Overall Survival of Patients with Melanoma of Unknown Primary Versus Melanoma of Known Primary Under Immunotherapy and Targeted Therapy: A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Protocol
2.2. Search
2.3. Selection of Studies
2.4. Data Extraction
2.5. Assessment of Study Quality and Risk of Bias
2.6. Data Synthesis and Statistical Analysis
3. Results
3.1. Study Selection
3.2. Study Characteristics
3.3. Individual Assessment of the Risk of Bias and Study Quality
3.4. Hazard Ratios and Pooled Analysis
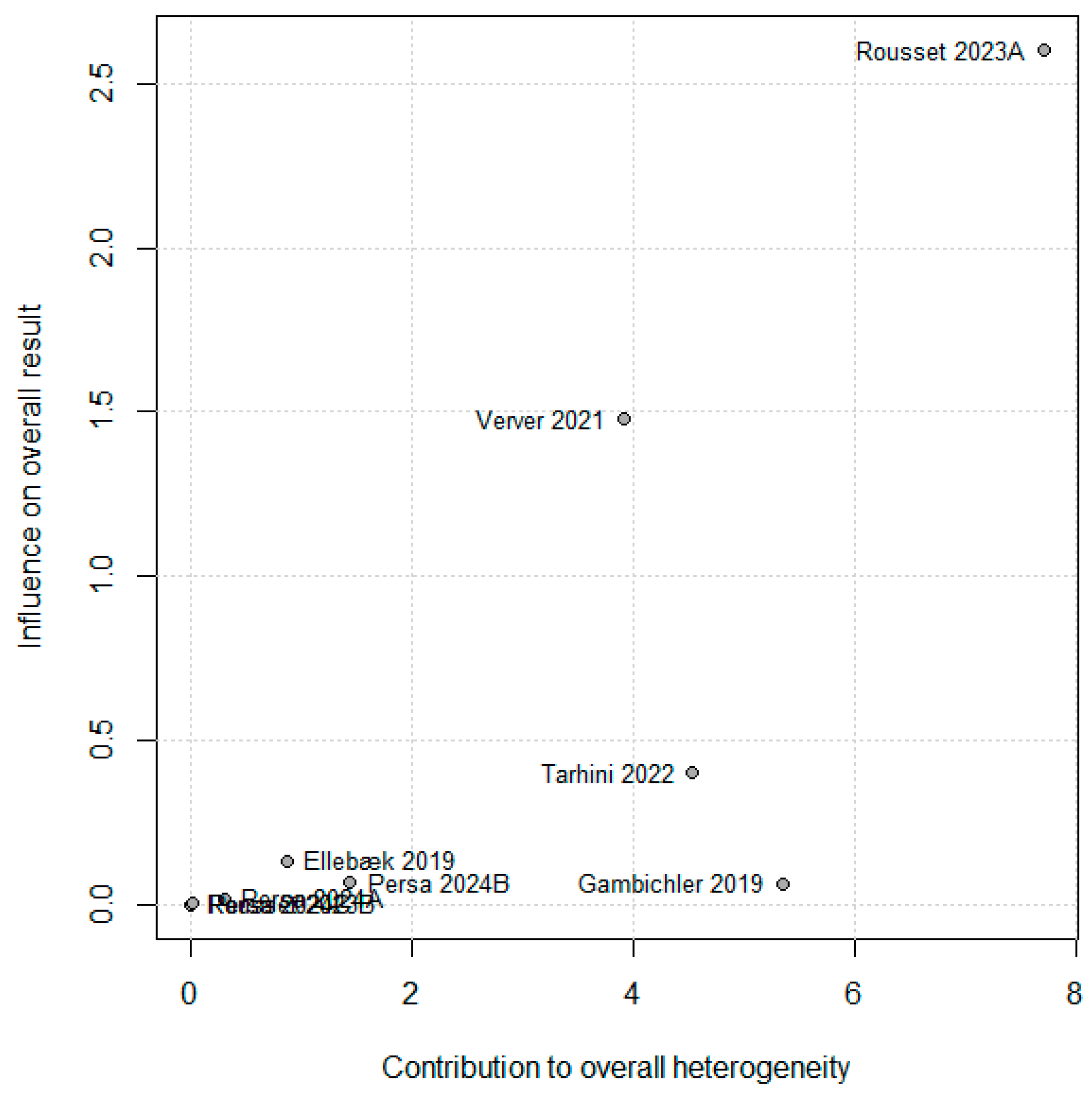
3.5. Heterogeneity and Baujat Analysis
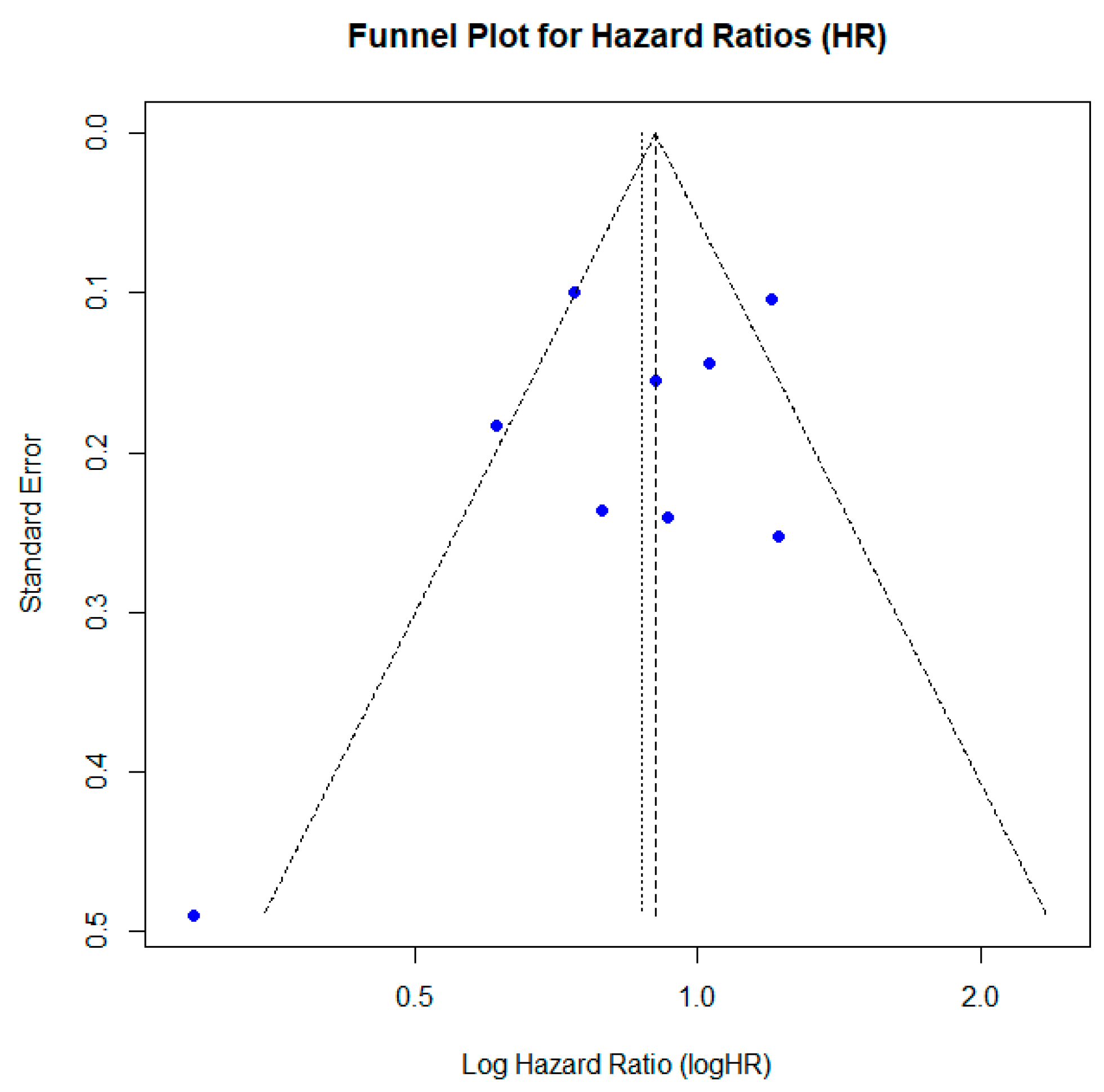
3.6. Publication Bias
3.7. Meta-Regression and Moderator Analysis
3.8. Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Boussios, S.; Rassy, E.; Samartzis, E.; Moschetta, M.; Sheriff, M.; Pérez-Fidalgo, J.A.; Pavlidis, N. Melanoma of unknown primary: New perspectives for an old story. Crit. Rev. Oncol. Hematol. 2021, 158, 103208. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.P.; Sheth, R.A.; Davis, C.; Medina, T. Combination Immunotherapy With Nivolumab Plus Ipilimumab in Melanoma of Unknown Primary. J. Clin. Oncol. 2025, 43, 907–911. [Google Scholar] [CrossRef] [PubMed]
- Andrade, J.P.; Wong, P.; O’Leary, M.P.; Parekh, V.; Amini, A.; Schoellhammer, H.F.; Margolin, K.A.; Afkhami, M.; Melstrom, L.G. Multidisciplinary Care for Melanoma of Unknown Primary: Experience in the Era of Molecular Profiling. Ann. Surg. Oncol. 2020, 27, 5240–5247. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Karakousis, G.C. Melanoma of unknown primary. J. Surg. Oncol. 