Applying the ‘Human Rights Model of Disability’ to Informed Consent: Experiences and Reflections from the SHAPES Project
Abstract
1. Introduction
The SHAPES Project
- (1)
- Smart Living Environment for Healthy Ageing at Home;
- (2)
- Improving In-Home and Community-based Care;
- (3)
- Medicine Control and Optimisation;
- (4)
- Psycho-social and Cognitive Stimulation Promoting Well-being;
- (5)
- Caring for Older Individuals with Neurodegenerative Diseases;
- (6)
- Physical Rehabilitation at Home;
- (7)
- Cross-border Health Data Exchange Supporting Mobility and Accessibility for Older Individuals.
2. The Human Rights Model of Disability as a Theoretical Framework
3. Participation of “Vulnerable Adults” in Research Projects: A Review of the Relevant Literature
3.1. Participatory Research
3.2. Participation of “Vulnerable Adults” in Research Projects
4. Conceptual, Legal, and Procedural Challenges: The Winding Road toward Enacting Participatory Research in line with the Human Rights Model
4.1. Conceptual Challenges
4.2. Legal Challenges
4.3. Procedural and Practical Challenges
5. How to Address Those Challenges? The Approach Enshrined in SHAPES
- Providing the legal basis for consent: ensuring that there are sufficient capabilities for asking consent as part of the service and that the consent is documented properly (obligatory), and building up a repository where consents can be collected centrally (optional, contingent upon value to the SHAPES project).
- Providing a process for the implementation of services for single end-users (older persons) and for the assessment of the suitability of the services from time to time (including a process to assess the digital literacy of the end-user and adapt the services according to end-user needs and capabilities). The process should include more time to discuss choices or have an advocate regarding important appointments in order to make notes and help the person understand or remember choices.
- Providing a detailed process, taking into consideration national laws, to determine if the older person is able to decide on accessing the services and, secondly, if they are able to give informed consent and re-consent for the collection of the information.
- Providing understandable and plain-language materials, instructions, and information in visual form (including information on each service and how it operates and what data it collects) and providing video-based instructions for the end-user (older persons).
