Biological Evaluation of Alkyl Triphenylphosphonium Ostruthin Derivatives as Potential Anti-Inflammatory Agents Targeting the Nuclear Factor κB Signaling Pathway in Human Lung Adenocarcinoma A549 Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cells
2.2. Reagents
2.3. Cell-ELISA
2.4. Cell Viability Assay
2.5. Reporter Assay
2.6. Flow Cytometry
2.7. Real-Time PCR
2.8. Western Blotting
2.9. ChIP Assay
2.10. Statistical Analysis
3. Results
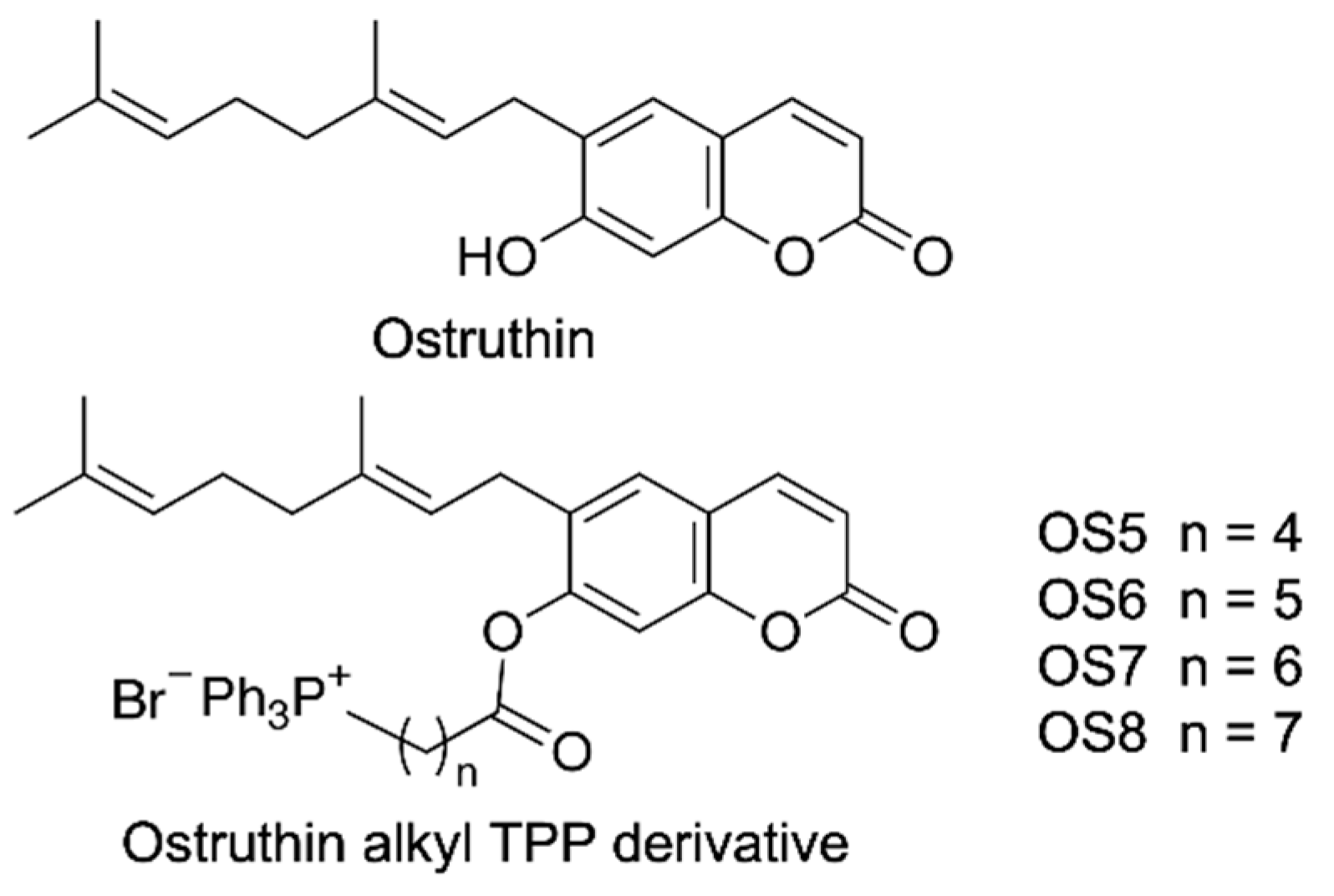
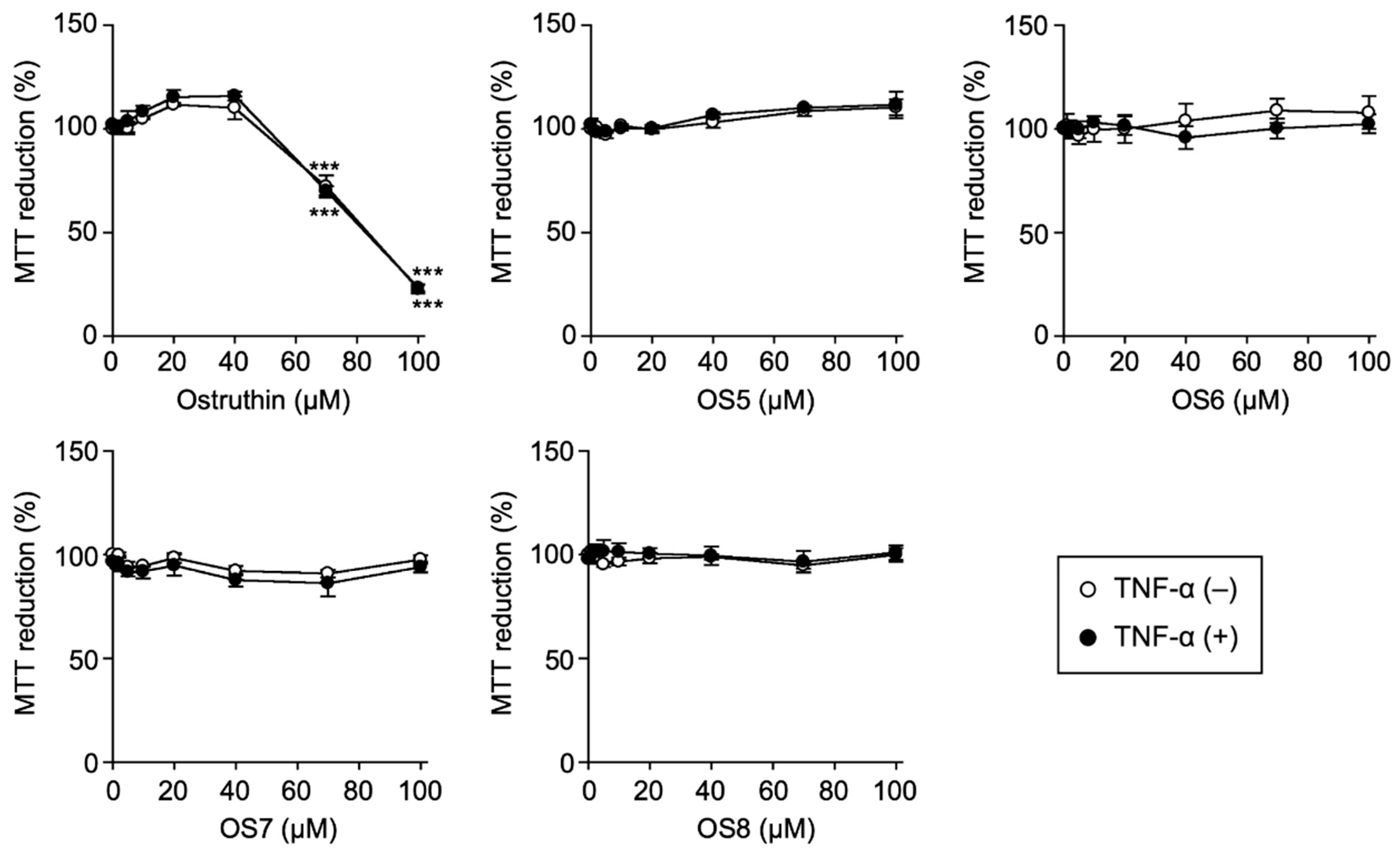
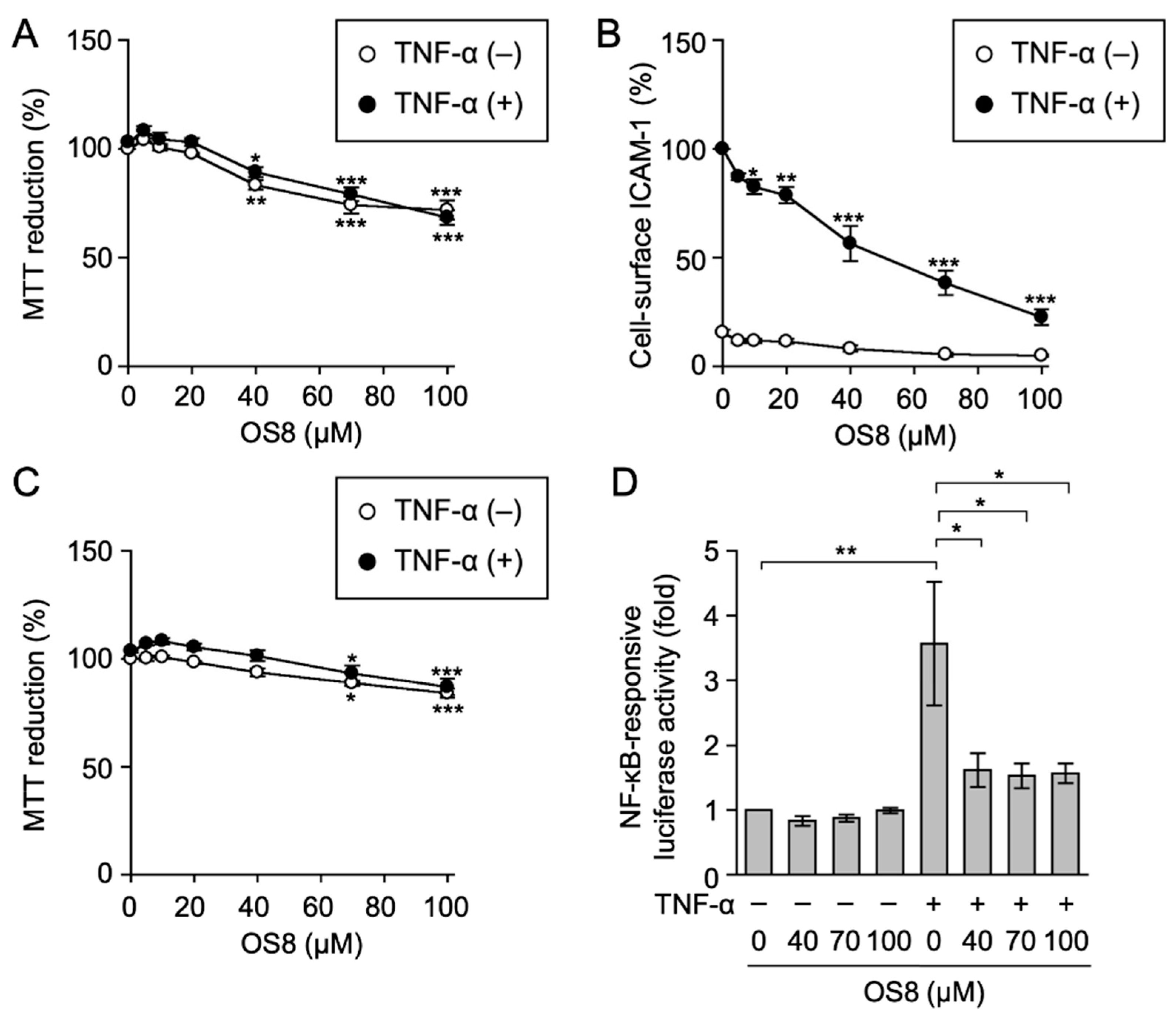
3.1. Biological Evaluation of Ostruthin and Its Alkyl TPP Derivatives on the Viability of A549 Cells
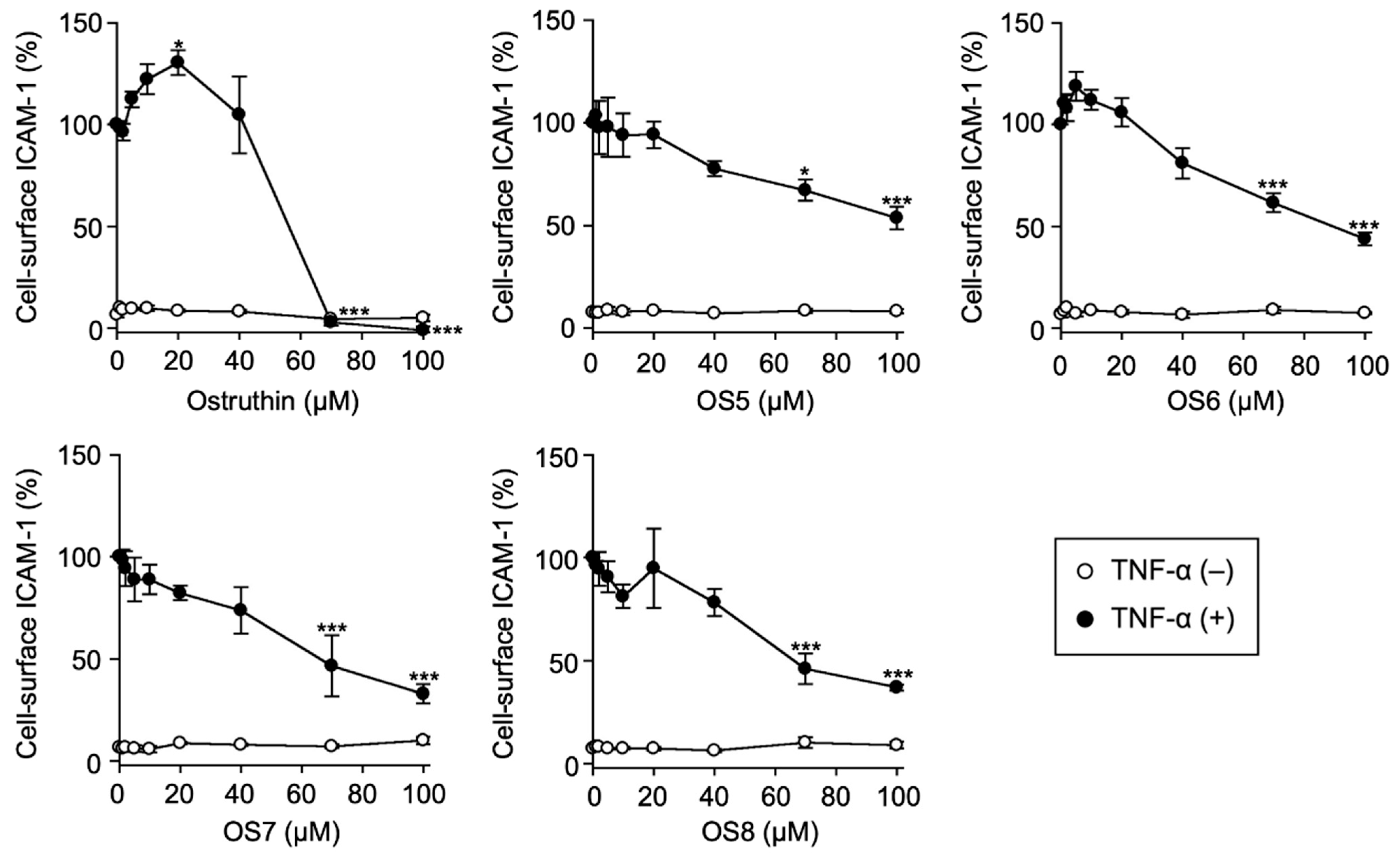
3.2. Biological Evaluation of Ostruthin and Its Alkyl TPP Derivatives on the TNF-α-Induced Expression of Cell-Surface ICAM-1 in A549 Cells
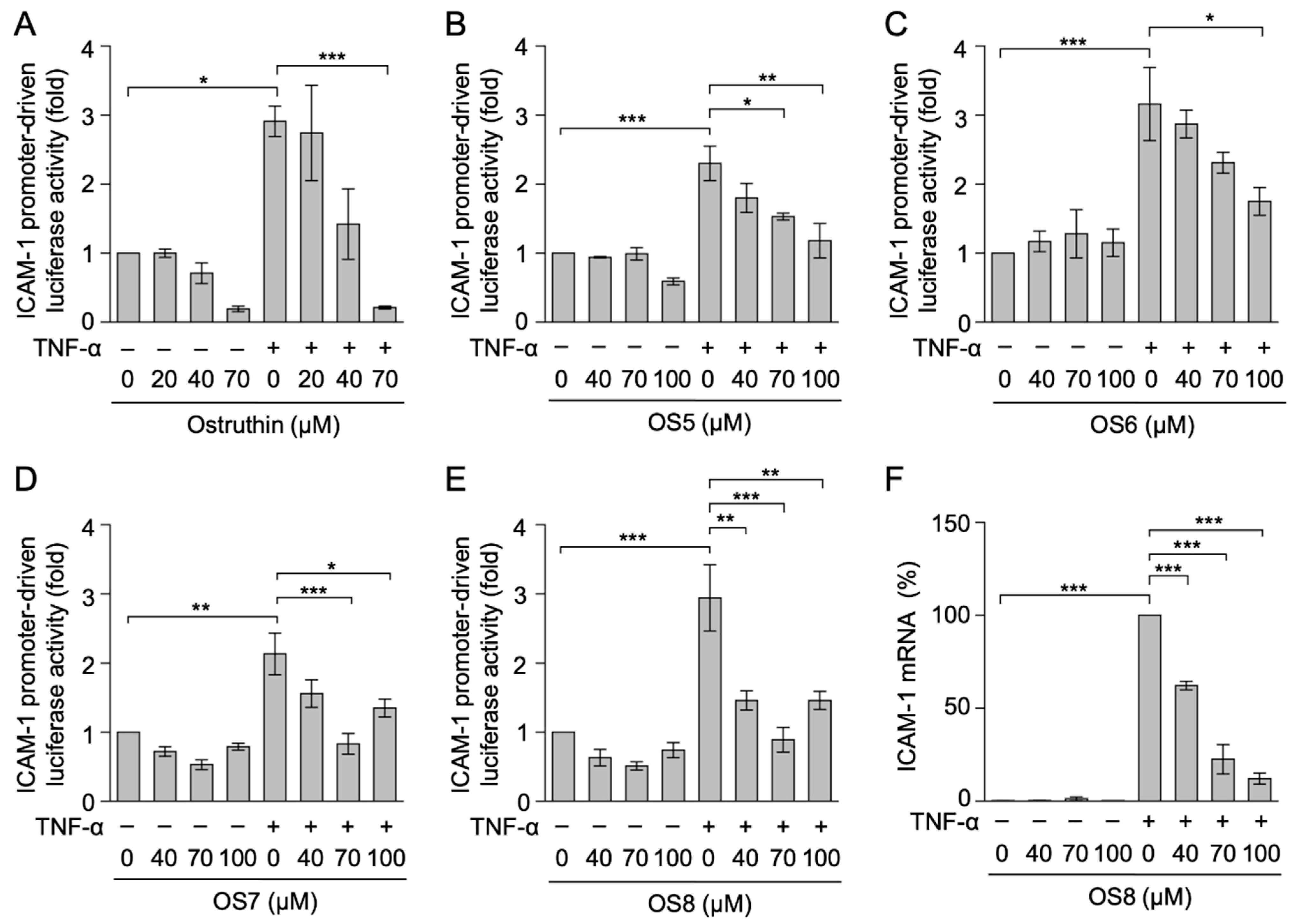
3.3. Biological Evaluation of Ostruthin and Its Alkyl TPP Derivatives on TNF-α-Induced ICAM-1 Promoter-Driven Luciferase Activity in A549 Cells
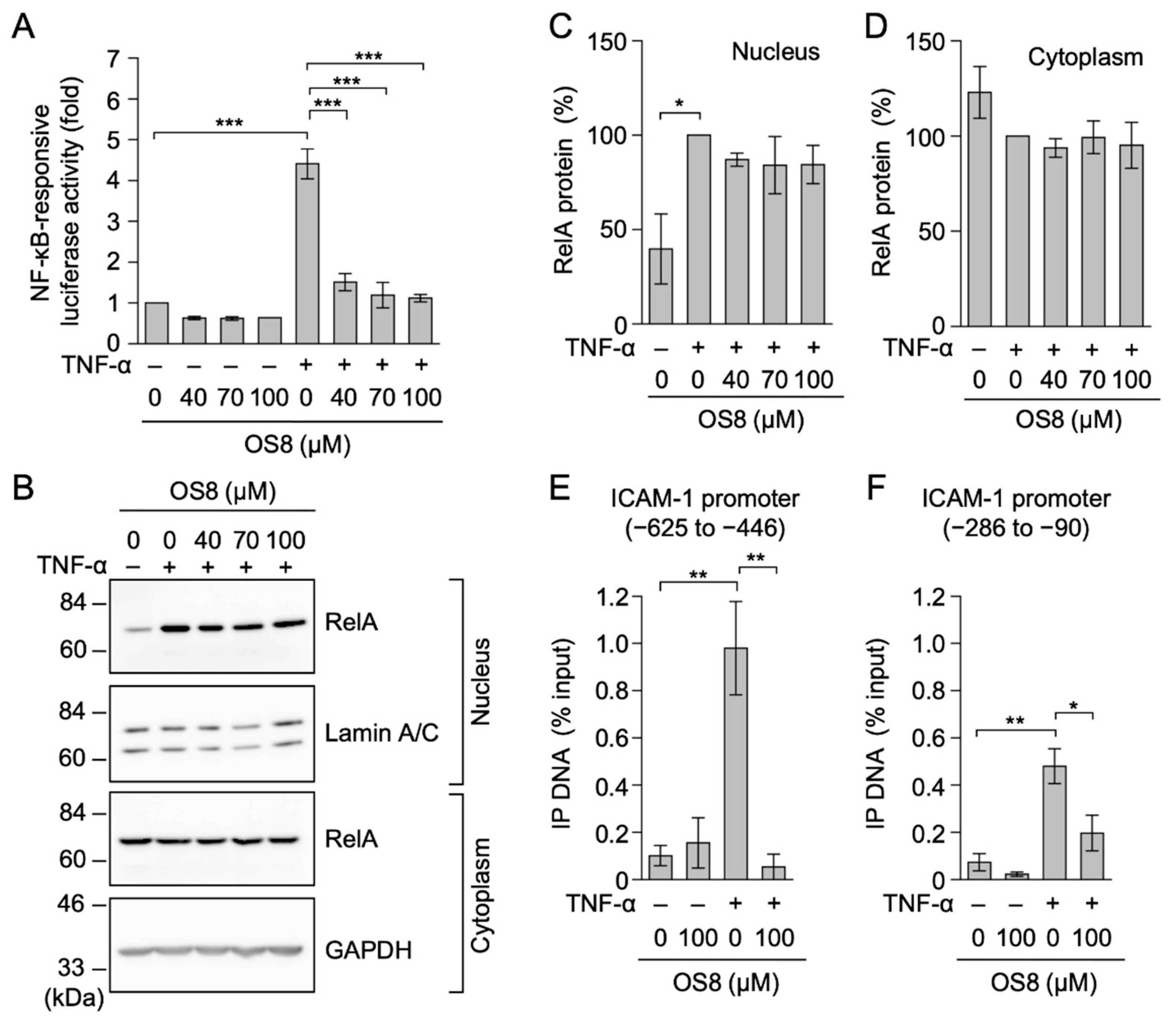
3.4. OS8 Prevented TNF-α-Induced ICAM-1 Expression and NF-κB-Responsive Luciferase Activity in HT-1080 Cells
3.5. OS8 Prevented the Binding of RelA to the ICAM-1 Promoter in TNF-α-Stimulated A549 Cells
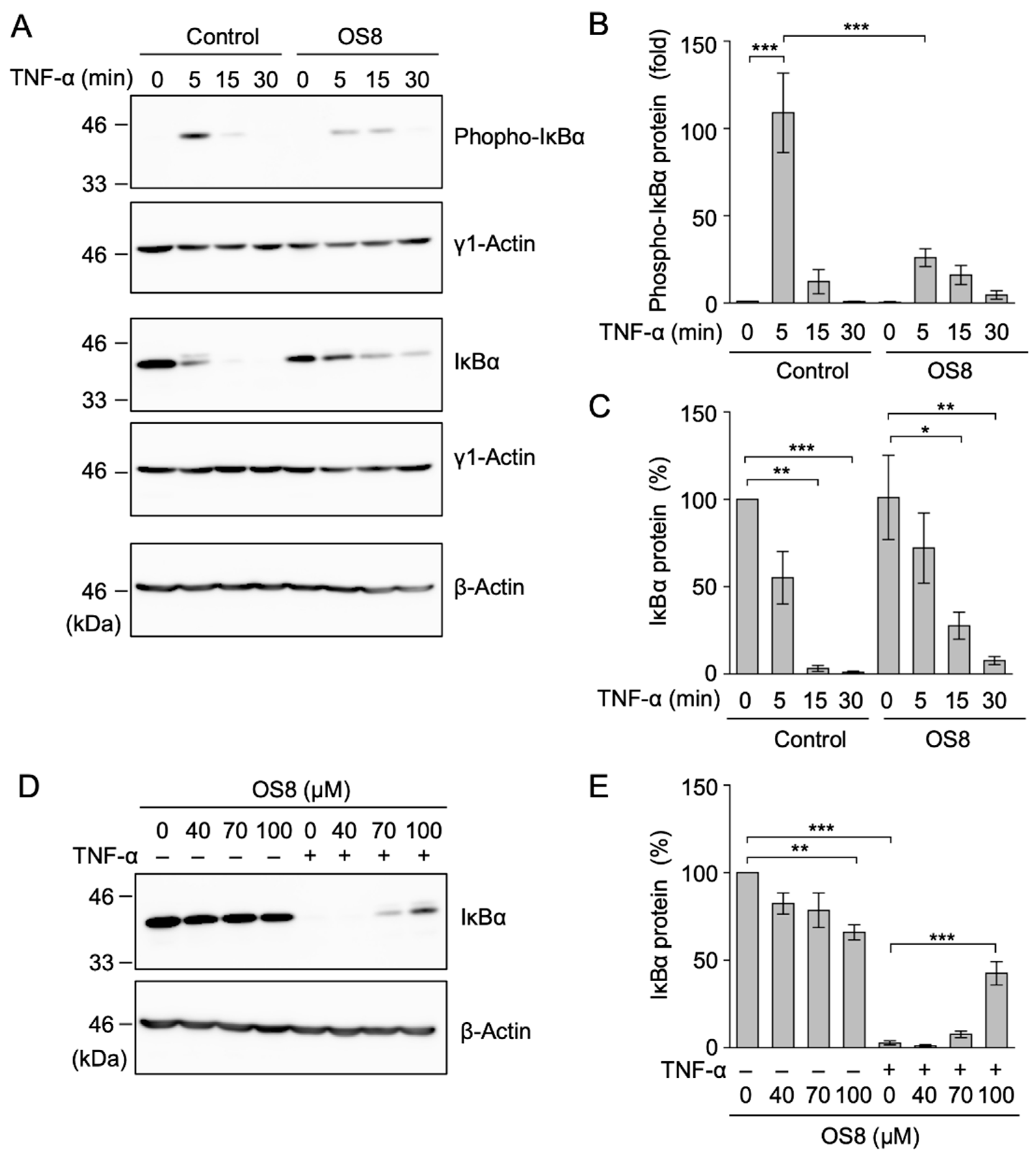
3.6. OS8 Inhibited the Phosphorylation of IκBα in TNF-α-Stimulated A549 Cells
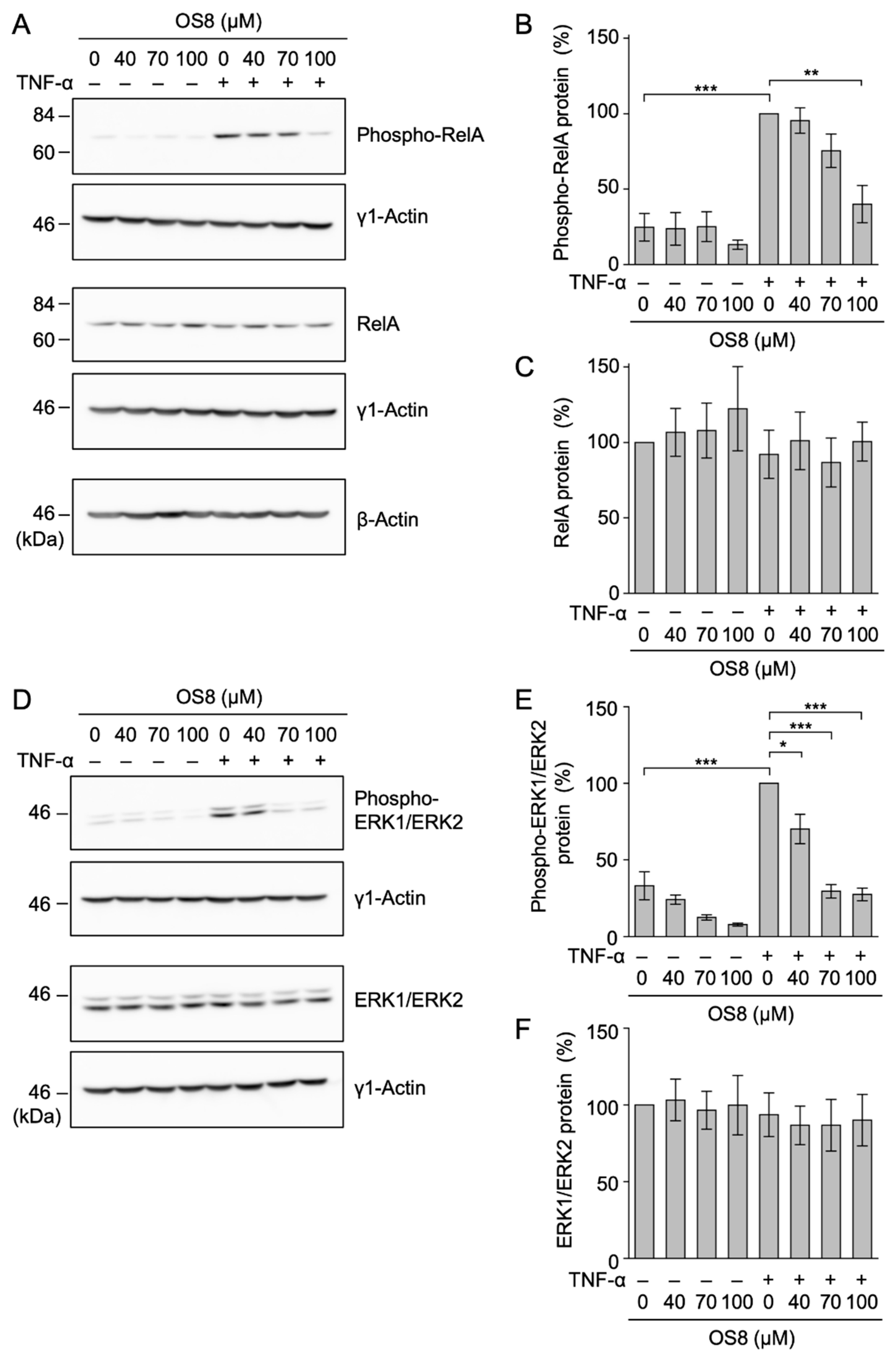
3.7. OS8 Inhibited the Phosphorylation of RelA and ERK1/ERK2 in TNF-α-Stimulated A549 Cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gerhardt, T.; Ley, K. Monocyte trafficking across the vessel wall. Cardiovasc. Res. 2015, 107, 321–330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vestweber, D. How leukocytes cross the vascular endothelium. Nat. Rev. Immunol. 2015, 15, 692–704. [Google Scholar] [CrossRef] [PubMed]
