Metagenomic Insights into the Composition and Function of Microbes Associated with the Rootzone of Datura inoxia
Abstract
:1. Introduction
2. Materials and Methods
3. Results
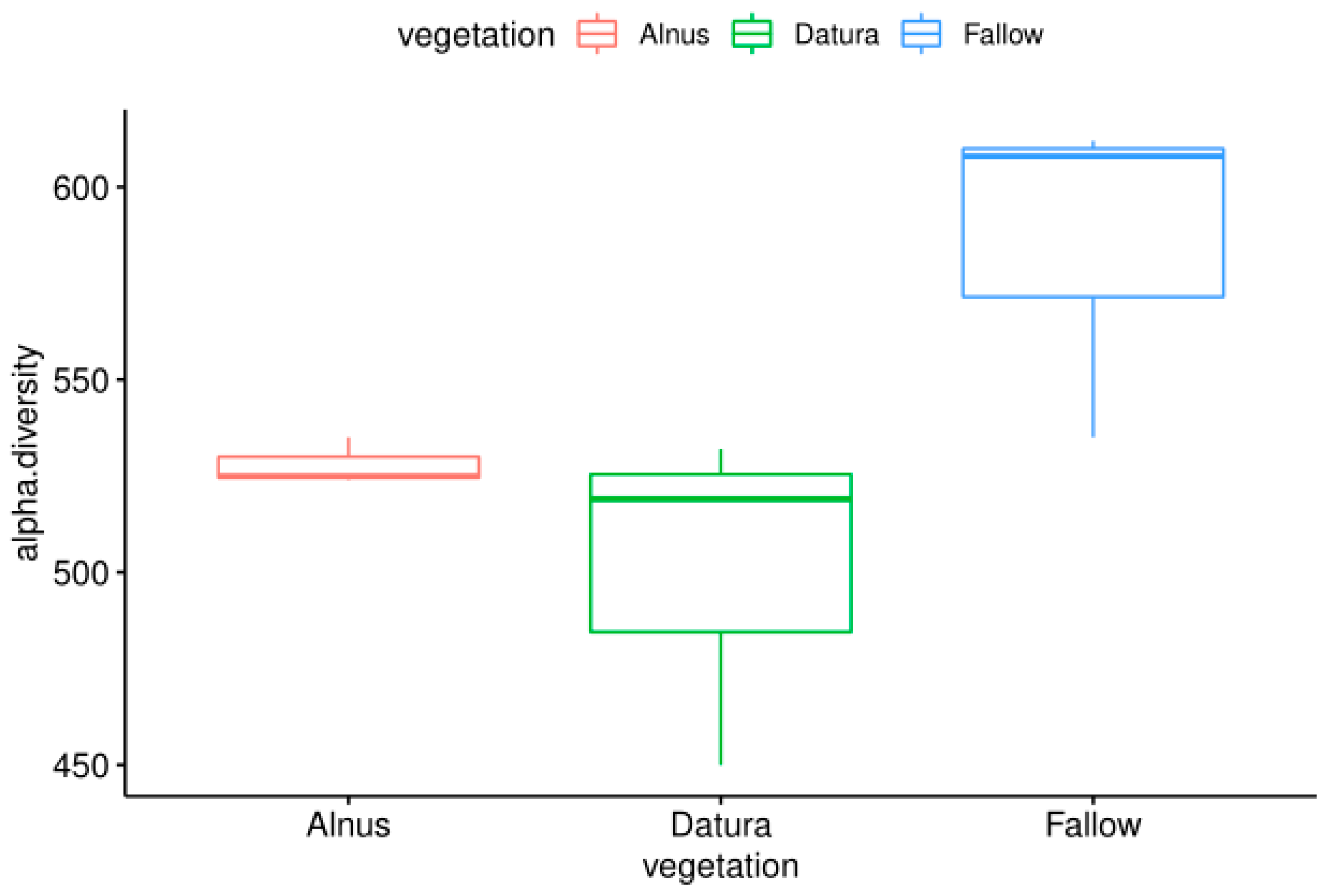
3.1. Soil Samples WGS and MG-RAST Taxonomic and Functional Assignments
3.2. Functional Analysis in STAMP
3.3. Purple Line Analysis and Comparison
3.4. Differential Abundance Analysis in DESeq2
3.5. Microbial Barcoding and Essential Oil Trial
4. Discussion
Genetic Engineering and the Flavonoid Biosynthesis Pathway
- Structural derivatives of phenyl-substituted propylbenzenes with a C15 backbone,
- Phenyl-substituted propylbenzene derivatives with a C16 skeleton, or
- Phenyl-substituted propylbenzenes condensed with C6-C3 lignan precursors to form flavonolignans.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sonia, P.; Siddiqui, A.A.; Dwivedi, J.; Sonia, V. Pharmacological properties of Datura stramonium L. as a potential medicinal tree: An overview. Asian Pac. J. Trop. Biomed. 2012, 2, 1002–1008. [Google Scholar] [CrossRef] [Green Version]
- Han, X.L.; Wang, H.; Zhang, Z.H.; Tan, Y.; Wang, J.H. Study on chemical constituents in seeds of Datura metel from Xinjiang. Zhong yao cai = Zhongyaocai = J. Chin. Med. Mater. 2015, 38, 1646–1648. [Google Scholar]
- Lemos Ari, S.O.; Florêncio Jônatas, R.; Pinto Nícolas, C.C.; Campos Lara, M.; Silva Thiago, P.; Grazul Richard, M.; Pinto Priscila, F.; Tavares Guilherme, D.; Elita, S.; Apolônio Ana Carolina, M.; et al. Antifungal Activity of the Natural Coumarin Scopoletin Against Planktonic Cells and Biofilms from a Multidrug-Resistant Candida tropicalis Strain. Front. Microbiol. 2020, 11, 1525. Available online: https://www.frontiersin.org/article/10.3389/fmicb.2020.01525 (accessed on 9 December 2021). [CrossRef] [PubMed]
- Bulcsu, S. Datura rituals in the vajramahabhairava-tantra. Acta Orient. Acad. Sci. Hung. 1994, 47, 409–416. Available online: www.jstor.org/stable/23658487 (accessed on 18 July 2021).
- Rajewski, A.; Carter-House, D.; Stajich, J.; Litt, A. Datura genome reveals duplications of psychoactive alkaloid biosynthetic genes and high mutation rate following tissue culture. BMC Genom. 2021, 22, 201. [Google Scholar] [CrossRef]
- Robinson, D.W.; Brown, K.; McMenemy, M.; Dennany, L.; Baker, M.J.; Allan, P.; Cartwright, C.; Bernard, J.; Sturt, F.; Kotoula, E.; et al. Datura quids at Pinwheel Cave, California, provide unambiguous confirmation of the ingestion of hallucinogens at a rock art site. Proc. Natl. Acad. Sci. USA 2020, 117, 31026–31037. [Google Scholar] [CrossRef] [PubMed]
