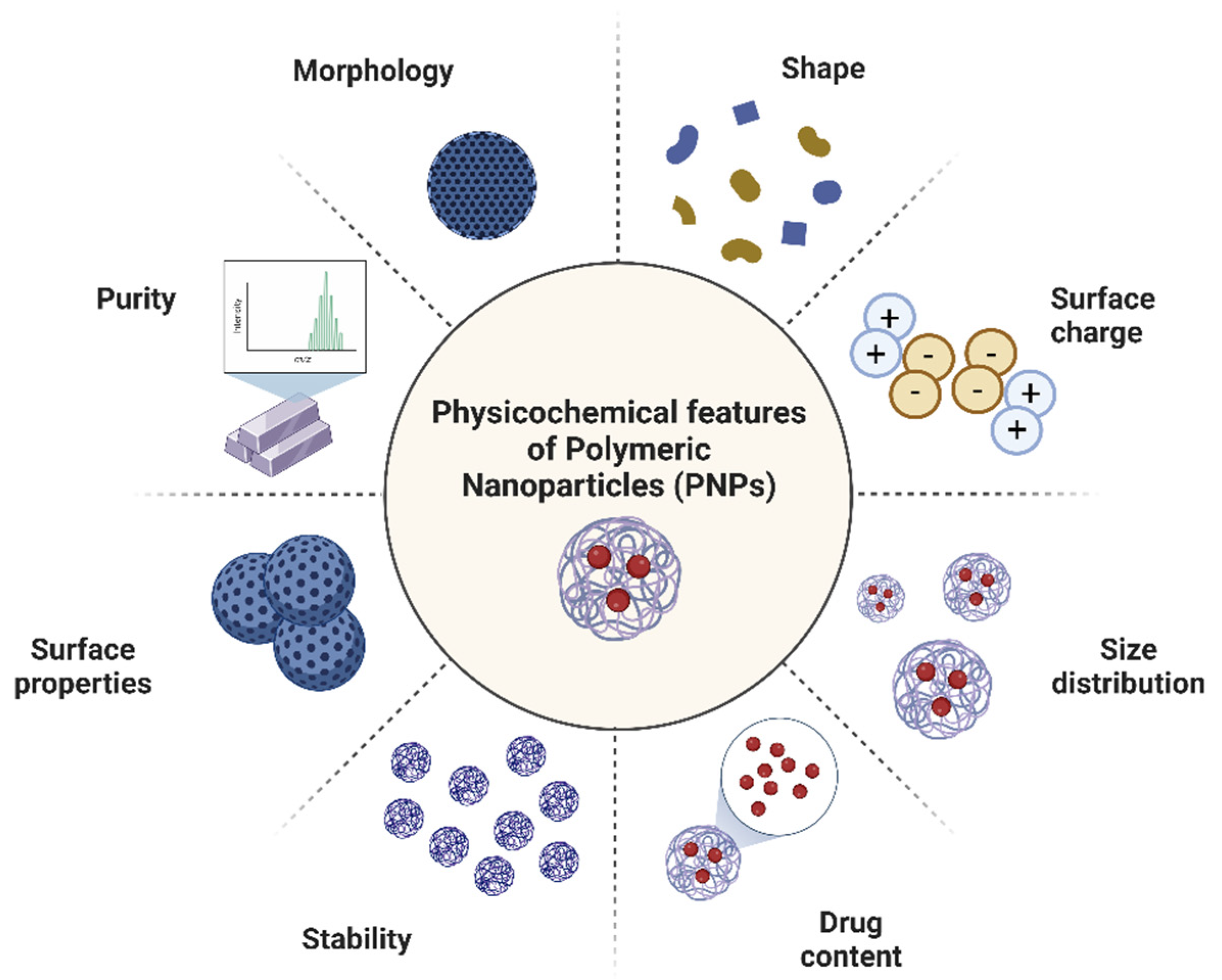
2. Methods Used for Nanoparticles Characterization
2.1. Particle Size Distribution and Morphological Characterizations
2.2. Chemical Composition and Structural Characterizations
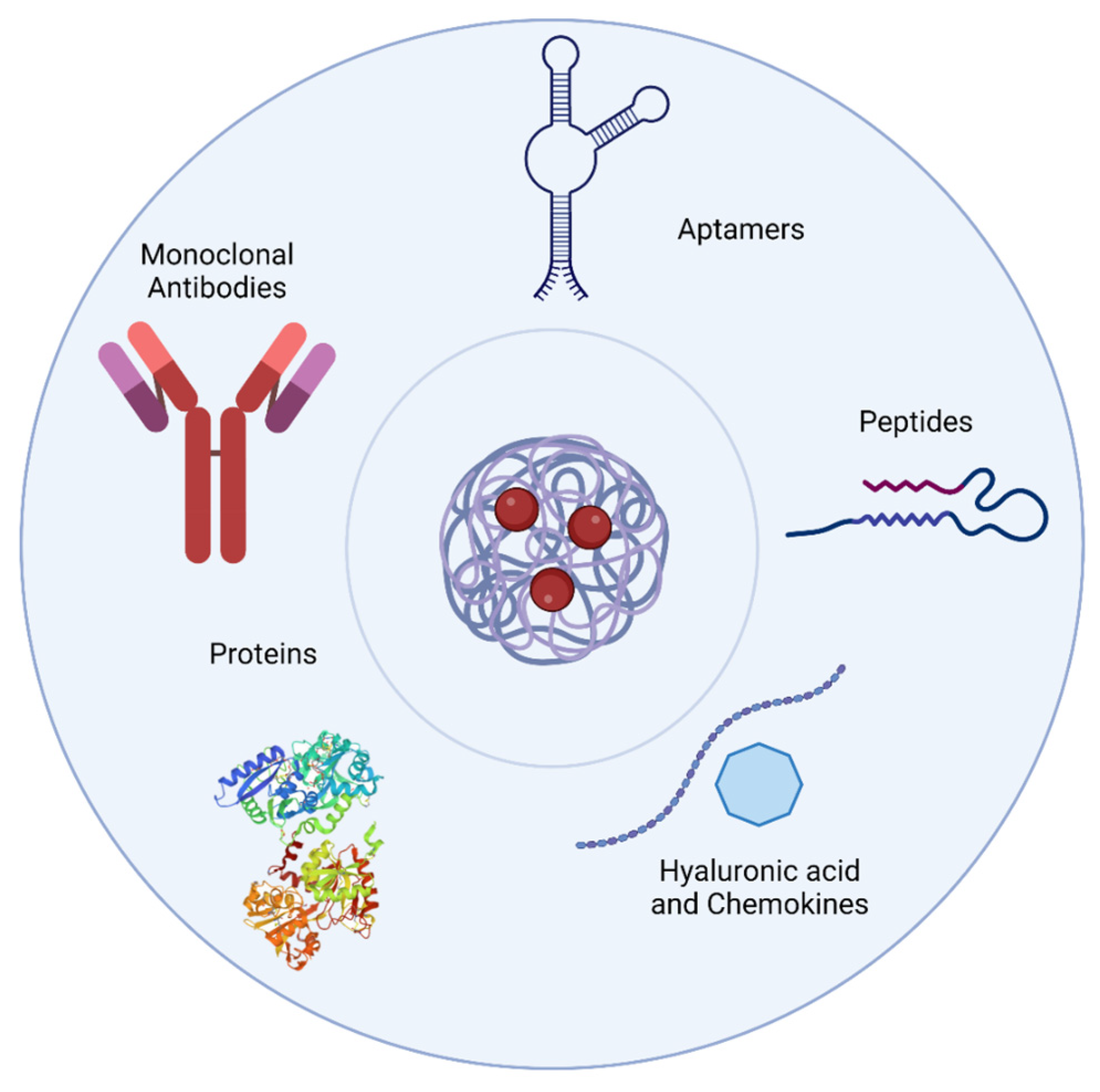
3. Targeting Strategies for PNPs Delivery
3.1. Monoclonal Antibodies
3.2. Oligonucleotide Aptamers
3.3. Peptides
3.4. Other Molecules
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Anselmo, A.C.; Mitragotri, S.; Mitragotri, S.; Paulson, J.A. Nanoparticles in the Clinic: An Update. Bioeng. Transl. Med. 2019, 4, e10143. [Google Scholar] [CrossRef] [PubMed]
- Wilczewska, A.Z.; Niemirowicz, K.; Markiewicz, K.H.; Car, H. Nanoparticles as Drug Delivery Systems. Pharmacol. Rep. 2012, 64, 1020–1037. [Google Scholar] [CrossRef]
- Polymeric Nanoparticles—Search Results—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/?term=polymeric+nanoparticles&filter=simsearch3.fft&filter=datesearch.y_10 (accessed on 14 November 2022).
- Calzoni, E.; Cesaretti, A.; Polchi, A.; Di Michele, A.; Tancini, B.; Emiliani, C. Biocompatible Polymer Nanoparticles for Drug Delivery Applications in Cancer and Neurodegenerative Disorder Therapies. J. Funct. Biomater. 2019, 10, 4. [Google Scholar] [CrossRef]
- Polymeric Nanoparticles Italy—Search Results—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/?term=polymeric+nanoparticles+Italy&filter=simsearch3.fft&filter=datesearch.y_10 (accessed on 14 November 2022).
- Peviani, M.; Palmiero, U.C.; Cecere, F.; Milazzo, R.; Moscatelli, D.; Biffi, A. Biodegradable polymeric nanoparticles administered in the cerebrospinal fluid: Brain biodistribution, preferential internalization in microglia and implications for cell-selective drug release. Biomaterials 2019, 209, 25–40. [Google Scholar] [CrossRef]
- Craparo, E.F.; Cabibbo, M.; Conigliaro, A.; Barreca, M.M.; Musumeci, T.; Giammona, G.; Cavallaro, G. Rapamycin-Loaded Polymeric Nanoparticles as an Advanced Formulation for Macrophage Targeting in Atherosclerosis. Pharmaceutics 2021, 13, 503. [Google Scholar] [CrossRef] [PubMed]
- Cavallo, E.; He, X.; Luzi, F.; Dominici, F.; Cerrutti, P.; Bernal, C.; Foresti, M.L.; Torre, L.; Puglia, D. UV Protective, Antioxidant, Antibacterial and Compostable Polylactic Acid Composites Containing Pristine and Chemically Modified Lignin Nanoparticles. Molecules 2020, 26, 126. [Google Scholar] [CrossRef] [PubMed]
- Abruzzo, A.; Giordani, B.; Miti, A.; Vitali, B.; Zuccheri, G.; Cerchiara, T.; Luppi, B.; Bigucci, F. Mucoadhesive and Mucopenetrating Chitosan Nanoparticles for Glycopeptide Antibiotic Administration. Int. J. Pharm. 2021, 606, 120874. [Google Scholar] [CrossRef] [PubMed]
- Georgouvelas, D.; Abdelhamid, H.N.; Li, J.; Edlund, U.; Mathew, A.P. All-Cellulose Functional Membranes for Water Treatment: Adsorption of Metal Ions and Catalytic Decolorization of Dyes. Carbohydr. Polym. 2021, 264, 118044. [Google Scholar] [CrossRef]
