Heart Energy Harvesting and Cardiac Bioelectronics: Technologies and Perspectives
Abstract
1. Introduction
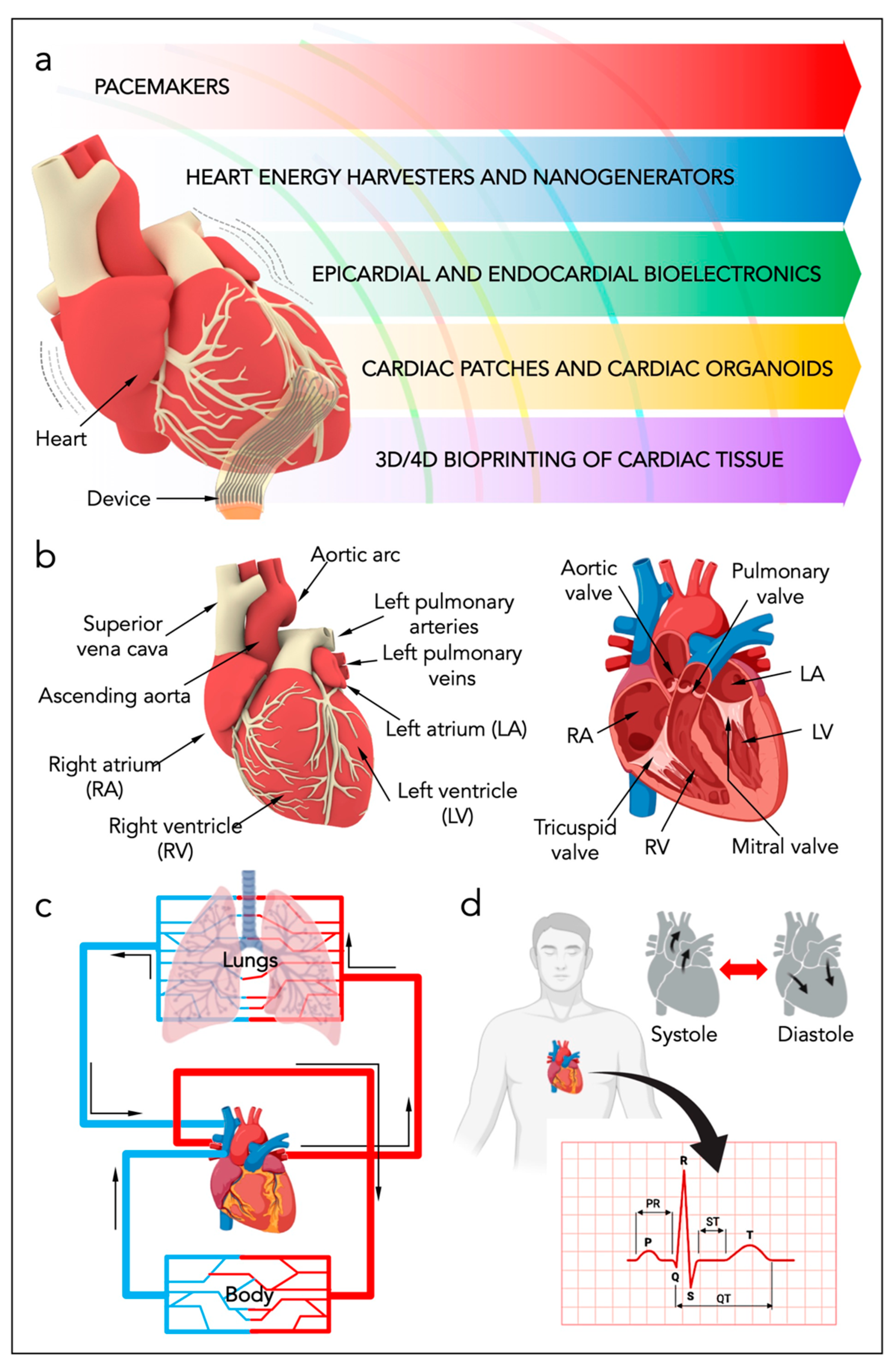
2. Heart Anatomy and Heart Energy
3. Standard Cardiac Technologies
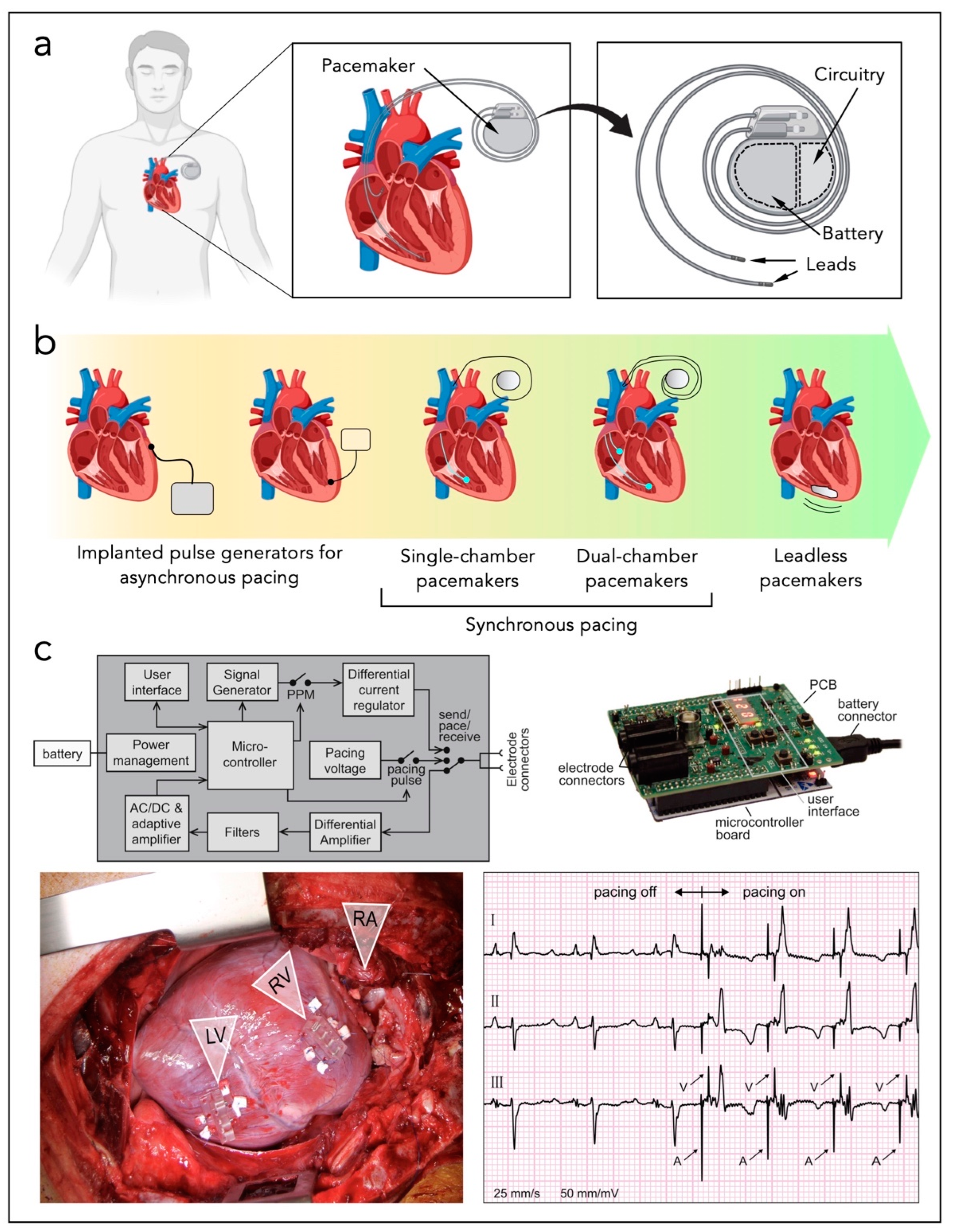
4. Micro-Devices for Heart Energy Harvesting
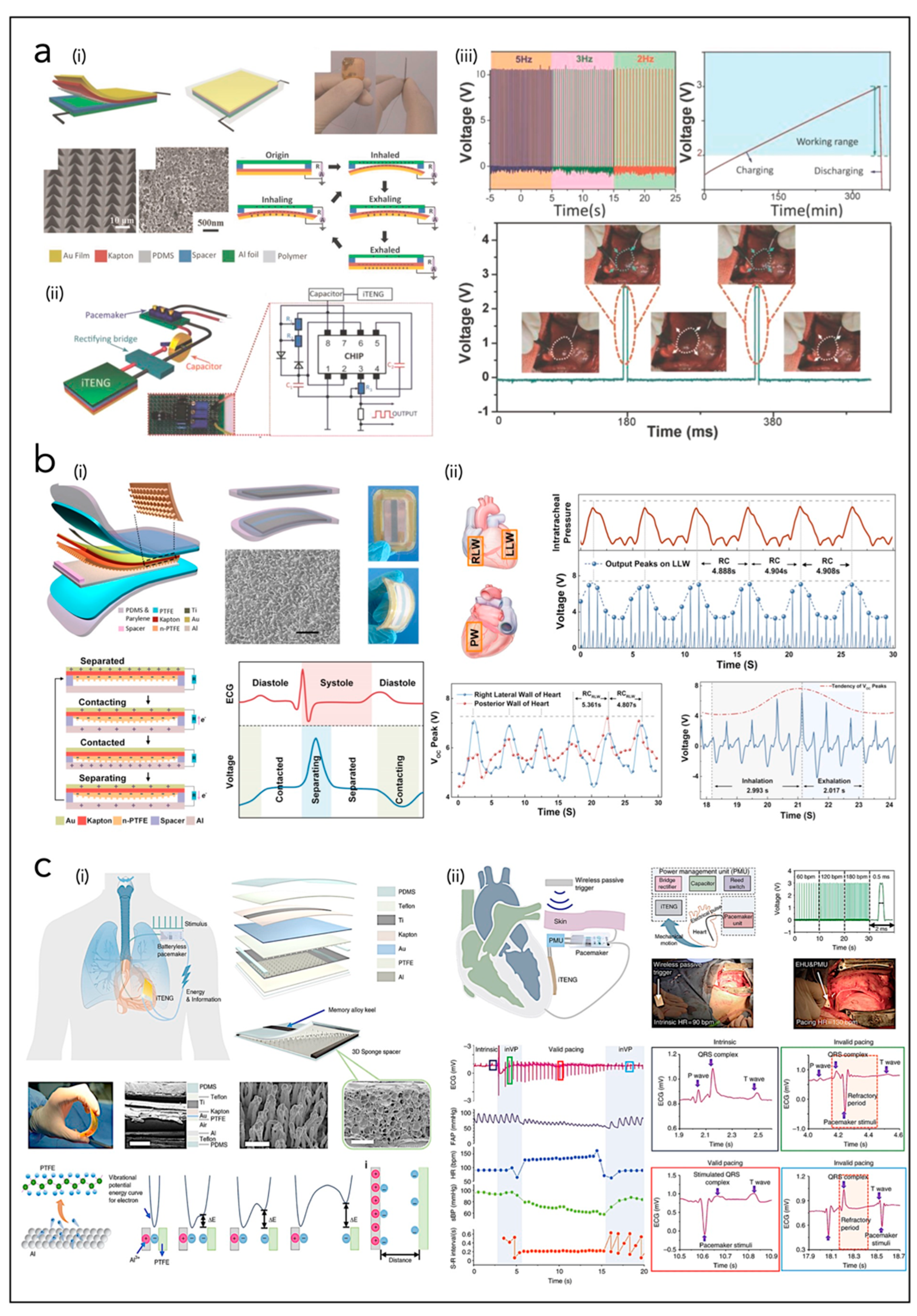
4.1. Piezoelectric Nanogenerators (PNGs)
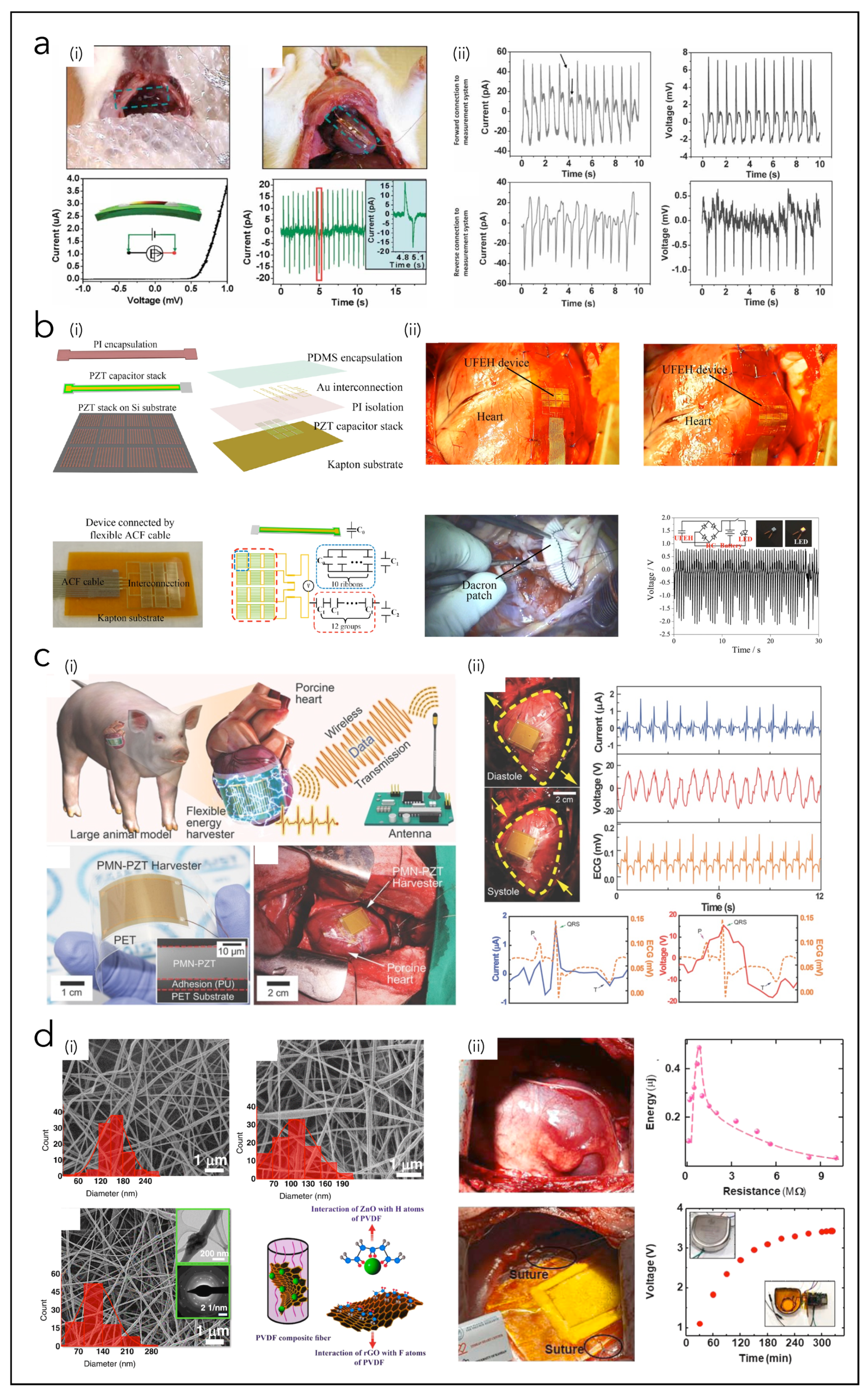
4.2. Triboelectric Nanogenerators (TNGs)
4.3. Hybrid Piezo/Triboelectric Nanogenerators (HNGs)
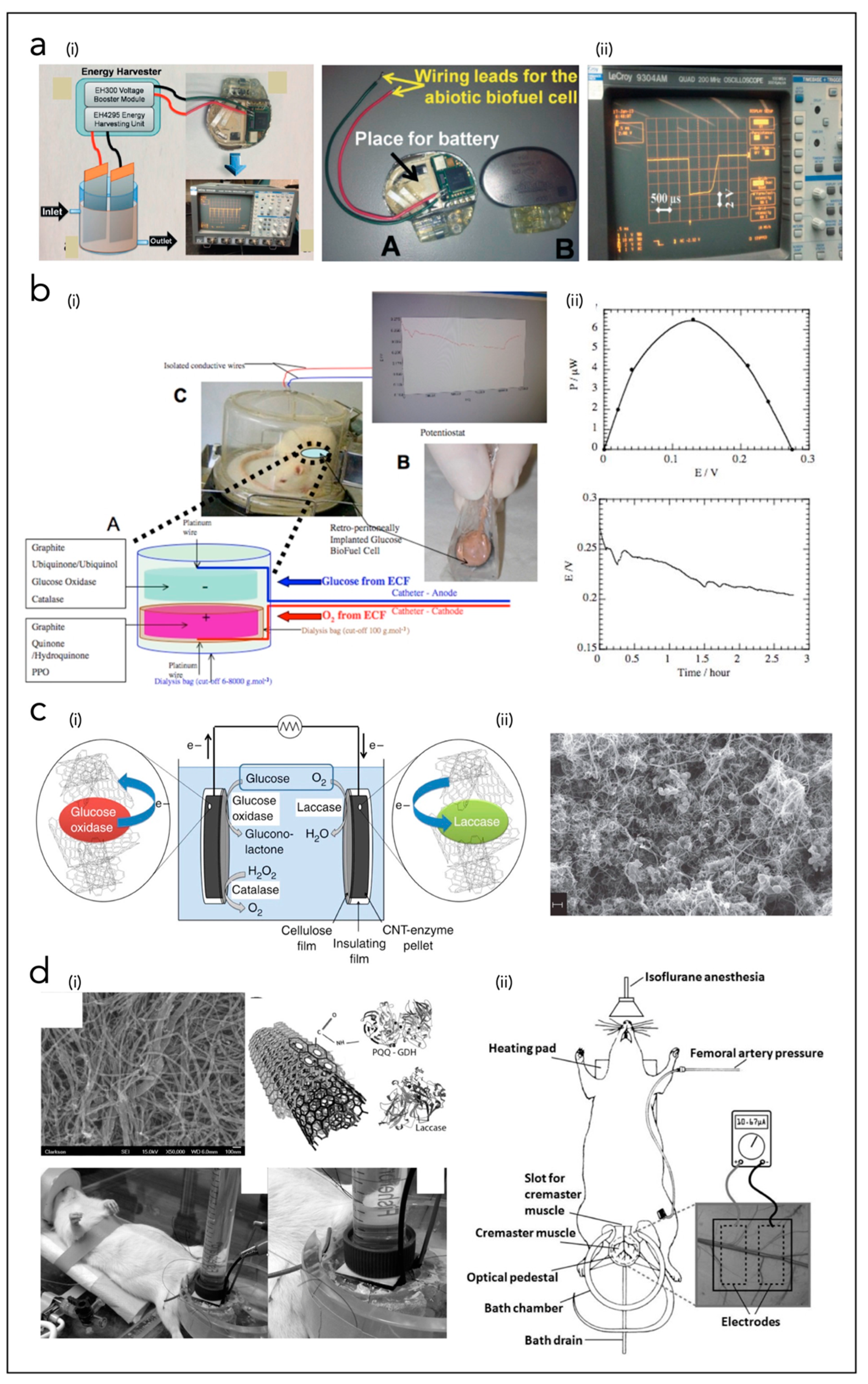
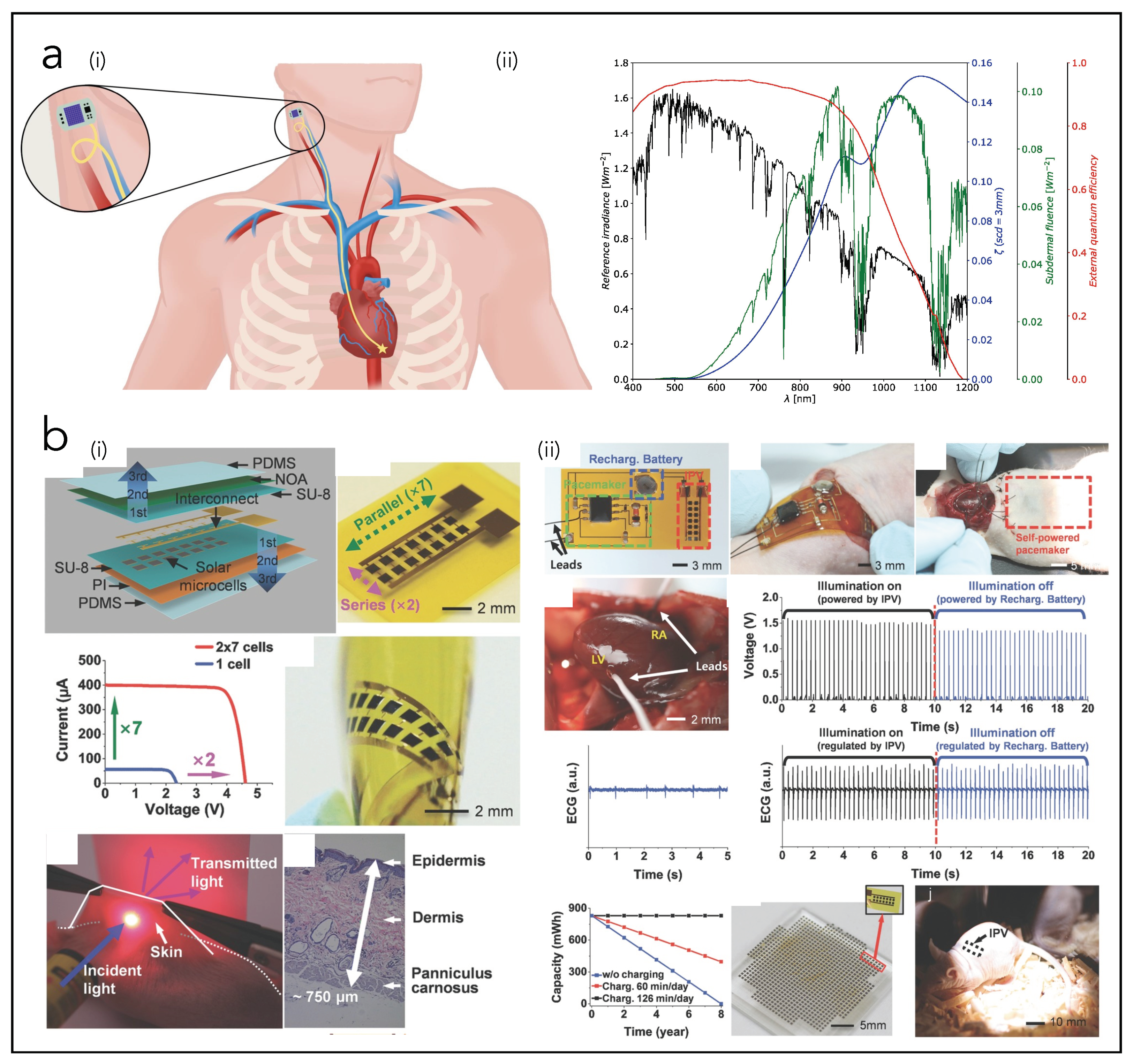
4.4. Solar Cells (SCs)
4.5. Biofuel Cells (BFCs)
5. Bioelectronic Systems with Cardiac Interfaces
5.1. In Vivo Epicardial Systems
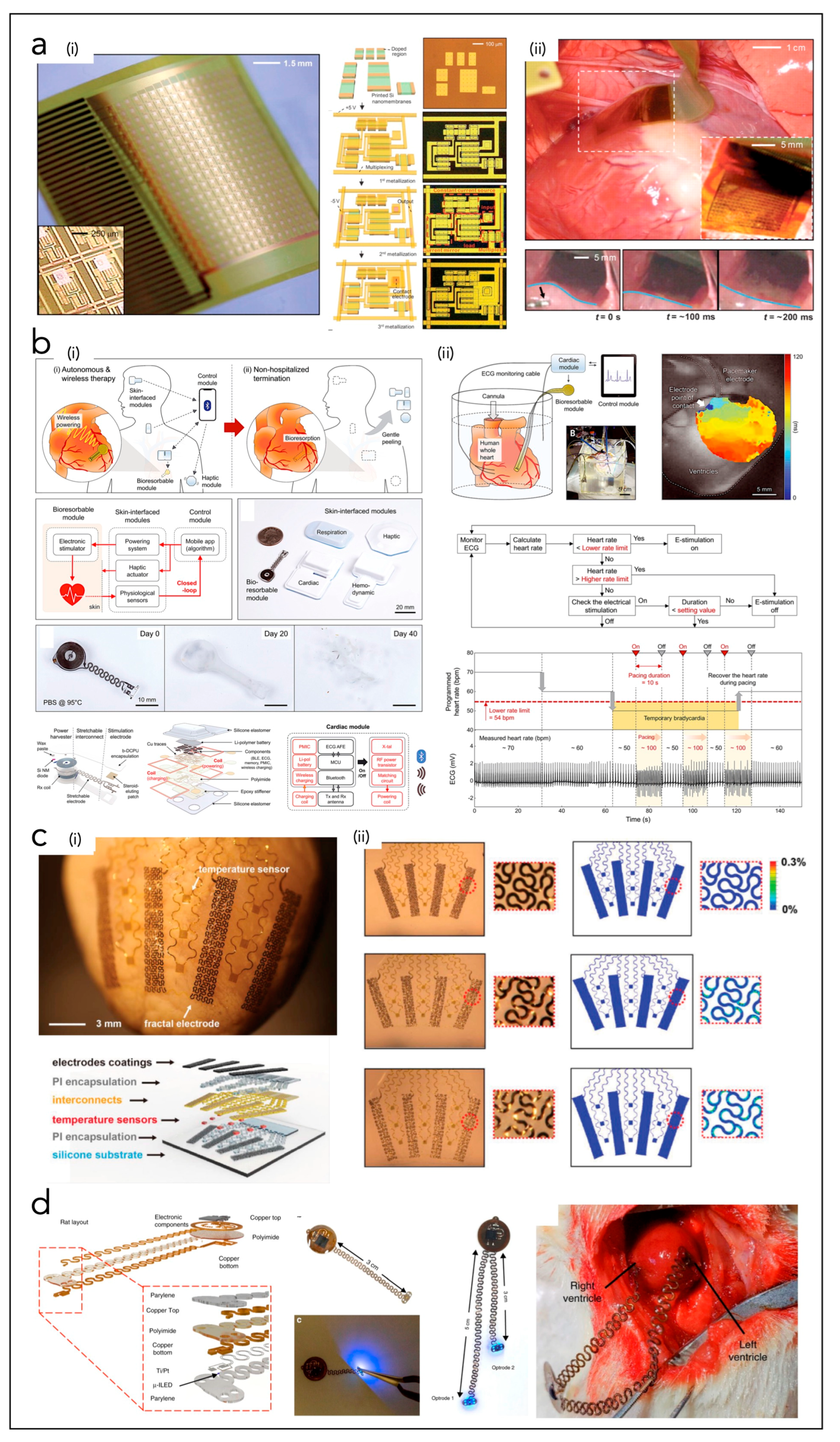
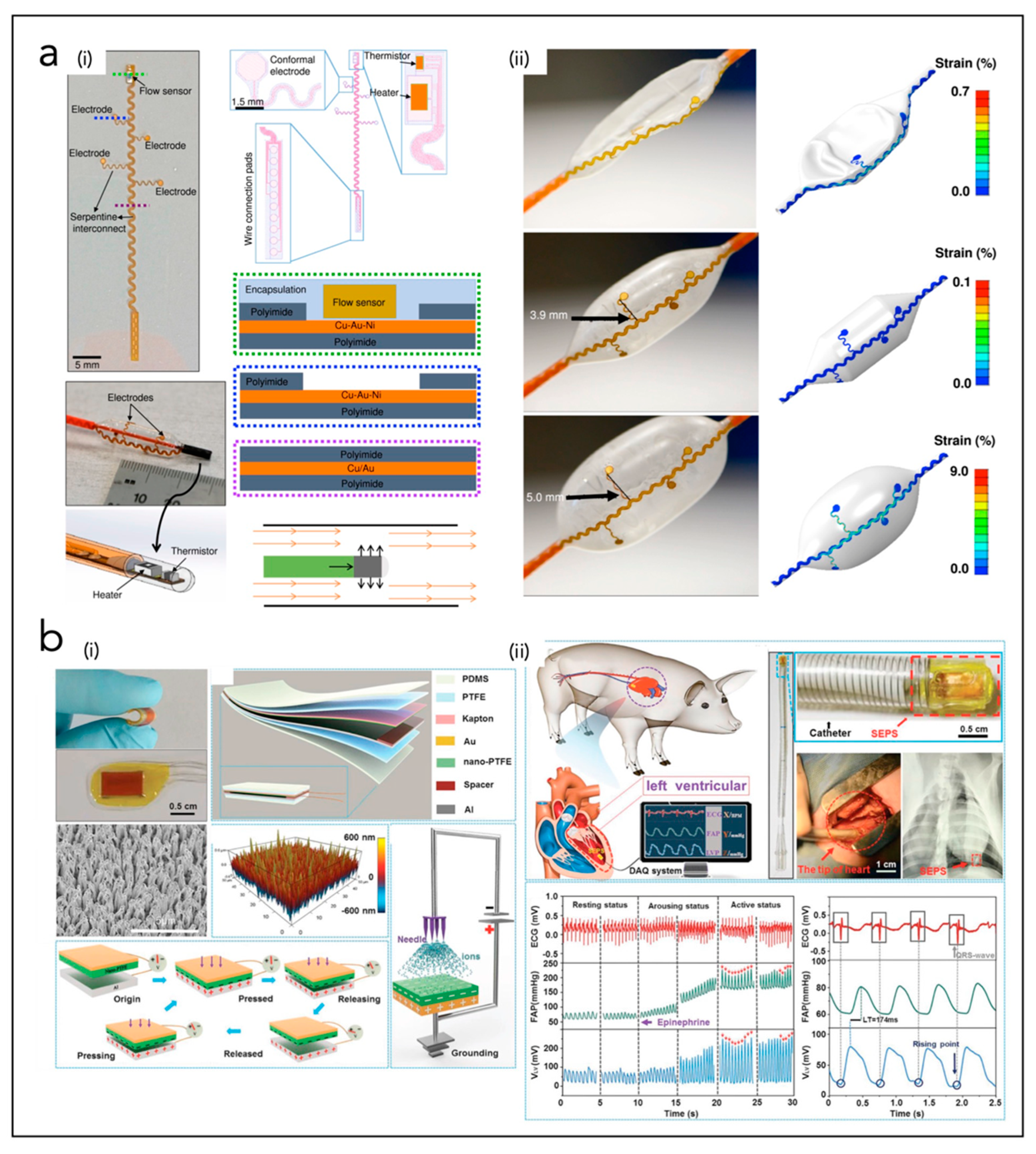
5.2. In Vivo Endocardial Systems
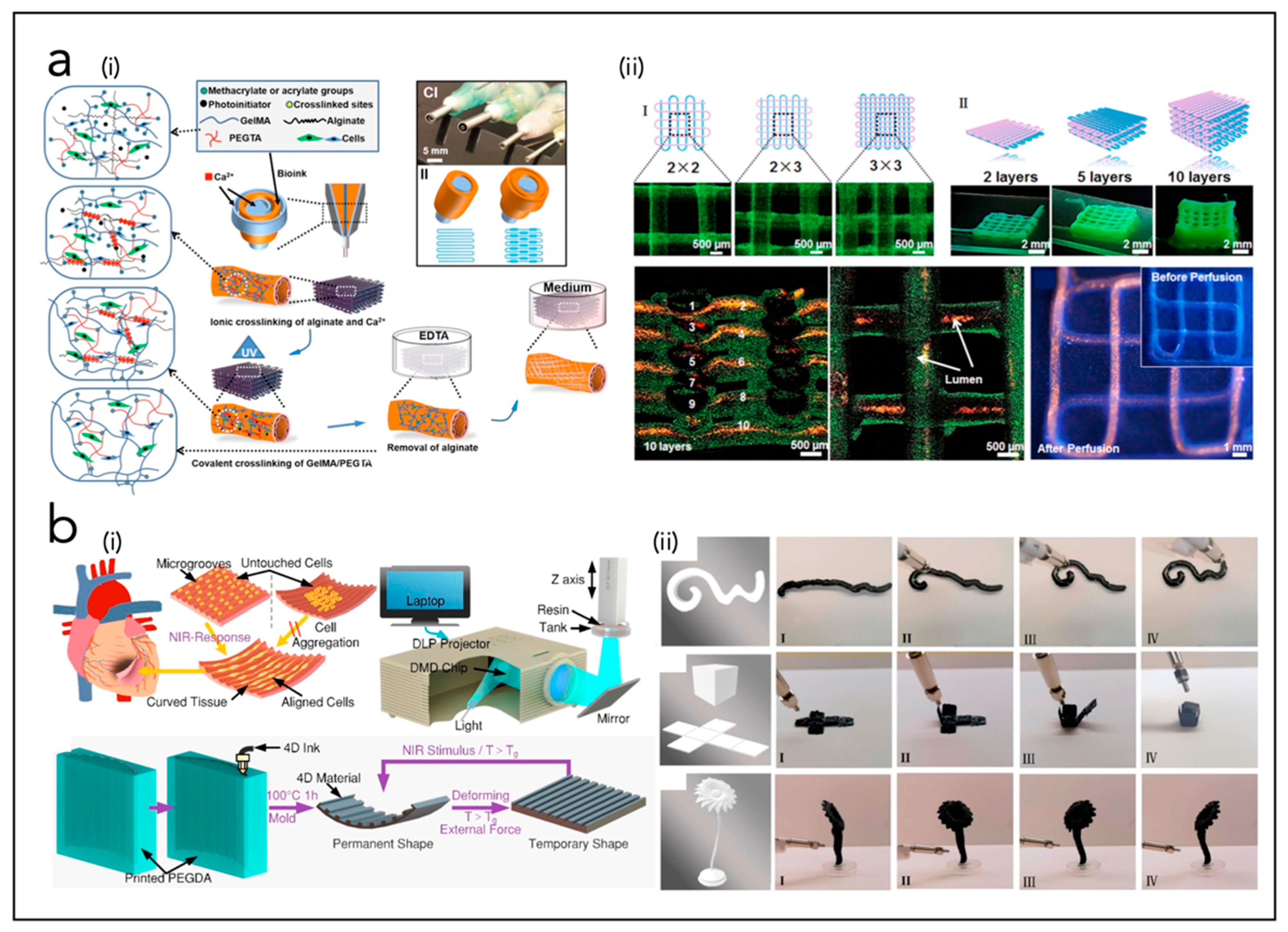
5.3. In Vitro Systems for Cardiac Patch
5.4. In Vitro Systems for Cardiac Organoids
6. Bioprinting of Cardiac Tissue
| Ref. | Type of Device | Materials | Output Performances/Working Principle | Application Field |
|---|---|---|---|---|
| Dagdeviren et al. [257] | PNG | PZT/Pt/Ti/SiO2 Ti/Pt, Cr/Au electrodes | VOC = 1 mV; ISC = 1 pA E = 0.001 pJ | EH from heart motion in rabbits |
| Xie et al. [258] | PNG | PMN-Pt | VOC = 3.2 V; ISC = 54 nA | EH from heart motion in rats |
| Hwang et al. [259] | PNG | PZT, Au, PET | VOC = 8.2 mV; ISC = 0.223 mA E = 1.8286 µJ | EH from heart motion in rats |
| Lu et al. [113] | PNG | PZT, Ti/Pt, SiO2, Si, polyimide | VOC = 3 V | EH from heart motion in pigs |
| Kim et al. [114] | PNG | PZT, PU, PET | VOC = 17.8 V; ISC = 1.74 µA E = 30.972 µJ | EH from heart motion in pigs |
| Li et al. [116] | PNG | PZT, Cr/Au, Be, PDMS | VOC = 20 V; ISC = 15 µA E = 300 µJ | EH from heart motion in pigs |
| Zhang et al. [115] | PNG | PVDF, Al | VOC = 1.5 mV; ISC = 0.3 mA E = 0.45 µJ | EH from vases in pigs |
| Li et al. [260] | PNG | ZnO, polyimide | VOC = 3 mV; ISC = 30 pA E = 0.06 pJ | EH from heart motion in rats |
| Dong et al. [118] | PNG | Cu, Au, Kapton, PVDF, porous PVDF | VOC = 0.5 V; ISC = 43 nA | EH from pacemaker leads |
| Azimi et al. [119] | PNG | PVDF-TrFE NFs, ZnO, rGO | E = 0.487 µJ | EH from heartbeat in dogs |
| Hyun Kim et al. [114] | PNG | PMN-PZT | VOC = 17.8 V; ISC = 1.74 µA | EH from heartbeat in pigs |
| Zheng et al. [33] | TNG | PTFE, Kapton, Au, Al | VOC = 14 V; ISC = 5 µA E = 70 µJ | EH from heart motion in pigs |
| Liu et al. [218] | TNG | PTFE, PDMS, Kapton, Au, Al | VOC = 0.008 V | EH from heart motion in pigs |
| Ouyang et al. [158] | TNG | PDMS, Ti, Kapton, Au, PTFE, Al | VOC = 65.2 V; ISC = 0.5 µA E = 32.6 µJ | EH from heart motion in pigs |
| Zheng et al. [156] | TNG | PDMS, Kapton, Au | VOC = 3.75 V; ISC = 0.14 µA E = 0.525 µJ | EH from breath in rats |
| Ryu et al. [160] | TNG | PVA-NH2, PFA | VOC = 3.75 V p = 4.9 µW/cm3 | EH from body motion in mongrels |
| Ma et al. [157] | TNG | PTFE, Kapton, Au | VOC = 10 V; ISC = 4 µA E = 40 µJ | EH from heat motion in pigs |
| Haeberlin et al. [44] | SC | Commercial modules | p = 1963 µW/cm2 (direct outdoor sunlight) p = 206 µW/cm2 (shade outdoors) p = 4 µW/cm2 (indoors) | EH from solar light to power pacemaker in pigs |
| Prominski et al. [174] | SC | Si NMs | - | Harvesting solar light for cardiac modulation in rats |
| Dan Tchin-Iou et al. [45] | SC | - | VOC = 4.5 V; ISC = 23 mA p = 80.6 mW | EH from solar light to charge artificial heart |
| Song et al. [171] | SC | PDMS, Polyimide, SU8, NOA, GaInP/GaAs | p = 647 µW | EH from solar light to power pacemaker in mice |
| Holade et al. [46] | BFC | AuxPty NPs, carbon black (CB), buckypaper | VOC = 0.35 V; ISC = 0.65 mAcm−2 p = 104 µW | EH from glucose oxidation to activate a pacemaker |
| Southcott et al. [47] | BFC | Buckypaper, CNTs, laccase PQQ-dependent glucose dehydrogenase | VOC = 0.47 V; ISC = 0.83 mAcm−2 | EH from glucose oxidation to activate a pacemaker |
| MacVittie et al. [191] | BFC | Buckypaper, CNTs, laccase PQQ-dependent glucose dehydrogenase | VOC = 3 V | EH from lobsters to power a pacemaker |
| Choi et al. [200] | BP/CLS | Si NM, Mo, b-DCPU, PLGA, elastomer | Transient closed-loop system for cardiac electrotherapy | |
| Viventi et al. [197] | Epi-CS | Si NM, PI | High-performance, conformal bioelectronic system used to measure cardiac electrophysiological activity | |
| Fang et al. [198] | Epi-CS | Si NM; SiO2 | Hermetically sealed array of 396 multiplexed capacitive sensors based on Si NM transistors | |
| Choi et al. [199] | Epi-CS | PLGA, W/Mg, SI NM diodes | Fully implantable and bioresorbable, leadless, battery-free and externally programmable cardiac pacemaker for the treatment of atrioventricular (AV) nodal heart block. | |
| Kim et al. [201] | Epi-CS | PI, PMMA, Cr/Au, silk, Ti/Pt | Stretchable sensors and actuators based on epicardial webs that can conformally wrap around the heart | |
| Xu et al. [202] | Epi-CS | Au, Si or INGaN, PI, PMMA, PET, PDMS | 3D multifunctional integumentary thin elastic membrane integrating 68 electrodes and custom-formed to envelope completely and conformally the heart, | |
| Xu et al. [203] | Epi-CS | Pt-Ir allow, PEDOT:PSS | Compliant, large-area, low-impedance electrodes for electrical stimulation and cardiac electrotherapy | |
| Lee et al. [204] | Epi-CS | Parylene, Au, PMC3A, PEDOT:PSS | Ultrathin, stretchable grid-patterned active OECT matrix directly attached to the surface of a rat’s beating heart. | |
| Park et al. [205] | Epi-CS | AgNWs, SBS rubber | Epicardial mesh for electromechanical cardioplasty and electrotherapy | |
| Choi et al. [206] | Epi-CS | Ag-Au, elastomers | 2D fan-shaped implantable devices with 42 multichannel electrodes and based on nanocomposites applied on the swine heart for continuous electrophysiological recording | |
| Gutruf et al. [207] | Epi-CS | Parylene, Ti/Pt, Cu, PI | Miniaturized wireless energy-harvesting and digital communication electronics implanted subdermally to provide pacing in rats | |
| Sim et al. [208] | Epi-CS | PDMS, AgNWs/PDMS, Ion gel, P3HT-NFs/PDMS | Epicardial bioelectronic stretchable patch with cardiac tissue-like softness, for spatiotemporal mapping of electrophysiological activity, strain/temperature sensing and electrical pacing on a beating porcine heart. | |
| Liu et al. [209] | Epi-CS | PEDOT:PSS, fluorinated elastomers | Fully elastic electrode with tissue-like softness with 64 electrodes (elastrode) | |
| Klinker et al. [215] | Endo-CS | PI, Cu/Au/Ni | Smart balloon catheter system containing stretchable electrodes and thermal-based blood flow sensors | |
| Kim et al. [201,216] | Endo-CS | PI, PMMA, Cr/Au, silk, Ti/Pt | Array of integrated stretchable RF electrodes and multimodal (temperature, flow, tactile) sensors on a balloon catheter | |
| Han et al. [217] | Endo-CS | Cr/Au, PI, SiO2, Ti/Cu | Balloon catheter-integrated soft multilayer electronic array for multiplexed sensing and actuation during cardiac surgery | |
| Liu et al. [218] | Endo-CS | PDMS, PTFE, Kapton, Au, Al | Flexible self-powered endocardial pressure sensor for real-time electrophysiology monitoring. | |
| Liu et al. [224] | CP | Si NWs, SU8, Cr/Pd | 3D interconnected microporous nanoelectronics networks assembled from 2D precursors, integrated with cardiac patch. | |
| Tian et al. [225] | CP | Si NWs, SU-8, alginate, electrospun NFs | Microporous, flexible and free-standing nanoelectronics scaffold based on field-effect transistors and integrated with natural or synthetic biomaterials. | |
| Dai et al. [226] | CP | Si NWs, SiO2/Si, Ni, SU8 meshes, PLGA NFs | Tissue-scaffold-mimicking 3D nanoelectronics array (64 electrodes) for real-time sub-millisecond recording of extracellular action potential in developing rat cardiac tissues | |
| Wang et al. [227] | CP | PI, Au, PVA, Ti, SiO2, Dragon Skin | 3D porous, flexible electronic scaffold formed through compressive buckling of 2D precursor structures, with microelectrodes integrated within thermoresponsive extracellular matrix-based hydrogels | |
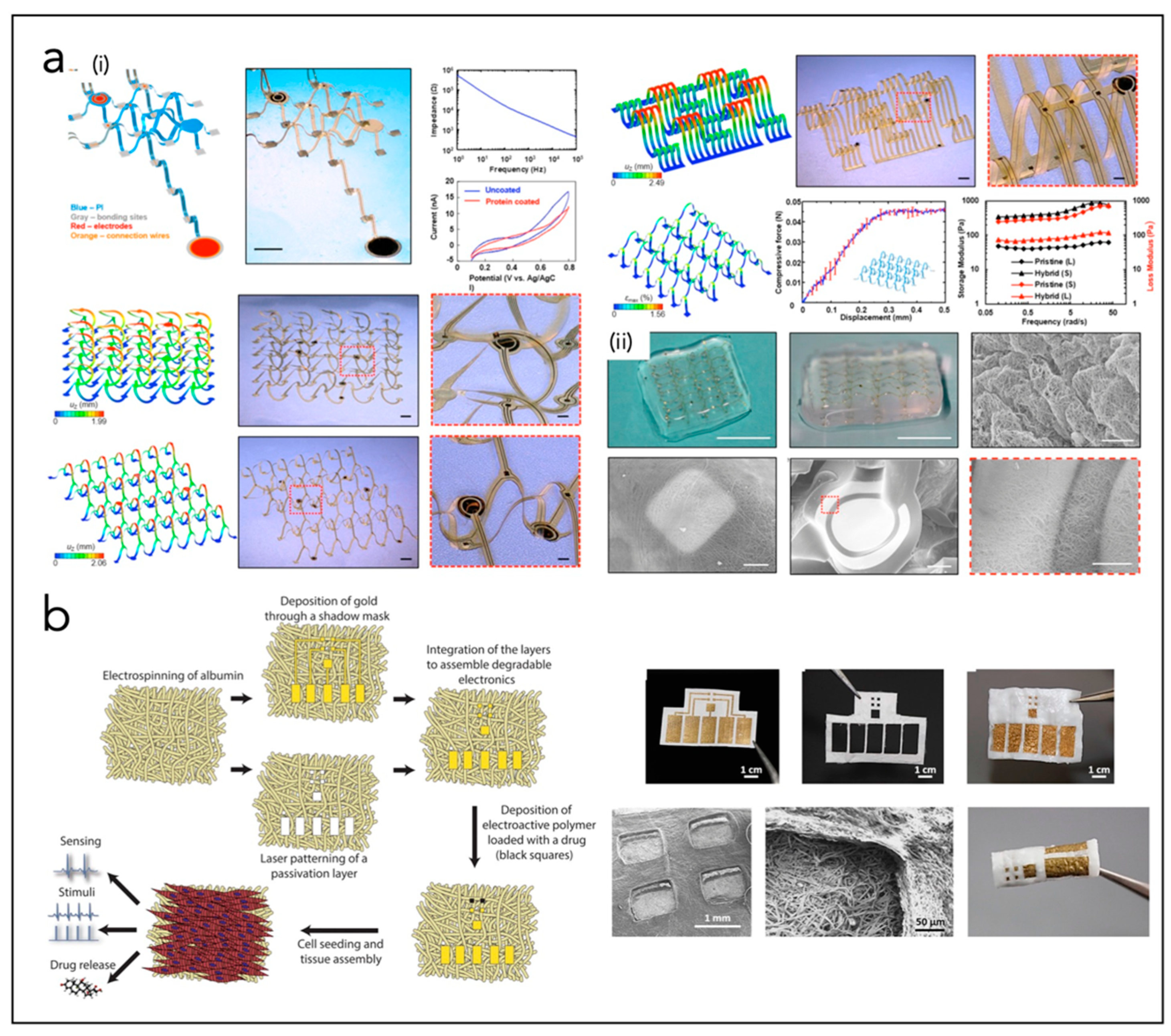
| Feiner et al. [228] | CP | SU8 mesh, Au, TiN, PCL NFs | Flexible, free-standing electronics (32 electrodes) and a 3D nanocomposite scaffold, integrated with a cardiac patch | |
| Feiner et al. [229] | CP | Electrospun albumin fibers, Au | Elastic, biodegradable, electronic scaffolds | |
| Lind et al. [230] | CP | Dextran, TPU, CB, PDMS, Ag | Multimaterial 3D-printed heart-on-chip electronics | |
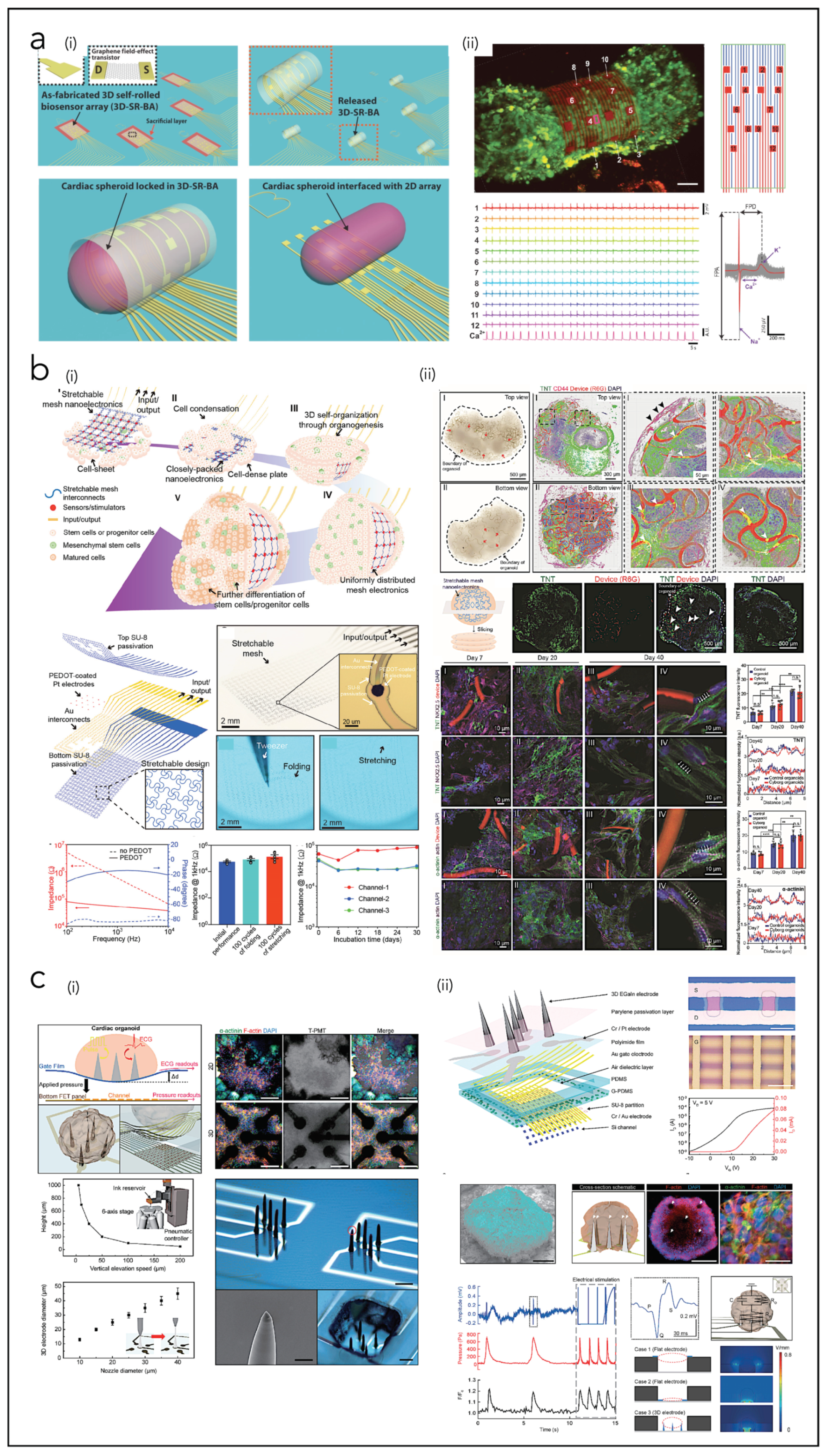
| Kalmykov et al. [239] | CO | SU8, Au, graphene | Flexible organ-on-electronic-chip device based on 3D self-rolled biosensor arrays | |
| Li et al. [240] | CO | SU8, PEDOT, Pt, Au | Cyborg human cardiac organoids based on the 3D assembly of stretchable sub-µm thick mesh nanoelectronics | |
| Kim et al. [241] | CO | EGaIn, Parylene, Cr/Pt, PI, Au, PDMS, SU8, Si | Multimodal mechanophysiological characterization of 3D human cardiac organoids | |
7. Conclusions and Future Challenges
Funding
Conflicts of Interest
References