- Van de Stolpe, A.; Van der Saag, P.T. Intercellular adhesion molecule-1. J. Mol. Med. 1996, 74, 13–33. [Google Scholar] [CrossRef] [PubMed]
- Collins, T.; Read, M.A.; Neish, A.S.; Whitley, M.Z.; Thanos, D.; Maniatis, T. Transcriptional regulation of endothelial cell adhesion molecules: NF-κB and cytokine-inducible enhancers. FASEB J. 1995, 9, 899–909. [Google Scholar] [CrossRef] [Green Version]
- Roebuck, K.A.; Finnegan, A. Regulation of intercellular adhesion molecule-1 (CD54) gene expression. J. Leukoc. Biol. 1999, 66, 876–888. [Google Scholar] [CrossRef]
- Bhoj, V.G.; Chen, Z.J. Ubiquitylation in innate and adaptive immunity. Nature 2009, 458, 430–437. [Google Scholar] [CrossRef]
- Hayden, M.S.; Ghosh, S. Regulation of NF-κB by TNF family cytokines. Semin. Immunol. 2014, 26, 253–266. [Google Scholar] [CrossRef] [Green Version]
- Karin, M.; Greten, F.R. NF-κB: Linking inflammation and immunity to cancer development and progression. Nat. Rev. Immunol. 2005, 5, 749–759. [Google Scholar] [CrossRef]
- Stefanachi, A.; Leonetti, F.; Pisani, L.; Catto, M.; Carotti, A. Coumarin: A natural, privileged and versatile scaffold for bioactive compounds. Molecules 2018, 23, 250. [Google Scholar] [CrossRef] [Green Version]
- Venugopala, K.N.; Rashmi, V.; Odhav, B. Review on natural coumarin lead compounds for their pharmacological activity. BioMed Res. Int. 2013, 2013, 963248. [Google Scholar] [CrossRef] [Green Version]
- Dang, P.H.; Le, T.H.; Phan, P.K.T.; Le, P.T.T.; Nguyen, M.T.T.; Nguyen, N.T. Two acridones and two coumarins from the roots of Paramignya trimera. Tetrahedron Lett. 2017, 58, 1553–1557. [Google Scholar] [CrossRef]
- Khalid, S.A.; Farouk, A.; Geary, T.G.; Jensen, J.B. Potential antimalarial candidates from African plants: An in vitro approach using Plasmodium falciparum. J. Ethnopharmacol. 1986, 15, 201–209. [Google Scholar] [CrossRef]
