Long-Term Changes in Four Populations of the Spiny Toad, Bufo spinosus, in Western France; Data from Road Mortalities
Abstract
:1. Introduction
- (1)
- What are the general long-term population trends in the study area?
- (2)
- Are there differences in long-term numbers between populations?—here defined as subpopulations migrating to different breeding ponds.
- (3)
- To what extent do the populations fluctuate on an annual basis?
- (4)
- (5)
- What impact did the restrictions on human movement due to the COVID-19 pandemic have on road mortalities during 2020 and 2021?
2. Methods
2.1. Study Area
2.2. Protocol
2.3. Statistical Analysis
3. Results
3.1. Total and Regional Counts
3.2. Monthly Counts
3.3. Frequency of Large Females
3.4. Annual Differences in SVL between Populations
3.5. Long-Term Annual Counts
3.6. Rainfall, Temperature and Annual Counts
3.7. Mortalities during 2020 and 2021
4. Discussion
4.1. General Considerations
4.2. Comparisons with Sympatric Amphibians
4.3. Rainfall and Temperature
4.4. Impact of Lockdown Restrictions
4.5. Concluding Remarks
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Colwell, R.K.; Dunn, R.R.; Harris, N.C. Coextinction and persistence of dependent species in a changing world. Annu. Rev. Ecol. Evol. Syst. 2012, 43, 183–203. [Google Scholar] [CrossRef] [Green Version]
- Beebee, T.J.C.; Griffiths, R.A. The amphibian decline crisis: A watershed for conservation biology? Biol. Conserv. 2005, 125, 271–285. [Google Scholar] [CrossRef]
- Wake, D.B. Declining amphibian populations. Science 1991, 253, 860. [Google Scholar] [CrossRef] [PubMed]
- Stuart, S.N.; Chanson, J.S.; Cox, N.A.; Young, B.E.; Rodrigues, A.S.L.; Fischmann, D.L.; Waller, R.W. Status and trends of amphibian declines and extinctions worldwide. Science 2004, 306, 1783–1786. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kyek, M.; Kaufmann, P.H.; Lindner, R. Differing long term trends for two common amphibian species (Bufo bufo and Rana temporaria) in alpine landscapes of Salzburg, Austria. PLoS ONE 2017, 12, e0187148. [Google Scholar] [CrossRef] [Green Version]
- Meek, R. Population trends of four species of amphibians in western France; results from a 15 year time series derived from road mortality counts. Acta Oecologica 2021, 110, 103713. [Google Scholar] [CrossRef]
- Pechmann, J.H.K.; Scott, D.E.; Semlitsch, R.D.; Caldwell, J.P.; Vitt, L.J.; Gibbons, J.W. Declining amphibian populations: The problem of separating human impacts from natural fluctuations. Science 1991, 253, 892–895. [Google Scholar] [CrossRef] [Green Version]
- Alford, R.A.; Richards, S.J. Global amphibian declines: A problem in applied ecology. Ann. Rev. Ecol. System. 1999, 30, 133–165. [Google Scholar] [CrossRef] [Green Version]
- Houlahan, J.E.; Findlay, C.S.; Schmidt, B.R.; Meyer, A.H.; Kuzmin, S.L. Quantitative evidence for global amphibian population declines. Nature 2000, 404, 752–755. [Google Scholar] [CrossRef]
- Green, D.M. The ecology of extinction: Population fluctuation and decline in amphibians. Biol. Conserv. 2003, 111, 331–343. [Google Scholar] [CrossRef]
- Green, D.M. Perspectives on amphibian declines: Defining the problem and searching for answers. In Amphibians in Decline: Canadian Studies of a Global Problem; Green, D.M., Ed.; Society for the Study of Amphibians and Reptiles, 1997; pp. 291–308. Available online: https://www.researchgate.net/publication/247167604_Perspectives_on_amphibian_population_declines_defining_the_problem_and_searching_for_answers (accessed on 4 July 2021).
