1. Introduction
Hydrogen atom [
1,
2,
3], electron [
4,
5] and energy [
6,
7,
8] transfer processes have been a subject of numerous theoretical and experimental studies. The exploration of these chemical processes has captivated the attention of researchers due to their significance as primary reactions in electronically excited molecules. Since the very first paper on photosensitization of naphthalene by the triplet of benzophenone in 1956 [
9], many studies have reported similar molecular systems [
10,
11]; however, the mechanism of interaction of triplet-sensitized molecules with ground-state donors still lacks full understanding. In other words, despite the wealth of studies on similar molecular systems, the interaction of triplet-sensitized molecules with ground-state donors remains an area requiring comprehensive exploration.
When the benzophenone–naphthalene system is irradiated using a 337 nm laser, only benzophenone is selectively excited, and two fundamental processes are energetically possible: energy transfer (Δ
ET ≈ −59 kJ/mol [
12]) and electron transfer (Δ
Gel ≈ −64 kJ/mol [
13,
14]). Yamaji and co-workers published a number of reports on triplet benzophenone-sensitized reactions with selected naphthalene derivatives (naphthols [
13,
15], naphtylamines [
12,
16] and methoxynaphtalenes [
3]). Their investigations addressed a crucial question: which mechanism predominates in the interaction between triplet-state benzophenone and naphthalene derivates anions—triplet energy transfer to naphthalene, resulting in excited triplet naphthalene, or electron/hydrogen transfer leading to the formation of respective radicals? Notably, the outcomes of these studies revealed that energy transfer and electron/hydrogen process are competitive to each other. According to the authors, the process mechanism’s path depends strongly on two factors: The first one is a solvent environment including the pH value [
13], polarity [
12,
16], and proticity as it is estimated that the hydrogen bonds in protic solvents induce electron/hydrogen transfer process, while in the aprotic solvents, the dominate process is triplet energy transfer [
16]. The second important factor is the naphthalene substituents employed as quenchers; their structure, including the presence of protonic hydrogens or function groups [
12], may significantly influence the occurrence of one of the two abovementioned processes [
3]. Building on this foundation, our current research focuses on the interaction between excited triplet 4-Carboxybenzophenone (CB, a water-soluble benzophenone analog) and 2-Naphthalene Sulfonate (NpSO
