1. Introduction
Over the years, concrete has dominated the construction industry due to the fact that its constituent materials are evenly distributed throughout the world, thereby making it easily available for use. Cement, which is a major component of concrete, has been used for centuries as a result of the hydraulic property that it possesses. However, the production of cement has drawn a lot of criticisms and concerns due to its contribution towards the release of carbon dioxide into the atmosphere. In 2016, cement production generated around 2.2 billion tons of CO
2—equivalent to 8% of the global total CO
2 emission [
1]. In order to reduce the CO
2 emission footprint in cement production, industrial by-products, also known as supplementary cementitious materials, are introduced in concrete mix proportions to partially replace Portland cement. One of such SCMs is ground granulated blast furnace slag (BFS), which is a by-product from iron or steel production. Alkali-activated materials, in which Portland cement is not used at all, have recently been demonstrated for their environmental-friendly nature [
2]. However, it is still difficult to use this type of material as a general-purpose concrete material.
In Japan, a blended cement incorporating BFS at a replacement ratio of around 40%, accounts for 20% of the total amount of cement used and has been earmarked as an environmentally-friendly product [
3]. This means the cement with GGBFS is the second popular cement next to ordinary Portland cement in this country. However, the effects of the kind and the contents of minor additives such as gypsum and limestone powder have not been made clear yet.
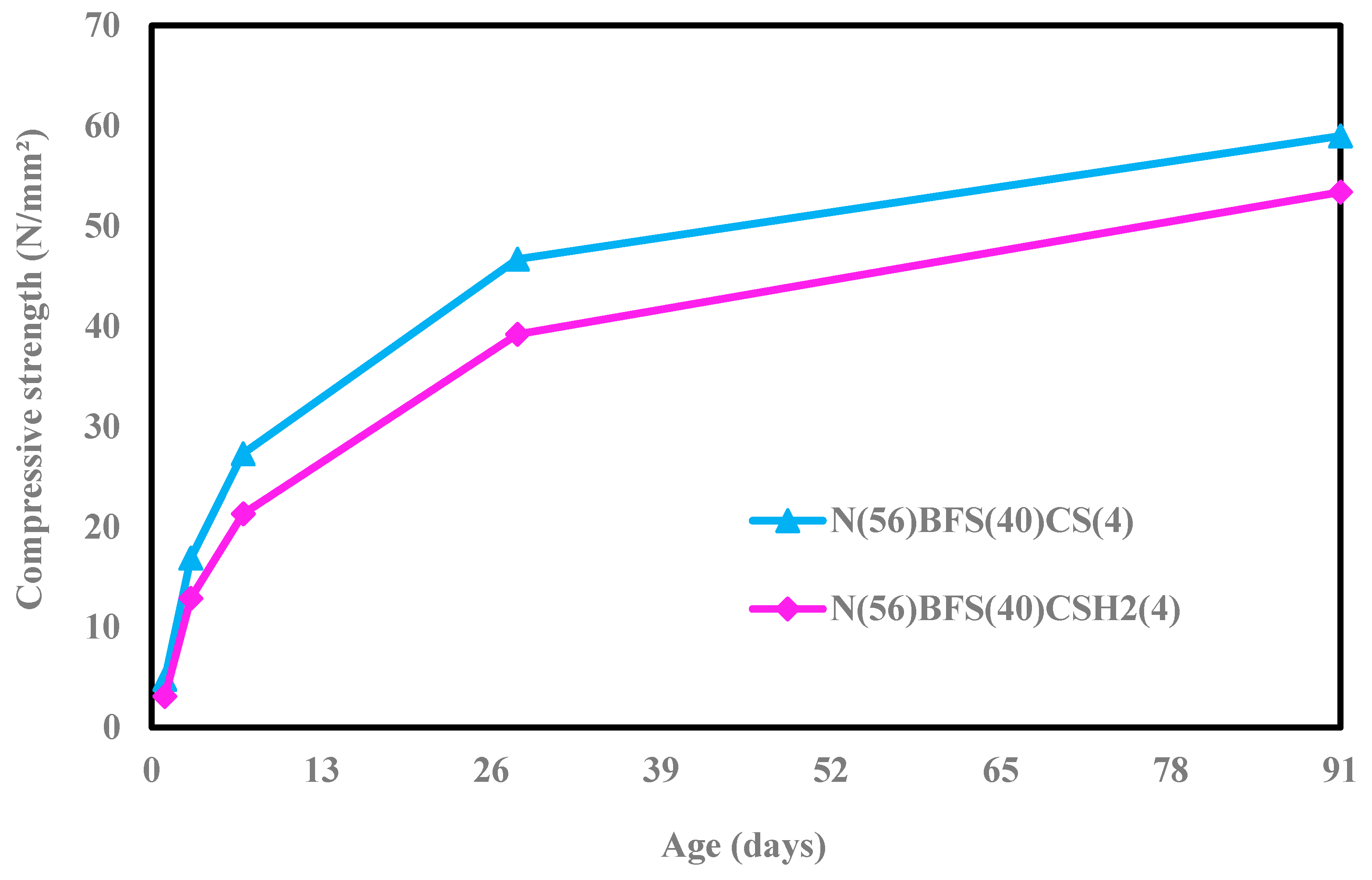
Gypsum is an important additive that plays a critical role in cement production. The additive ingredient present in gypsum is calcium sulfate. Gypsum exists in many forms, such as anhydrous gypsum, dihydrate gypsum, and hemihydrate, with different solubility rates, which are very important in determining the strength gain and setting of concrete. Hemihydrate has the highest solubility, about five times that of gypsum, while natural anhydrite is the least soluble. The quantity or optimum amount of SO
3 to be used in concrete need to be specified since it affects not only setting but compressive strength and expansion. The minimum content of SO
3 required to control setting is typically around 2% [
4]. In a study, it was reported that the C
3S reaction amount increased when gypsum was incorporated in a concrete mix containing ggbfs and the increase was thought to have affected the strength enhancement of the mix containing gypsum [
5]. Another research studied compressive strength development by adding gypsum and observed that gypsum addition produced fine ettringite crystals and removed large pores, resulting in significant pore-size refinement and enhancing strength development. However, excessive gypsum addition led to a reduction in strength due to expansion [