- An explanation of why the research is being conducted.
- Who is organising and funding the research?
- Why am I being asked to take part?
- How will the research be conducted?
- What will happen if I agree to take part?
- What are the benefits/What are the risks?
- Is the research confidential?
- Information on data protection (specifically GDPR regulations).
- Where can I get further information?
- How do I withdraw consent?
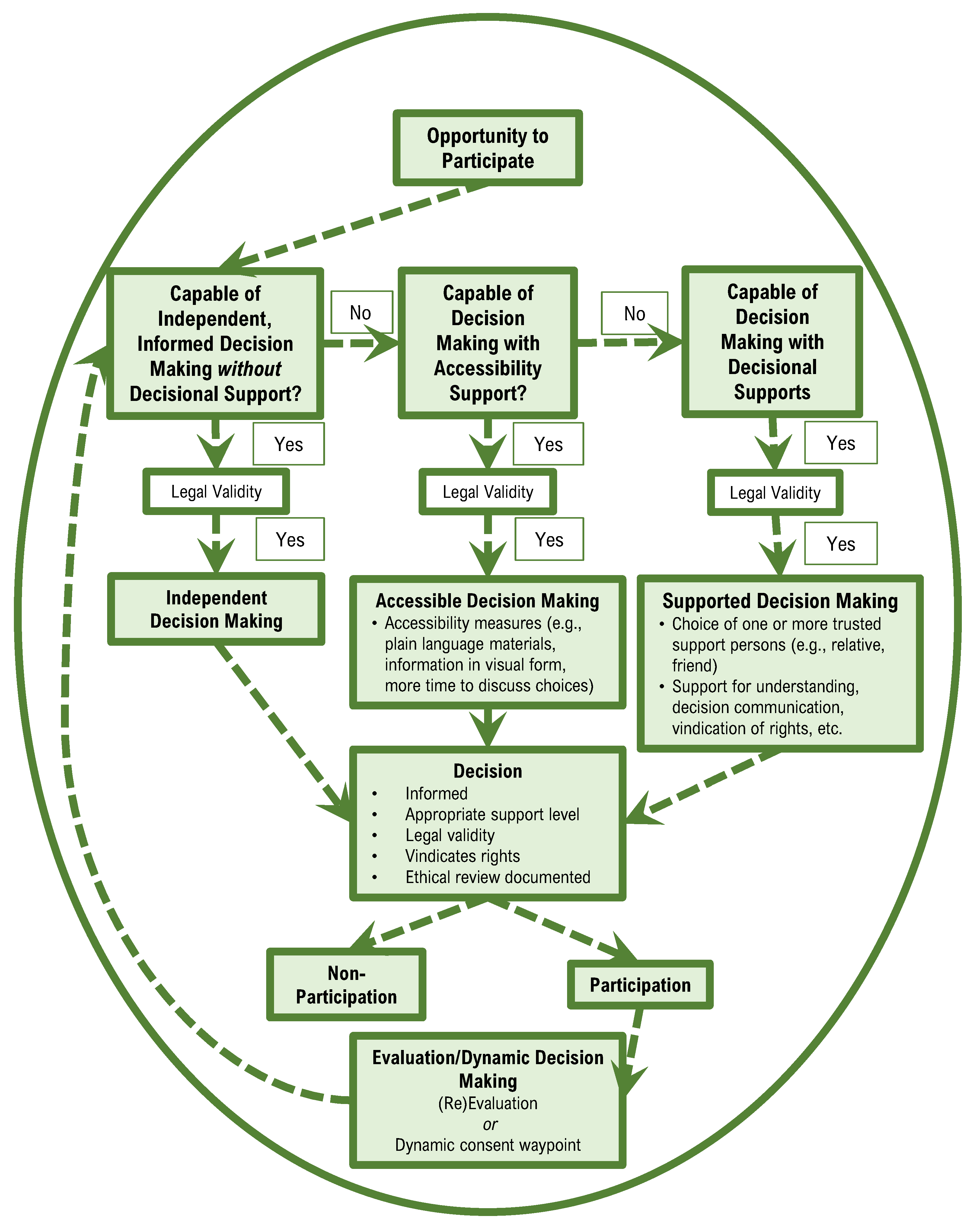
5.1. Guidelines for Informed Consent Processes
5.1.1. Rights-Based Approach
5.1.2. Accessibility Measures
5.1.3. Allow for Decision-Making Supports
5.1.4. Decision-Making as a Cyclical Process and (Re) Evaluation
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- European Commission; Statistical Office of the European Union. Ageing Europe: Looking at the Lives of Older People in the EU: 2020 Edition; Publications Office: Luxembourg, 2020. [Google Scholar]
- European Commission. Report from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions on the Impact of Demographic Change. (COM (2020) 241 Final); European Commission: Brussels, Belgium, 2020. [Google Scholar]
- World Health Organization. International Classification of Functioning, Disability and Health; World Health Organization: Geneva, Switzerland, 2001. [Google Scholar]
- United Nations Special Rapporteur on the Rights of Persons with Disabilities. Report of the Special Rapporteur on the Rights of Persons with Disabilities (A/74/186); United Nations: New York, NY, USA, 2019. [Google Scholar]
- United Nations. Convention on the Rights of Persons with Disabilities; Treaty Series; United Nations: New York, NY, USA, 2006; Volume 3. [Google Scholar]
- Broderick, A. The Long and Winding Road to Equality and Inclusion for Persons with Disabilities: The United Nations Convention on the Rights of Persons with Disabilities; School of Human Rights Research Series; Intersentia: Cambridge, UK, 2015; ISBN 978-1-78068-358-4. [Google Scholar]
- Dute, J. Should Substituted Decision-Making Be Abolished? Eur. J. Health Law 2015, 22, 315–320. [Google Scholar] [CrossRef]
- Waddington, L. Exploring Vulnerability in EU Law: An Analysis of ‘Vulnerability’ in EU Criminal Law and Consumer Protection Law. Eur. Law Rev. 2020, 45, 779–801. [Google Scholar]
- Fineman, M.A. The Vulnerable Subject: Anchoring Equality in the Human Condition. Yale J. Law Fem. 2008, 20, 1–23. Available online: http://hdl.handle.net/20.500.13051/6993 (accessed on 10 January 2023).