- Scherer, R.D.; Tracey, J.A. A power analysis for the use of counts of egg masses to monitor wood frog (Lithobates sylvaticus) populations. Herpetol. Conserv. Biol. 2011, 6, 81–90. [Google Scholar]
- Cooke, A.S.; Sparks, T.H. Population declines of common toads (Bufo bufo): The contribution of road traffic and monitoring value of casualty counts. Herpetol. Soc. Bull. 2004, 88, 13–26. [Google Scholar]
- Meyer, A.H.; Schmidt, B.R.; Grossenbacher, K. Analysis of three amphibian populations with quarter century long time series. Proc. R. Soc. 1998, 265, 523–528. [Google Scholar] [CrossRef]
- Meek, R. Patterns of amphibian road-kills in the Vendée region of Western France. Herpetol. J. 2012, 22, 51–58. [Google Scholar]
- Meek, R. Temporal trends in agile frog Rana dalmatina numbers: Results from a long term study in western France. Herpetol. J. 2018, 28, 117–122. [Google Scholar]
- Mallick, S.A.; Hocking, G.J.; Driessen, M.M. Road kills of the eastern barred bandicoot (Perameles gunnii) in Tasmania: An index of abundance. Wildl. Res. 1998, 2, 139–145. [Google Scholar] [CrossRef]
- Baker, P.; Harris, S.; Robertson, C.; Saunders, G.; White, P. Is it possible to monitor mammal population changes from counts of road traffic casualties? An analysis using Bristol’s red foxes Vulpes vulpes as an example. Mamm. Rev. 2004, 34, 115–130. [Google Scholar] [CrossRef]
- Widenmaier, K.; Fahrig, L. Inferring white-tailed deer (Odocoileus virginianus) population dynamics from wildlife collisions in the City of Ottawa. In Proceedings of the 2005 International Conference on Ecology and Transportation, San Diego, CA, USA, 29 August–2 September 2005; Irwin, C.L., Garrett, P., McDermott, K.P., Eds.; Center for Transportation and the Environment: San Diego, CA, USA, 2006; pp. 589–602. [Google Scholar]
- Battisti, C.; Amori, G.; De Felici, S.; Luiselli, L.; Zapparoli, M. Mammal roadkilling from a Mediterranean area in central Italy: Evidence from an atlas dataset. Rend. Lincei 2012, 2, 217–223. [Google Scholar] [CrossRef]
- Meek, R. Temporal trends in Podarcis muralis and Lacerta bilineata populations in a fragmented landscape in Western France: Results from a 14 year time series. Herpetol. J. 2020, 30, 19–25. [Google Scholar] [CrossRef] [Green Version]
- Rugiero, L.; Capula, M.; Capizzi, D.; Amori, G.; Milana, G.; Lai, M.; Luiselli, L. Long-term observations on the number of roadkilled Zamenis longissimus (Laurenti, 1768) in a hilly area of central Italy. Herpetozoa 2018, 30, 212–217. [Google Scholar]