- Schinkovitz, A.; Gibbons, S.; Stavri, M.; Cocksedge, M.J.; Bucar, F. Ostruthin: An antimycobacterial coumarin from the roots of Peucedanum ostruthium. Planta Med. 2003, 69, 369–371. [Google Scholar] [CrossRef] [Green Version]
- Li, F.; Okamura, Y.; Dibwe, D.F.; Awale, S.; Kadota, S.; Tezuka, Y. Anti-austerity agents from Rhizoma et Radix Notopterygii (Qianghuo). Planta Med. 2012, 78, 796–799. [Google Scholar] [CrossRef] [Green Version]
- Tuan Anh, H.L.; Kim, D.C.; Ko, W.; Ha, T.M.; Nhiem, N.X.; Yen, P.H.; Tai, B.H.; Truong, L.H.; Long, V.N.; Gioi, T.; et al. Anti-inflammatory coumarins from Paramignya trimera. Pharm. Biol. 2017, 55, 1195–1201. [Google Scholar] [CrossRef] [Green Version]
- Zwirchmayr, J.; Grienke, U.; Hummelbrunner, S.; Seigner, J.; de Martin, R.; Dirsch, V.M.; Rollinger, J.M. A Biochemometric approach for the identification of in vitro anti-inflammatory constituents in masterwort. Biomolecules 2020, 10, 679. [Google Scholar] [CrossRef] [PubMed]
- Dang, P.H.; Dao, T.H.X.; Le, V.T.; Nguyen, C.M.; Ly, T.T.; Nguyen, H.X.; Le, T.H.; Do, T.N.V.; Nguyen, M.T.T.; Sun, S.; et al. Synthesis of alkyl triphenylphosphonium ostruthin derivatives as potential cytotoxic candidates. ChemistrySelect 2020, 5, 12636–12640. [Google Scholar] [CrossRef]
- Zielonka, J.; Joseph, J.; Sikora, A.; Hardy, M.; Ouari, O.; Vasquez-Vivar, J.; Cheng, G.; Lopez, M.; Kalyanaraman, B. Mitochondrial-targeted triphenylphosphonium-based compounds: Syntheses, mechanisms of action, and therapeutic and diagnostic applications. Chem. Rev. 2017, 117, 10043–10120. [Google Scholar] [CrossRef] [PubMed]
- Yamada, Y.; Taketani, S.; Osada, H.; Kataoka, T. Cytotrienin A, a translation inhibitor that induces ectodomain shedding of TNF receptor 1 via activation of ERK and p38 MAP kinase. Eur. J. Pharmacol. 2011, 667, 113–119. [Google Scholar] [CrossRef]
