Post-COVID Kawasaki-like Multisystem Inflammatory Syndrome Complicated by Herpes Simplex Virus-1 in a Two-Year-Old Child
Abstract
:1. Introduction
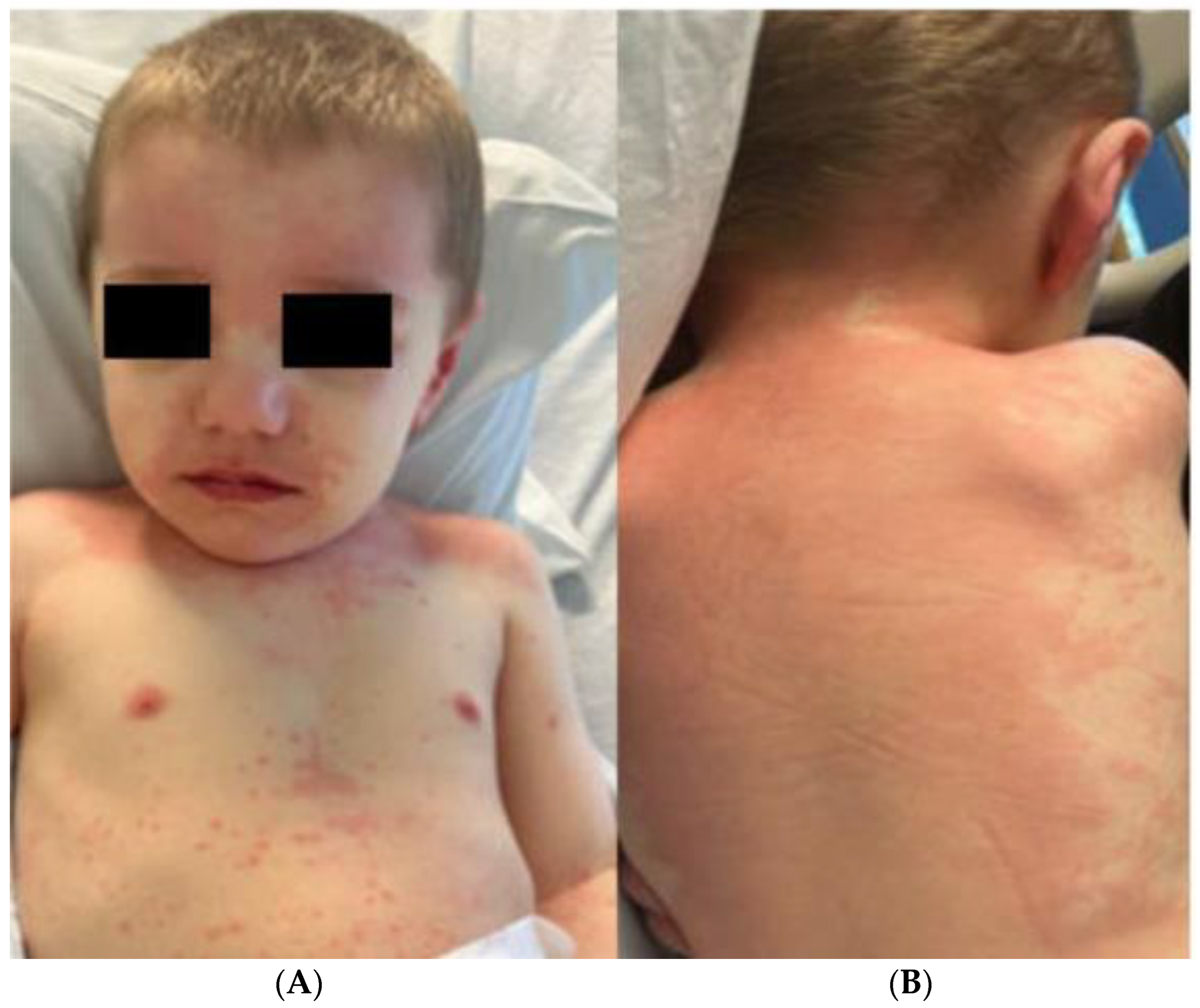
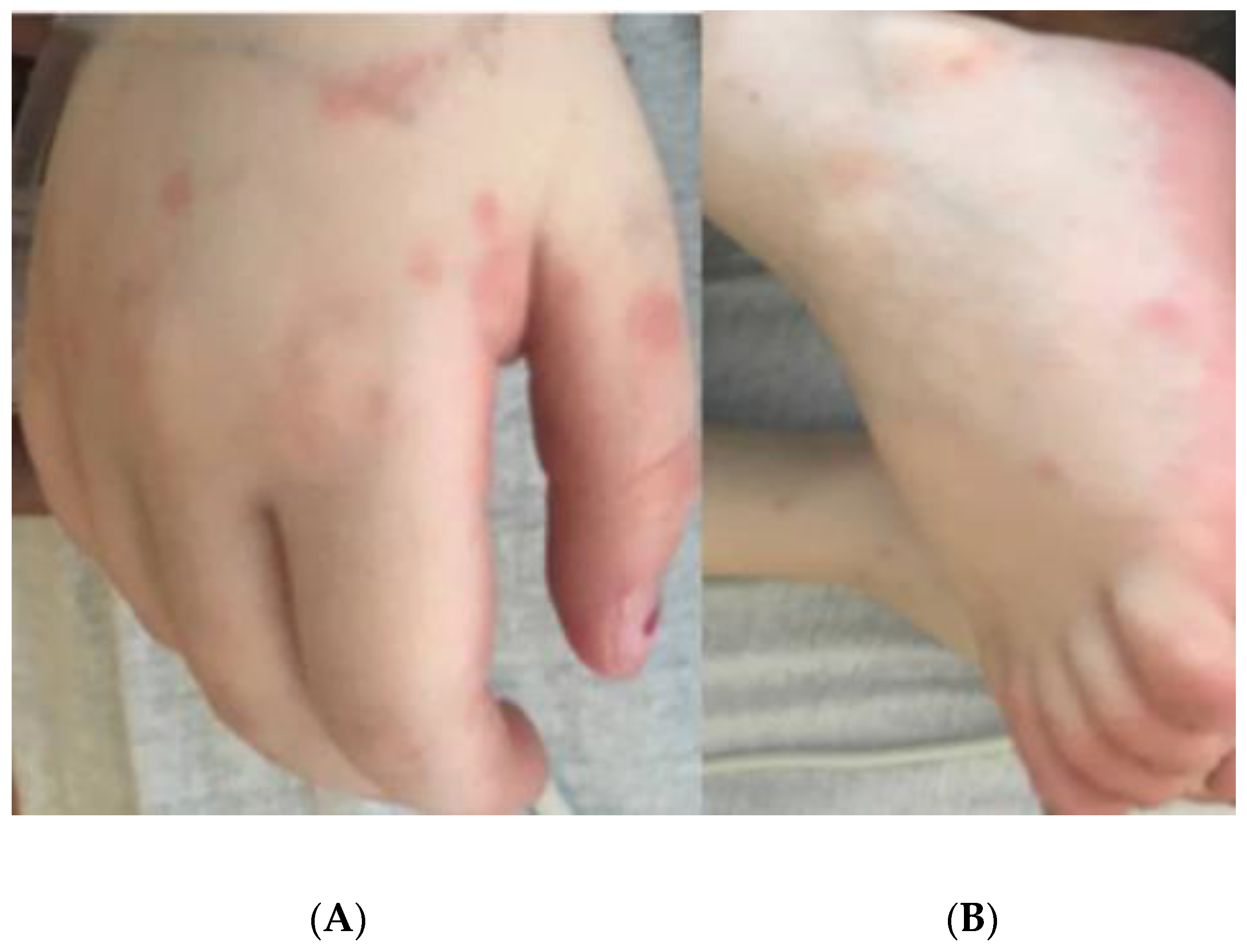
2. Case Report
3. Discussion
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Rothan, H.A.; Byrareddy, S.N. The potential threat of multisystem inflammatory syndrome in children during the COVID-19 pandemic. Pediatr. Allergy Immunol. 2021, 32, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Ramaswamy, A.; Brodsky, N.N.; Sumida, T.S.; Comi, M.; Asashima, H.; Hoehn, K.B.; Li, N.; Liu, Y.; Shah, A.; Ravindra, N.G.; et al. Post-infectious inflammatory disease in MIS-C features elevated cytotoxicity signatures and autoreactivity that correlates with severity. medRxiv 2021, 2020-12. [Google Scholar] [CrossRef]
- Sethuraman, N.; Jeremiah, S.S.; Ryo, A. Interpreting diagnostic tests for SARS-CoV-2. JAMA 2020, 323, 2249–2251. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.; Zhang, X.; Li, R.; Zheng, M.; Yang, S.; Dai, L.; Wu, A.; Hu, C.; Huang, Y.; Xie, M.; et al. Overexpression of the SARS-CoV-2 receptor ACE2 is induced by cigarette smoke in bronchial and alveolar epithelia. J. Pathol. 