- Lai, Y.-C.; Hsiao, Y.-C.; Wu, H.-M.; Wang, Z.L. Waterproof Fabric-Based Multifunctional Triboelectric Nanogenerator for Universally Harvesting Energy from Raindrops, Wind, and Human Motions and as Self-Powered Sensors. Adv. Sci. 2019, 6, 1801883. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Li, X.; Yin, J.; Xu, Y.; Fei, W.; Xue, M.; Wang, Q.; Zhou, J.; Guo, W. Emerging hydrovoltaic technology. Nat. Nanotechnol. 2018, 13, 1109–1119. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Wang, X.; Wang, Z.L. Microfibre–nanowire hybrid structure for energy scavenging. Nature 2008, 451, 809–813. [Google Scholar] [CrossRef]
- Wang, Z.L.; Song, J. Piezoelectric Nanogenerators Based on Zinc Oxide Nanowire Arrays. Science 2006, 312, 242–246. [Google Scholar] [CrossRef]
- Wang, Z.L. Catch wave power in floating nets. Nature 2017, 542, 159–160. [Google Scholar] [CrossRef]
- Wang, Z.L. Self-Powered Nanosensors and Nanosystems. Adv. Mater. 2012, 24, 280–285. [Google Scholar] [CrossRef]
- Wu, Z.; Cheng, T.; Wang, Z.L. Self-Powered Sensors and Systems Based on Nanogenerators. Sensors 2020, 20, 2925. [Google Scholar] [CrossRef]
- Wang, Z.L. Energy harvesting for self-powered nanosystems. Nano Res. 2008, 1, 1–8. [Google Scholar] [CrossRef]
- Pu, X.; Li, L.; Liu, M.; Jiang, C.; Du, C.; Zhao, Z.; Hu, W.; Wang, Z.L. Wearable Self-Charging Power Textile Based on Flexible Yarn Supercapacitors and Fabric Nanogenerators. Adv. Mater. 2015, 28, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Sim, H.J.; Choi, C.; Kim, S.H.; Kim, K.M.; Lee, C.J.; Kim, Y.T.; Lepró, X.; Baughman, R.H.; Kim, S.J. Stretchable Triboelectric Fiber for Self-powered Kinematic Sensing Textile. Sci. Rep. 2016, 6, 35153. [Google Scholar] [CrossRef]
- Nielsen, M. Batteryless Pacemaker Could Use Heart’s Energy for Power; AIP Publishing LLC: Melville, NY, USA, 2021. [Google Scholar]
- Sun, M.; Li, Z.; Yang, C.; Lv, Y.; Yuan, L.; Shang, C.; Liang, S.; Guo, B.; Liu, Y.; Li, Z.; et al. Nanogenerator-based devices for biomedical applications. Nano Energy 2021, 89, 106461. [Google Scholar] [CrossRef]
- Helseth, L.E.; Guo, X.D. Contact Electrification and Energy Harvesting Using Periodically Contacted and Squeezed Water Droplets. Langmuir 2015, 31, 3269–3276. [Google Scholar] [CrossRef] [PubMed]
- Mariello, M.; Guido, F.; Mastronardi, V.M.; Madaro, F.; Mehdipour, I.; Todaro, M.T.; Rizzi, F.; De Vittorio, M. Chapter 13—Micro- and Nanodevices for Wind Energy Harvesting. In Nano Tools and Devices for Enhanced Renewable Energy; Devasahayam, S., Hussain, C.M., Eds.; Micro and Nano Technologies; Elsevier: Newark, NJ, USA, 2021; pp. 291–374. ISBN 978-0-12-821709-2. [Google Scholar]
- Mariello, M.; Guido, F.; Mastronardi, V.; Todaro, M.; Desmaële, D.; De Vittorio, M. Nanogenerators for harvesting mechanical energy conveyed by liquids. Nano Energy 2018, 57, 141–156. [Google Scholar] [CrossRef]
- Zhang, Z.; Du, K.; Chen, X.; Xue, C.; Wang, K. An air-cushion triboelectric nanogenerator integrated with stretchable electrode for human-motion energy harvesting and monitoring. Nano Energy 2018, 53, 108–115. [Google Scholar] [CrossRef]
- Mitcheson, P.D.; Yeatman, E.M.; Rao, G.K.; Holmes, A.S.; Green, T.C. Energy Harvesting from Human and Machine Motion for Wireless Electronic Devices. Proc. IEEE 2008, 96, 1457–1486. [Google Scholar] [CrossRef]
- Guido, F.; Qualtieri, A.; Algieri, L.; Lemma, E.D.; De Vittorio, M.; Todaro, M.T. AlN-based flexible piezoelectric skin for energy harvesting from human motion. Microelectron. Eng. 2016, 159, 174–178. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, H.; Lin, Z.-H.; Zhou, Y.S.; Jing, Q.; Su, Y.; Yang, J.; Chen, J.; Hu, C.; Wang, Z.L. Human Skin Based Triboelectric Nanogenerators for Harvesting Biomechanical Energy and as Self-Powered Active Tactile Sensor System. ACS Nano 2013, 7, 9213–9222. [Google Scholar] [CrossRef]
- Shi, Q.; Wang, H.; Wu, H.; Lee, C. Self-powered triboelectric nanogenerator buoy ball for applications ranging from environment monitoring to water wave energy farm. Nano Energy 2017, 40, 203–213. [Google Scholar] [CrossRef]
- Olsen, M.; Zhang, R.; Örtegren, J.; Andersson, H.; Yang, Y.; Olin, H. Frequency and voltage response of a wind-driven fluttering triboelectric nanogenerator. Sci. Rep. 2019, 9, 5543. [Google Scholar] [CrossRef]
- Shi, Q.; Zhang, Z.; Chen, T.; Lee, C. Minimalist and multi-functional human machine interface (HMI) using a flexible wearable triboelectric patch. Nano Energy 2019, 62, 355–366. [Google Scholar] [CrossRef]
- O’Flynn, B.; Sanchez-Torres, J.; Tedesco, S.; Walsh, M. Challenges in the Development of Wearable Human Machine Interface Systems. In Proceedings of the 2019 IEEE International Electron Devices Meeting (IEDM), San Francisco, CA, USA, 7–11 December 2019; pp. 10.4.1–10.4.4. [Google Scholar]
- Sun, Z.; Zhu, M.; Lee, C. Progress in the Triboelectric Human–Machine Interfaces (HMIs)-Moving from Smart Gloves to AI/Haptic Enabled HMI in the 5G/IoT Era. Nanoenergy Adv. 2021, 1, 81–120. [Google Scholar] [CrossRef]
- Gerratt, A.P.; Michaud, H.O.; Lacour, S.P. Elastomeric Electronic Skin for Prosthetic Tactile Sensation. Adv. Funct. Mater. 2015, 25, 2287–2295. [Google Scholar] [CrossRef]
- Brunelli, D.; Tadesse, A.M.; Vodermayer, B.; Nowak, M.; Castellini, C. Low-Cost Wearable Multichannel Surface EMG Acquisition for Prosthetic Hand Control. In Proceedings of the 2015 6th International Workshop on Advances in Sensors and Interfaces (IWASI), Gallipoli, Italy, 18–19 June 2015; pp. 94–99. [Google Scholar]
- Akaydin, H.D.; Elvin, N.; Andreopoulos, Y. Energy Harvesting from Highly Unsteady Fluid Flows using Piezoelectric Materials. J. Intell. Mater. Syst. Struct. 2010, 21, 1263–1278. [Google Scholar] [CrossRef]
- Dagdeviren, C.; Joe, P.; Tuzman, O.L.; Park, K.-I.; Lee, K.J.; Shi, Y.; Huang, Y.; Rogers, J.A. Recent progress in flexible and stretchable piezoelectric devices for mechanical energy harvesting, sensing and actuation. Extreme Mech. Lett. 2016, 9, 269–281. [Google Scholar] [CrossRef]
- Choi, A.Y.; Lee, C.J.; Park, J.; Kim, D.; Kim, Y.T. Corrugated Textile based Triboelectric Generator for Wearable Energy Harvesting. Sci. Rep. 2017, 7, 45583. [Google Scholar] [CrossRef]
- Chen, X.; Shao, J.; An, N.; Li, X.; Tian, H.; Xu, C.; Ding, Y. Self-powered flexible pressure sensors with vertically well-aligned piezoelectric nanowire arrays for monitoring vital signs. J. Mater. Chem. C 2015, 3, 11806–11814. [Google Scholar] [CrossRef]
- Zhang, X.-S.; Han, M.-D.; Wang, R.-X.; Zhu, F.-Y.; Li, Z.-H.; Wang, W.; Zhang, H.-X. Frequency-Multiplication High-Output Triboelectric Nanogenerator for Sustainably Powering Biomedical Microsystems. Nano Lett. 2013, 13, 1168–1172. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.-Y.; Lee, W.-H.; Hwang, H.-G.; Kim, D.-H.; Kim, J.-H.; Lee, S.-H.; Nahm, S. Resistive Switching Memory Integrated with Nanogenerator for Self-Powered Bioimplantable Devices. Adv. Funct. Mater. 2016, 26, 5211–5221. [Google Scholar] [CrossRef]
- Zheng, Q.; Zhang, H.; Shi, B.; Xue, X.; Liu, Z.; Jin, Y.; Ma, Y.; Zou, Y.; Wang, X.; An, Z.; et al. In Vivo Self-Powered Wireless Cardiac Monitoring via Implantable Triboelectric Nanogenerator. ACS Nano 2016, 10, 6510–6518. [Google Scholar] [CrossRef]
- Gu, Y.; Zhang, T.; Chen, H.; Wang, F.; Pu, Y.; Gao, C.; Li, S. Mini Review on Flexible and Wearable Electronics for Monitoring Human Health Information. Nanoscale Res. Lett. 2019, 14, 263. [Google Scholar] [CrossRef]
- Fan, F.R.; Tang, W.; Wang, Z.L. Flexible Nanogenerators for Energy Harvesting and Self-Powered Electronics. Adv. Mater. 2016, 28, 4283–4305. [Google Scholar] [CrossRef] [PubMed]
- Khan, U.; Kim, S.-W. Triboelectric Nanogenerators for Blue Energy Harvesting. ACS Nano 2016, 10, 6429–6432. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-H.; Lee, K.Y.; Kumar, B.; Tien, N.T.; Lee, N.-E.; Kim, S.-W. Highly sensitive stretchable transparent piezoelectric nanogenerators. Energy Environ. Sci. 2012, 6, 169–175. [Google Scholar] [CrossRef]
- Mariello, M.; Scarpa, E.; Algieri, L.; Guido, F.; Mastronardi, V.M.; Qualtieri, A.; De Vittorio, M. Mechanical energy harvesting through a novel flexible contact-separation mode triboelectric nanogenerator based on metallized porous PDM and Parylene-C. In Proceedings of the 19th International Conference on Micro and Nanotechnology for Power Generation and Energy Conversion Applications (PowerMEMS), Krakow, Poland, 2–6 December 2019. [Google Scholar] [CrossRef]
- Wang, A.; Wu, C.; Pisignano, D.; Wang, Z.L.; Persano, L. Polymer nanogenerators: Opportunities and challenges for large-scale applications. J. Appl. Polym. Sci. 2018, 135, 45674. [Google Scholar] [CrossRef]
- Todaro, M.T.; Guido, F.; Algieri, L.; Mastronardi, V.M.; Desmaele, D.; Epifani, G.; De Vittorio, M. Biocompatible, Flexible, and Compliant Energy Harvesters Based on Piezoelectric Thin Films. IEEE Trans. Nanotechnol. 2018, 17, 220–230. [Google Scholar] [CrossRef]
- Ryu, H.; Yoon, H.; Kim, S. Hybrid Energy Harvesters: Toward Sustainable Energy Harvesting. Adv. Mater. 2019, 31, e1802898. [Google Scholar] [CrossRef] [PubMed]
- Mariello, M. Recent Advances on Hybrid Piezo-Triboelectric Bio-Nanogenerators: Materials, Architectures and Circuitry. Nanoenergy Adv. 2022, 2, 64–109. [Google Scholar] [CrossRef]