- Mitsuda, S.; Yokomichi, T.; Yokoigawa, J.; Kataoka, T. Ursolic acid, a natural pentacyclic triterpenoid, inhibits intracellular trafficking of proteins and induces accumulation of intercellular adhesion molecule-1 linked to high-mannose-type glycans in the endoplasmic reticulum. FEBS Open Bio 2014, 4, 229–239. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fukuhara, S.; Tanigaki, R.; Kimura, K.I.; Kataoka, T. Kujigamberol interferes with pro-inflammatory cytokine-induced expression of and N-glycan modifications to cell adhesion molecules at different stages in human umbilical vein endothelial cells. Int. Immunopharmacol. 2018, 62, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Dohrman, A.; Kataoka, T.; Cuenin, S.; Russell, J.Q.; Tschopp, J.; Budd, R.C. Cellular FLIP (long form) regulates CD8+ T cell activation through caspase-8-dependent NF-κB activation. J. Immunol. 2005, 174, 5270–5278. [Google Scholar] [CrossRef] [Green Version]
- Matsuda, I.; Matsuo, K.; Matsushita, Y.; Haruna, Y.; Niwa, M.; Kataoka, T. The C-terminal domain of the long form of cellular FLICE-inhibitory protein (c-FLIPL) inhibits the interaction of the caspase 8 prodomain with the receptor-interacting protein 1 (RIP1) death domain and regulates caspase 8-dependent nuclear factor κB (NF-κB) activation. J. Biol. Chem. 2014, 289, 3876–3887. [Google Scholar] [PubMed] [Green Version]
- Vo, T.N.; Sasaki, S.; Miyake, Y.; Nguyen, N.T.; Dang, P.H.; Nguyen, M.T.T.; Kataoka, T. α-Conidendrin inhibits the expression of intercellular adhesion molecule-1 induced by tumor necrosis factor-α in human lung adenocarcinoma A549 cells. Eur. J. Pharmacol. 2021, 890, 173651. [Google Scholar] [CrossRef]