- Rania, A.B.A.; Jabnoun-Khiareddine, H.; Nefzi, A.; Mokni-Tlili, S.; Daami-Remadi, M. Endophytic bacteria from Datura metel for plant growth promotion and bioprotection against Fusarium wilt in tomato. Biocontrol Sci. Technol. 2016, 26, 1139–1165. [Google Scholar] [CrossRef]
- Irmer, S.; Podzun, N.; Langel, D.; Heidemann, F.; Kaltenegger, E.; Schemmerling, B.; Geilfus, C.M.; Zörb, C.; Ober, D. New aspect of plant–rhizobia interaction: Alkaloid biosynthesis in Crotalaria depends on nodulation. Proc. Natl. Acad. Sci. USA 2015, 112, 4164–4169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhuyar, P.; Rahim, M.H.; Sundararaju, S.; Maniam, G.P.; Govindan, N. Antioxidant and antibacterial activity of red seaweed Kappaphycus alvarezii against pathogenic bacteria. Glob. J. Environ. Sci. Manag. 2019, 6, 47–58. [Google Scholar]
- Xia, Y.; Dong, M.; Yu, L.; Kong, L.; Seviour, R.; Kong, Y. Compositional and Functional Profiling of the Rhizosphere Microbiomes of the Invasive Weed Ageratina adenophora and Native Plants. PeerJ 2021, 9, E10844. [Google Scholar] [CrossRef] [PubMed]
- Sang, M.K.; Kim, K.D. The volatile-producing Flavobacterium johnsoniae strain GSE09 shows biocontrol activity against Phytophthora capsici in pepper. J. Appl. Microbiol. 2012, 113, 383–398. [Google Scholar] [CrossRef]
- Carrión, V.J.; Perez-Jaramillo, J.; Cordovez, V.; Tracanna, V.; De Hollander, M.; Ruiz-Buck, D.; Mendes, L.W.; Van Ijcken, W.F.J.; Gomez-Exposito, R.; Elsayed, S.S.; et al. Pathogen-induced activation of disease-suppressive functions in the endophytic root microbiome. Science 2019, 366, 606–612. [Google Scholar] [CrossRef] [PubMed]
- Miriam, L.; Gabriel, C.M.; Asaf, L. The Role of Secretion Systems, Effectors, and Secondary Metabolites of Beneficial Rhizobacteria in Interactions with Plants and Microbes. Front. Plant Sci. 2020, 11, 1718. Available online: https://www.frontiersin.org/article/10.3389/fpls.2020.589416 (accessed on 26 July 2021).
- del Barrio-Duque, A.; Ley, J.; Samad, A.; Antonielli, L.; Sessitsch, A.; Compant, S. Beneficial Endophytic Bacteria-Serendipita indica Interaction for Crop Enhancement and Resistance to Phytopathogens. Front. Microbiol. 2019, 10, 2888. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.S.; Facchini, P.J. Molecular Origins of Functional Diversity in Benzylisoquinoline Alkaloid Methyltransferases. Front. Plant Sci. 2019, 10, 1058. [Google Scholar] [CrossRef] [Green Version]