- Mariello, M.; Fachechi, L.; Guido, F.; De Vittorio, M. Multifunctional sub-100 µm thickness flexible piezo/triboelectric hybrid water energy harvester based on biocompatible AlN and soft parylene C-PDMS-Ecoflex™. Nano Energy 2021, 83, 105811. [Google Scholar] [CrossRef]
- Haeberlin, A.; Zurbuchen, A.; Walpen, S.; Schaerer, J.; Niederhauser, T.; Huber, C.; Tanner, H.; Servatius, H.; Seiler, J.; Haeberlin, H.; et al. The first batteryless, solar-powered cardiac pacemaker. Heart Rhythm 2015, 12, 1317–1323. [Google Scholar] [CrossRef]
- Tchin-Iou, A.D.; Min, B. Design of the Solar Cell System for Recharging the External Battery of the Totally-Implantable Artificial Heart. Int. J. Artif. Organs 1999, 22, 823–826. [Google Scholar] [CrossRef]
- Holade, Y.; MacVittie, K.; Conlon, T.; Guz, N.; Servat, K.; Napporn, T.W.; Kokoh, K.B.; Katz, E. Pacemaker Activated by an Abiotic Biofuel Cell Operated in Human Serum Solution. Electroanalysis 2014, 26, 2445–2457. [Google Scholar] [CrossRef]
- Southcott, M.; MacVittie, K.; Halámek, J.; Halámková, L.; Jemison, W.D.; Lobel, R.; Katz, E. A pacemaker powered by an implantable biofuel cell operating under conditions mimicking the human blood circulatory system—Battery not included. Phys. Chem. Chem. Phys. 2013, 15, 6278–6283. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Boudreaux, B.; Chaturvedi, R.; Romanosky, S.; Downing, B. The Internet of Bodies: Opportunities, Risks, and Governance; RAND Corporation: Santa Monica, CA, USA, 2020. [Google Scholar]
- Al-Suhaimi, E.A.; Aljafary, M.A.; Alfareed, T.M.; Alshuyeh, H.A.; Alhamid, G.M.; Sonbol, B.; Almofleh, A.; Alkulaifi, F.M.; Altwayan, R.K.; Alharbi, J.N.; et al. Nanogenerator-Based Sensors for Energy Harvesting from Cardiac Contraction. Front. Energy Res. 2022, 10, 900534. [Google Scholar] [CrossRef]
- Muñoz, H.R.; Sacco, C.M. Cardiac mechanical energy and effects on the arterial tree. J. Cardiothorac. Vasc. Anesth. 1997, 11, 289–298. [Google Scholar] [CrossRef]
- Papp, L. Isn’t the heart the source of energy for blood circulation? “The heart doesn’t know the basic laws of physics”. Orv. Hetil. 2008, 149, 1443–1447. [Google Scholar] [CrossRef]
- Thudichum, I.L.W. On the Cause of the Emptiness of the Arteries after Death. Assoc. Med. J. 1855, 3, 122–127. [Google Scholar] [CrossRef]
- Manteuffel-Szoege, L. Energy Sources of Blood Circulation and the Mechanical Action of the Heart. Thorax 1960, 15, 47–53. [Google Scholar] [CrossRef]
- Furst, B. Hemodynamics of the Early Embryo Circulation. In The Heart and Circulation: An Integrative Model; Furst, B., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 21–29. ISBN 978-3-030-25062-1. [Google Scholar]
- Li, Z.; Pollack, G.H. Surface-induced flow: A natural microscopic engine using infrared energy as fuel. Sci. Adv. 2020, 6, eaba0941. [Google Scholar] [CrossRef]
- Moreno, A.; Walton, R.; Constantin, M.; Bernus, O.; Vigmond, E.J.; Bayer, J.D. Wide-area low-energy surface stimulation of large mammalian ventricular tissue. Sci. Rep. 2019, 9, 15863. [Google Scholar] [CrossRef]
- Zurbuchen, A.; Pfenniger, A.; Stahel, A.; Stoeck, C.T.; Vandenberghe, S.; Koch, V.M.; Vogel, R. Energy Harvesting from the Beating Heart by a Mass Imbalance Oscillation Generator. Ann. Biomed. Eng. 2013, 41, 131–141. [Google Scholar] [CrossRef]
- Moerke, C.; Wolff, A.; Ince, H.; Ortak, J.; Öner, A. New strategies for energy supply of cardiac implantable devices. Herzschrittmachertherapie Elektrophysiologie 2022, 33, 224–231. [Google Scholar] [CrossRef]
- Aquilina, O. A brief history of cardiac pacing. Images Paediatr. Cardiol. 2006, 8, 17–81. [Google Scholar] [PubMed]
- Van Hemel, N.M.; Van Der Wall, E.E. 8 October 1958, D Day for the implantable pacemaker. Neth. Heart J. 2008, 16, S3–S4. [Google Scholar] [CrossRef] [PubMed]
- Parsonnet, V.; Driller, J.; Cook, D.; Rizvi, S.A. Thirty-One Years of Clinical Experience with “Nuclear-Powered” Pacemakers. Pacing Clin. Electrophysiol. 2006, 29, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Mond, H.G.; Freitag, G. The Cardiac Implantable Electronic Device Power Source: Evolution and Revolution. Pacing Clin. Electrophysiol. 2014, 37, 1728–1745. [Google Scholar] [CrossRef] [PubMed]
- Biffi, M.; Bertini, M.; Saporito, D.; Ziacchi, M.; Martignani, C.; Diemberger, I.; Boriani, G. Actual Pacemaker Longevity: The Benefit of Stimulation by Automatic Capture Verification. Pacing Clin. Electrophysiol. 2010, 33, 873–881. [Google Scholar] [CrossRef]
- Untereker, D.F.; Schmidt, C.L.; Jain, G.; Tamirisa, P.A.; Hossick-Schott, J.; Viste, M. 8—Power Sources and Capacitors for Pacemakers and Implantable Cardioverter-Defibrillators. In Clinical Cardiac Pacing, Defibrillation and Resynchronization Therapy, 5th ed.; Ellenbogen, K.A., Wilkoff, B.L., Kay, G.N., Lau, C.-P., Auricchio, A., Eds.; Elsevier: Philadelphia, PA, USA, 2017; pp. 251–269. ISBN 978-0-323-37804-8. [Google Scholar]
- Hauser, R.G.; Casey, S.A.; Gitter, C.B.; Tang, C.Y.; Abdelhadi, R.H.; Gornick, C.C.; Stanberry, L.; Sengupta, J.D. Reliability and longevity of implantable defibrillators. J. Interv. Card. Electrophysiol. 2021, 62, 507–518. [Google Scholar] [CrossRef]
- Hauser, R.G.; Hayes, D.L.; Kallinen, L.M.; Cannom, D.S.; Epstein, A.E.; Almquist, A.K.; Song, S.L.; Tyers, G.F.O.; Vlay, S.C.; Irwin, M. Clinical experience with pacemaker pulse generators and transvenous leads: An 8-year prospective multicenter study. Heart Rhythm. 2007, 4, 154–160. [Google Scholar] [CrossRef]
- Baddour, L.M.; Epstein, A.E.; Erickson, C.C.; Knight, B.P.; Levison, M.E.; Lockhart, P.B.; Masoudi, F.A.; Okum, E.J.; Wilson, W.R.; Beerman, L.B.; et al. Update on Cardiovascular Implantable Electronic Device Infections and Their Management. Circulation 2010, 121, 458–477. [Google Scholar] [CrossRef]
- Basar, N.; Cagli, K.; Basar, O.; Sen, N.; Gurel, O.M.; Akpinar, I.; Ozlu, M.F.; Okten, S. Upper-extremity deep vein thrombosis and downhill esophageal varices caused by long-term pacemaker implantation. Tex. Heart Inst. J. 2010, 37, 714–716. [Google Scholar]
- Miller, M.A.; Neuzil, P.; Dukkipati, S.R.; Reddy, V.Y. Leadless Cardiac Pacemakers: Back to the Future. J. Am. Coll. Cardiol. 2015, 66, 1179–1189. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.Y.; Knops, R.E.; Sperzel, J.; Miller, M.A.; Petru, J.; Simon, J.; Sediva, L.; De Groot, J.R.; Tjong, F.V.; Jacobson, P.; et al. Permanent Leadless Cardiac Pacing: Results of the LEADLESS Trial. Circulation 2014, 129, 1466–1471. [Google Scholar] [CrossRef] [PubMed]
- Piccini, J.P.; Stromberg, K.; Jackson, K.P.; Laager, V.; Duray, G.Z.; El-Chami, M.; Ellis, C.R.; Hummel, J.; Jones, D.R.; Kowal, R.C.; et al. Long-term outcomes in leadless Micra transcatheter pacemakers with elevated thresholds at implantation: Results from the Micra Transcatheter Pacing System Global Clinical Trial. Heart Rhythm. 2017, 14, 685–691. [Google Scholar] [CrossRef] [PubMed]
- Bereuter, L.; Gysin, M.; Kueffer, T.; Kucera, M.; Niederhauser, T.; Fuhrer, J.; Heinisch, P.; Zurbuchen, A.; Obrist, D.; Tanner, H.; et al. Leadless Dual-Chamber Pacing: A Novel Communication Method for Wireless Pacemaker Synchronization. JACC Basic Transl. Sci. 2018, 3, 813–823. [Google Scholar] [CrossRef] [PubMed]
- Ansari, M.H.; Karami, M.A. Piezoelectric energy harvesting from heartbeat vibrations for leadless pacemakers. J. Phys. Conf. Ser. 2015, 660, 012121. [Google Scholar] [CrossRef]
- Cingolani, E.; Goldhaber, J.; Marbán, E. Next-generation pacemakers: From small devices to biological pacemakers. Nat. Rev. Cardiol. 2018, 15, 139–150. [Google Scholar] [CrossRef]
- Goto, H.; Sugiura, T.; Kazui, T. Feasibility of the Automatic Generating System (AGS) for Quartz Watches as a Leadless Pacemaker Power Source: A Preliminary Report. In Proceedings of the 20th Annual International Conference of the IEEE Engineering in Medicine and Biology Society. Vol.20 Biomedical Engineering Towards the Year 2000 and Beyond (Cat. No.98CH36286), Hong Kong, China, 1 November 1998; Volume 1, pp. 417–419. [Google Scholar]
- Kim, S.-G.; Priya, S.; Kanno, I. Piezoelectric MEMS for energy harvesting. MRS Bull. 2012, 37, 1039–1050. [Google Scholar] [CrossRef]
- Tian, W.; Ling, Z.; Yu, W.; Shi, J. A Review of MEMS Scale Piezoelectric Energy Harvester. Appl. Sci. 2018, 8, 645. [Google Scholar] [CrossRef]
- Li, P.; Ryu, J.; Hong, S. Piezoelectric/Triboelectric Nanogenerators for Biomedical Applications; IntechOpen: London, UK, 2019; ISBN 978-1-83881-060-3. [Google Scholar]
- Curie, J.; Curie, P. Phénomènes électriques des cristaux hémièdres à faces inclinées. J. Phys. Theor. Appl. 1882, 1, 245–251. [Google Scholar] [CrossRef]
- Kim, H.S.; Kim, J.-H.; Kim, J. A review of piezoelectric energy harvesting based on vibration. Int. J. Precis. Eng. Manuf. 2011, 12, 1129–1141. [Google Scholar] [CrossRef]
- Wang, P.; Du, H. ZnO thin film piezoelectric MEMS vibration energy harvesters with two piezoelectric elements for higher output performance. Rev. Sci. Instrum. 2015, 86, 075002. [Google Scholar] [CrossRef] [PubMed]