3−). Employing 337 nm Nanosecond Laser Flash Photolysis, we aim to contribute novel insights into the intricacies of these triplet interactions and unveil potential nuances in the triplet-sensitized reactions within this specific molecular system. In this communication, we report the interactions inherent to this system by closely examining the kinetic aspects of the investigated processes.
2. Experimental
All chemicals (4-Carboxybenzophenone and 2-Naphthalene Sulfonate Sodium salt) were purchased commercially from Sigma Aldrich (St. Louis, MO, USA) at the highest purity grade available; these compounds were used without further purification. Water was purified with a Millipore Milli-Q system (Bedford, MA, USA).
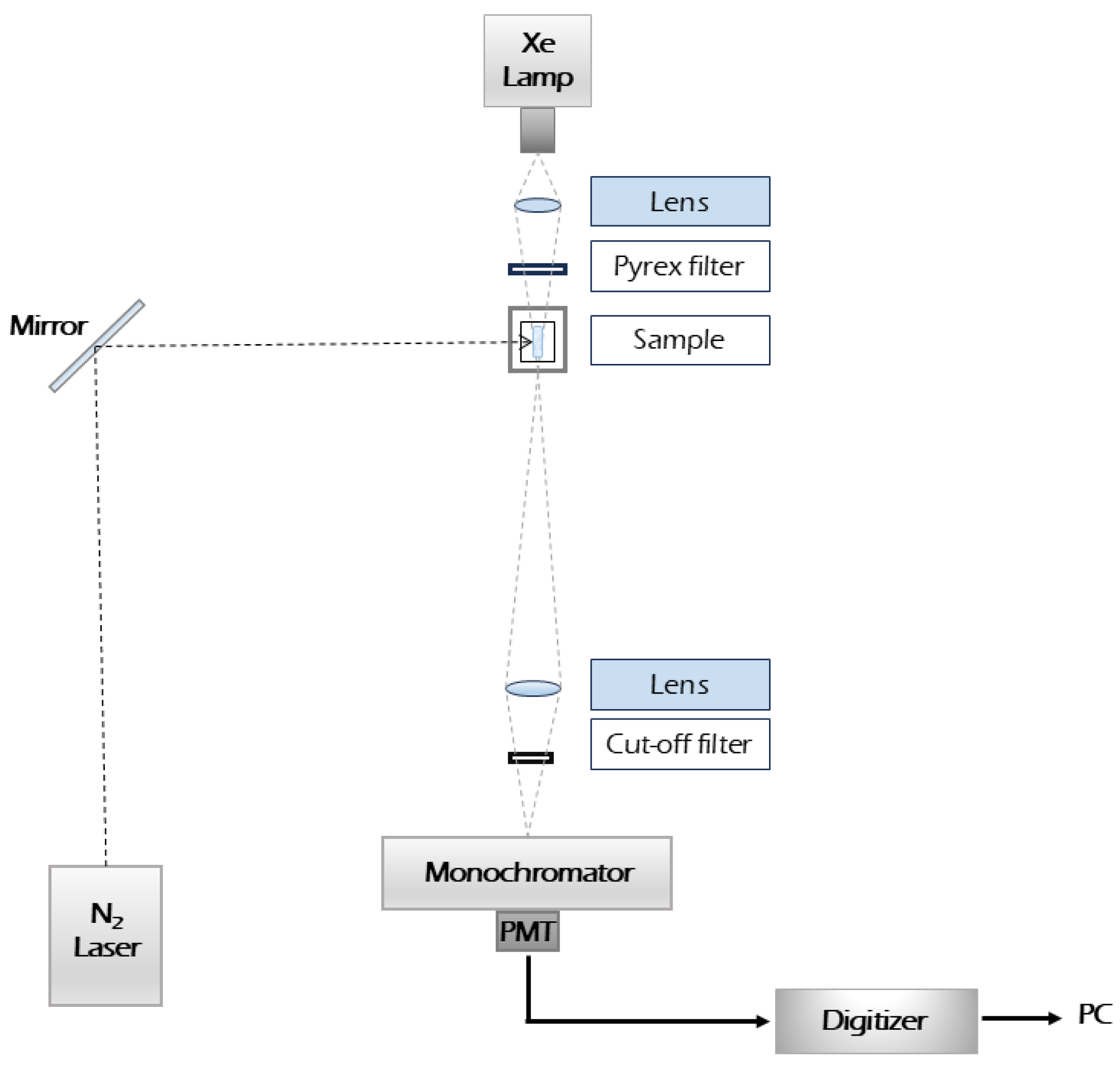
The Laser Flash Photolysis (LFP) experiments were conducted using a setup housed at the University of Notre Dame Radiation Laboratory [
17]. A nitrogen laser, specifically the Laser Photonics PRA/Model UV-24 (Laser Photonics, Lake Mary, FL, USA), served as the excitation source at a wavelength of 337.1 nm and at a repetition rate of 10 Hz [
18]. Transient absorbances at preselected wavelengths were monitored by a detection system consisting of a monochromator Spex 270 M (Queens, NY, USA), a photomultiplier tube Hamamatsu R955 (Iwata City, Japan), and a pulsed xenon lamp (1 kW) as the monitoring light source. The signal from the photomultiplier was processed by a LeCroy 7200 digital storage oscilloscope (Chestnut Ridge, NY, USA) and a PC-compatible computer [
17]. Rectangular quartz cells (0.5 × 1 cm) with a path length of 0.5 cm for the monitoring beam were used. Typically, 10 laser shots were averaged for each kinetic trace. To optimize signal quality, a configuration involving the laser and the Xe lamp beams was implemented, as illustrated in
Figure 1. The laser beam and the analyzing light were set to pass through the same section of the cell. A critical factor in ensuring accurate measurements was the relative cross-sectional size of the laser beam compared to the analyzing beam. This was achieved by positioning the analyzing beam at a right angle (90°) to the laser beam. Notably, the energy deposited during each pulse was controlled to be approximately 1.5 mJ/pulse, and the pulse duration was maintained at approximately 8 ns. Kinetic traces were recorded over a spectral range between 360 and 720 nm, with data points acquired at 10 nm intervals. These kinetic traces were subsequently employed to construct detailed time-resolved absorption spectra, providing valuable insights into the dynamic behavior of the studied reactants. To remove any traces of oxygen from the solutions, they were subjected to a thorough 15–20 min bubbling process with high-purity argon. This measure aimed to mitigate any potential interference from oxygen in the experimental results.