- Ta, Q.; Ting, J.; Harwood, S.; Browning, N.; Simm, A.; Ross, K.; Olier, I.; Al-Kassas, R. Chitosan Nanoparticles for Enhancing Drugs and Cosmetic Components Penetration through the Skin. Eur. J. Pharm. Sci. 2021, 160, 105765. [Google Scholar] [CrossRef]
- Perinelli, D.R.; Palmieri, G.F.; Cespi, M.; Bonacucina, G. Encapsulation of Flavours and Fragrances into Polymeric Capsules and Cyclodextrins Inclusion Complexes: An Update. Molecules 2020, 25, 5878. [Google Scholar] [CrossRef]
- Alam, M.T.; Parvez, N.; Sharma, P.K. FDA-Approved Natural Polymers for Fast Dissolving Tablets. J. Pharm. 2014, 2014, 952970. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, A.; Giuliano, E.; Venkateswararao, E.; Fresta, M.; Bulotta, S.; Awasthi, V.; Cosco, D. Biodegradable Polymeric Nanoparticles for Drug Delivery to Solid Tumors. Front. Pharmacol. 2021, 12, 601626. [Google Scholar] [CrossRef] [PubMed]
- Lacroce, E.; Rossi, F. Polymer-Based Thermoresponsive Hydrogels for Controlled Drug Delivery. Expert Opin. Drug Deliv. 2022, 19, 1203–1215. [Google Scholar] [CrossRef]
- Crucho, C.I.C.; Barros, M.T. Polymeric Nanoparticles: A Study on the Preparation Variables and Characterization Methods. Mater. Sci. Eng. C 2017, 80, 771–784. [Google Scholar] [CrossRef] [PubMed]
- Zielinska, A.; Carreiró, F.; Oliveira, A.M.; Neves, A.; Pires, B.; Nagasamy Venkatesh, D.; Durazzo, A.; Lucarini, M.; Eder, P.; Silva, A.M.; et al. Polymeric Nanoparticles: Production, Characterization, Toxicology and Ecotoxicology. Molecules 2020, 25, 3731. [Google Scholar] [CrossRef]
- Barhoum, A.; García-Betancourt, M.L.; Rahier, H.; Van Assche, G. Physicochemical Characterization of Nanomaterials: Polymorph, Composition, Wettability, and Thermal Stability. In Emerging Applications of Nanoparticles and Architecture Nanostructures; Elsevier: Amsterdam, The Netherlands, 2018; pp. 255–278. [Google Scholar] [CrossRef]
- Lin, P.C.; Lin, S.; Wang, P.C.; Sridhar, R. Techniques for Physicochemical Characterization of Nanomaterials. Biotechnol. Adv. 2014, 32, 711–726. [Google Scholar] [CrossRef]
- Villetti, M.A.; Clementino, A.R.; Dotti, I.; Ebani, P.R.; Quarta, E.; Buttini, F.; Sonvico, F.; Bianchera, A.; Borsali, R. Design and Characterization of Maltoheptaose- b-Polystyrene Nanoparticles, as a Potential New Nanocarrier for Oral Delivery of Tamoxifen. Molecules 2021, 26, 6507. [Google Scholar] [CrossRef]
- Rebanda, M.M.; Bettini, S.; Blasi, L.; Gaballo, A.; Ragusa, A.; Quarta, A.; Piccirillo, C. Poly(l-Lactide-Co-Caprolactone-Co-Glycolide)-Based Nanoparticles as Delivery Platform: Effect of the Surfactants on Characteristics and Delivery Efficiency. Nanomaterials 2022, 12, 1550. [Google Scholar] [CrossRef]
- Adel, A.M.; Al-Shemy, M.T.; Diab, M.A.; El-Sakhawy, M.; Toro, R.G.; Montanari, R.; de Caro, T.; Caschera, D. Fabrication of Packaging Paper Sheets Decorated with Alginate/Oxidized Nanocellulose-silver Nanoparticles Bio-Nanocomposite. Int. J. Biol. Macromol. 2021, 181, 612–620. [Google Scholar] [CrossRef]