2019, 119, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.M.; Choi, Y.Y.; Kim, D.S.; Lee, J.H.; Jang, H.S.; Lee, J.H.; Kim, H.; Oh, B.H.; Roh, M.R.; Nam, K.A.; et al. Metastatic melanomas of unknown primary show better prognosis than those of known primary: A systematic review and meta-analysis of observational studies. J. Am. Acad. Dermatol. 2015, 72, 59–70. [Google Scholar] [CrossRef] [PubMed]
- Verver, D.; van der Veldt, A.; van Akkooi, A.; Verhoef, C.; Grünhagen, D.J.; Louwman, W.J. Treatment of melanoma of unknown primary in the era of immunotherapy and targeted therapy: A Dutch population-based study. Int. J. Cancer 2019, 146, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Hughes, M.C.; Wright, A.; Barbour, A.; Thomas, J.; Smithers, B.M.; Green, A.C.; Khosrotehrani, K. Patients undergoing lymphadenectomy for stage III melanomas of known or unknown primary site do not differ in outcome. Int. J. Cancer 2013, 133, 3000–3007. [Google Scholar] [CrossRef] [PubMed]
- Amin, M.B.; Edge, S.B.; Greene, F.L.; Byrd, D.R.; Brookland, R.K.; Washington, M.K.; Gershenwald, J.E.; Compton, C.C.; Hess, K.R.; Sullivan, D.C.; et al. AJCC Cancer Staging Manua, 8th ed.; Springer: New York, NY, USA, 2017. [Google Scholar]
- Smith, D.L.; Patel, P.; Lewis, K.G. Histopathologic evidence of immune-mediated regression in primary cutaneous melanoma. J. Investig. Dermatol. 2017, 137, 948–955. [Google Scholar]
- Jones, M.N.; Zhang, T.; Harris, S.; Kim, A.G. De novo nodal melanoma arising from ectopic nevus cell aggregates: A clinicopathologic study. Am. J. Surg. Pathol. 2018, 42, 156–162. [Google Scholar]