3. Results and Discussion
The 337 nm laser excitation of the aqueous solution containing CB (2 mM) and NpSO
3− sodium salt (1 mM) leads to the selective excitation of the former; naphthalene has a negligible absorption at 337 nm (see
Figure S1). Direct excitation of NpSO
3− is observed, but it results in very small concentrations of transients (see
Figure S2) which can be neglected. Both triplet energy transfer from the excited
3CB* to NpSO
3− and electron transfer from NpSO
3− to
3CB* are possible since the triplet energy of NpSO
3− is smaller than that of CB (Δ
ET = Δ
ET (NpSO
3−) − Δ
ET (CB) = –59 kJ/mol [
12,
13]). The free energy change in electron transfer of the latter process is approximately Δ
Gel ≈ –64 kJ/mol. This exothermic value for Δ
Gel is based on the following parameters:
Eox(NpSO
3−) = 0.5 V [
19],
Ered(CB) = –1.83 V [
20] and
ET(CB) = 69 kcal/mol [
14].
The CB...NpSO
3− system has been studied using 337 nm laser photolysis in order to reveal its mechanism.
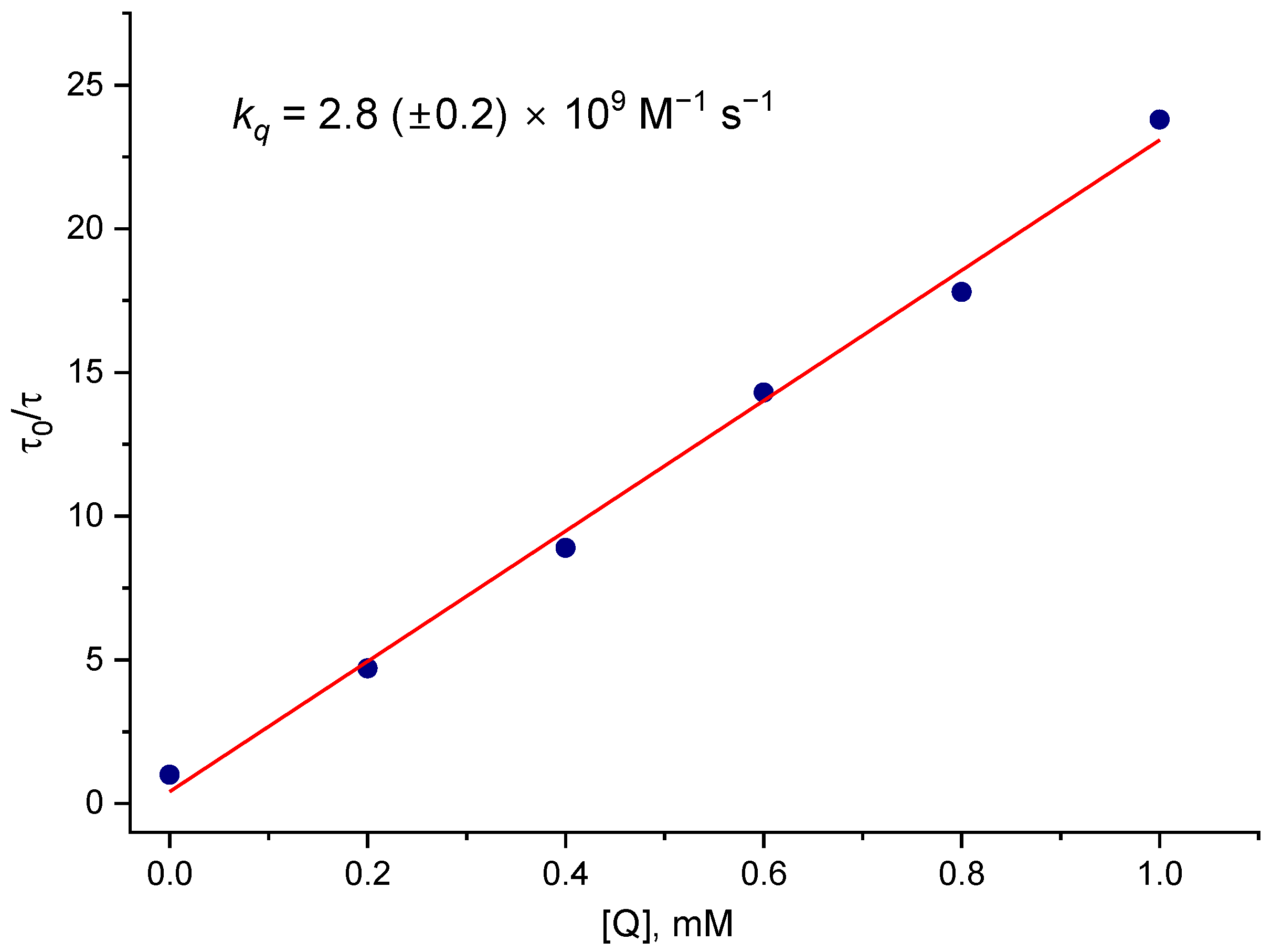
Figure 2 presents the Stern–Volmer plot of τ
0/τ vs. various concentrations of the quencher [NpSO
3−] obtained from Laser Flash Photolysis experiments. From the slope of the Stern-Volmer equation, Equation (1), (where τ
0 and τ are the lifetimes of
3CB* in the absence and in the presence of the quencher (NpSO
3−), respectively, and [Q] is the quencher molar concentration, and
kq is the quenching rate constant; the CB triplet lifetime in the absence of a quencher has been measured to be τ
0 = 6.2 µs for an argon-saturated solution) the quenching rate constant
kq = 2.8 × 10
9 M
−1s
−1 was obtained, suggesting a collisional quenching process.
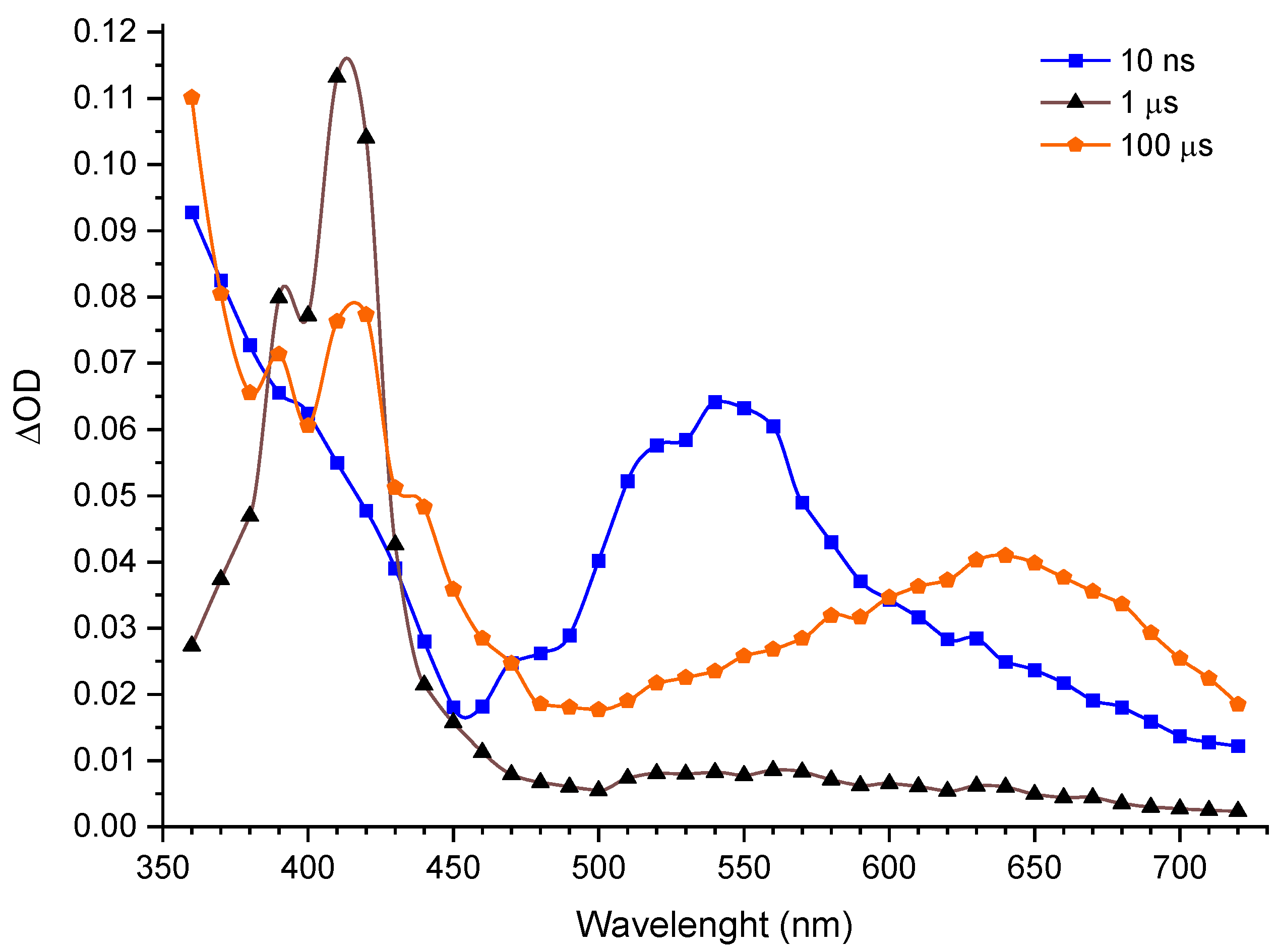
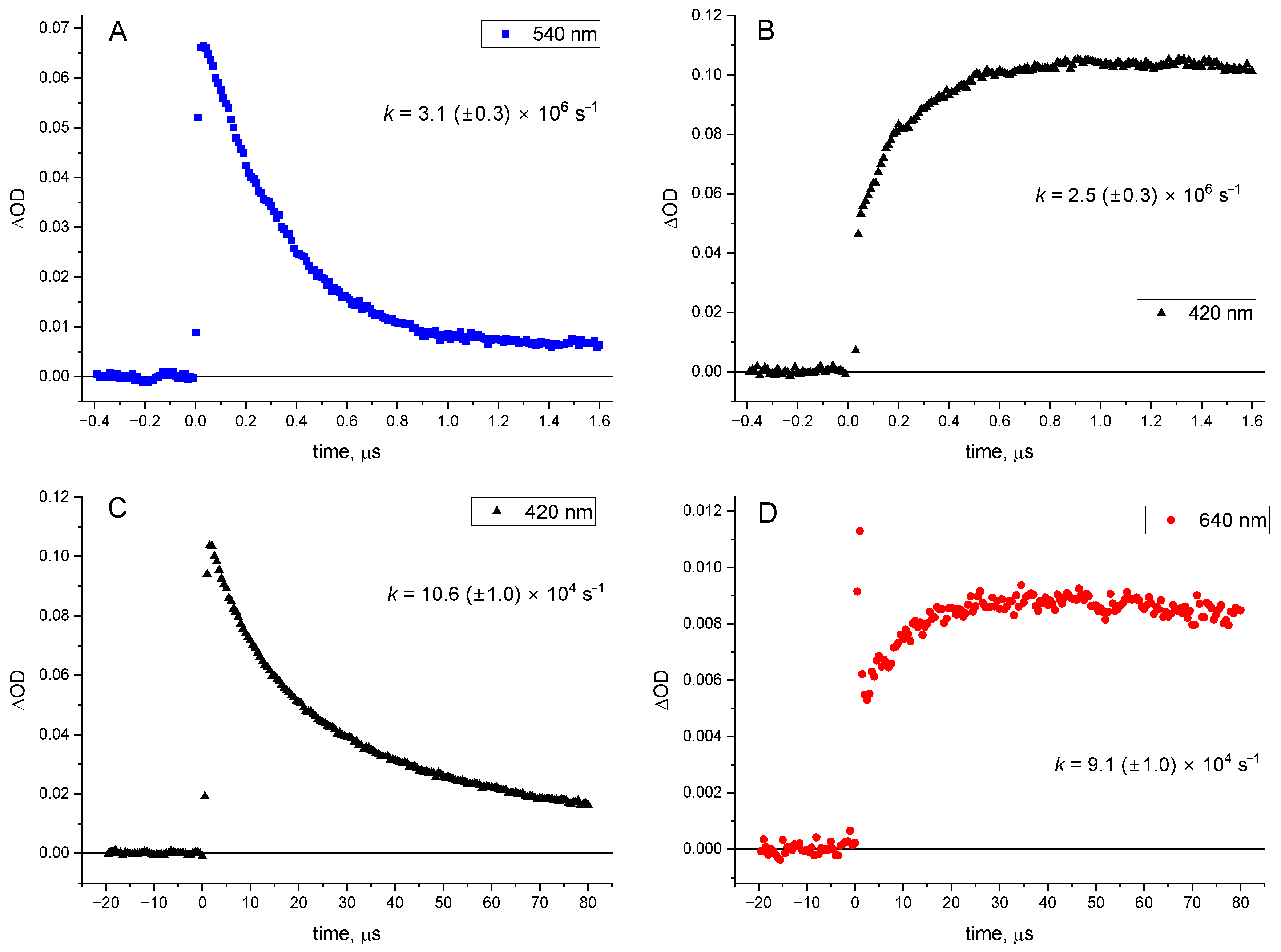
As depicted in
Figure 3,
Figure 4 and