- Beckmann, C.; Shine, R. Do the numbers and locations of road-killed anuran carcasses accurately reflect impacts of vehicular traffic? J. Wildl. Manag. 2014, 79, 92–101. [Google Scholar] [CrossRef]
- Gotelli, N.J.; Ellison, A.M. A Primer of Ecological Statistics; Sinauer Associates: Sunderland, MA, USA, 2004; p. 510. [Google Scholar]
- Meek, R.; Jolley, E. Body temperatures of the common toad, Bufo bufo, in the Vendee, France. Herpetol. Bull. 2006, 95, 21–24. [Google Scholar]
- Heusser, H. Uber die Beziehungen der Erdkröte (Bufo bufo L.) zu ihrem Laichplatz. II. Behaviour 1960, 16, 93–109. [Google Scholar] [CrossRef]
- Heusser, H. Die Lebensweise der Erdkrote, Bufo bufo (L.); Das Orientierungsproblem. Rev. Suisse Zool. 1969, 76, 443–518. [Google Scholar] [PubMed]
- Haapanen, A. Site tenacity of the common toad, Bufo bufo (L). Ann. Zool. Fenn. 1974, 11, 251–252. [Google Scholar]
- Sinsch, U. Seasonal changes in migratory behaviour of the toad Bufo bufo; direction and magnitude of movements. Oecologia 1998, 76, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Reading, C.J.; Loman, J.; Madsen, T. Breeding pond fidelity in the common toad Bufo bufo. J. Zool. 1991, 225, 201–211. [Google Scholar] [CrossRef]
- Zuiderwijk, A.; Janssen, I. Results of 14 Years Reptile Monitoring in the Netherlands. In Proceedings of the 6th World Congress of Herpetology, Manaus, Brazil, 18 August 2008. [Google Scholar]
- Cruickshank, S.S.; Ozgul, A.; Zumbach, S.; Schmidt, B.R. Quantifying population declines based on presence only records for Red List assessments. Conserv. Biol. 2016, 30, 1112–1121. [Google Scholar] [CrossRef]
- Gittins, S.P.; Parker, A.G.; Slater, F.M. Population characteristics of the common toad (Bufo bufo) visiting a breeding site in mid -Wales. J. Anim. Ecol. 1980, 49, 161–173. [Google Scholar] [CrossRef]
- Gittins, S.P. The breeding migration of the common toad (Bufo bufo) to a pond in Mid-Wales. J. Zool. Lond. 1983, 199, 555–562. [Google Scholar] [CrossRef]
- Griffiths, R.A.; Harrison, J.D.; Gittins, S.P. The breeding migrations of amphibians at Llysdinam pond, Wales: 1981–1985. In Studies in Herpetology; Rocek, Z., Ed.; Charles University: Prague, Czech Republic, 1986; pp. 543–546. [Google Scholar]
- Kuhn, J. Lebensgeschichte und demographie von erdkrötenweibchen Bufo bufo (L.). Z. Feldherpelopie 1994, 1, 3–87. [Google Scholar]
- Schabetsberger, R.; Langer, H.; Jersabek, M.; Goldschmid, A. On age structure and longevity in two populations of Bufo bufo (LINNAEUS, 1758) at high altitude breeding sites in Austria. Herpetozoa 2000, 13, 187–191. [Google Scholar]