- Colzani, B.; Pandolfi, L.; Hoti, A.; Iovene, P.A.; Natalello, A.; Avvakumova, S.; Colombo, M.; Prosperi, D. Investigation of Antitumor Activities of Trastuzumab Delivered by PLGA Nanoparticles. Int. J. Nanomed. 2018, 13, 957–973. [Google Scholar] [CrossRef]
- Gallo, E.; Diaferia, C.; Rosa, E.; Smaldone, G.; Morelli, G.; Accardo, A. Peptide-Based Hydrogels and Nanogels for Delivery of Doxorubicin. Int. J. Nanomed. 2021, 16, 1617–1630. [Google Scholar] [CrossRef] [PubMed]
- Palamà, I.E.; Di Maria, F.; Zangoli, M.; D’amone, S.; Manfredi, G.; Barsotti, J.; Lanzani, G.; Ortolani, L.; Salatelli, E.; Gigli, G.; et al. Enantiopure Polythiophene Nanoparticles. Chirality Dependence of Cellular Uptake, Intracellular Distribution and Antimicrobial Activity. RSC Adv. 2019, 9, 23036–23044. [Google Scholar] [CrossRef] [PubMed]
- Todaro, B.; Moscardini, A.; Luin, S. Pioglitazone-Loaded PLGA Nanoparticles: Towards the Most Reliable Synthesis Method. Int. J. Mol. Sci. 2022, 23, 2522. [Google Scholar] [CrossRef] [PubMed]
- Turino, L.N.; Stella, B.; Dosio, F.; Luna, J.A.; Barresi, A.A. Nanoparticles Obtained by Confined Impinging Jet Mixer: Poly(Lactide-Co-Glycolide) vs. Poly-ε-Caprolactone. Drug Dev. Ind. Pharm. 2018, 44, 934–941. [Google Scholar] [CrossRef]
- Chirio, D.; Peira, E.; Sapino, S.; Dianzani, C.; Barge, A.; Muntoni, E.; Morel, S.; Gallarate, M. Stearoyl-Chitosan Coated Nanoparticles Obtained by Microemulsion Cold Dilution Technique. Int. J. Mol. Sci. 2018, 19, 3833. [Google Scholar] [CrossRef]
- Piazzini, V.; Vasarri, M.; Degl’Innocenti, D.; Guastini, A.; Barletta, E.; Salvatici, M.C.; Bergonzi, M.C. Comparison of Chitosan Nanoparticles and Soluplus Micelles to Optimize the Bioactivity of Posidonia Oceanica Extract on Human Neuroblastoma Cell Migration. Pharmaceutics 2019, 11, 655. [Google Scholar] [CrossRef]
- Abruzzo, A.; Croatti, V.; Zuccheri, G.; Pasquale Nicoletta, F.; Sallustio, V.; Corazza, E.; Vitali, B.; Cerchiara, T.; Luppi, B.; Bigucci, F. Drug-in-Cyclodextrin-in-Polymeric Nanoparticles: A Promising Strategy for Rifampicin Administration. Eur. J. Pharm. Biopharm. 2022, 180, 190–200. [Google Scholar] [CrossRef]
- Rossi, S.; Vigani, B.; Puccio, A.; Bonferoni, M.C.; Id, G.S.; Ferrari, F. Chitosan Ascorbate Nanoparticles for the Vaginal Delivery of Antibiotic Drugs in Atrophic Vaginitis. Mar. Drugs 2017, 15, 319. [Google Scholar] [CrossRef]
- Gagliardi, A.; Paolino, D.; Iannone, M.; Palma, E.; Fresta, M.; Cosco, D. Sodium Deoxycholate-Decorated Zein Nanoparticles for a Stable Colloidal Drug Delivery System. Int. J. Nanomed. 2018, 13, 601–614. [Google Scholar] [CrossRef]
- Mineo, P.G.; Foti, C.; Vento, F.; Montesi, M.; Panseri, S.; Piperno, A.; Scala, A. Salinomycin-Loaded PLA Nanoparticles: Drug Quantification by GPC and Wave Voltammetry and Biological Studies on Osteosarcoma Cancer Stem Cells. Anal. Bioanal. Chem. 2020, 412, 4681–4690. [Google Scholar] [CrossRef]
- Palmioli, A.; La Ferla, B. Glycofunctionalization of Poly(Lactic- Co-Glycolic Acid) Polymers: Building Blocks for the Generation of Defined Sugar-Coated Nanoparticles. Org. Lett. 2018, 20, 3509–3512. [Google Scholar] [CrossRef] [PubMed]