6]. An experiment was conducted to compare compressive strength development using two forms of calcium sulfates, namely natural gypsum (CaSO
4·2H
2O) and anhydrite (CaSO
4). The test results indicated that the higher optimum strength was achieved in the mix containing anhydrite as the retarder because it contained less pore volume, lower average pore diameter, and higher solubility [
7].
Limestone powder can be used to replace Portland cement in concrete production, and it acts as a filler with some beneficial effects on some concrete properties, such as workability, permeability, etc. Limestone powder mainly consists of calcium carbonates also called calcite and its reaction undergoes three sequences when added to Portland cement: 1. Ettringite forms through consumption of mono-sulphate; 2. When mono-sulphate is exhausted, mono-carbonate forms through consumption of hermi-carbonate; and 3. When the hermi-carbonate phase is used up, additional calcium carbonate does not react but persists as a stable phase [
8]. The first two reactions give limestone powder its filler properties while the third reaction makes limestone powder a ‘diluent’ in that porosity increases due to further addition of carbonates. In Japan, the maximum limestone powder replacement level is 5%. On setting time, a study reported that the addition of limestone powder led to an acceleration of setting time [
9,
10]. A substantial decrease in initial and final setting time after inter-grounding an additional 5% of limestone with cement clinker containing 5% gypsum has been reported [
11]. Limestone powder addition can have both positive and negative effects on compressive strength. Limestone powder was added to a concrete mix containing 20% and 25 wt% BFS and compressive strength was found to increase [
12]. At limestone replacements up to 5%, there is an increase in early-age strength as a result of improved particle packing and an increase in cement hydration [
13]. When limestone powder addition was 15%, concrete reduced in strength as the limestone content increased [
14,
15].
The objective of this research is to improve the properties of general-purpose cement with BFS by modifying composition of minor additives. Fresh properties of concrete such as slump, setting time, and compressive strength cast at two different ambient temperatures were experimentally investigated on concrete mixture proportions containing ground granulated blast furnace slag, incorporating two different forms of gypsum, namely anhydrous gypsum and dihydrate gypsum, using variable SO3 contents and limestone powder. By curing the mix proportions at two different temperatures of 20 °C and 10 °C, the formwork removal time was also estimated using “maturity equation”.