- Alfredo, B.L.; Daniel, C.; Susana, R.; Laura, C.L.; Rosana, S.; Castellani Lucas, G.; Torres, T.G.; Susana, B. Transfer of the Symbiotic Plasmid of Rhizobium etli CFN42 to Endophytic Bacteria Inside Nodules. Front. Microbiol. 2020, 11, 1752. [Google Scholar]
- Kado, C.I.; Kelly, B. Transformation of Actinomycetes. Agrobacterium Protoc. 2006, 2, 395–402. [Google Scholar]
- Wu, W.; Chen, W.; Liu, S.; Wu, J.; Zhu, Y.; Qin, L.; Zhu, B. Beneficial Relationships between Endophytic Bacteria and Medicinal Plants. Front. Plant Sci. 2021, 12, 646146. [Google Scholar] [CrossRef]
- Flores-Bustamante, Z.R.; Rivera-Orduna, F.N.; Martínez-Cárdenas, A.; Flores-Cotera, L.B. Microbial Paclitaxel: Advances and Perspectives. J. Antibiot. 2010, 63, 460–467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oukala, N.; Aissat, K.; Pastor, V. Bacterial Endophytes: The Hidden Actor in Plant Immune Responses against Biotic Stress. Plants 2021, 10, 1012. [Google Scholar] [CrossRef]
- Chen, H.; Wu, H.; Yan, B.; Zhao, H.; Liu, F.; Zhang, H.; Sheng, Q.; Miao, F.; Liang, Z. Core Microbiome of Medicinal Plant Salvia miltiorrhiza Seed: A Rich Reservoir of Beneficial Microbes for Secondary Metabolism? Int. J. Mol. Sci. 2018, 19, 672. [Google Scholar] [CrossRef] [Green Version]
- You, C.; Qin, D.; Wang, Y.; Lan, W.; Li, Y.; Yu, B.; Peng, Y.; Xu, J.; Dong, J. Plant Triterpenoids Regulate Endophyte Community to Promote Medicinal Plant Schisandra sphenanthera Growth and Metabolites Accumulation. J. Fungi 2021, 7, 788. [Google Scholar] [CrossRef]
- Comeau, A.M.; Douglas, G.M.; Langille, M.G.I. Microbiome Helper: A Custom and Streamlined Workflow for Microbiome Research. mSystems 2017, 2, e00127-16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ul-Hasan, S.; Bowers, R.M.; Figueroa-Montiel, A.; Licea-Navarro, A.F.; Beman, J.M.; Woyke, T. Community ecology across bacteria, archaea and microbial eukaryotes in the sediment and seawater of coastal Puerto Nuevo, Baja California. PLoS ONE 2019, 14, e0212355. [Google Scholar] [CrossRef] [Green Version]
- Soil Survey Staff, Natural Resources Conservation Service, United States Department of Agriculture. Web Soil Survey. Available online: http://websoilsurvey.sc.egov.usda.gov/ (accessed on 14 October 2021).