- Kim, S.J.; Park, H.J.; Choi, Y.J. Challenges in detecting occult mucosal and ocular melanoma primaries in melanoma of unknown primary. Melanoma Res. 2019, 29, 499–505. [Google Scholar]
- Ellebæk, E.; Bastholt, L.; Schmidt, H.; Svane, I.M.; Donia, M. The real-world outcome of metastatic melanoma: Unknown primary vs. known cutaneous. Int. J. Cancer 2019, 145, 3173–3174. [Google Scholar] [CrossRef] [PubMed]
- Gambichler, T.; Chatzipantazi, M.; Schröter, U.; Stockfleth, E.; Gedik, C. Patients with melanoma of unknown primary show better outcome under immune checkpoint inhibitor therapy than patients with known primary: Preliminary results. Oncoimmunology 2019, 8, e1677139. [Google Scholar] [CrossRef] [PubMed]
- Verver, D.; Grünhagen, D.J.; van Akkooi, A.C.J.; Aarts, M.J.B.; van den Berkmortel, F.W.P.J.; van den Eertwegh, A.J.M.; de Groot, J.W.B.; Boers-Sonderen, M.J.; Haanen, J.B.A.G.; Hospers, G.A.P.; et al. Clinical outcome of patients with metastatic melanoma of unknown primary in the era of novel therapy. Cancer Immunol. Immunother. 2021, 70, 3123–3135. [Google Scholar] [CrossRef] [PubMed]
- Tarhini, A.A.; Lee, S.J.; Tan, A.C.; El Naqa, I.M.; Stephen Hodi, F.; Butterfield, L.H.; LaFramboise, W.A.; Storkus, W.J.; Karunamurthy, A.D.; Conejo-Garcia, J.R.; et al. Improved prognosis and evidence of enhanced immunogenicity in tumor and circulation of high-risk melanoma patients with unknown primary. J. Immunother. Cancer 2022, 10, e004310. [Google Scholar] [CrossRef] [PubMed]