- Algieri, L.; Todaro, M.T.; Guido, F.; Mastronardi, V.; Desmaële, D.; Qualtieri, A.; Giannini, C.; Sibillano, T.; De Vittorio, M. Flexible Piezoelectric Energy-Harvesting Exploiting Biocompatible AlN Thin Films Grown onto Spin-Coated Polyimide Layers. ACS Appl. Energy Mater. 2018, 1, 5203–5210. [Google Scholar] [CrossRef]
- Eom, C.-B.; Trolier-McKinstry, S. Thin-film piezoelectric MEMS. MRS Bull. 2012, 37, 1007–1017. [Google Scholar] [CrossRef]
- Wasa, K.; Matsushima, T.; Adachi, H.; Kanno, I.; Kotera, H. Thin-Film Piezoelectric Materials For a Better Energy Harvesting MEMS. J. Microelectromech. Syst. 2012, 21, 451–457. [Google Scholar] [CrossRef]
- Won, S.S.; Sheldon, M.; Mostovych, N.; Kwak, J.; Chang, B.-S.; Ahn, C.W.; Kingon, A.I.; Kim, I.W.; Kim, S.-H. Piezoelectric poly(vinylidene fluoride trifluoroethylene) thin film-based power generators using paper substrates for wearable device applications. Appl. Phys. Lett. 2015, 107, 202901. [Google Scholar] [CrossRef]
- Fang, J.; Wang, X.; Lin, T. Electrical power generator from randomly oriented electrospun poly(vinylidene fluoride) nanofibre membranes. J. Mater. Chem. 2011, 21, 11088–11091. [Google Scholar] [CrossRef]
- Chen, X.; Xu, S.; Yao, N.; Shi, Y. 1.6 V Nanogenerator for Mechanical Energy Harvesting Using PZT Nanofibers. Nano Lett. 2010, 10, 2133–2137. [Google Scholar] [CrossRef]
- Bai, S.; Xu, Q.; Gu, L.; Ma, F.; Qin, Y.; Wang, Z.L. Single crystalline lead zirconate titanate (PZT) nano/micro-wire based self-powered UV sensor. Nano Energy 2012, 1, 789–795. [Google Scholar] [CrossRef]
- Todaro, M.T.; Guido, F.; Mastronardi, V.M.; Desmaele, D.; Epifani, G.; Algieri, L.; De Vittorio, M. Piezoelectric MEMS vibrational energy harvesters: Advances and outlook. Microelectron. Eng. 2017, 183–184, 23–26. [Google Scholar] [CrossRef]
- Singh, H.H.; Khare, N. Flexible ZnO-PVDF/PTFE based piezo-tribo hybrid nanogenerator. Nano Energy 2018, 51, 216–222. [Google Scholar] [CrossRef]
- Wang, Z.; Narita, F. Corona Poling Conditions for Barium Titanate/Epoxy Composites and their Unsteady Wind Energy Harvesting Potential. Adv. Eng. Mater. 2019, 21, 1900169. [Google Scholar] [CrossRef]
- Clementi, G.; Lombardi, G.; Margueron, S.; Suarez, M.A.; Lebrasseur, E.; Ballandras, S.; Imbaud, J.; Lardet-Vieudrin, F.; Gauthier-Manuel, L.; Dulmet, B.; et al. LiNbO3 films—A low-cost alternative lead-free piezoelectric material for vibrational energy harvesters. Mech. Syst. Signal Process. 2021, 149, 107171. [Google Scholar] [CrossRef]
- Bartasyte, A.; Margueron, S.; Baron, T.; Oliveri, S.; Boulet, P. Toward High-Quality Epitaxial LiNbO3and LiTaO3Thin Films for Acoustic and Optical Applications. Adv. Mater. Interfaces 2017, 4, 1600998. [Google Scholar] [CrossRef]
- Kosec, M.; Malič, B.; Benčan, A.; Rojac, T. KNN-Based Piezoelectric Ceramics. In Piezoelectric and Acoustic Materials for Transducer Applications; Safari, A., Akdoğan, E.K., Eds.; Springer: Boston, MA, USA, 2008; pp. 81–102. ISBN 978-0-387-76540-2. [Google Scholar]
- Qualtieri, A.; Rizzi, F.; Todaro, M.; Passaseo, A.; Cingolani, R.; De Vittorio, M. Stress-driven AlN cantilever-based flow sensor for fish lateral line system. Microelectron. Eng. 2011, 88, 2376–2378. [Google Scholar] [CrossRef]
- Caro, M.A.; Zhang, S.; Riekkinen, T.; Ylilammi, M.; Moram, M.A.; Lopez-Acevedo, O.; Molarius, J.; Laurila, T. Piezoelectric coefficients and spontaneous polarization of ScAlN. J. Phys. Condens. Matter 2015, 27, 245901. [Google Scholar] [CrossRef] [PubMed]
- Koblmueller, G.; Averbeck, R.; Geelhaar, L.; Riechert, H.; Hösler, W.; Pongratz, P. Growth diagram and morphologies of AlN thin films grown by molecular beam epitaxy. J. Appl. Phys. 2003, 93, 9591–9596. [Google Scholar] [CrossRef]
- Chen, D.; Colas, J.; Mercier, F.; Boichot, R.; Charpentier, L.; Escape, C.; Balat-Pichelin, M.; Pons, M. High temperature properties of AlN coatings deposited by chemical vapor deposition for solar central receivers. Surf. Coat. Technol. 2019, 377, 124872. [Google Scholar] [CrossRef]
- Mariello, M.; Blad, T.; Mastronardi, V.; Madaro, F.; Guido, F.; Staufer, U.; Tolou, N.; De Vittorio, M. Flexible piezoelectric AlN transducers buckled through package-induced preloading for mechanical energy harvesting. Nano Energy 2021, 85, 105986. [Google Scholar] [CrossRef]
- Mariello, M.; Guido, F.; Algieri, L.; Mastronardi, V.M.; Qualtieri, A.; Pisanello, F.; De Vittorio, M. Microstructure and Electrical Properties of Novel Piezo-Optrodes Based on Thin-Film Piezoelectric Aluminium Nitride for Sensing. IEEE Trans. Nanotechnol. 2021, 20, 10–19. [Google Scholar] [CrossRef]
- Akiyama, M.; Morofuji, Y.; Kamohara, T.; Nishikubo, K.; Tsubai, M.; Fukuda, O.; Ueno, N. Flexible piezoelectric pressure sensors using oriented aluminum nitride thin films prepared on polyethylene terephthalate films. J. Appl. Phys. 2006, 100, 114318. [Google Scholar] [CrossRef]
- Mariello, M.; Fachechi, L.; Guido, F.; De Vittorio, M. Conformal, Ultra-thin Skin-Contact-Actuated Hybrid Piezo/Triboelectric Wearable Sensor Based on AlN and Parylene-Encapsulated Elastomeric Blend. Adv. Funct. Mater. 2021, 31, 2101047. [Google Scholar] [CrossRef]
- Signore, M.A.; Rescio, G.; De Pascali, C.; Iacovacci, V.; Dario, P.; Leone, A.; Quaranta, F.; Taurino, A.; Siciliano, P.; Francioso, L. Fabrication and characterization of AlN-based flexible piezoelectric pressure sensor integrated into an implantable artificial pancreas. Sci. Rep. 2019, 9, 17130. [Google Scholar] [CrossRef] [PubMed]
- Jackson, N.; Keeney, L.; Mathewson, A. Flexible-CMOS and biocompatible piezoelectric AlN material for MEMS applications. Smart Mater. Struct. 2013, 22, 115033. [Google Scholar] [CrossRef]
- Hu, X.; Yang, F.; Guo, M.; Pei, J.; Zhao, H.; Wang, Y. Fabrication of polyimide microfluidic devices by laser ablation based additive manufacturing. Microsyst. Technol. 2020, 26, 1573–1583. [Google Scholar] [CrossRef]
- Sun, Y.; Lacour, S.P.; Brooks, R.A.; Rushton, N.; Fawcett, J.; Cameron, R.E. Assessment of the biocompatibility of photosensitive polyimide for implantable medical device use. J. Biomed. Mater. Res. Part A 2009, 90, 648–655. [Google Scholar] [CrossRef]
- Pi, Z.; Zhang, J.; Wen, C.; Zhang, Z.-B.; Wu, D. Flexible piezoelectric nanogenerator made of poly(vinylidenefluoride-co-trifluoroethylene) (PVDF-TrFE) thin film. Nano Energy 2014, 7, 33–41. [Google Scholar] [CrossRef]
- Mariello, M.; Qualtieri, A.; Mele, G.; De Vittorio, M. Metal-Free Multilayer Hybrid PENG Based on Soft Electrospun/-Sprayed Membranes with Cardanol Additive for Harvesting Energy from Surgical Face Masks. ACS Appl. Mater. Interfaces 2021, 13, 20606–20621. [Google Scholar] [CrossRef]
- Chiu, Y.-Y.; Lin, W.-Y.; Wang, H.-Y.; Huang, S.-B.; Wu, M.-H. Development of a piezoelectric polyvinylidene fluoride (PVDF) polymer-based sensor patch for simultaneous heartbeat and respiration monitoring. Sensors Actuators A Phys. 2013, 189, 328–334. [Google Scholar] [CrossRef]
- Bae, J.-H.; Chang, S.-H. PVDF-based ferroelectric polymers and dielectric elastomers for sensor and actuator applications: A review. Funct. Compos. Struct. 2019, 1, 012003. [Google Scholar] [CrossRef]
- Liu, J.; Yu, D.; Zheng, Z.; Huangfu, G.; Guo, Y. Lead-free BiFeO3 film on glass fiber fabric: Wearable hybrid piezoelectric-triboelectric nanogenerator. Ceram. Int. 2021, 47, 3573–3579. [Google Scholar] [CrossRef]
- Li, Z.; Zhu, G.; Yang, R.; Wang, A.; Wang, Z.L. Muscle-Driven In Vivo Nanogenerator. Adv. Mater. 2010, 22, 2534–2537. [Google Scholar] [CrossRef] [PubMed]
- Lu, B.; Chen, Y.; Ou, D.; Chen, H.; Diao, L.; Zhang, W.; Zheng, J.; Ma, W.; Sun, L.; Feng, X. Ultra-flexible Piezoelectric Devices Integrated with Heart to Harvest the Biomechanical Energy. Sci. Rep. 2015, 5, 16065. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Shin, H.J.; Lee, H.; Jeong, C.K.; Park, H.; Hwang, G.-T.; Lee, H.-Y.; Joe, D.J.; Han, J.H.; Lee, S.H.; et al. In Vivo Self-Powered Wireless Transmission Using Biocompatible Flexible Energy Harvesters. Adv. Funct. Mater. 2017, 27, 1700341. [Google Scholar] [CrossRef]
- Zhang, H.; Zhang, X.-S.; Cheng, X.; Liu, Y.; Han, M.; Xue, X.; Wang, S.; Yang, F.; Smitha, A.S.; Zhang, H.; et al. A flexible and implantable piezoelectric generator harvesting energy from the pulsation of ascending aorta: In vitro and in vivo studies. Nano Energy 2015, 12, 296–304. [Google Scholar] [CrossRef]
- Li, N.; Yi, Z.; Ma, Y.; Xie, F.; Huang, Y.; Tian, Y.; Dong, X.; Liu, Y.; Shao, X.; Jin, L.; et al. Direct Powering a Real Cardiac Pacemaker by Natural Energy of a Heartbeat. ACS Nano 2019, 13, 2822–2830. [Google Scholar] [CrossRef] [PubMed]
- Natta, L.; Mastronardi, V.M.; Guido, F.; Algieri, L.; Puce, S.; Pisano, F.; Rizzi, F.; Pulli, R.; Qualtieri, A.; De Vittorio, M. Soft and flexible piezoelectric smart patch for vascular graft monitoring based on Aluminum Nitride thin film. Sci. Rep. 2019, 9, 8392. [Google Scholar] [CrossRef]
- Dong, L.; Han, X.; Xu, Z.; Closson, A.B.; Liu, Y.; Wen, C.; Liu, X.; Escobar, G.P.; Oglesby, M.; Feldman, M.; et al. Flexible Porous Piezoelectric Cantilever on a Pacemaker Lead for Compact Energy Harvesting. Adv. Mater. Technol. 2019, 4, 1800148. [Google Scholar] [CrossRef]
- Azimi, S.; Golabchi, A.; Nekookar, A.; Rabbani, S.; Amiri, M.H.; Asadi, K.; Abolhasani, M.M. Self-powered cardiac pacemaker by piezoelectric polymer nanogenerator implant. Nano Energy 2021, 83, 105781. [Google Scholar] [CrossRef]