- Gigli, V.; Tortolini, C.; Capecchi, E.; Angeloni, A.; Lenzi, A.; Antiochia, R. Novel Amperometric Biosensor Based on Tyrosinase/Chitosan Nanoparticles for Sensitive and Interference-Free Detection of Total Catecholamine. Biosensors 2022, 12, 519. [Google Scholar] [CrossRef] [PubMed]
- Khadjavi, A.; Stura, I.; Prato, M.; Minero, V.G.; Panariti, A.; Rivolta, I.; Gulino, G.R.; Bessone, F.; Giribaldi, G.; Quaglino, E.; et al. “In Vitro”, “In Vivo” and “In Silico” Investigation of the Anticancer Effectiveness of Oxygen-Loaded Chitosan-Shelled Nanodroplets as Potential Drug Vector. Pharm. Res. 2018, 35, 75. [Google Scholar] [CrossRef] [PubMed]
- Venuta, A.; Moret, F.; Dal Poggetto, G.; Esposito, D.; Fraix, A.; Avitabile, C.; Ungaro, F.; Malinconico, M.; Sortino, S.; Romanelli, A.; et al. Shedding Light on Surface Exposition of Poly(Ethylene Glycol) and Folate Targeting Units on Nanoparticles of Poly(ε-Caprolactone) Diblock Copolymers: Beyond a Paradigm. Eur. J. Pharm. Sci. 2018, 111, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Bonaccorso, A.; Musumeci, T.; Carbone, C.; Vicari, L.; Lauro, M.R.; Puglisi, G. Revisiting the Role of Sucrose in PLGA-PEG Nanocarrier for Potential Intranasal Delivery. Pharm. Dev. Technol. 2018, 23, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Crivelli, B.; Bari, E.; Perteghella, S.; Catenacci, L.; Sorrenti, M.; Mocchi, M.; Faragò, S.; Tripodo, G.; Prina-Mello, A.; Torre, M.L. Silk Fibroin Nanoparticles for Celecoxib and Curcumin Delivery: ROS-Scavenging and Anti-Inflammatory Activities in an in Vitro Model of Osteoarthritis. Eur. J. Pharm. Biopharm. 2019, 137, 37–45. [Google Scholar] [CrossRef]
- Trombino, S.; Poerio, T.; Curcio, F.; Piacentini, E.; Cassano, R. Production of α-Tocopherol-Chitosan Nanoparticles by Membrane Emulsification. Molecules 2022, 27, 2319. [Google Scholar] [CrossRef]
- Zuppolini, S.; Maya, I.C.; Diodato, L.; Guarino, V.; Borriello, A.; Ambrosio, L. Self-Associating Cellulose-Graft-Poly(ε-Caprolactone) to Design Nanoparticles for Drug Release. Mater. Sci. Eng. C 2020, 108, 110385. [Google Scholar] [CrossRef]
- Chiesa, E.; Dorati, R.; Conti, B.; Modena, T.; Cova, E.; Meloni, F.; Genta, I. Hyaluronic Acid-Decorated Chitosan Nanoparticles for CD44-Targeted Delivery of Everolimus. Int. J. Mol. Sci. 2018, 19, 2310. [Google Scholar] [CrossRef]
- Marinelli, L.; Ciulla, M.; Ritsema, J.A.S.; van Nostrum, C.F.; Cacciatore, I.; Dimmito, M.P.; Palmerio, F.; Orlando, G.; Robuffo, I.; Grande, R.; et al. Preparation, Characterization, and Biological Evaluation of a Hydrophilic Peptide Loaded on PEG-PLGA Nanoparticles. Pharmaceutics 2022, 14, 1821. [Google Scholar] [CrossRef]
- Trapani, A.; Cometa, S.; De Giglio, E.; Corbo, F.; Cassano, R.; Di Gioia, M.L.; Trombino, S.; Hossain, M.N.; Di Gioia, S.; Trapani, G.; et al. Novel Nanoparticles Based on N, O-Carboxymethyl Chitosan-Dopamine Amide Conjugate for Nose-to-Brain Delivery. Pharmaceutics 2022, 14, 147. [Google Scholar] [CrossRef] [PubMed]