- Harada, M.; Morimoto, K.; Kondo, T.; Hiramatsu, R.; Okina, Y.; Muko, R.; Matsuda, I.; Kataoka, T. Quinacrine inhibits ICAM-1 transcription by blocking DNA binding of the NF-κB subunit p65 and sensitizes human lung adenocarcinoma A549 cells to TNF-α and the Fas ligand. Int. J. Mol. Sci. 2017, 18, 2603. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wan, M.; Liu, J.; Ouyang, X. Nucleotide-binding oligomerization domain 1 regulates Porphyromonas gingivalis-induced vascular cell adhesion molecule 1 and intercellular adhesion molecule 1 expression in endothelial cell through NF-κB pathway. J. Periodont. Res. 2015, 50, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lian, F.; Zhu, Y.; Xia, M.; Wang, Q.; Ling, W.; Wang, X.D. Cyanidin-3-O-β-glucoside inhibits LPS-induced expression of inflammatory mediators through decreasing IκBα phosphorylation in THP-1 cells. Inflamm. Res. 2010, 59, 723–730. [Google Scholar] [CrossRef]
- Hirano, S.; Kataoka, T. Deoxynivalenol induces ectodomain shedding of TNF receptor 1 and thereby inhibits the TNF-α-induced NF-κB signaling pathway. Eur. J. Pharmacol. 2013, 701, 144–151. [Google Scholar] [CrossRef]
- Fukuoka, N.; Harada, M.; Nishida, A.; Ito, Y.; Shiota, H.; Kataoka, T. Eomesodermin promotes interferon-γ expression and binds to multiple conserved noncoding sequences across the Ifng locus in mouse thymoma cell lines. Genes Cells 2016, 21, 146–162. [Google Scholar] [CrossRef]
- Xue, J.; Thippegowda, P.B.; Hu, G.; Bachmaier, K.; Christman, J.W.; Malik, A.B.; Tiruppathi, C. NF-κB regulates thrombin-induced ICAM-1 gene expression in cooperation with NFAT by binding to the intronic NF-κB site in the ICAM-1 gene. Physiol. Genom. 2009, 38, 42–53. [Google Scholar] [CrossRef] [Green Version]
- Tanigaki, R.; Takahashi, R.; Nguyen, M.T.T.; Nguyen, N.T.; Do, T.V.N.; Nguyen, H.X.; Kataoka, T. 4-Hydroxypanduratin A and isopanduratin A inhibit tumor necrosis factor α-stimulated gene expression and the nuclear factor κB-dependent signaling pathway in human lung adenocarcinoma A549 cells. Biol. Pharm. Bull. 2019, 42, 26–33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, Z.; Kang, W.; Wang, L.; Sun, B.; Ma, J.; Zheng, C.; Sun, J.; Tian, Z.; Yang, X.; Xiao, W. E2F1 renders prostate cancer cell resistant to ICAM-1 mediated antitumor immunity by NF-κB modulation. Mol. Cancer 2014, 13, 84. [Google Scholar] [CrossRef] [Green Version]