- Xu, Z.; Jin, C.; Cabe, A.; Escobedo, D.; Gruslova, A.; Jenney, S.; Closson, A.B.; Dong, L.; Chen, Z.; Feldman, M.D.; et al. Implantable Cardiac Kirigami-Inspired Lead-Based Energy Harvester Fabricated by Enhanced Piezoelectric Composite Film. Adv. Healthc. Mater. 2021, 10, e2002100. [Google Scholar] [CrossRef]
- Niu, S.; Wang, Z.L. Theoretical systems of triboelectric nanogenerators. Nano Energy 2015, 14, 161–192. [Google Scholar] [CrossRef]
- Fan, F.-R.; Tian, Z.-Q.; Wang, Z.L. Flexible triboelectric generator. Nano Energy 2012, 1, 328–334. [Google Scholar] [CrossRef]
- Niu, S.; Liu, Y.; Wang, S.; Lin, L.; Zhou, Y.S.; Hu, Y.; Wang, Z.L. Theoretical Investigation and Structural Optimization of Single-Electrode Triboelectric Nanogenerators. Adv. Funct. Mater. 2014, 24, 3332–3340. [Google Scholar] [CrossRef]
- Henniker, J. Triboelectricity in Polymers. Nature 1962, 196, 474. [Google Scholar] [CrossRef]
- Luo, J.; Wang, Z.L. Recent progress of triboelectric nanogenerators: From fundamental theory to practical applications. EcoMat 2020, 2, e12059. [Google Scholar] [CrossRef]
- Wang, Z.; Lin, L.; Chen, J.; Niu, S.; Zi, Y. Triboelectric Nanogenerators; Green Energy and Technology; Springer International Publishing: Atlanta, GA, USA, 2016; ISBN 978-3-319-40038-9. [Google Scholar]
- Zou, H.; Zhang, Y.; Guo, L.; Wang, P.; He, X.; Dai, G.; Zheng, H.; Chen, C.; Wang, A.C.; Xu, C.; et al. Quantifying the triboelectric series. Nat. Commun. 2019, 10, 1427. [Google Scholar] [CrossRef]
- Ibrahim, M.; Jiang, J.; Wen, Z.; Sun, X. Surface Engineering for Enhanced Triboelectric Nanogenerator. Nanoenergy Adv. 2021, 1, 58–80. [Google Scholar] [CrossRef]
- Zhang, H.; Yao, L.; Quan, L.; Zheng, X. Theories for triboelectric nanogenerators: A comprehensive review. Nanotechnol. Rev. 2020, 9, 610–625. [Google Scholar] [CrossRef]
- Narasimulu, A.A.; Zhao, P.; Soin, N.; Prashanthi, K.; Ding, P.; Chen, J.; Dong, S.; Chen, L.; Zhou, E.; Montemagno, C.D.; et al. Significant triboelectric enhancement using interfacial piezoelectric ZnO nanosheet layer. Nano Energy 2017, 40, 471–480. [Google Scholar] [CrossRef]
- Ravichandran, A.N.; Ramuz, M.; Blayac, S. Increasing surface charge density by effective charge accumulation layer inclusion for high-performance triboelectric nanogenerators. MRS Commun. 2019, 9, 682–689. [Google Scholar] [CrossRef]
- Kim, H.-J.; Yim, E.-C.; Kim, J.-H.; Kim, S.-J.; Park, J.-Y.; Oh, I.-K. Bacterial Nano-Cellulose Triboelectric Nanogenerator. Nano Energy 2017, 33, 130–137. [Google Scholar] [CrossRef]
- Wen, R.; Guo, J.; Yu, A.; Zhang, K.; Kou, J.; Zhu, Y.; Zhang, Y.; Li, B.-W.; Zhai, J. Remarkably enhanced triboelectric nanogenerator based on flexible and transparent monolayer titania nanocomposite. Nano Energy 2018, 50, 140–147. [Google Scholar] [CrossRef]
- Kil Yun, B.; Kim, H.S.; Ko, Y.J.; Murillo, G.; Jung, J.H. Interdigital electrode based triboelectric nanogenerator for effective energy harvesting from water. Nano Energy 2017, 36, 233–240. [Google Scholar] [CrossRef]
- Mariello, M.; Scarpa, E.; Algieri, L.; Guido, F.; Mastronardi, V.M.; Qualtieri, A.; De Vittorio, M. Novel Flexible Triboelectric Nanogenerator based on Metallized Porous PDMS and Parylene C. Energies 2020, 13, 1625. [Google Scholar] [CrossRef]
- Qian, Y.; Lyu, Z.; Kim, D.-H.; Kang, D.J. Enhancing the output power density of polydimethylsiloxane-based flexible triboelectric nanogenerators with ultrathin nickel telluride nanobelts as a co-triboelectric layer. Nano Energy 2021, 90, 106536. [Google Scholar] [CrossRef]
- Qian, Y.; Kang, D.J. Large-Area High-Quality AB-Stacked Bilayer Graphene on h-BN/Pt Foil by Chemical Vapor Deposition. ACS Appl. Mater. Interfaces 2018, 10, 29069–29075. [Google Scholar] [CrossRef]
- Harnchana, V.; Van Ngoc, H.; He, W.; Rasheed, A.; Park, H.; Amornkitbamrung, V.; Kang, D.J. Enhanced Power Output of a Triboelectric Nanogenerator using Poly(dimethylsiloxane) Modified with Graphene Oxide and Sodium Dodecyl Sulfate. ACS Appl. Mater. Interfaces 2018, 10, 25263–25272. [Google Scholar] [CrossRef]
- Kim, D.Y.; Kim, H.S.; Kong, D.S.; Choi, M.; Kim, H.B.; Lee, J.-H.; Murillo, G.; Lee, M.; Kim, S.S.; Jung, J.H. Floating buoy-based triboelectric nanogenerator for an effective vibrational energy harvesting from irregular and random water waves in wild sea. Nano Energy 2018, 45, 247–254. [Google Scholar] [CrossRef]
- Chen, J.; Yang, J.; Li, Z.; Fan, X.; Zi, Y.; Jing, Q.; Guo, H.; Wen, Z.; Pradel, K.C.; Niu, S.; et al. Networks of Triboelectric Nanogenerators for Harvesting Water Wave Energy: A Potential Approach toward Blue Energy. ACS Nano 2015, 9, 3324–3331. [Google Scholar] [CrossRef]
- Wang, Z.L.; Jiang, T.; Xu, L. Toward the blue energy dream by triboelectric nanogenerator networks. Nano Energy 2017, 39, 9–23. [Google Scholar] [CrossRef]
- Xiong, J.; Lin, M.-F.; Wang, J.; Gaw, S.L.; Parida, K.; Lee, P.S. Wearable All-Fabric-Based Triboelectric Generator for Water Energy Harvesting. Adv. Energy Mater. 2017, 7, 1701243. [Google Scholar] [CrossRef]
- Luo, J.; Tang, W.; Fan, F.R.; Liu, C.; Pang, Y.; Cao, G.; Wang, Z.L. Transparent and Flexible Self-Charging Power Film and Its Application in a Sliding Unlock System in Touchpad Technology. ACS Nano 2016, 10, 8078–8086. [Google Scholar] [CrossRef] [PubMed]
- Luo, J.; Wang, Z.; Xu, L.; Wang, A.C.; Han, K.; Jiang, T.; Lai, Q.; Bai, Y.; Tang, W.; Fan, F.R.; et al. Flexible and durable wood-based triboelectric nanogenerators for self-powered sensing in athletic big data analytics. Nat. Commun. 2019, 10, 5147. [Google Scholar] [CrossRef] [PubMed]
- Gorham, W.F. A New, General Synthetic Method for the Preparation of Linear Poly-p-xylylenes. J. Polym. Sci. Part A-1 Polym. Chem. 1966, 4, 3027–3039. [Google Scholar] [CrossRef]
- Mariello, M.; Guido, F.; Mastronardi, V.M.; De Donato, F.; Salbini, M.; Brunetti, V.; Qualtieri, A.; Rizzi, F.; De Vittorio, M. Captive-air-bubble aerophobicity measurements of antibiofouling coatings for underwater MEMS devices. Nanomater. Nanotechnol. 2019, 9, 1847980419862075. [Google Scholar] [CrossRef]
- Mariello, M.; Guido, F.; Mastronardi, V.M.; Giannuzzi, R.; Algieri, L.; Qualteri, A.; Maffezzoli, A.; De Vittorio, M. Reliability of Protective Coatings for Flexible Piezoelectric Transducers in Aqueous Environments. Micromachines 2019, 10, 739. [Google Scholar] [CrossRef]
- Golda-Cepa, M.; Brzychczy-Wloch, M.; Engvall, K.; Aminlashgari, N.; Hakkarainen, M.; Kotarba, A. Microbiological investigations of oxygen plasma treated parylene C surfaces for metal implant coating. Mater. Sci. Eng. C 2015, 52, 273–281. [Google Scholar] [CrossRef]
- Chen, T.-N.; Wuu, D.-S.; Wu, C.-C.; Chiang, C.-C.; Chen, Y.-P.; Horng, R.-H. Improvements of Permeation Barrier Coatings Using Encapsulated Parylene Interlayers for Flexible Electronic Applications. Plasma Process. Polym. 2007, 4, 180–185. [Google Scholar] [CrossRef]
- Applerot, G.; Abu-Mukh, R.; Irzh, A.; Charmet, J.; Keppner, H.; Laux, E.; Guibert, G.; Gedanken, A. Decorating Parylene-Coated Glass with ZnO Nanoparticles for Antibacterial Applications: A Comparative Study of Sonochemical, Microwave, and Microwave-Plasma Coating Routes. ACS Appl. Mater. Interfaces 2010, 2, 1052–1059. [Google Scholar] [CrossRef]
- Cieślik, M.; Engvall, K.; Pan, J.; Kotarba, A. Silane–parylene coating for improving corrosion resistance of stainless steel 316L implant material. Corros. Sci. 2011, 53, 296–301. [Google Scholar] [CrossRef]
- Lo, H.-W.; Tai, Y.-C. Parylene-based electret power generators. J. Micromech. Microeng. 2008, 18, 104006. [Google Scholar] [CrossRef]
- Genter, S.; Langhof, T.; Paul, O. Electret-based Out-Of-Plane Micro Energy Harvester with Parylene-C Serving as the Electret and Spring Material. Procedia Eng. 2015, 120, 341–344. [Google Scholar] [CrossRef][Green Version]
- Calcagnile, P.; Blasi, L.; Rizzi, F.; Qualtieri, A.; Athanassiou, A.; Gogolides, E.; De Vittorio, M. Parylene C Surface Functionalization and Patterning with pH-Responsive Microgels. ACS Appl. Mater. Interfaces 2014, 6, 15708–15715. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zheng, Q.; Wang, Z.L.; Li, Z. Nanogenerator-Based Self-Powered Sensors for Wearable and Implantable Electronics. Research 2020, 2020, 8710686. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Shi, B.; Fan, F.; Wang, X.; Yan, L.; Yuan, W.; Wang, S.; Liu, H.; Li, Z.; Wang, Z.L. In Vivo Powering of Pacemaker by Breathing-Driven Implanted Triboelectric Nanogenerator. Adv. Mater. 2014, 26, 5851–5856. [Google Scholar] [CrossRef]
- Ma, Y.; Zheng, Q.; Liu, Y.; Shi, B.; Xue, X.; Ji, W.; Liu, Z.; Jin, Y.; Zou, Y.; An, Z.; et al. Self-Powered, One-Stop, and Multifunctional Implantable Triboelectric Active Sensor for Real-Time Biomedical Monitoring. Nano Lett. 2016, 16, 6042–6051. [Google Scholar] [CrossRef]
- Ouyang, H.; Liu, Z.; Li, N.; Shi, B.; Zou, Y.; Xie, F.; Ma, Y.; Li, Z.; Li, H.; Zheng, Q.; et al. Symbiotic cardiac pacemaker. Nat. Commun. 2019, 10, 1821. [Google Scholar] [CrossRef]