- Chen, Y.; Chen, Y.; Shi, C.; Huang, Z.; Zhang, Y.; Li, S.; Li, Y.; Ye, J.; Yu, C.; Li, Z.; et al. SOAPnuke: A MapReduce acceleration-supported software for integrated quality control and preprocessing of high-throughput sequencing data. GigaScience 2018, 7, gix120. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meyer, F.; Paarmann, D.; D’Souza, M.; Olson, R.; Glass, E.M.; Kubal, M. The metagenomics RAST server—A public resource for the automatic phylogenetic and functional analysis of metagenomes. BMC Bioinform. 2008, 9, 386. [Google Scholar] [CrossRef] [Green Version]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef] [Green Version]
- Parks, D.H.; Tyson, G.W.; Hugenholtz, P.; Beiko, R.G. STAMP: Statistical analysis of taxonomic and functional profiles. Bioinformatics 2014, 30, 3123–3124. [Google Scholar] [CrossRef] [Green Version]
- Hilgert, U.; McKay, S.; Khalfan, M.; Williams, J.; Ghiban, C.; Micklos, D. DNA Subway: Making Genome Analysis Egalitarian. In Proceedings of the 2014 Annual Conference on Extreme Science and Engineering Discovery Environment, Atlanta, GA, USA, 13–18 July 2014; Association for Computing Machinery: New York, NY, USA, 2014; Volume 70, pp. 1–3. [Google Scholar] [CrossRef]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Lozupone, C.A.; Turnbaugh, P.J.; Fierer, N.; Knight, R. Global patterns of 16S rRNA diversity at a depth of millions of sequences per sample. Proc. Natl. Acad. Sci. USA 2011, 108 (Suppl. 1), 4516–4522. [Google Scholar] [CrossRef] [Green Version]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Huntley, J.; Fierer, N. Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J. 2012, 6, 1621–1624. [Google Scholar] [CrossRef] [Green Version]
- Clair, S.S.; Saraylou, M.; Maine, E. Introduction to Soil Science Lab Manual v1. Protocols.io 2019. [Google Scholar] [CrossRef]
- Yoon, S.H.; Ha, S.M.; Kwon, S.; Lim, J.; Kim, Y.; Seo, H.; Chun, J. Introducing EzBioCloud: A taxonomically united database of 16S rRNA and whole genome assemblies. Int. J. Syst. Evol. Microbiol. 2017, 67, 1613–1617. [Google Scholar] [CrossRef]
- Kryvtsova, M.V.; Trush, K.; Koščová, J.; Eftimova, J. Antimicrobial, antioxidant and some biochemical properties of arnica montanal. Bull Probl. Biol. Med. 2019, 3, 268. [Google Scholar] [CrossRef]
- Eudes, A.; Mouille, M.; Robinson, D.S.; Benites, V.T.; Wang, G.; Roux, L.; Tsai, Y.-L.; Baidoo, E.E.K.; Chiu, T.-Y.; Heazlewood, J.L.; et al. Exploiting members of the BAHD acyltransferase family to synthesize multiple hydroxycinnamate and benzoate conjugates in yeast. Microb. Cell Factories 2016, 15, 198. [Google Scholar] [CrossRef] [Green Version]
- Jendresen, C.B.; Stahlhut, S.G.; Li, M.J.; Gaspar, P.; Siedler, S.; Foörster, J.; Maury, J.; Borodina, I.; Nielsen, A.T. Highly Active and Specific Tyrosine Ammonia-Lyases from Diverse Origins Enable Enhanced Production of Aromatic Compounds in Bacteria and Saccharomyces cerevisiae. Appl. Environ. Microbiol. 2015, 81, 4458–4476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Friedrich, I.; Hollensteiner, J.; Schneider, D.; Poehlein, A.; Hertel, R.; Daniel, R. First Complete Genome Sequences of Janthinobacterium lividum EIF1 and EIF2 and Their Comparative Genome Analysis. Genome Biol. Evol. 2020, 12, 1782–1788. [Google Scholar] [CrossRef]
- Dara, S.K. Biopesticides: Categories and use strategies for IPM and IRM. E-J. Entomol. Biolog. 2021. Available online: https://ucanr.edu/blogs/blogcore/postdetail.cfm?postnum=46134 (accessed on 29 December 2021).