- Herring, J. Vulnerable Adults and the Law; Oxford University Press: Oxford, UK, 2016; ISBN 978-0-19-873727-8. [Google Scholar]
- Hardicre, J. Valid Informed Consent in Research: An Introduction. Br. J. Nurs. 2014, 23, 564–567. [Google Scholar] [CrossRef]
- Appelbaum, P.S.; Lidz, C.W.; Meisel, A. Informed Consent: Legal Theory and Clinical Practice; Oxford University Press: Oxford, UK, 1987. [Google Scholar]
- O’Neill, O. Some Limits of Informed Consent. J. Med. Ethics 2003, 29, 4–7. [Google Scholar] [CrossRef] [PubMed]
- Tori, K.; Kalligeros, M.; Shehadeh, F.; Khader, R.; Nanda, A.; van Aalst, R.; Chit, A.; Mylonakis, E. The Process of Obtaining Informed Consent to Research in Long Term Care Facilities (LTCFs): An Observational Clinical Study. Medicine 2020, 99, e20225. [Google Scholar] [CrossRef]
- Perlin, M.L. A Change Is Gonna Come: The Implications of the United Nations Convention on the Rights of Persons with Disabilities for the Domestic Practice of Constitutional Mental Disability Law. North. Ill. Univ. Law Rev. 2009, 29, 483–498. [Google Scholar]
- Perlin, M.L. “The Ladder of the Law Has No Top and No Bottom”: How Therapeutic Jurisprudence Can Give Life to International Human Rights. Int. J. Law Psychiatry 2014, 37, 535–542. [Google Scholar] [CrossRef]
- Skarstad, K.; Stein, M.A. Mainstreaming Disability in the United Nations Treaty Bodies. J. Hum. Rights 2018, 17, 1–24. [Google Scholar] [CrossRef]
- Degener, T. A New Human Rights Model of Disability. In The United Nations Convention on the Rights of Persons with Disabilities; Della Fina, V., Cera, R., Palmisano, G., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 41–59. ISBN 978-3-319-43788-0. [Google Scholar]
- Lawson, A.; Beckett, A.E. The Social and Human Rights Models of Disability: Towards a Complementarity Thesis. Int. J. Hum. Rights 2021, 25, 348–379. [Google Scholar] [CrossRef]
- Mitra, S. The Capability Approach and Disability. J. Disabil. Policy Stud. 2006, 16, 236–247. [Google Scholar] [CrossRef]
- Degener, T. A Human Rights Model of Disability. In Routledge Handbook of Disability Law and Human Rights; Blanck, P., Flynn, E., Eds.; Routledge: London, UK, 2016; pp. 31–49. [Google Scholar]
- Degener, T. Disability in a Human Rights Context. Laws 2016, 5, 35. [Google Scholar] [CrossRef]
- Degener, T. 10 Years of Convention on the Rights of Persons with Disabilities. Neth. Q. Hum. Rights 2017, 35, 152–157. [Google Scholar] [CrossRef]
- United Nations Committee on the Rights of Persons with Disabilities. Concluding Observations on the Initial Report of Bulgaria (CRPD/C/BGR/CO/1); United Nations: New York, NY, USA, 2018. [Google Scholar]
- General Comment No. 6 (2018) on Equality and Non-Discrimination. CRPD/C/GC/6. 2018. Available online: https://digitallibrary.un.org/record/1626976 (accessed on 10 January 2023).
- MacLachlan, M.; Mannan, H. Is Disability a Health Problem? Soc. Inclusion 2013, 1, 139–141. [Google Scholar] [CrossRef]
- World Health Organization. Declaration of Alma Ata. International Conference on Primary Health Care, Alma-Ata, USSR, 6–12 September 1978; WHO: Geneva, Switzerland, 1978. [Google Scholar]
- Beresford, P.; Russo, J. Patient and Public Involvement in Research. In Achieving Person-Centred Health Systems; Nolte, E., Merkur, S., Anell, A., Eds.; Cambridge University Press: Cambridge, UK, 2020; pp. 145–172. ISBN 978-1-108-85546-4. [Google Scholar]
- Jagosh, J.; Macaulay, A.C.; Pluye, P.; Salsberg, J.; Bush, P.L.; Henderson, J.; Sirett, E.; Wong, G.; Cargo, M.; Herbert, C.P.; et al. Uncovering the Benefits of Participatory Research: Implications of a Realist Review for Health Research and Practice: A Realist Review for Health Research and Practice. Milbank Q. 2012, 90, 311–346. [Google Scholar] [CrossRef]
- Cargo, M.; Mercer, S.L. The Value and Challenges of Participatory Research: Strengthening Its Practice. Annu. Rev. Public Health 2008, 29, 325–350. [Google Scholar] [CrossRef]
- National Institute for Health and Care Research Briefing Notes for Researchers—Public Involvement in NHS, Health and Social Care Research. Available online: https://www.nihr.ac.uk/documents/briefing-notes-for-researchers-public-involvement-in-nhs-health-and-social-care-research/27371#Ethics (accessed on 12 May 2022).