- De Negri Atanasio, G.; Ferrari, P.F.; Campardelli, R.; Perego, P.; Palombo, D. Poly (Lactic- Co-Glycolic Acid) Nanoparticles and Nanoliposomes for Protein Delivery in Targeted Therapy: A Comparative In Vitro Study. Polymers 2020, 12, 2566. [Google Scholar] [CrossRef] [PubMed]
- Antonelli, F.; Galotta, G.; Sidoti, G.; Zikeli, F.; Nisi, R.; Davidde Petriaggi, B.; Romagnoli, M. Cellulose and Lignin Nano-Scale Consolidants for Waterlogged Archaeological Wood. Front. Chem. 2020, 8, 32. [Google Scholar] [CrossRef]
- Bilia, A.R.; Bergonzi, M.C.; Isacchi, B.; Antiga, E.; Caproni, M. Curcumin Nanoparticles Potentiate Therapeutic Effectiveness of Acitrein in Moderate-to-Severe Psoriasis Patients and Control Serum Cholesterol Levels. J. Pharm. Pharmacol. 2018, 70, 919–928. [Google Scholar] [CrossRef] [PubMed]
- Lionetto, F.; Corcione, C.E.; Rizzo, A.; Maffezzoli, A. Production and Characterization of Polyethylene Terephthalate Nanoparticles. Polymers 2021, 13, 3745. [Google Scholar] [CrossRef]
- Sanvito, T.; Bigini, P.; Cavanna, M.V.; Fiordaliso, F.; Violatto, M.B.; Talamini, L.; Salmona, M.; Milani, P.; Potenza, M.A.C. Single Particle Extinction and Scattering Optical Method Unveils in Real Time the Influence of the Blood Components on Polymeric Nanoparticles. Nanomedicine 2017, 13, 2597–2603. [Google Scholar] [CrossRef]
- Ding, S.; Attia, M.F.; Wallyn, J.; Taddei, C.; Serra, C.A.; Anton, N.; Kassem, M.; Schmutz, M.; Er-Rafik, M.; Messaddeq, N.; et al. Microfluidic-Assisted Production of Size-Controlled Superparamagnetic Iron Oxide Nanoparticles-Loaded Poly(Methyl Methacrylate) Nanohybrids. Langmuir 2018, 34, 1981–1991. [Google Scholar] [CrossRef]
- Rotem, R.; Micale, A.; Rizzuto, M.A.; Migliavacca, M.; Giustra, M.; Salvioni, L.; Tasin, F.; Prosperi, D.; Colombo, M. Modeling the Interaction of Amphiphilic Polymer Nanoparticles with Biomembranes to Guide Rational Design of Drug Delivery Systems. Colloids Surf. B Biointerfaces 2020, 196, 111366. [Google Scholar] [CrossRef]
- Foglietta, F.; Spagnoli, G.C.; Muraro, M.G.; Ballestri, M.; Guerrini, A.; Ferroni, C.; Aluigi, A.; Sotgiu, G.; Varchi, G. Anticancer Activity of Paclitaxel-Loaded Keratin Nanoparticles in Two-Dimensional and Perfused Three-Dimensional Breast Cancer Models. Int. J. Nanomed. 2018, 13, 4847–4867. [Google Scholar] [CrossRef]
- Banik, B.L.; Fattahi, P.; Brown, J.L. Polymeric Nanoparticles: The Future of Nanomedicine. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2016, 8, 271–299. [Google Scholar] [CrossRef]
- Mansfield, E.; Kaiser, D.L.; Fujita, D.; Van de Voorde, M. Metrology and Standardization for Nanotechnology: Protocols and Industrial Innovations. Available online: https://books.google.it/books?hl=it&lr=&id=strwDQAAQBAJ&oi=fnd&pg=PR27&dq=lisabeth+Mansfield,+Debra+L.+Kaiser,+Daisuke+Fujita,+Marcel+Van+de+Voorde+Metrology+and+Standardization+for+Nanotechnology:+Protocols+and+Industrial+Innovations%3B&ots=YGVpXC7gi8&si (accessed on 8 November 2022).