- Perkins, N.D. Post-translational modifications regulating the activity and function of the nuclear factor kappa B pathway. Oncogene 2006, 25, 6717–6730. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hoesel, B.; Schmid, J.A. The complexity of NF-κB signaling in inflammation and cancer. Mol. Cancer 2013, 12, 86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Christian, F.; Smith, E.L.; Carmody, R.J. The regulation of NF-κB subunits by phosphorylation. Cells 2016, 5, 12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, B.; Wang, Z.; Liu, Y.; Xu, S.; Huang, G.; Xiong, Y.; Zhang, S.; Xu, L.; Deng, X.; Guan, S. Immunosuppressive activity of daphnetin, one of coumarin derivatives, is mediated through suppression of NF-κB and NFAT signaling pathways in mouse T cells. PLoS ONE 2014, 9, e96502. [Google Scholar] [CrossRef]
- Xu, K.; Guo, L.; Bu, H.; Wang, H. Daphnetin inhibits high glucose-induced extracellular matrix accumulation, oxidative stress and inflammation in human glomerular mesangial cells. J. Pharm. Sci. 2019, 139, 91–97. [Google Scholar] [CrossRef]
- Qi, J.; Ojika, M.; Sakagami, Y. Differentiation in a rat PC12 cell line induced by ostruthin and (–)-bornyl ferulate, constituents of a Chinese herbal medicine. Biosci. Biotechnol. Biochem. 1999, 63, 1501–1502. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Scarlett, C.J. Cytotoxic activity of extracts and fractions from Paramignya trimera root and Phyllanthus amarus against pancreatic cancer cell lines. J. Cancer Res. Ther. 2019, 15, 245–249. [Google Scholar] [CrossRef]