- Luna, V.A.; King, D.S.; Gulledge, J.; Cannons, A.C.; Amuso, P.T.; Cattani, J. Susceptibility of Bacillus anthracis, Bacillus cereus, Bacillus mycoides, Bacillus pseudomycoides and Bacillus thuringiensis to 24 antimicrobials using Sensititre automated microbroth dilution and Etest agar gradient diffusion methods. J. Antimicrob. Chemother. 2007, 60, 555–567. [Google Scholar] [CrossRef] [Green Version]
- Baek, K.; Choi, A. Draft genome sequence of the polysaccharide degrading bacterium Cytophagaceae strain SJW1-29. Korean J. Microbiol. 2020, 56, 422–425. [Google Scholar] [CrossRef]
- Maria, M.; Mucha, Â.; Ferreira, H.; Gonçalves, B.; Bacelar, E.; Marques, G. Comparative study of plant growth-promoting bacteria on the physiology, growth, and fruit quality of strawberry. J. Sci. Food Agric. 2019, 99, 5341–5349. [Google Scholar]
- Kolton, M.; Erlacher, A.; Berg, G.; Cytryn, E. Ch 9: The Flavobacterium genus in the Plant Holobiont: Ecological, Physiological, and Applicative Insights. In Microbial Models from Environmental to Industrial Sustainabilty; Springer: Singapore, 2016. [Google Scholar]
- Yonekura-Sakakibara, K.; Higashi, Y.; Nakabayashi, R. The Origin and Evolution of Plant Flavonoid Metabolism. Front. Plant Sci. 2019, 10, 943. [Google Scholar] [CrossRef] [Green Version]
- Abe, I. Biosynthesis of medicinally important plant metabolites by unusual type III polyketide synthases. J. Nat. Med. 2020, 74, 639–646. [Google Scholar] [CrossRef]
- Schlatter, D.; Kinkel, L.; Thomashow, L.; Weller, D.; Paulitz, T. Disease Suppressive Soils: New Insights from the Soil Microbiome. Phytopathol. Rev. 2017, 107, 1284–1297. [Google Scholar] [CrossRef] [Green Version]
- Nordberg, H.; Cantor, M.; Dusheyko, S.; Hua, S.; Poliakov, A.; Shabalov, I.; Smirnova, T.; Grigoriev, I.V.; Dubchak, I. The genome portal of the Department of Energy Joint Genome Institute: 2014 updates. Nucleic Acids Res. 2014, 42, D26–D31. [Google Scholar] [CrossRef] [PubMed]
- Fadiji, A.E.; Babalola, O.O. Elucidating Mechanisms of Endophytes Used in Plant Protection and Other Bioactivities with Multifunctional Prospects. Front. Bioeng. Biotechnol. 2020, 8, 467. [Google Scholar] [CrossRef]
- Bedewitz, M.A.; Jones, A.D.; D’Auria, J.C.; Barry, C.S. Tropinone synthesis via an atypical polyketide synthase and P450 mediated cyclization. Nat. Commun. 2018, 9, 5281. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.P.; Fang, C.; Ma, X.; Wang, L.; Yang, J.; Luo, J.; Huang, S.X. Tropane alkaloids biosynthesis involves an unusual type III polyketide synthase and non-enzymatic condensation. Nat. Commun. 2019, 10, 4036. [Google Scholar] [CrossRef] [Green Version]
- Huccetogullari, D.; Luo, Z.W.; Lee, S.Y. Metabolic engineering of microorganisms for production of aromatic compounds. Microb. Cell Factories 2019, 18, 41. [Google Scholar] [CrossRef] [PubMed]