- Mourdikoudis, S.; Pallares, R.M.; Thanh, N.T.K. Characterization Techniques for Nanoparticles: Comparison and Complementarity upon Studying Nanoparticle Properties. Nanoscale 2018, 10, 12871–12934. [Google Scholar] [CrossRef] [PubMed]
- Martella, E.; Ferroni, C.; Guerrini, A.; Ballestri, M.; Columbaro, M.; Santi, S.; Sotgiu, G.; Serra, M.; Donati, D.M.; Lucarelli, E.; et al. Functionalized Keratin as Nanotechnology-Based Drug Delivery System for the Pharmacological Treatment of Osteosarcoma. Int. J. Mol. Sci. 2018, 19, 3670. [Google Scholar] [CrossRef] [PubMed]
- Dazon, C.; Witschger, O.; Bau, S.; Fierro, V.; Llewellyn, P.L. Nanomaterial Identification of Powders: Comparing Volume Specific Surface Area, X-Ray Diffraction and Scanning Electron Microscopy Methods. Environ. Sci. Nano 2019, 6, 152–162. [Google Scholar] [CrossRef]
- Scimeca, M.; Bischetti, S.; Lamsira, H.K.; Bonfiglio, R.; Bonanno, E. Energy Dispersive X-Ray (EDX) Microanalysis: A Powerful Tool in Biomedical Research and Diagnosis. Eur. J. Histochem. 2018, 62. [Google Scholar] [CrossRef] [PubMed]
- Moreau, L.M.; Ha, D.H.; Zhang, H.; Hovden, R.; Muller, D.A.; Robinson, R.D. Defining Crystalline/Amorphous Phases of Nanoparticles through X-Ray Absorption Spectroscopy and X-Ray Diffraction: The Case of Nickel Phosphide. Chem. Mater. 2013, 25, 2394–2403. [Google Scholar] [CrossRef]
- Khan, I.; Saeed, K.; Khan, I. Nanoparticles: Properties, Applications and Toxicities. Arab. J. Chem. 2019, 12, 908–931. [Google Scholar] [CrossRef]
- Todaro, B.; Begarani, F.; Sartori, F.; Luin, S. Is Raman the Best Strategy towards the Development of Non-Invasive Continuous Glucose Monitoring Devices for Diabetes Management? Front. Chem. 2022, 10, 994272. [Google Scholar] [CrossRef]
- Ranjbar, B.; Gill, P. Circular Dichroism Techniques: Biomolecular and Nanostructural Analyses—A Review. Chem. Biol. Drug Des. 2009, 74, 101–120. [Google Scholar] [CrossRef]
- Mansfield, E.; Tyner, K.M.; Poling, C.M.; Blacklock, J.L. Determination of Nanoparticle Surface Coatings and Nanoparticle Purity Using Microscale Thermogravimetric Analysis. Anal. Chem. 2014, 86, 1478–1484. [Google Scholar] [CrossRef]
- Bonferoni, M.C.; Rassu, G.; Gavini, E.; Sorrenti, M.; Catenacci, L.; Torre, M.L.; Perteghella, S.; Ansaloni, L.; Maestri, M.; Giunchedi, P. Electrochemotherapy of Deep-Seated Tumors: State of Art and Perspectives as Possible “EPR Effect Enhancer” to Improve Cancer Nanomedicine Efficacy. Cancers 2021, 13, 4437. [Google Scholar] [CrossRef]
- Ottonelli, I.; Duskey, J.T.; Genovese, F.; Pederzoli, F.; Caraffi, R.; Valenza, M.; Tosi, G.; Vandelli, M.A.; Ruozi, B. Quantitative Comparison of the Protein Corona of Nanoparticles with Different Matrices. Int. J. Pharm. X 2022, 4, 100136. [Google Scholar] [CrossRef] [PubMed]
- Casalini, T.; Limongelli, V.; Schmutz, M.; Som, C.; Jordan, O.; Wick, P.; Borchard, G.; Perale, G. Molecular Modeling for Nanomaterial-Biology Interactions: Opportunities, Challenges, and Perspectives. Front. Bioeng. Biotechnol. 2019, 7, 268. [Google Scholar] [CrossRef]
- Jain, K.K. Personalized Immuno-Oncology. Med. Princ. Pract. 2021, 30, 1–16. [Google Scholar] [CrossRef]
- Duskey, J.T.; Rinaldi, A.; Ottonelli, I.; Caraffi, R.; De Benedictis, C.A.; Sauer, A.K.; Tosi, G.; Vandelli, M.A.; Ruozi, B.; Grabrucker, A.M. Glioblastoma Multiforme Selective Nanomedicines for Improved Anti-Cancer Treatments. Pharmaceutics 2022, 14, 1450. [Google Scholar] [CrossRef] [PubMed]
- Le Basle, Y.; Chennell, P.; Tokhadze, N.; Astier, A.; Sautou, V. Physicochemical Stability of Monoclonal Antibodies: A Review. J. Pharm. Sci. 2020, 109, 169–190. [Google Scholar] [CrossRef] [PubMed]
- Troisi, R.; Sica, F. Aptamers: Functional-Structural Studies and Biomedical Applications. Int. J. Mol. Sci. 2022, 23, 4796. [Google Scholar] [CrossRef]
- Agnello, L.; Camorani, S.; Fedele, M.; Cerchia, L. Aptamers and Antibodies: Rivals or Allies in Cancer Targeted Therapy? Explor. Target. Anti-Tumor Ther. 2021, 2, 107–121. [Google Scholar] [CrossRef]
- Camorani, S.; Fedele, M.; Zannetti, A.; Cerchia, L. TNBC Challenge: Oligonucleotide Aptamers for New Imaging and Therapy Modalities. Pharmaceuticals 2018, 11, 123. [Google Scholar] [CrossRef]