- Niu, S.; Wang, X.; Yi, F.; Zhou, Y.S.; Wang, Z.L. A universal self-charging system driven by random biomechanical energy for sustainable operation of mobile electronics. Nat. Commun. 2015, 6, 8975. [Google Scholar] [CrossRef]
- Ryu, H.; Park, H.-M.; Kim, M.-K.; Kim, B.; Myoung, H.S.; Kim, T.Y.; Yoon, H.-J.; Kwak, S.S.; Kim, J.; Hwang, T.H.; et al. Self-rechargeable cardiac pacemaker system with triboelectric nanogenerators. Nat. Commun. 2021, 12, 4374. [Google Scholar] [CrossRef]
- Jiang, W.; Li, H.; Liu, Z.; Li, Z.; Tian, J.; Shi, B.; Zou, Y.; Ouyang, H.; Zhao, C.; Zhao, L.; et al. Fully Bioabsorbable Natural-Materials-Based Triboelectric Nanogenerators. Adv. Mater. 2018, 30, e1801895. [Google Scholar] [CrossRef]
- Hinchet, R.; Yoon, H.-J.; Ryu, H.; Kim, M.-K.; Choi, E.-K.; Kim, D.-S.; Kim, S.-W. Transcutaneous ultrasound energy harvesting using capacitive triboelectric technology. Science 2019, 365, 491–494. [Google Scholar] [CrossRef]
- Shawon, S.M.A.Z.; Sun, A.X.; Vega, V.S.; Chowdhury, B.D.; Tran, P.; Carballo, Z.D.; Tolentino, J.A.; Li, J.; Rafaqut, M.S.; Danti, S.; et al. Piezo-tribo dual effect hybrid nanogenerators for health monitoring. Nano Energy 2021, 82, 105691. [Google Scholar] [CrossRef]
- Dong, L.; Jin, C.; Closson, A.B.; Trase, I.; Richards, H.R.; Chen, Z.; Zhang, J.X.J. Cardiac Cardiac energy harvesting and sensing based on piezoelectric and triboelectric designs. Nano Energy 2020, 76, 105076. [Google Scholar] [CrossRef]
- Shi, B.; Zheng, Q.; Jiang, W.; Yan, L.; Wang, X.; Liu, H.; Yao, Y.; Li, Z.; Wang, Z.L. A Packaged Self-Powered System with Universal Connectors Based on Hybridized Nanogenerators. Adv. Mater. 2016, 28, 846–852. [Google Scholar] [CrossRef]
- Ippili, S.; Jella, V.; Thomas, A.; Yoon, S.-G. The Recent Progress on Halide Perovskite-Based Self-Powered Sensors Enabled by Piezoelectric and Triboelectric Effects. Nanoenergy Adv. 2021, 1, 3–31. [Google Scholar] [CrossRef]
- Wu, T.; Redoute, J.-M.; Yuce, M.R. A Wireless Implantable Sensor Design With Subcutaneous Energy Harvesting for Long-Term IoT Healthcare Applications. IEEE Access 2018, 6, 35801–35808. [Google Scholar] [CrossRef]
- Chen, Z.; Law, M.-K.; Mak, P.-I.; Martins, R.P. A Single-Chip Solar Energy Harvesting IC Using Integrated Photodiodes for Biomedical Implant Applications. IEEE Trans. Biomed. Circuits Syst. 2017, 11, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Haeberlin, A.; Zurbuchen, A.; Schaerer, J.; Wagner, J.; Walpen, S.; Huber, C.; Haeberlin, H.; Fuhrer, J.; Vogel, R. Successful pacing using a batteryless sunlight-powered pacemaker. EP Eur. 2014, 16, 1534–1539. [Google Scholar] [CrossRef]
- Burks, S.; Kolcun, J.P.; Wang, M.Y. Subcutaneous Solar Cells Harvest Energy for Pulse Generation. Neurosurgery 2017, 81, N24. [Google Scholar] [CrossRef]
- Song, K.; Han, J.H.; Lim, T.; Kim, N.; Shin, S.; Kim, J.; Choo, H.; Jeong, S.; Kim, Y.-C.; Wang, Z.L.; et al. Subdermal Flexible Solar Cell Arrays for Powering Medical Electronic Implants. Adv. Healthc. Mater. 2016, 5, 1572–1580. [Google Scholar] [CrossRef]
- Bereuter, L.; Williner, S.; Pianezzi, F.; Bissig, B.; Buecheler, S.; Burger, J.; Vogel, R.; Zurbuchen, A.; Haeberlin, A. Energy Harvesting by Subcutaneous Solar Cells: A Long-Term Study on Achievable Energy Output. Ann. Biomed. Eng. 2017, 45, 1172–1180. [Google Scholar] [CrossRef]
- Tholl, M.; Akarçay, H.; Tanner, H.; Niederhauser, T.; Zurbuchen, A.; Frenz, M.; Haeberlin, A. Subdermal solar energy harvesting—A new way to power autonomous electric implants. Appl. Energy 2020, 269, 114948. [Google Scholar] [CrossRef]
- Prominski, A.; Shi, J.; Li, P.; Yue, J.; Lin, Y.; Park, J.; Tian, B.; Rotenberg, M.Y. Porosity-based heterojunctions enable leadless optoelectronic modulation of tissues. Nat. Mater. 2022, 21, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Wu, F.; Redoute, J.-M.; Yuce, M.R. An Autonomous Wireless Body Area Network Implementation Towards IoT Connected Healthcare Applications. IEEE Access 2017, 5, 11413–11422. [Google Scholar] [CrossRef]
- Shuvo, M.H.; Titirsha, T.; Amin, N.; Islam, S.K. Energy Harvesting in Implantable and Wearable Medical Devices for Enduring Precision Healthcare. Energies 2022, 15, 7495. [Google Scholar] [CrossRef]
- Dieffenderfer, J.P.; Beppler, E.; Novak, T.; Whitmire, E.; Jayakumar, R.; Randall, C.; Qu, W.; Rajagopalan, R.; Bozkurt, A. Solar Powered Wrist Worn Acquisition System for Continuous Photoplethysmogram Monitoring. In Proceedings of the 2014 36th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Chicago, IL, USA, 26–30 August 2014; pp. 3142–3145. [Google Scholar]
- Cosnier, S.; Le Goff, A.; Holzinger, M. Towards glucose biofuel cells implanted in human body for powering artificial organs: Review. Electrochem. Commun. 2013, 38, 19–23. [Google Scholar] [CrossRef]
- Drake, R.F.; Kusserow, B.K.; Messinger, S.; Matsuda, S. A tissue implantable fuel cell power supply. Trans. Am. Soc. Artif. Intern. Organs 1970, 16, 199–205. [Google Scholar] [PubMed]
- Simons, P.; Schenk, S.A.; Gysel, M.A.; Olbrich, L.F.; Rupp, J.L.M. A Ceramic-Electrolyte Glucose Fuel Cell for Implantable Electronics. Adv. Mater. 2022, 34, 2109075. [Google Scholar] [CrossRef]
- Holzinger, M.; Le Goff, A.; Cosnier, S. Carbon nanotube/enzyme biofuel cells. Electrochim. Acta 2012, 82, 179–190. [Google Scholar] [CrossRef]
- Gellett, W.; Kesmez, M.; Schumacher, J.; Akers, N.; Minteer, S.D. Biofuel Cells for Portable Power. Electroanalysis 2010, 22, 727–731. [Google Scholar] [CrossRef]
- Heller, A. Miniature biofuel cells. Phys. Chem. Chem. Phys. 2004, 6, 209–216. [Google Scholar] [CrossRef]
- Mano, N.; Mao, F.; Heller, A. Characteristics of a Miniature Compartment-less Glucose−O2 Biofuel Cell and Its Operation in a Living Plant. J. Am. Chem. Soc. 2003, 125, 6588–6594. [Google Scholar] [CrossRef] [PubMed]
- Cinquin, P.; Gondran, C.; Giroud, F.; Mazabrard, S.; Pellissier, A.; Boucher, F.; Alcaraz, J.-P.; Gorgy, K.; Lenouvel, F.; Mathé, S.; et al. A Glucose BioFuel Cell Implanted in Rats. PLoS ONE 2010, 5, e10476. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, M.; Ritzmann, R.E.; Lee, I.; Pollack, A.J.; Scherson, D. An Implantable Biofuel Cell for a Live Insect. J. Am. Chem. Soc. 2012, 134, 1458–1460. [Google Scholar] [CrossRef] [PubMed]
- Halámková, L.; Halámek, J.; Bocharova, V.; Szczupak, A.; Alfonta, L.; Katz, E. Implanted Biofuel Cell Operating in a Living Snail. J. Am. Chem. Soc. 2012, 134, 5040–5043. [Google Scholar] [CrossRef]
- Zebda, A.; Gondran, C.; Le Goff, A.; Holzinger, M.; Cinquin, P.; Cosnier, S. Mediatorless high-power glucose biofuel cells based on compressed carbon nanotube-enzyme electrodes. Nat. Commun. 2011, 2, 370. [Google Scholar] [CrossRef]
- Castorena-Gonzalez, J.A.; Foote, C.; MacVittie, K.; Halámek, J.; Halámková, L.; Martinez-Lemus, L.A.; Katz, E. Biofuel Cell Operating in Vivo in Rat. Electroanalysis 2013, 25, 1579–1584. [Google Scholar] [CrossRef]
- Sales, F.C.P.F.; Iost, R.M.; Martins, M.V.A.; Almeida, M.C.; Crespilho, F.N. An intravenous implantable glucose/dioxygen biofuel cell with modified flexible carbon fiber electrodes. Lab Chip 2013, 13, 468–474. [Google Scholar] [CrossRef]
- MacVittie, K.; Halámek, J.; Halámková, L.; Southcott, M.; Jemison, W.D.; Lobel, R.; Katz, E. From “cyborg” lobsters to a pacemaker powered by implantable biofuel cells. Energy Environ. Sci. 2012, 6, 81–86. [Google Scholar] [CrossRef]
- Barton, S.C.; Gallaway, J.; Atanassov, P. Enzymatic Biofuel Cells for Implantable and Microscale Devices. Chem. Rev. 2004, 104, 4867–4886. [Google Scholar] [CrossRef]
- Tang, X.; He, Y.; Liu, J. Soft bioelectronics for cardiac interfaces. Biophys. Rev. 2022, 3, 011301. [Google Scholar] [CrossRef]
- Kim, D.-H.; Ahn, J.-H.; Choi, W.M.; Kim, H.-S.; Kim, T.-H.; Song, J.; Huang, Y.Y.; Liu, Z.; Lu, C.; Rogers, J.A. Stretchable and Foldable Silicon Integrated Circuits. Science 2008, 320, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Rogers, J.A.; Lagally, M.G.; Nuzzo, R.G. Synthesis, assembly and applications of semiconductor nanomembranes. Nature 2011, 477, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Phan, H.-P.; Zhong, Y.; Nguyen, T.-K.; Park, Y.; Dinh, T.K.; Song, E.; Vadivelu, R.K.; Masud, M.K.; Li, J.; Shiddiky, M.J.A.; et al. Long-Lived, Transferred Crystalline Silicon Carbide Nanomembranes for Implantable Flexible Electronics. ACS Nano 2019, 13, 11572–11581. [Google Scholar] [CrossRef]
- Viventi, J.; Kim, D.-H.; Moss, J.D.; Kim, Y.-S.; Blanco, J.A.; Annetta, N.; Hicks, A.; Xiao, J.; Huang, Y.; Callans, D.J.; et al. A Conformal, Bio-Interfaced Class of Silicon Electronics for Mapping Cardiac Electrophysiology. Sci. Transl. Med. 2010, 2, 24ra22. [Google Scholar] [CrossRef]
- Fang, H.; Yu, K.J.; Gloschat, C.; Yang, Z.; Song, E.; Chiang, C.-H.; Zhao, J.; Won, S.M.; Xu, S.; Trumpis, M.; et al. Capacitively coupled arrays of multiplexed flexible silicon transistors for long-term cardiac electrophysiology. Nat. Biomed. Eng. 2017, 1, 0038. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.S.; Yin, R.T.; Pfenniger, A.; Koo, J.; Avila, R.; Lee, K.B.; Chen, S.W.; Lee, G.; Li, G.; Qiao, Y.; et al. Fully implantable and bioresorbable cardiac pacemakers without leads or batteries. Nat. Biotechnol. 2021, 39, 1228–1238. [Google Scholar] [CrossRef]