- The UniProt Consortium. UniProt: The universal protein knowledgebase in 2021. Nucleic Acids Res. 2021, 49, D1. [Google Scholar]
- Krey Karol, L.; Nabity Paul, D.; Blubaugh Carmen, K.; Zhen, F.; Van Leuven James, T. Organic Farming Sharpens Plant Defenses in the Field. Front. Sustain. Food Syst. 2020, 4, 97. Available online: https://www.frontiersin.org/article/10.3389/fsufs.2020.00097 (accessed on 29 December 2021). [CrossRef] [PubMed]




| Map Unit Name | pH | CEC | % Sand | % Silt | % Clay | % OM |
|---|---|---|---|---|---|---|
| Cropley-Urban Land Complex, 0 to 2% | 7.9 | 37.5 | 22.1 | 27.9 | 50 | 1.5 |
| Sample No. | Field | Colony Color | Morphology | Antibiotic Used | Media | Absorbed CR |
|---|---|---|---|---|---|---|
| 1 | Marquis C | Transl yellow | Unkown | NA | Nutrient agar | NA |
| 2 | Marquis D | White/tan | Unkown | Penicillin | Nutrient agar | NA |
| 3 | Field 28 | White/tan | Unkown | Penicillin | Nutrient agar | NA |
| 4 | Marquis A | Yellow | Unkown | Streptomycin | Nutrient agar | NA |
| 5 | Marquis A | Yellow | Raised | NA | Nutrient agar | NA |
| 6 | Arboretum | White | Spreading | NA | TYES-CR | absorbed cr |
| 7 | Marquis A | Transl yellow | Raised | NA | Nutrient agar | NA |
| 8 | Marquis A | White | Gliding | NA | Nutrient agar | NA |
| 9 | Marquis A | Transl yellow | Gliding | NA | Nutrient agar | NA |
| 10 | Marquis A | Yellow | Flat | NA | Nutrient agar | NA |
| 11 | Marquis A | Transl Yellow | Highly motile at 4 °C | NA | Nutrient agar | NA |
| 12 | Arboretum | Clear/tan | Mucoid | NA | TYES | Intense pink |
| 13 | Arboretum | White | Flat | NA | TYES | Dark red |
| 14 | Marquis A | Yellow | Raised | NA | Nutrient agar | NA |
| 15 | Arboretum | Clear/tan | Small mucoid | NA | TYES | Light pink |
| 16 | Marquis A | Clear/tan | Mucoid | NA | TYES | NA |
| 17 | Marquis A | Transl yellow | Mucoid | NA | TYES | NA |
| 18 | Arboretum | Clear/tan | Gliding | NA | TYES | Abs cr |
| 19 | Arboretum | White | Spreading | NA | TYES | Abs cr ring |
| 20 | Marquis A | Transl yellow | Raised, wrinkled | NA | 1/2 NA + AC | NA |
| 21 | Marquis A | Transl yellow | Small colony | NA | 1/2 NA +AC | NA |
| 22 | Marquis A | Transl yellow | Small colony | Griseofulvin | ISP-6 | NA |
| 23 | Marquis A | Yellow/orange | raised | Griseofulvin | ISP-6 | NA |
| 24 | Marquis A | White/grey | Large colony | Griseofulvin | ISP-6 | NA |
| 25 | Marquis A | Transl yellow | Wrinkled | Griseofulvin | ISP-6 | NA |
| OTUID | Genus | log2FoldChange | p-adj. |
|---|---|---|---|
| OTU149 | Cavemovirus | −8.74069944 | 1.46 × 10−11 |
| OTU494 | Pantoea | −3.382739635 | 5.9 × 10−18 |
| OTU168 | Chryseobacterium | −2.894493584 | 0.000172 |