- Joa, H.; Vogl, S.; Atanasov, A.G.; Zehl, M.; Nakel, T.; Fakhrudin, N.; Heiss, E.K.; Picker, P.; Urban, E.; Wawrosch, C.; et al. Identification of ostruthin from Peucedanum ostruthium rhizomes as an inhibitor of vascular smooth muscle cell proliferation. J. Nat. Prod. 2011, 74, 1513–1516. [Google Scholar] [CrossRef]
- Nguyen, V.T.; Sakoff, J.A.; Scarlett, C.J. Physicochemical, antioxidant, and cytotoxic properties of Xao tam phan (Paramignya trimera) root extract and its fractions. Chem. Divers. 2017, 14, e1600396. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.T.; Sakoff, J.A.; Scarlett, C.J. Physicochemical properties, antioxidant, and anti-proliferative capacities of dried leaf and its extract from Xao tam phan (Paramignya trimera). Chem. Biodivers. 2017, 14, e1600498. [Google Scholar] [CrossRef]
- Cai, Y.; Bennett, D.; Nair, R.V.; Ceska, O.; Ashwood-Smith, M.J.; DiGiovanni, J. Inhibition and inactivation of murine hepatic ethoxy- and petoxyresorufin O-dealkylase by naturally occurring coumarins. Chem. Res. Toxicol. 1993, 6, 872–879. [Google Scholar] [CrossRef]
- Urbain, A.; Marston, A.; Hostettmann, K. Coumarins from Peucedanum ostruthium as inhibitors of acetylcholinesterase. Pharm. Biol. 2005, 43, 647–650. [Google Scholar] [CrossRef] [Green Version]
- Trinh, D.H.; Tran, P.H.; Trinh, B.T.D.; Nguyen, H.T.; Nguyen, H.D.; Ha, L.D.; Nguyen, L.H.D. Coumarins and acridone alkaloids with α-glucosidase inhibitory and antioxidant activity from the roots of Paramignya trimera. Phytochem. Lett. 2020, 35, 94–98. [Google Scholar] [CrossRef]
- Joseph, A.; Thuy, T.T.T.; Thanh, L.T.; Okada, M. Antidepressive and anxiolytic effects of ostruthin, a TREK-1 channel activator. PLoS ONE 2018, 13, e0201092. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, J.; Nakano, H.; Sakurai, H.; Colburn, N.H. Insufficient p65 phosphorylation at S536 specifically contributes to the lack of NF-κB activation and transformation in resistant JB6 cells. Carcinogenesis 2004, 25, 1991–2003. [Google Scholar] [CrossRef] [PubMed] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vo, N.T.; Kusagawa, E.; Nakano, K.; Moriwaki, C.; Miyake, Y.; Haruyama, S.; Fukuhara, S.; Nguyen, N.T.; Dang, P.H.; Nguyen, M.T.T.; et al. Biological Evaluation of Alkyl Triphenylphosphonium Ostruthin Derivatives as Potential Anti-Inflammatory Agents Targeting the Nuclear Factor κB Signaling Pathway in Human Lung Adenocarcinoma A549 Cells. BioChem 2021, 1, 107-121. https://doi.org/10.3390/biochem1020010
Vo NT, Kusagawa E, Nakano K, Moriwaki C, Miyake Y, Haruyama S, Fukuhara S, Nguyen NT, Dang PH, Nguyen MTT, et al. Biological Evaluation of Alkyl Triphenylphosphonium Ostruthin Derivatives as Potential Anti-Inflammatory Agents Targeting the Nuclear Factor κB Signaling Pathway in Human Lung Adenocarcinoma A549 Cells. BioChem. 2021; 1(2):107-121. https://doi.org/10.3390/biochem1020010
Chicago/Turabian StyleVo, Nghia Trong, Eiichi Kusagawa, Kaori Nakano, Chihiro Moriwaki, Yasunobu Miyake, Sayaka Haruyama, Sayuri Fukuhara, Nhan Trung Nguyen, Phu Hoang Dang, Mai Thanh Thi Nguyen, and et al. 2021. "Biological Evaluation of Alkyl Triphenylphosphonium Ostruthin Derivatives as Potential Anti-Inflammatory Agents Targeting the Nuclear Factor κB Signaling Pathway in Human Lung Adenocarcinoma A549 Cells" BioChem 1, no. 2: 107-121. https://doi.org/10.3390/biochem1020010
APA StyleVo, N. T., Kusagawa, E., Nakano, K., Moriwaki, C., Miyake, Y., Haruyama, S., Fukuhara, S., Nguyen, N. T., Dang, P. H., Nguyen, M. T. T., & Kataoka, T. (2021). Biological Evaluation of Alkyl Triphenylphosphonium Ostruthin Derivatives as Potential Anti-Inflammatory Agents Targeting the Nuclear Factor κB Signaling Pathway in Human Lung Adenocarcinoma A549 Cells. BioChem, 1(2), 107-121. https://doi.org/10.3390/biochem1020010