- Choi, Y.S.; Jeong, H.; Yin, R.T.; Avila, R.; Pfenniger, A.; Yoo, J.; Lee, J.Y.; Tzavelis, A.; Lee, Y.J.; Chen, S.W.; et al. A transient, closed-loop network of wireless, body-integrated devices for autonomous electrotherapy. Science 2022, 376, 1006–1012. [Google Scholar] [CrossRef]
- Kim, D.-H.; Ghaffari, R.; Lu, N.; Wang, S.; Lee, S.P.; Keum, H.; D’Angelo, R.; Klinker, L.; Su, Y.; Lu, C.; et al. Electronic sensor and actuator webs for large-area complex geometry cardiac mapping and therapy. Proc. Natl. Acad. Sci. USA 2012, 109, 19910–19915. [Google Scholar] [CrossRef]
- Xu, L.; Gutbrod, S.R.; Bonifas, A.P.; Su, Y.; Sulkin, M.S.; Lu, N.; Chung, H.-J.; Jang, K.-I.; Liu, Z.; Ying, M.; et al. 3D multifunctional integumentary membranes for spatiotemporal cardiac measurements and stimulation across the entire epicardium. Nat. Commun. 2014, 5, 3329. [Google Scholar] [CrossRef]
- Xu, L.; Gutbrod, S.R.; Ma, Y.; Petrossians, A.; Liu, Y.; Webb, R.C.; Fan, J.A.; Yang, Z.; Xu, R.; Whalen, J.J., III; et al. Materials and Fractal Designs for 3D Multifunctional Integumentary Membranes with Capabilities in Cardiac Electrotherapy. Adv. Mater. 2015, 27, 1731–1737. [Google Scholar] [CrossRef]
- Lee, W.; Kobayashi, S.; Nagase, M.; Jimbo, Y.; Saito, I.; Inoue, Y.; Yambe, T.; Sekino, M.; Malliaras, G.G.; Yokota, T.; et al. Nonthrombogenic, stretchable, active multielectrode array for electroanatomical mapping. Sci. Adv. 2018, 4, eaau2426. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Choi, S.; Janardhan, A.H.; Lee, S.-Y.; Raut, S.; Soares, J.; Shin, K.; Yang, S.; Lee, C.; Kang, K.-W.; et al. Electromechanical cardioplasty using a wrapped elasto-conductive epicardial mesh. Sci. Transl. Med. 2016, 8, 344ra86. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.; Han, S.I.; Jung, D.; Hwang, H.J.; Lim, C.; Bae, S.; Park, O.K.; Tschabrunn, C.M.; Lee, M.; Bae, S.Y.; et al. Highly conductive, stretchable and biocompatible Ag–Au core–sheath nanowire composite for wearable and implantable bioelectronics. Nat. Nanotechnol. 2018, 13, 1048–1056. [Google Scholar] [CrossRef] [PubMed]
- Gutruf, P.; Yin, R.T.; Lee, K.B.; Ausra, J.; Brennan, J.A.; Qiao, Y.; Xie, Z.; Peralta, R.; Talarico, O.; Murillo, A.; et al. Wireless, battery-free, fully implantable multimodal and multisite pacemakers for applications in small animal models. Nat. Commun. 2019, 10, 5742. [Google Scholar] [CrossRef]
- Sim, K.; Ershad, F.; Zhang, Y.; Yang, P.; Shim, H.; Rao, Z.; Lu, Y.; Thukral, A.; Elgalad, A.; Xi, Y.; et al. An epicardial bioelectronic patch made from soft rubbery materials and capable of spatiotemporal mapping of electrophysiological activity. Nat. Electron. 2020, 3, 775–784. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, X.; Liu, Y.; Rodrigo, M.; Loftus, P.D.; Aparicio-Valenzuela, J.; Zheng, J.; Pong, T.; Cyr, K.J.; Babakhanian, M.; et al. Intrinsically stretchable electrode array enabled in vivo electrophysiological mapping of atrial fibrillation at cellular resolution. Proc. Natl. Acad. Sci. USA 2020, 117, 14769–14778. [Google Scholar] [CrossRef]
- Liu, J.; Wang, J.; Zhang, Z.; Molina-Lopez, F.; Wang, G.-J.N.; Schroeder, B.C.; Yan, X.; Zeng, Y.; Zhao, O.; Tran, H.; et al. Fully stretchable active-matrix organic light-emitting electrochemical cell array. Nat. Commun. 2020, 11, 3362. [Google Scholar] [CrossRef] [PubMed]
- Pappone, C.; Santinelli, V. Multielectrode basket catheter: A new tool for curing atrial fibrillation? Heart Rhythm 2006, 3, 385–386. [Google Scholar] [CrossRef]
- Kim, D.-H.; Ghaffari, R.; Lu, N.; Rogers, J.A. Flexible and Stretchable Electronics for Biointegrated Devices. Annu. Rev. Biomed. Eng. 2012, 14, 113–128. [Google Scholar] [CrossRef]
- Oesterlein, T.; Frisch, D.; Loewe, A.; Seemann, G.; Schmitt, C.; Dössel, O.; Luik, A. Basket-Type Catheters: Diagnostic Pitfalls Caused by Deformation and Limited Coverage. BioMed Res. Int. 2016, 2016, e5340574. [Google Scholar] [CrossRef]
- Su, Y.; Liu, Z.; Wang, S.; Ghaffari, R.; Kim, D.-H.; Hwang, K.-C.; Rogers, J.A.; Huang, Y. Mechanics of stretchable electronics on balloon catheter under extreme deformation. Int. J. Solids Struct. 2014, 51, 1555–1561. [Google Scholar] [CrossRef][Green Version]
- Klinker, L.; Lee, S.; Work, J.; Wright, J.; Ma, Y.; Ptaszek, L.; Webb, R.C.; Liu, C.; Sheth, N.; Mansour, M.; et al. Balloon catheters with integrated stretchable electronics for electrical stimulation, ablation and blood flow monitoring. Extreme Mech. Lett. 2015, 3, 45–54. [Google Scholar] [CrossRef]
- Kim, D.H.; Lu, N.; Ghaffari, R.; Kim, Y.-S.; Lee, S.P.; Xu, L.; Wu, J.; Kim, R.-H.; Song, J.; Liu, Z.; et al. Materials for multifunctional balloon catheters with capabilities in cardiac electrophysiological mapping and ablation therapy. Nat. Mater. 2011, 10, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Han, M.; Chen, L.; Aras, K.; Liang, C.; Chen, X.; Zhao, H.; Li, K.; Faye, N.R.; Sun, B.; Kim, J.-H.; et al. Catheter-integrated soft multilayer electronic arrays for multiplexed sensing and actuation during cardiac surgery. Nat. Biomed. Eng. 2020, 4, 997–1009. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Ma, Y.; Ouyang, H.; Shi, B.; Li, N.; Jiang, D.; Xie, F.; Qu, D.; Zou, Y.; Huang, Y.; et al. Transcatheter Self-Powered Ultrasensitive Endocardial Pressure Sensor. Adv. Funct. Mater. 2019, 29, 1807560. [Google Scholar] [CrossRef]
- Dvir, T.; Timko, B.; Brigham, M.; Naik, S.R.; Karajanagi, S.S.; Levy, O.; Jin, H.; Parker, K.K.; Langer, R.; Kohane, D.S. Nanowired three-dimensional cardiac patches. Nat. Nanotechnol. 2011, 6, 720–725. [Google Scholar] [CrossRef]
- Mei, X.; Cheng, K. Recent Development in Therapeutic Cardiac Patches. Front. Cardiovasc. Med. 2020, 7, 610364. [Google Scholar] [CrossRef]
- Chantre, C.O.; Gonzalez, G.M.; Ahn, S.; Cera, L.; Campbell, P.H.; Hoerstrup, S.P.; Parker, K.K. Porous Biomimetic Hyaluronic Acid and Extracellular Matrix Protein Nanofiber Scaffolds for Accelerated Cutaneous Tissue Repair. ACS Appl. Mater. Interfaces 2019, 11, 45498–45510. [Google Scholar] [CrossRef]
- Yang, Q.; Wei, T.; Yin, R.T.; Wu, M.; Xu, Y.; Koo, J.; Choi, Y.S.; Xie, Z.; Chen, S.W.; Kandela, I.; et al. Photocurable bioresorbable adhesives as functional interfaces between flexible bioelectronic devices and soft biological tissues. Nat. Mater. 2021, 20, 1559–1570. [Google Scholar] [CrossRef]
- Li, J.; Celiz, A.D.; Yang, J.; Yang, Q.; Wamala, I.; Whyte, W.; Seo, B.R.; Vasilyev, N.V.; Vlassak, J.J.; Suo, Z.; et al. Tough adhesives for diverse wet surfaces. Science 2017, 357, 378–381. [Google Scholar] [CrossRef]
- Liu, J.; Xie, C.; Dai, X.; Jin, L.; Zhou, W.; Lieber, C.M. Multifunctional three-dimensional macroporous nanoelectronic networks for smart materials. Proc. Natl. Acad. Sci. USA 2013, 110, 6694–6699. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Liu, J.; Dvir, T.; Jin, L.; Tsui, J.H.; Qing, Q.; Suo, Z.; Langer, R.; Kohane, D.S.; Lieber, C.M. Macroporous nanowire nanoelectronic scaffolds for synthetic tissues. Nat. Mater. 2012, 11, 986–994. [Google Scholar] [CrossRef] [PubMed]
- Dai, X.; Zhou, W.; Gao, T.; Liu, J.; Lieber, C.M. Three-dimensional mapping and regulation of action potential propagation in nanoelectronics-innervated tissues. Nat. Nanotechnol. 2016, 11, 776–782. [Google Scholar] [CrossRef]
- Wang, X.; Feiner, R.; Luan, H.; Zhang, Q.; Zhao, S.; Zhang, Y.; Han, M.; Li, Y.; Sun, R.; Wang, H.; et al. Three-dimensional electronic scaffolds for monitoring and regulation of multifunctional hybrid tissues. Extreme Mech. Lett. 2020, 35, 100634. [Google Scholar] [CrossRef]
- Feiner, R.; Engel, L.; Fleischer, S.; Malki, M.; Gal, I.; Shapira, A.; Shacham-Diamand, Y.; Dvir, T. Engineered hybrid cardiac patches with multifunctional electronics for online monitoring and regulation of tissue function. Nat. Mater. 2016, 15, 679–685. [Google Scholar] [CrossRef]
- Feiner, R.; Fleischer, S.; Shapira, A.; Kalish, O.; Dvir, T. Multifunctional degradable electronic scaffolds for cardiac tissue engineering. J. Control. Release 2018, 281, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Lind, J.U.; Busbee, T.A.; Valentine, A.D.; Pasqualini, F.S.; Yuan, H.; Yadid, M.; Park, S.-J.; Kotikian, A.; Nesmith, A.P.; Campbell, P.H.; et al. Instrumented cardiac microphysiological devices via multimaterial three-dimensional printing. Nat. Mater. 2017, 16, 303–308. [Google Scholar] [CrossRef]
- Drakhlis, L.; Biswanath, S.; Farr, C.-M.; Lupanow, V.; Teske, J.; Ritzenhoff, K.; Franke, A.; Manstein, F.; Bolesani, E.; Kempf, H.; et al. Human heart-forming organoids recapitulate early heart and foregut development. Nat. Biotechnol. 2021, 39, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Hofer, M.; Lutolf, M.P. Engineering organoids. Nat. Rev. Mater. 2021, 6, 402–420. [Google Scholar] [CrossRef]
- Lewis-Israeli, Y.R.; Wasserman, A.H.; Gabalski, M.A.; Volmert, B.D.; Ming, Y.; Ball, K.A.; Yang, W.; Zou, J.; Ni, G.; Pajares, N.; et al. Self-assembling human heart organoids for the modeling of cardiac development and congenital heart disease. Nat. Commun. 2021, 12, 5142. [Google Scholar] [CrossRef]