- Rousset, P.; Dalle, S.; Mortier, L.; Dereure, O.; Dalac, S.; Dutriaux, C.; Leccia, M.T.; Legoupil, D.; Brunet-Possenti, F.; De Quatrebarbes, J.; et al. Impact of systemic therapies in metastatic melanoma of unknown primary: A study from MELBASE, a French multicentric prospective cohort. J. Am. Acad. Dermatol. 2023, 88, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Persa, O.D.; Hassel, J.C.; Steeb, T.; Erdmann, M.; Karimi, B.; Stege, H.; Klespe, K.C.; Schatton, K.; Tomsitz, D.; Rübben, A.; et al. Brief Communication: Treatment Outcomes for Advanced Melanoma of Unknown Primary Compared With Melanoma With Known Primary. J. Immunother. 2024, 47, 384–387. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Critical Appraisal Skills Programme. CASP Cohort Study Checklist. Available online: https://casp-uk.net/casp-tools-checklists/cohort-study-checklist/ (accessed on 9 March 2025).

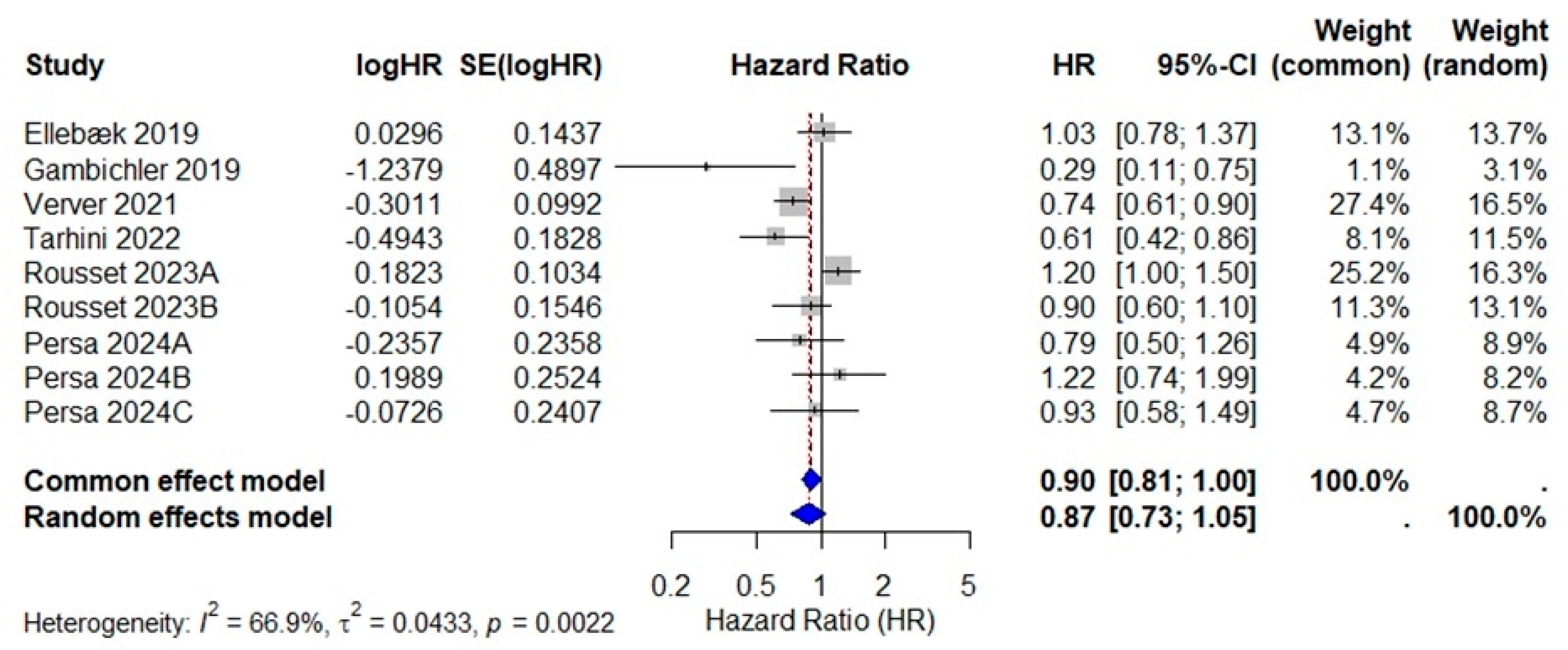
| # | Study (Country) Stage | Treatment | HR OS §/MSS $ | 95% CI (Lower-Upper) | MUP n | MKP n |
|---|---|---|---|---|---|---|
| 1 | Ellebæk 2019 [12] § (Denmark) unresectable IIIC/IV (M1a or M1b) | * ICI | 1.03 | 0.78–1.37 | 80 | 496 |
| 2 | Gambichler 2019 [13] $ (Germany) unresectable IIIC/IV (M1a or M1b) | * ICI | 0.29 | 0.11–0.75 | 9 | 32 |
| 3 | Verver 2021 [14] § (Netherlands) unresectable IIIC/IV (M1a or M1b) | * ICI or TT | 0.74 | 0.61–0.90 | 385 | 2321 |
| 4 | Tarhini 2022 [15] § (USA) resected IIIB, IIIC, IV (M1a or M1b) | * CTLA-4i or hd-INF | 0.61 | 0.42–0.86 | 214 | 1669 |
| 5 A B | Rousset 2023 [16] § (France) unresectable IIIC/IV (M1a or M1b) | * ICI * TT | 1.20 0.90 | 1.00–1.50 0.60–1.10 | 165 86 | 1059 488 |