| OTU332 | Janthinobacterium | −2.396801275 | 5.53 × 10−6 |
| OTU307 | Herbaspirillum | −2.385130454 | 2.86 × 10−8 |
| OTU505 | Pedobacter | −2.384332893 | 6 × 10−5 |
| OTU247 | Erwinia | −2.324319999 | 3.84 × 10−35 |
| OTU738 | unclassified (derived from Flavobacteriaceae) | −2.299754251 | 0.00078 |
| OTU273 | Gemmata | −2.299386567 | 3.76 × 10−19 |
| OTU308 | Herminiimonas | −2.1461305 | 2.3 × 10−5 |
| OTU693 | Variovorax | −2.033564378 | 1.25 × 10−7 |
| OTU759 | unclassified (derived from Verrucomicrobia subdivision 3) | −1.966909323 | 5.46 × 10−18 |
| OTU169 | Chthoniobacter | −1.924054848 | 5.19 × 10−11 |
| OTU440 | Mucilaginibacter | −1.920113231 | 6.47 × 10−5 |
| OTU263 | Fluoribacter | −1.819462381 | 0.006919 |
| OTU569 | Riemerella | −1.783874932 | 0.008796 |
| OTU488 | Oxalobacter | −1.775720869 | 1.2 × 10−6 |
| OTU339 | Klebsiella | −1.680008901 | 0.00538 |
| OTU277 | Geodermatophilus | −1.67277933 | 0.000157 |
| OTU463 | Nitrospira | −1.638446217 | 0.000467 |
| OTU262 | Flavobacterium | −1.633855026 | 0.007125 |
| OTU156 | Chitinophaga | −1.588995265 | 0.000676 |
| OTU696 | Verrucomicrobium | −1.523905486 | 3.61 × 10−5 |
| OTU234 | Dyadobacter | −1.505907596 | 0.005228 |
| OTU480 | Opitutus | −1.500266429 | 5.62 × 10−5 |
| OTU598 | Serratia | −1.489698167 | 2.49 × 10−10 |
| OTU421 | Methylobacterium | −1.448043272 | 2.59 × 10-12 |
| OTU136 | Candidatus Solibacter | −1.428079685 | 1.1 × 10−15 |
| OTU624 | Stigmatella | −1.392596868 | 6.52 × 10−7 |
| OTU751 | unclassified (derived from Proteobacteria) | −1.373840383 | 0.003172 |
| OTU180 | Conexibacter | −1.373047754 | 0.000188 |
| OTU651 | Terriglobus | −1.362530369 | 6.27 × 10−16 |
| OTU532 | Polaromonas | −1.340071098 | 9.35 × 10−6 |
| OTU727 | unclassified (derived from Candidatus Poribacteria) | −1.330571533 | 0.00172 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Senn, S.; Pangell, K.; Bowerman, A.L. Metagenomic Insights into the Composition and Function of Microbes Associated with the Rootzone of Datura inoxia. BioTech 2022, 11, 1. https://doi.org/10.3390/biotech11010001
Senn S, Pangell K, Bowerman AL. Metagenomic Insights into the Composition and Function of Microbes Associated with the Rootzone of Datura inoxia. BioTech. 2022; 11(1):1. https://doi.org/10.3390/biotech11010001
Chicago/Turabian StyleSenn, Savanah, Kelly Pangell, and Adrianna L. Bowerman. 2022. "Metagenomic Insights into the Composition and Function of Microbes Associated with the Rootzone of Datura inoxia" BioTech 11, no. 1: 1. https://doi.org/10.3390/biotech11010001
APA StyleSenn, S., Pangell, K., & Bowerman, A. L. (2022). Metagenomic Insights into the Composition and Function of Microbes Associated with the Rootzone of Datura inoxia. BioTech, 11(1), 1. https://doi.org/10.3390/biotech11010001