- Fudge, N.; Wolfe, C.D.A.; McKevitt, C. Involving Older People in Health Research. Age Ageing 2007, 36, 492–500. [Google Scholar] [CrossRef]
- James, H.; Buffel, T. Co-Research with Older People: A Systematic Literature Review. Ageing Soc. 2022, 1–27. [Google Scholar] [CrossRef]
- Di Lorito, C.; Godfrey, M.; Dunlop, M.; Bosco, A.; Pollock, K.; Wardt, V.; Harwood, R.H. Adding to the Knowledge on Patient and Public Involvement: Reflections from an Experience of Co-research with Carers of People with Dementia. Health Expect 2020, 23, 691–706. [Google Scholar] [CrossRef]
- Beresford, P. Public Participation in Health and Social Care: Exploring the Co-Production of Knowledge. Front. Sociol. 2019, 3, 41. [Google Scholar] [CrossRef]
- Baldwin, J.N.; Napier, S.; Neville, S.; Wright-St Clair, V.A. Impacts of Older People’s Patient and Public Involvement in Health and Social Care Research: A Systematic Review. Age Ageing 2018, 47, 801–809. [Google Scholar] [CrossRef] [PubMed]
- Tullo, E.S.; Robinson, L.; Newton, J. Comparing the Perceptions of Academics and Members of the Public about Patient and Public Involvement in Ageing Research. Age Ageing 2015, 44, 533–536. [Google Scholar] [CrossRef]
- Abma, T.A.; Nierse, C.J.; Widdershoven, G.A.M. Patients as Partners in Responsive Research: Methodological Notions for Collaborations in Mixed Research Teams. Qual. Health Res. 2009, 19, 401–415. [Google Scholar] [CrossRef]
- Green, G. Power to the People: To What Extent Has Public Involvement in Applied Health Research Achieved This? Res. Involv. Engagem. 2016, 2, 28. [Google Scholar] [CrossRef] [PubMed]
- Goodwin, M. Vulnerable Subjects: Why Does Informed Consent Matter? J. Law Med. Ethics 2016, 44, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Dalpé, G.; Thorogood, A.; Knoppers, B.M. A Tale of Two Capacities: Including Children and Decisionally Vulnerable Adults in Biomedical Research. Front. Genet. 2019, 10, 289. [Google Scholar] [CrossRef]
- MacLachlann, M.; Mji, G.; Chataika, T.; Wazakili, M.; Dube, A.K.; Mulumba, M.; Massah, B.; Wakene, D.; Kallon, F.; Maughan, M. Facilitating Disability Inclusion in Poverty Reduction Processes: Group Consensus Perspectives from Disability Stakeholders in Uganda, Malawi, Ethiopia, and Sierra Leone. Disabil. Glob. South 2014, 1, 107–127. [Google Scholar]
- Jongsma, K.; Bos, W.; van de Vathorst, S. Morally Relevant Similarities and Differences Between Children and Dementia Patients as Research Subjects: Representation in Legal Documents and Ethical Guidelines: Children and Dementia Patients as Research Subjects. Bioethics 2015, 29, 662–670. [Google Scholar] [CrossRef]
- Mannheim, I.; Schwartz, E.; Xi, W.; Buttigieg, S.C.; McDonnell-Naughton, M.; Wouters, E.J.M.; van Zaalen, Y. Inclusion of Older Adults in the Research and Design of Digital Technology. Int. J. Environ. Res. Public Health 2019, 16, 3718. [Google Scholar] [CrossRef]
- Gove, D.; Diaz-Ponce, A.; Georges, J.; Moniz-Cook, E.; Mountain, G.; Chattat, R.; Øksnebjerg, L. The European Working Group of People with Dementia Alzheimer Europe’s Position on Involving People with Dementia in Research through PPI (Patient and Public Involvement). Aging Ment. Health 2018, 22, 723–729. [Google Scholar] [CrossRef]