Figure S3, the excitation of CB generates a quickly decaying absorption band (
kobs = 3.1 × 10
6 s
−1), peaking at around 540 nm and indicating the formation of the CB triplet [
21]. This decay is accompanied with a growth of an absorption at 410 nm; this transient absorption is formed with a very similar rate (within experimental error, see
Figure 4 for details) as the decay of
3CB* (
kobs = 2.5 × 10
6 s
−1). Its spectral shape with a sharp peak in the 410–420 nm region is a characteristic feature of the naphthalene triplet [
22], clearly indicating that the primary process in the investigated system is triplet energy transfer to form
3(NpSO
3−)*. This transient decays with a first-order rate constant of
kobs = 10.6 × 10
4 s
−1, and a new, broad absorption signal builds up at around 640 nm on the same time-scale (
kobs = 9.1 × 10
4 s
−1). The latter signal (see
Figure 4D) is most likely due to the formation of CB radical anion (CB
•−) [
22], resulting from an electron transfer from triplet naphthalene
3(NpSO
3−)* to the CB ground state.
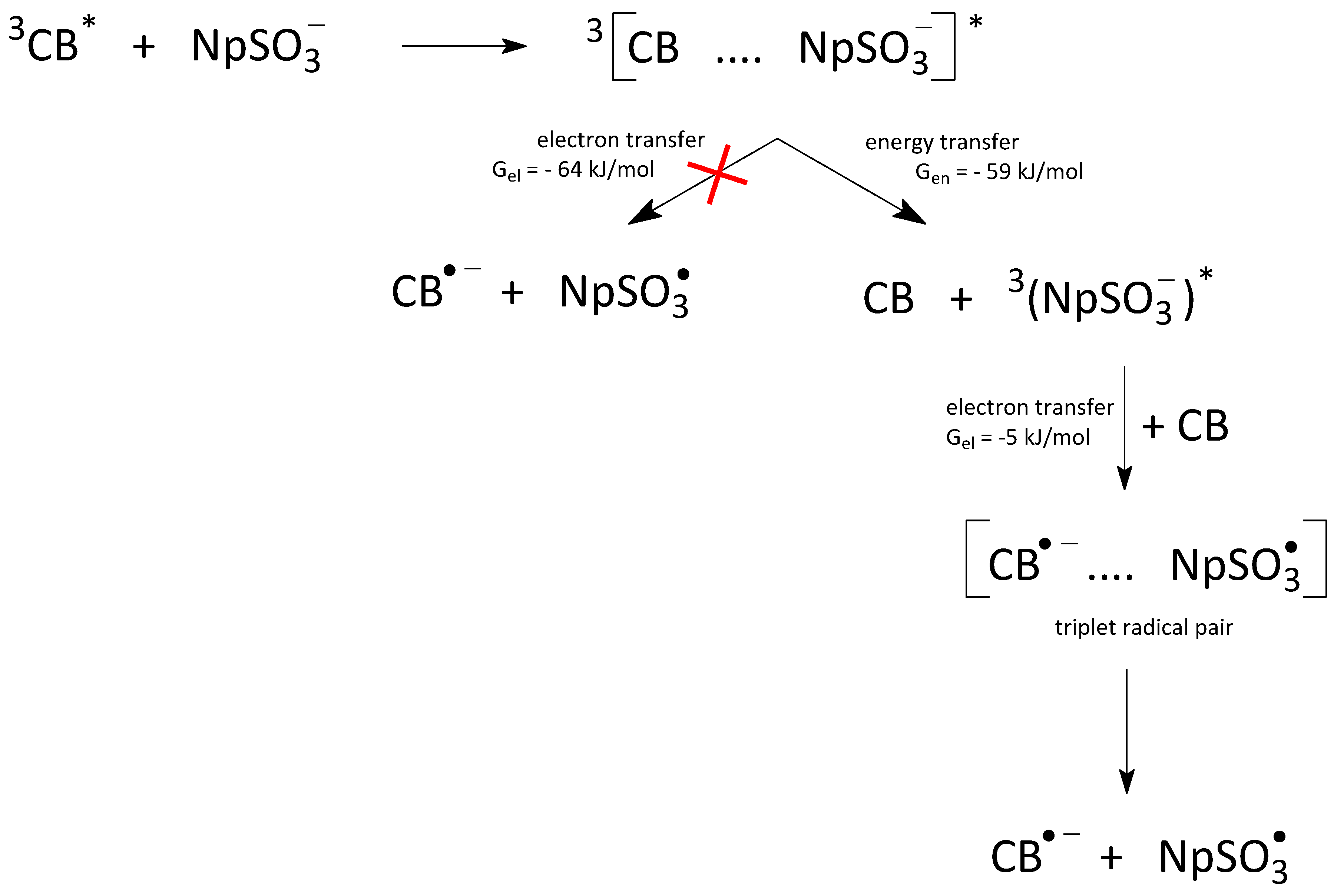
As mentioned previously, in aqueous solutions (pH ≈ 7), two competitive molecular interactions are possible between a triplet-excited state of 4-Carboxybenzophenone (
3CB*) and 2-Naphthalene Sulfonate anion (NpSO
3−): (i) electron transfer from NpSO
3− to
3CB* and (ii) energy transfer from
3CB* to NpSO
3−, which can be monitored with the Laser Flash Photolysis (LFP) technique. After excitation with a nitrogen laser pulse (λ = 337 nm), a broad band is observed, with the absorption maximum at 535 nm. With the specifically fixed excitation, only CB molecules are excited to the triplet state during the experiment, while NpSO
3− does not absorb in this spectral range. This transient absorption band decays, while a sharp peak builds up at around 420 nm, with similar kinetic parameters to those of the
3CB* decay. The signal most likely originates from the triplet-excited
3(NpSO
3−)* as a result of energy transfer from
3CB* to the naphthalene derivate. Subsequently,
3(NpSO
3−)* transiently decays by transferring electrons to the CB ground state, yielding a radical anion, CB
•−, which can be observed as a wide band with a maximum at around 640 nm, with a relatively long lifetime (in the range of milliseconds) along with the NpSO
3• radical. The NpSO
3• radical absorbs in the same range as CB
•− (see
Figure 3), making it difficult for direct observation. It should be noted that the secondary electron transfer process from the excited triplet
3(NpSO
3−)* to the ground-state CB is also energetically feasible at
Gel ≈ −5 kJ/mol (value calculated based on thermodynamic data given above). The proposed reaction mechanism in the CB...NpSO
3− system is presented in
Scheme 1.