- Wallace, H.; Badawy, G.M.I.; Wallace, B.M.N. Amphibian sex determination and sex reversal. Cell. Mol. Life Sci. 1999, 55, 901–909. [Google Scholar] [CrossRef] [PubMed]
- Matsuba, C.; Miura, I.; Merilä, J. Disentangling genetic vs. environmental causes of sex determination in the common frog, Rana temporaria. BMC Genet. 2008, 9, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, M.A.; Green, D.M. Dispersal and the metapopulation paradigm in amphibian ecology and conservation: Are all amphibian populations metapopulations? Ecography 2005, 28, 110–128. [Google Scholar] [CrossRef]
- Pechmann, J.H.K.; Estes, R.A.; David, E.; Scott, D.E.; Whitfield Gibbons, J. Amphibian colonization and use of ponds created for trial mitigation of wetland loss. Wetlands 2001, 21, 93–111. [Google Scholar] [CrossRef]
- Petranka, J.W.; Kennedy, C.A.; Murray, S.S. Response of amphibians to restoration of a southern appalachian wetland: A long-term analysis of community dynamics. Wetlands 2003, 3, 1030–1042. [Google Scholar] [CrossRef]
- Gittins, S.P.; Kennedy, R.I.; Williams, R. Aspects of the population age- structure of the common toad (Bufo bufo) at Llandrindod Wells lake, mid-Wales. Br. J. Herpetol. 1985, 6, 447–449. [Google Scholar]
- Ficetola, G.F.; Siesa, M.E.; Manenti, R.; Bottoni, L.; De Bernardi, L. Early assessment of the impact of alien species: Differential consequences of an invasive crayfish on adult and larval amphibians. Divers. Distrib. 2011, 17, 1141–1151. [Google Scholar] [CrossRef]
- Semlitsch, R.D.; Scott, D.E.; Pechmann, J.H.K.; Gibbons, J.W. Structure and dynamics of an amphibian community: Evidence from a 16-year study of a natural pond. In Long-Term Studies of Vertebrate Communities; Cody, M.L., Smallwood, J.A., Eds.; Academic Press: San Diego, CA, USA, 1996; pp. 217–248. [Google Scholar]
- Bragg, A.N. Population fluctuation in the amphibian fauna of Cleveland County, Oklahoma during the past twenty-five years. Southwest. Nat. 1960, 5, 165–169. [Google Scholar] [CrossRef]
- Bragg, A.N. Decline in toad populations in central Oklahoma. Proc. Oklahoma Acad. Sci. 1954, 33, 70. [Google Scholar]
- Zipkin, E.F.; DiRenzo, G.V.; Ray, J.M.; Rossman, S.; Lips, K.R. Tropical snake diversity collapses after widespread amphibian loss. Science 2020, 367, 814–816. [Google Scholar] [CrossRef] [PubMed]