- Littlechild, R.; Tanner, D.; Hall, K. Co-Research with Older People: Perspectives on Impact. Qual. Soc. Work 2015, 14, 18–35. [Google Scholar] [CrossRef]
- Waite, J.; Poland, F.; Charlesworth, G. Facilitators and Barriers to Co-Research by People with Dementia and Academic Researchers: Findings from a Qualitative Study. Health Expect. 2019, 22, 761–771. [Google Scholar] [CrossRef] [PubMed]
- Morbey, H.; Harding, A.J.E.; Swarbrick, C.; Ahmed, F.; Elvish, R.; Keady, J.; Williamson, P.R.; Reilly, S.T. Involving People Living with Dementia in Research: An Accessible Modified Delphi Survey for Core Outcome Set Development. Trials 2019, 20, 12. [Google Scholar] [CrossRef]
- Shepherd, V. Research Involving Adults Lacking Capacity to Consent: The Impact of Research Regulation on ‘Evidence Biased’ Medicine. BMC Med. Ethics 2016, 17, 55. [Google Scholar] [CrossRef] [PubMed]
- Knaapen, L.; Lehoux, P. Three Conceptual Models of Patient and Public Involvement in Standard-Setting: From Abstract Principles to Complex Practice. Sci. Cult. 2016, 25, 239–263. [Google Scholar] [CrossRef]
- Madden, M.; Speed, E. Beware Zombies and Unicorns: Toward Critical Patient and Public Involvement in Health Research in a Neoliberal Context. Front. Sociol. 2017, 2, 7. [Google Scholar] [CrossRef]
- International Collaboration for Participatory Health Research. What Is Participatory Health Research? International Collaboration for Participatory Health Research: Berlin, Germany, 2013. [Google Scholar]
- Nilsson, A. Who Gets to Decide? Right to Legal Capacity for Persons with Intellectual and Psychosocial Disabilities; The Council of Europe Commissioner for Human Rights: Strasbourg, France, 2012. [Google Scholar]
- European Convention on Human Rights. 34. Available online: https://www.echr.coe.int/documents/convention_eng.pdf (accessed on 10 January 2023).
- European Law Institute. The Protection of Adults in International Situations; ELI: Vienna, Austria, 2020. [Google Scholar]
- Keys, M. Legal Capacity Law Reform in Europe: An Urgent Challenge. Eur. Yearb. Disabil. Law 2009, 1, 59–88. [Google Scholar]
- Mental Disability Advocacy Centre. Legal Capacity in Europe: A Call to Action to Governments and to the EU; MDAC: Budapest, Hungary, 2013. [Google Scholar]
- United Nations Committee on the Rights of Persons with Disabilities. Concluding Observations on the Initial Report of the Czech Republic (CRPD/C/CZE/CO/1); United Nations: New York, NY, USA, 2015. [Google Scholar]
- United Nations Committee on the Rights of Persons with Disabilities. Concluding Observations on the Initial Periodic Report of Hungary, Adopted by the Committee at Its Eighth Session (CRPD/C/HUN/CO/1); United Nations: New York, NY, USA, 2012. [Google Scholar]
- Council of the Notariats of the European Union Protective Measures for Minors and Vulnerable Adults in 22 European Countries. Available online: http://www.the-vulnerable.eu/Home.aspx (accessed on 10 January 2023).
- Franzina, P.; Long, J.; The Protection of Vulnerable Adults in EU Member States. The Added Value of EU Action in the Light of The Hague Adults Convention. In Protection of Vulnerable Adults—European Added Value Assessment; European Parliament, Directorate-General for Parliamentary Research Services, Salm, C., Eds.; 2016; Available online: https://data.europa.eu/doi/10.2861/664256 (accessed on 10 January 2023).