Yamaji and co-workers conducted experiments on an analogous system:
3BP*...2-naphtol (NpO
−), and they reported their results in their work in 1995 [
13]. According to them, out of two energetically conceivable primary processes: (i) triplet–triplet energy transfer and (ii) electron transfer from NpO
− to
3BP*, only the latter was experimentally observed. The authors attribute the occurrence of electron transfer process (instead of the energy transfer process) with Dexter’s electron exchange mechanism [
13,
23]. In this mechanism the primary process is an electron transfer, resulting in the triplet ionic pair
3(BP
•−...NpO
•)*. The obtained excimer decomposes rapidly, yielding respective radicals, before the transfer of the back electron (a necessary process, according to the Dexter electron exchange mechanism). This decomposition was explained by the authors as the weak Coulombic attraction in the complex, specifically inert NpO
• radical and BP
•− radical anion interactions were considered less effective than cation–anion interactions, resulting in a more efficient formation of BP
•− and NpO
• radicals.
Focusing on the structure of the excited complex
3CB*…NpSO
3− in our study, we note a similarity to the
3(BP
•−...NpO
•)* complex in which no Coulombic attractions are evident to stabilize the structures. In our system, however, the only observed primary process is energy transfer, as indicated by its rate constant of
kq = 2.8 × 10
9 dm
3 mol
−1 s
−1, with no evidence of products from the electron transfer process. This observation clearly contrasts the findings of Yamaji et al. [
13]. In the investigated system, the initial energy/electron transfer competition is controlled by photoenergy transfer, possibly due to geometrical constraints in a polar solvent environment. The final electron transfer process is relatively slow (
kobs = 10.6 × 10
4 s
−1) due to its low driving force of just ≈ −5 kJ/mol.
Conversely, Yamaji et al. revealed in their 1993 paper [
15] that the energy transfer deactivation path is significantly solvent-dependent, with the rate of energy transfer increasing with the polarity of the solvent. Notably, the
3BP*…NpO
− experiments were conducted in an acetonitrile and water mixture, which is less polar than the purified Milli-Q water used in our study. The authors explain that in more polar solvents, the energy gap of
3BP decreases due to spin–orbital coupling, thereby increasing the likelihood of the energy transfer reaction from
3BP*. This solvent-dependent behavior provides valuable context for the contrasting results observed between the two systems and emphasizes the nuanced interplay between the excited-state processes and the solvent environment.
4. Conclusions
In our investigation aimed at elucidating the complex interactions between the excited state of 4-Carboxybenzophenone (CB) and 2-Naphthalene Sulfonate (NpSO3−), we initiated a targeted excitation of the photosensitizer (CB) to its triplet state using the 337 nm Nanosecond Laser Flash Photolysis (LFP). This excitation facilitated the intricate interplay between CB and NpSO3−. Our experimental findings provided valuable insights into the primary processes governing this interaction, particularly focusing on electron transfer and energy transfer mechanisms.
Contrary to the anticipated scenarios, the experimental results demonstrated the prevalence of energy transfer from ³CB* to NpSO3−, rather than the electron transfer process. Subsequent to this energy transfer event, an electron transfer from the triplet naphthalene ³(NpSO3−)* to the ground state of CB ensued. This secondary reaction step led to the emergence of a distinct transient absorption band at around 640 nm, signaling the presence of CB•−. Significantly, the formation of this radical anion occurred at a considerably slower rate compared to the products of the primary energy transfer process, introducing a temporal complexity to the observed reactions.
The experimental observations presented here starkly contrast with the results reported by Yamaji et al. in 1995. The differing reactivity observed for the two naphthalene derivatives, NpSO3− and NpO−, investigated in their studies, can likely be attributed to subtle disparities in their redox properties. This nuanced interplay between the photosensitizer and quencher molecules highlights the sensitivity of these reactions to the specific molecular structures and electronic configurations of the species involved. It is also crucial to acknowledge that the experimental conditions for the two systems (Yamaji et al. 3BP*…NpO− and our study on 3CB*…NpSO3−) were conducted in solvents with differing polarities. This environmental contrast adds an extra layer of complexity as solvent polarity can significantly influence the direction and efficiency of the ³CB* deactivation process. The solvent environment thus emerges as a critical factor influencing the dynamics of the reaction and requires careful consideration in interpreting the observed results. This exploration into the excited-state dynamics of CB and NpSO3− not only enhances our understanding of their interactions but also underscores the intricate role played by solvent conditions in shaping the outcomes of these chemical processes.