- Agnello, L.; Tortorella, S.; d’Argenio, A.; Carbone, C.; Camorani, S.; Locatelli, E.; Auletta, L.; Sorrentino, D.; Fedele, M.; Zannetti, A.; et al. Optimizing Cisplatin Delivery to Triple-Negative Breast Cancer through Novel EGFR Aptamer-Conjugated Polymeric Nanovectors. J. Exp. Clin. Cancer Res. 2021, 40, 239. [Google Scholar] [CrossRef]
- Camorani, S.; Tortorella, S.; Agnello, L.; Spanu, C.; d’Argenio, A.; Nilo, R.; Zannetti, A.; Locatelli, E.; Fedele, M.; Comes Franchini, M.; et al. Aptamer-Functionalized Nanoparticles Mediate PD-L1 SiRNA Delivery for Effective Gene Silencing in Triple-Negative Breast Cancer Cells. Pharmaceutics 2022, 14, 2225. [Google Scholar] [CrossRef]
- Andrieu, J.; Re, F.; Russo, L.; Nicotra, F. Phage-Displayed Peptides Targeting Specific Tissues and Organs. J. Drug Target. 2019, 27, 555–565. [Google Scholar] [CrossRef] [PubMed]
- D’Annessa, I.; Di Leva, F.S.; La Teana, A.; Novellino, E.; Limongelli, V.; Di Marino, D. Bioinformatics and Biosimulations as Toolbox for Peptides and Peptidomimetics Design: Where Are We? Front. Mol. Biosci. 2020, 7, 66. [Google Scholar] [CrossRef] [PubMed]
- Rios De La Rosa, J.M.; Spadea, A.; Donno, R.; Lallana, E.; Lu, Y.; Puri, S.; Caswell, P.; Lawrence, M.J.; Ashford, M.; Tirelli, N. Microfluidic-Assisted Preparation of RGD-Decorated Nanoparticles: Exploring Integrin-Facilitated Uptake in Cancer Cell Lines. Sci. Rep. 2020, 10, 14505. [Google Scholar] [CrossRef] [PubMed]
- Bognanni, N.; Viale, M.; Distefano, A.; Tosto, R.; Bertola, N.; Loiacono, F.; Ponassi, M.; Spinelli, D.; Pappalardo, G.; Vecchio, G. Cyclodextrin Polymers as Delivery Systems for Targeted Anti-Cancer Chemotherapy. Molecules 2021, 26, 6046. [Google Scholar] [CrossRef] [PubMed]
- Todaro, B.; Achilli, S.; Liet, B.; Laigre, E.; Tiertant, C.; Goyard, D.; Berthet, N.; Renaudet, O. Structural Influence of Antibody Recruiting Glycodendrimers (ARGs) on Antitumoral Cytotoxicity. Biomater. Sci. 2021, 9, 4076–4085. [Google Scholar] [CrossRef] [PubMed]
- Liet, B.; Laigre, E.; Goyard, D.; Todaro, B.; Tiertant, C.; Boturyn, D.; Berthet, N.; Renaudet, O. Multifunctional Glycoconjugates for Recruiting Natural Antibodies against Cancer Cells. Chem. Eur. J. 2019, 25, 15508–15515. [Google Scholar] [CrossRef]
- Del Grosso, A.; Galliani, M.; Angella, L.; Santi, M.; Tonazzini, I.; Parlanti, G.; Signore, G.; Cecchini, M. Brain-Targeted Enzyme-Loaded Nanoparticles: A Breach through the Blood-Brain Barrier for Enzyme Replacement Therapy in Krabbe Disease. Sci. Adv. 2019, 5, eaax7462. [Google Scholar] [CrossRef]
- Galliani, M.; Santi, M.; Grosso, A.D.; Cecchettini, A.; Santorelli, F.M.; Hofmann, S.L.; Lu, J.; Angella, L.; Cecchini, M.; Signore, G. Cross Linked Enzyme Aggregates as Versatile Tool for Enzyme Delivery: Application to Polymeric Nanoparticles. Bioconjug. Chem. 2018, 29, 2225–2231. [Google Scholar] [CrossRef]
- Galliani, M.; Tremolanti, C.; Signore, G. Nanocarriers for Protein Delivery to the Cytosol: Assessing the Endosomal Escape of Poly(Lactide-Co-Glycolide)-Poly(Ethylene Imine) Nanoparticles. Nanomaterials 2019, 9, 652. [Google Scholar] [CrossRef]
- Palombarini, F.; Masciarelli, S.; Incocciati, A.; Liccardo, F.; Di Fabio, E.; Iazzetti, A.; Fabrizi, G.; Fazi, F.; Macone, A.; Bonamore, A.; et al. Self-Assembling Ferritin-Dendrimer Nanoparticles for Targeted Delivery of Nucleic Acids to Myeloid Leukemia Cells. J. Nanobiotechnol. 2021, 19, 172. [Google Scholar] [CrossRef]
- Pisani, A.; Donno, R.; Gennari, A.; Cibecchini, G.; Catalano, F.; Marotta, R.; Pompa, P.P.; Tirelli, N.; Bardi, G. CXCL12-PLGA/Pluronic Nanoparticle Internalization Abrogates CXCR4-Mediated Cell Migration. Nanomaterials 2020, 10, 2304. [Google Scholar] [CrossRef] [PubMed]
- Santi, M.; Maccari, G.; Mereghetti, P.; Voliani, V.; Rocchiccioli, S.; Ucciferri, N.; Luin, S.; Signore, G. Rational Design of a Transferrin-Binding Peptide Sequence Tailored to Targeted Nanoparticle Internalization. Bioconjug. Chem. 2016, 28, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Li, X.; Dong, D.; Zhang, B.; Xue, Y.; Shang, P. Transferrin Receptor 1 in Cancer: A New Sight for Cancer Therapy. Am. J. Cancer Res. 2018, 8, 916–931. [Google Scholar] [PubMed]
- Jose, S.; Cinu, T.A.; Sebastian, R.; Shoja, M.H.; Aleykutty, N.A.; Durazzo, A.; Lucarini, M.; Santini, A.; Souto, E.B. Transferrin-Conjugated Docetaxel-PLGA Nanoparticles for Tumor Targeting: Influence on MCF-7 Cell Cycle. Polymers 2019, 11, 1905. [Google Scholar] [CrossRef] [PubMed]