- Knoppers, B.M.; Zawati, M.H.; Sénécal, K. Return of Genetic Testing Results in the Era of Whole-Genome Sequencing. Nat. Rev. Genet 2015, 16, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Tozzi, A.E.; Cinelli, G. Informed consent and artificial intelligence applied to RCT and COVID-19. BioLaw J. Riv. BioDiritto 2021, 2S, 97–108. [Google Scholar] [CrossRef]
- van Delden, J.J.M.; van der Graaf, R. Revised CIOMS International Ethical Guidelines for Health-Related Research Involving Humans. JAMA 2017, 317, 135–136. [Google Scholar] [CrossRef]
- Macioce, F. Informed consent and group vulnerability in the context of the pandemic. BioLaw J. Riv. BioDiritto 2021, 2S, 17–33. [Google Scholar] [CrossRef]
- Browne, J.; Dorris, E.R. What Can We Learn From a Human-Rights Based Approach to Disability for Public and Patient Involvement in Research? Front. Rehabilit. Sci. 2022, 3, 878231. [Google Scholar] [CrossRef] [PubMed]
- Sarlio-Siintola, S.; Aalto, J.; Alapuranen, N.; Ferri, D.; Haapaniemi, H.; Kesäniemi, E.; Kärpänen, T.; Lampi, I.; Nikula, K.; Pietikäinen, M.; et al. D8.14—Final SHAPES Ethical Framework Final Version; SHAPES—Smart and Healthy Ageing through People Engaging in Supportive Systems Consortium: Maynooth, Ireland, 2021. [Google Scholar]
- Seidel, K.; Labor, M.; Lombard-Vance, R.; McEvoy, E.; Cooke, M.; D’Arino, L.; Desmond, D.; Ferri, D.; Franke, P.; Gheno, I.; et al. Implementation of a Pan-European Ecosystem and an Interoperable Platform for Smart and Healthy Ageing in Europe: An Innovation Action Research Protocol. Open Res. Eur. 2022, 2, 85. [Google Scholar] [CrossRef]
- All European Academies. The European Code of Conduct for Research Integrity; Revised; All European Academies: Berlin, Germany, 2017. [Google Scholar]
- Arstein-Kerslake, A. Legal Capacity and Supported Decision-Making: Respecting Rights and Empowering People (University of Melbourne Law School Legal Studies Research Paper No. 736). 2016. Available online: https://doi.org/10.2139/ssrn.2818153 (accessed on 10 January 2023).
- Iacono, T.; Murray, V. Issues of Informed Consent in Conducting Medical Research Involving People with Intellectual Disability. J. Appl. Res. Int. Dis. 2003, 16, 41–51. [Google Scholar] [CrossRef]
- Budin-Ljøsne, I.; Teare, H.J.A.; Kaye, J.; Beck, S.; Bentzen, H.B.; Caenazzo, L.; Collett, C.; D’Abramo, F.; Felzmann, H.; Finlay, T.; et al. Dynamic Consent: A Potential Solution to Some of the Challenges of Modern Biomedical Research. BMC Med. Ethics 2017, 18, 4. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lombard-Vance, R.; Soye, E.; Ferri, D.; McEvoy, E.; MacLachlan, M.; Sarlio-Siintola, S. Applying the ‘Human Rights Model of Disability’ to Informed Consent: Experiences and Reflections from the SHAPES Project. Disabilities 2023, 3, 28-47. https://doi.org/10.3390/disabilities3010003
Lombard-Vance R, Soye E, Ferri D, McEvoy E, MacLachlan M, Sarlio-Siintola S. Applying the ‘Human Rights Model of Disability’ to Informed Consent: Experiences and Reflections from the SHAPES Project. Disabilities. 2023; 3(1):28-47. https://doi.org/10.3390/disabilities3010003
Chicago/Turabian StyleLombard-Vance, Richard, Evelyn Soye, Delia Ferri, Emma McEvoy, Malcolm MacLachlan, and Sari Sarlio-Siintola. 2023. "Applying the ‘Human Rights Model of Disability’ to Informed Consent: Experiences and Reflections from the SHAPES Project" Disabilities 3, no. 1: 28-47. https://doi.org/10.3390/disabilities3010003
APA StyleLombard-Vance, R., Soye, E., Ferri, D., McEvoy, E., MacLachlan, M., & Sarlio-Siintola, S. (2023). Applying the ‘Human Rights Model of Disability’ to Informed Consent: Experiences and Reflections from the SHAPES Project. Disabilities, 3(1), 28-47. https://doi.org/10.3390/disabilities3010003