- Manenti, R.; Mori, E.; Di Canio, V.; Mercurio, S.; Picone, M.; Ca, M.; Brambilla, M.; Ficetola, G.F.; Rubolini, D. The good, the bad and the ugly of COVID-19 lockdown effects on wildlife conservation: Insights from the first European locked down country. Biol. Conserv. 2020, 249, 108728. [Google Scholar] [CrossRef] [PubMed]
- Mougey, T. Des tunnels pour batraciens. Cour. Nat. 1996, 155, 22–28. [Google Scholar]
- Mcinroy, C.; Rose, T. Trialling amphibian ladders within roadside gullypots in Angus, Scotland: 2014 impact study. Herpetol. Bull. 2015, 132, 15–19. [Google Scholar]




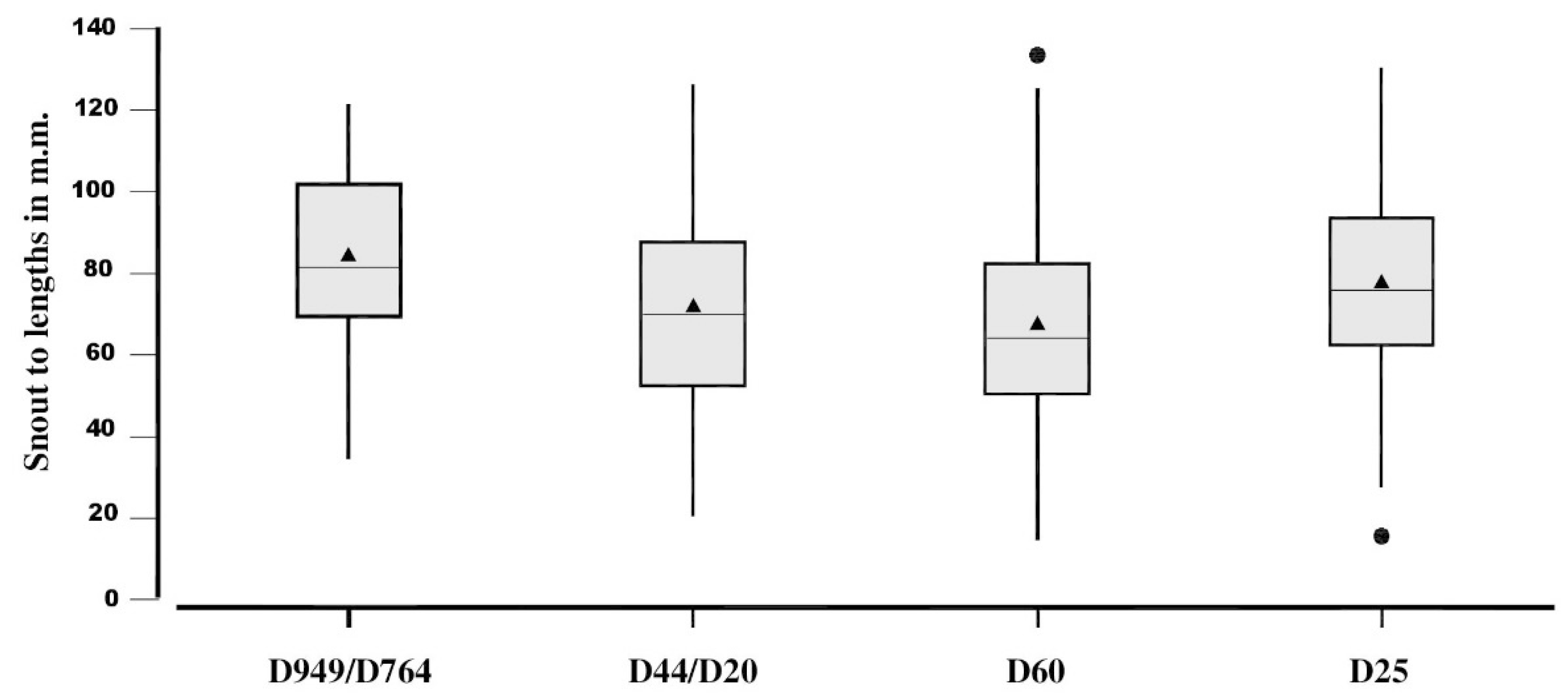

| D949/D746 | D44/D127 | D60 | D25 | |
|---|---|---|---|---|
| Means | 83.7 ± 20.2 | 71.3 ± 73.8 | 66.9 ± 22.2 | 77.3 ± 23.1 |
| Skewness | −0.07 | 0.24 | 0.47 | −0.08 |
| n | 187 | 347 | 386 | 359 |
| Area | m | ±SE | t | P | Mean JK m | Mean JK Std Error | P | Influence Function |
|---|---|---|---|---|---|---|---|---|
| D949/D764 | −0.07 | 0.04 | 1.58 | 0.14 | −0.07 | 0.046 | 0.14 | 2007 (L) |
| D44/D127 | 0.05 | 0.04 | 1.41 | 0.18 | 0.03 | 0.055 | 0.20 | 2015 (L) |
| D60 | 0.15 | 0.03 | 4.84 | <0.0001 | 0.15 | 0.031 | <0.0001 | 2010 (H) |
| D25 | 0.23 | 0.04 | 6.20 | <0.0001 | 0.22 | 0.039 | <0.0001 | 2008 (L) |
| 2 | λ Rate Parameter | x Random Variable | P (λ = x) | P (x > λ) |
| D949/764 | 12 | 1 | <1% | >99.9% |
| D44/127 | 15 | 5 | <1% | >99.9% |
| D60 | 24 | 8 | <1% | >99.9% |
| D25 | 22 | 15 | 0.03% | 92.3% |
| 2020 total n | 29 | |||
| λ Rate Parameter | x Random Variable | P (λ = x) | P (x > λ) | |
| D949/764 | 12 | 1 | <1% | >99.9% |
| D44/127 | 15 | 4 | <1% | >99.9% |
| D60 | 24 | 5 | <1% | >99.9% |
| D25 | 22 | 11 | 0.004% | >99.9% |
| 2021 total n | 21 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meek, R. Long-Term Changes in Four Populations of the Spiny Toad, Bufo spinosus, in Western France; Data from Road Mortalities. Conservation 2022, 2, 248-261. https://doi.org/10.3390/conservation2020017
Meek R. Long-Term Changes in Four Populations of the Spiny Toad, Bufo spinosus, in Western France; Data from Road Mortalities. Conservation. 2022; 2(2):248-261. https://doi.org/10.3390/conservation2020017
Chicago/Turabian StyleMeek, Roger. 2022. "Long-Term Changes in Four Populations of the Spiny Toad, Bufo spinosus, in Western France; Data from Road Mortalities" Conservation 2, no. 2: 248-261. https://doi.org/10.3390/conservation2020017
APA StyleMeek, R. (2022). Long-Term Changes in Four Populations of the Spiny Toad, Bufo spinosus, in Western France; Data from Road Mortalities. Conservation, 2(2), 248-261. https://doi.org/10.3390/conservation2020017