- Yong, K.W.; Yuen, D.; Chen, M.Z.; Porter, C.J.H.; Johnston, A.P.R. Pointing in the Right Direction: Controlling the Orientation of Proteins on Nanoparticles Improves Targeting Efficiency. Nano Lett. 2019, 19, 1827–1831. [Google Scholar] [CrossRef] [PubMed]
- Chiesa, E.; Greco, A.; Riva, F.; Dorati, R.; Conti, B.; Modena, T.; Genta, I. CD44-Targeted Carriers: The Role of Molecular Weight of Hyaluronic Acid in the Uptake of Hyaluronic Acid-Based Nanoparticles. Pharmaceuticals 2022, 15, 103. [Google Scholar] [CrossRef]
- Morillas-Becerril, L.; Peta, E.; Gabrielli, L.; Russo, V.; Lubian, E.; Nodari, L.; Ferlin, M.G.; Scrimin, P.; Palù, G.; Barzon, L.; et al. Multifunctional, CD44v6-Targeted ORMOSIL Nanoparticles Enhance Drugs Toxicity in Cancer Cells. Nanomaterials 2020, 10, 298. [Google Scholar] [CrossRef]

| Characterization Techniques | NPs Characteristics Analyzed | References |
|---|---|---|
| Atomic Force Microscopy (AFM) | Size and size distribution Shape of nanomaterials Structure of nanomaterials Surface charge of nanomaterials Aggregation Surface properties Dispersion of nanomaterials | [20,21,22] |
| Circular Dichroism (CD) | Structure of nanomaterials Stability of nanomaterials Surface properties of nanomaterials (coupled with ELISA) Protein corona (conformation) | [23,24,25] |
| Dynamic Light Scattering (DLS) | Hydrodynamic size and size distribution Protein corona (thickness and density) | [26,27,28,29,30,31,32,33,34] |
| Energy Dispersive X-Ray (EDX) | Composition of nanomaterials Concentration of nanomaterials | [35] |
| Fluorescence Correlation Spectroscopy (FCS) | Critical association concentration determination Size and size distribution Drug content In vitro drug release | [36,37,38] |
| Fourier Transform Infrared Spectroscopy (FTIR) | Structure and conformation of bioconjugates Functional group analysis Surface properties of nanomaterials | [21,22,39,40,41,42,43,44] |
| Nuclear Magnetic Resonance (NMR) | Structure of nanomaterials Composition of nanomaterials Purity of nanomaterials Conformational change | [37,40] |
| Nanoparticle Tracking Analysis (NTA) | Size and size distribution | [20] |
| Raman Spectroscopy (RS) | Structure and conformation of bioconjugates Functional group analysis | [22] |
| Scanning Electron Microscopy (SEM) | Size and size distribution Shape of nanomaterials Aggregation of nanomaterials | [39,40,41,42,45,46,47,48] |
| Single Particle Extinction and Scattering (SPES) | Size and size distribution | [49] |
| Thermogravimetric Analysis (TGA) | Drug-polymer interactions Stability of nanomaterials Purity of nanomaterials | [21,22,39,41,50] |
| Transmission Electron Microscopy (TEM) | Size and size distribution Shape heterogeneity of nanomaterials Dispersion/Aggregation of nanomaterials Protein corona (thickness and density) | [21,23,36,41,42,43,47,50,51,52] |
| Ultraviolet–Visible Spectroscopy (UV-vis) | Size and size distribution | [23,24] |
| X-Ray Photoelectron Spectroscopy (XPS) | Surface properties of nanomaterials | [44] |
| X-Ray Diffraction (XRD) | Size and size distribution Shape of nanomaterials Structure of nanomaterials | [22,47,48] |
| Zeta Potential | Stability of nanomaterials Surface charge of nanomaterials | [21,26] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Todaro, B.; Santi, M. Characterization and Functionalization Approaches for the Study of Polymeric Nanoparticles: The State of the Art in Italian Research. Micro 2023, 3, 9-21. https://doi.org/10.3390/micro3010002
Todaro B, Santi M. Characterization and Functionalization Approaches for the Study of Polymeric Nanoparticles: The State of the Art in Italian Research. Micro. 2023; 3(1):9-21. https://doi.org/10.3390/micro3010002
Chicago/Turabian StyleTodaro, Biagio, and Melissa Santi. 2023. "Characterization and Functionalization Approaches for the Study of Polymeric Nanoparticles: The State of the Art in Italian Research" Micro 3, no. 1: 9-21. https://doi.org/10.3390/micro3010002
APA StyleTodaro, B., & Santi, M. (2023). Characterization and Functionalization Approaches for the Study of Polymeric Nanoparticles: The State of the Art in Italian Research. Micro, 3(1), 9-21. https://doi.org/10.3390/micro3010002