- Richards, D.J.; Coyle, R.C.; Tan, Y.; Jia, J.; Wong, K.; Toomer, K.; Menick, D.R.; Mei, Y. Inspiration from heart development: Biomimetic development of functional human cardiac organoids. Biomaterials 2017, 142, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Lei, W.; Hu, S. Cardiac organoid—A promising perspective of preclinical model. Stem Cell Res. Ther. 2021, 12, 272. [Google Scholar] [CrossRef] [PubMed]
- Mills, R.; Hudson, J. An in vitro model of myocardial infarction. Nat. Biomed. Eng. 2020, 4, 366–367. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.S.; Aleman, J.; Shin, S.R.; Kilic, T.; Kim, D.; Shaegh, S.A.M.; Massa, S.; Riahi, R.; Chae, S.; Hu, N.; et al. Multisensor-integrated organs-on-chips platform for automated and continual in situ monitoring of organoid behaviors. Proc. Natl. Acad. Sci. USA 2017, 114, E2293–E2302. [Google Scholar] [CrossRef] [PubMed]
- Le Floch, P.; Li, Q.; Lin, Z.; Zhao, S.; Liu, R.; Tasnim, K.; Jiang, H.; Liu, J. Stretchable Mesh Nanoelectronics for 3D Single-Cell Chronic Electrophysiology from Developing Brain Organoids. Adv. Mater. 2022, 34, 2106829. [Google Scholar] [CrossRef]
- Kalmykov, A.; Huang, C.; Bliley, J.; Shiwarski, D.; Tashman, J.; Abdullah, A.; Rastogi, S.K.; Shukla, S.; Mataev, E.; Feinberg, A.W.; et al. Organ-on-e-chip: Three-dimensional self-rolled biosensor array for electrical interrogations of human electrogenic spheroids. Sci. Adv. 2019, 5, eaax0729. [Google Scholar] [CrossRef]
- Li, Q.; Nan, K.; Le Floch, P.; Lin, Z.; Sheng, H.; Blum, T.S.; Liu, J. Cyborg Organoids: Implantation of Nanoelectronics via Organogenesis for Tissue-Wide Electrophysiology. Nano Lett. 2019, 19, 5781–5789. [Google Scholar] [CrossRef]
- Kim, M.; Hwang, J.C.; Min, S.; Park, Y.-G.; Kim, S.; Kim, E.; Seo, H.; Chung, W.G.; Lee, J.; Cho, S.-W.; et al. Multimodal Characterization of Cardiac Organoids Using Integrations of Pressure-Sensitive Transistor Arrays with Three-Dimensional Liquid Metal Electrodes. Nano Lett. 2022, 22, 7892–7901. [Google Scholar] [CrossRef]
- Jafari, A.; Ajji, Z.; Mousavi, A.; Naghieh, S.; Bencherif, S.A.; Savoji, H. Latest Advances in 3D Bioprinting of Cardiac Tissues. Adv. Mater. Technol. 2022, 7, 2101636. [Google Scholar] [CrossRef]
- Kato, B.; Wisser, G.; Agrawal, D.K.; Wood, T.; Thankam, F.G. 3D bioprinting of cardiac tissue: Current challenges and perspectives. J. Mater. Sci. Mater. Med. 2021, 32, 54. [Google Scholar] [CrossRef]
- Jia, W.; Gungor-Ozkerim, P.S.; Zhang, Y.S.; Yue, K.; Zhu, K.; Liu, W.; Pi, Q.; Byambaa, B.; Dokmeci, M.R.; Shin, S.R.; et al. Direct 3D bioprinting of perfusable vascular constructs using a blend bioink. Biomaterials 2016, 106, 58–68. [Google Scholar] [CrossRef] [PubMed]
- Sanjuan-Alberte, P.; Whitehead, C.; Jones, J.N.; Silva, J.C.; Carter, N.; Kellaway, S.; Hague, R.J.; Cabral, J.M.; Ferreira, F.C.; White, L.J.; et al. Printing biohybrid materials for bioelectronic cardio-3D-cellular constructs. iScience 2022, 25, 104552. [Google Scholar] [CrossRef]
- Tamay, D.G.; Usal, T.D.; Alagoz, A.S.; Yucel, D.; Hasirci, N.; Hasirci, V. 3D and 4D Printing of Polymers for Tissue Engineering Applications. Front. Bioeng. Biotechnol. 2019, 7, 164. [Google Scholar] [CrossRef] [PubMed]
- Saska, S.; Pilatti, L.; Blay, A.; Shibli, J. Bioresorbable Polymers: Advanced Materials and 4D Printing for Tissue Engineering. Polymers 2021, 13, 563. [Google Scholar] [CrossRef]
- Cox-Pridmore, D.M.; Castro, F.A.; Silva, S.R.P.; Camelliti, P.; Zhao, Y. Emerging Bioelectronic Strategies for Cardiovascular Tissue Engineering and Implantation. Small 2022, 18, 2105281. [Google Scholar] [CrossRef] [PubMed]
- Yong, U.; Kang, B.; Jang, J. 3D bioprinted and integrated platforms for cardiac tissue modeling and drug testing. Essays Biochem. 2021, 65, 545–554. [Google Scholar] [CrossRef]
- Miao, S.; Castro, N.; Nowicki, M.; Xia, L.; Cui, H.; Zhou, X.; Zhu, W.; Lee, S.-J.; Sarkar, K.; Vozzi, G.; et al. 4D printing of polymeric materials for tissue and organ regeneration. Mater. Today 2017, 20, 577–591. [Google Scholar] [CrossRef]
- Wang, Y.; Cui, H.; Wang, Y.; Xu, C.; Esworthy, T.J.; Hann, S.Y.; Boehm, M.; Shen, Y.-L.; Mei, D.; Zhang, L.G. 4D Printed Cardiac Construct with Aligned Myofibers and Adjustable Curvature for Myocardial Regeneration. ACS Appl. Mater. Interfaces 2021, 13, 12746–12758. [Google Scholar] [CrossRef]
- Ma, S.; Jiang, Z.; Wang, M.; Zhang, L.; Liang, Y.; Zhang, Z.; Ren, L.; Ren, L. 4D printing of PLA/PCL shape memory composites with controllable sequential deformation. Bio-Design Manuf. 2021, 4, 867–878. [Google Scholar] [CrossRef]
- Pedron, S.; van Lierop, S.; Horstman, P.; Penterman, R.; Broer, D.J.; Peeters, E. Stimuli Responsive Delivery Vehicles for Cardiac Microtissue Transplantation. Adv. Funct. Mater. 2011, 21, 1624–1630. [Google Scholar] [CrossRef]
- Abraham, W.T.; Perl, L. Implantable Hemodynamic Monitoring for Heart Failure Patients. J. Am. Coll. Cardiol. 2017, 70, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Xie, X.; Duan, Y.; Wang, L.; Cheng, Z.; Cheng, J. A review of impedance measurements of whole cells. Biosens. Bioelectron. 2015, 77, 824–836. [Google Scholar] [CrossRef]
- Vashistha, R.; Dangi, A.K.; Kumar, A.; Chhabra, D.; Shukla, P. Futuristic biosensors for cardiac health care: An artificial intelligence approach. 3 Biotech 2018, 8, 358. [Google Scholar] [CrossRef] [PubMed]
- Dagdeviren, C.; Yang, B.D.; Su, Y.; Tran, P.L.; Joe, P.; Anderson, E.; Xia, J.; Doraiswamy, V.; Dehdashti, B.; Feng, X.; et al. Conformal piezoelectric energy harvesting and storage from motions of the heart, lung, and diaphragm. Proc. Natl. Acad. Sci. USA 2014, 111, 1927–1932. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Qian, X.; Li, N.; Cui, D.; Zhang, H.; Xu, Z. An experimental study on a piezoelectric vibration energy harvester for self-powered cardiac pacemakers. Ann. Transl. Med. 2021, 9, 880. [Google Scholar] [CrossRef] [PubMed]
- Hwang, G.-T.; Byun, M.; Jeong, C.K.; Lee, K.J. Flexible Piezoelectric Thin-Film Energy Harvesters and Nanosensors for Biomedical Applications. Adv. Healthc. Mater. 2015, 4, 646–658. [Google Scholar] [CrossRef]
- Sun, J.; Yang, A.; Zhao, C.; Liu, F.; Li, Z. Recent progress of nanogenerators acting as biomedical sensors in vivo. Science Bulletin. 2019, 64, 1336–1347. [Google Scholar] [CrossRef]
- Yu, Y.; Li, Z.; Wang, Y.; Gong, S.; Wang, X. Sequential Infiltration Synthesis of Doped Polymer Films with Tunable Electrical Properties for Efficient Triboelectric Nanogenerator Development. Adv. Mater. 2015, 27, 4938–4944. [Google Scholar] [CrossRef]
- Wang, H.; Xu, L.; Wang, Z. Advances of High-Performance Triboelectric Nanogenerators for Blue Energy Harvesting. Nanoenergy Adv. 2021, 1, 32–57. [Google Scholar] [CrossRef]
- Pongampai, S.; Charoonsuk, T.; Pinpru, N.; Pulphol, P.; Vittayakorn, W.; Pakawanit, P.; Vittayakorn, N. Triboelectric-piezoelectric hybrid nanogenerator based on BaTiO3-Nanorods/Chitosan enhanced output performance with self-charge-pumping system. Compos. Part B Eng. 2021, 208, 108602. [Google Scholar] [CrossRef]
- Mariello, M.; Kim, K.; Wu, K.; Lacour, S.P.; Leterrier, Y. Recent Advances in Encapsulation of Flexible Bioelectronic Implants: Materials, Technologies, and Characterization Methods. Adv. Mater. 2022, 34, e2201129. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Li, B.; Jia, W.; Zhu, Z. Predicting output performance of triboelectric nanogenerators using deep learning model. Nano Energy 2022, 93, 106830. [Google Scholar] [CrossRef]

| Heart Energy Harvesters (HEHs) | Working Principle | Advantages | Disadvantages |
|---|---|---|---|
| Piezoelectric nanogenerators (PNGs) | The energy generation is provided by the intrinsic properties of piezoelectric materials which convert even quasi-static mechanical deformation into electrical signals and power. |
|
|
| Triboelectric nanogenerators (TNGs) | The energy generation is provided by the contact or sliding between two dissimilar materials with different surface properties. |
|
|
| Hybrid nanogenerators (HNGs) | The energy generation is provided by the hybridization of piezoelectricity and triboelectricity in a single device. |
|
|
| Solar cells (SCs) | The energy generation is provided by the interaction of light with the materials of the SC. |
|
|
| Biofuel Cells (BFCs) | The energy generation is provided by electron transfer during electrochemical reactions. |
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mariello, M. Heart Energy Harvesting and Cardiac Bioelectronics: Technologies and Perspectives. Nanoenergy Adv. 2022, 2, 344-385. https://doi.org/10.3390/nanoenergyadv2040018
Mariello M. Heart Energy Harvesting and Cardiac Bioelectronics: Technologies and Perspectives. Nanoenergy Advances. 2022; 2(4):344-385. https://doi.org/10.3390/nanoenergyadv2040018
Chicago/Turabian StyleMariello, Massimo. 2022. "Heart Energy Harvesting and Cardiac Bioelectronics: Technologies and Perspectives" Nanoenergy Advances 2, no. 4: 344-385. https://doi.org/10.3390/nanoenergyadv2040018
APA StyleMariello, M. (2022). Heart Energy Harvesting and Cardiac Bioelectronics: Technologies and Perspectives. Nanoenergy Advances, 2(4), 344-385. https://doi.org/10.3390/nanoenergyadv2040018