| 6 A B C | Persa 2024 [17] § (Germany) unresectable IIIC/IV (M1a or M1b) | * PD-1i * PD-1i + CTLA-4i * BRAFi + MEKi | 0.79 1.22 0.93 | 0.50–1.26 0.74–1.99 0.58–1.49 | 142 152 101 | 142 152 101 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gambichler, T.; Gaertner, P.C.; Abu Rached, N.; Susok, L.; Weyer-Fahlbusch, S.S. Overall Survival of Patients with Melanoma of Unknown Primary Versus Melanoma of Known Primary Under Immunotherapy and Targeted Therapy: A Systematic Review and Meta-Analysis. Dermato 2025, 5, 15. https://doi.org/10.3390/dermato5030015
Gambichler T, Gaertner PC, Abu Rached N, Susok L, Weyer-Fahlbusch SS. Overall Survival of Patients with Melanoma of Unknown Primary Versus Melanoma of Known Primary Under Immunotherapy and Targeted Therapy: A Systematic Review and Meta-Analysis. Dermato. 2025; 5(3):15. https://doi.org/10.3390/dermato5030015
Chicago/Turabian StyleGambichler, Thilo, Priyanka C. Gaertner, Nessr Abu Rached, Laura Susok, and Sera S. Weyer-Fahlbusch. 2025. "Overall Survival of Patients with Melanoma of Unknown Primary Versus Melanoma of Known Primary Under Immunotherapy and Targeted Therapy: A Systematic Review and Meta-Analysis" Dermato 5, no. 3: 15. https://doi.org/10.3390/dermato5030015
APA StyleGambichler, T., Gaertner, P. C., Abu Rached, N., Susok, L., & Weyer-Fahlbusch, S. S. (2025). Overall Survival of Patients with Melanoma of Unknown Primary Versus Melanoma of Known Primary Under Immunotherapy and Targeted Therapy: A Systematic Review and Meta-Analysis. Dermato, 5(3), 15. https://doi.org/10.3390/dermato5030015