2021, 253, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Jindal, A.K.; Pilania, R.K. Diagnosis of Kawasaki disease. Int. J. Rheum. Dis. 2018, 21, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Tong, T.; Yao, X.; Lin, Z.; Tao, Y.; Xu, J.; Xu, X.; Fang, Z.; Geng, Z.; Fu, S.; Wang, W.; et al. Similarities and differences between MIS-C and KD: A systematic review and meta-analysis. Pediatr. Rheumatol. 2022, 20, 112. [Google Scholar] [CrossRef] [PubMed]
- Hufnagel, M.; Armann, J.; Jakob, A.; Doenhardt, M.; Diffloth, N.; Hospach, A.; Schneider, D.T.; Trotter, A.; Roessler, M.; Schmitt, J.; et al. A comparison of pediatric inflammatory multisystem syndrome temporarily-associated with SARS-CoV-2 and Kawasaki disease. Sci. Rep. 2023, 13, 1173. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.Y.; Xu, B.W.; Du, J.B. Similarities and differences between multiple inflammatory syndrome in children associated with COVID-19 and Kawasaki disease: Clinical presentations, diagnosis, and treatment. World J. Pediatr. 2021, 17, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Yeo, W.S.; Ng, Q.X. Distinguishing between typical Kawasaki disease and multisystem inflammatory syndrome in children (MIS-C) associated with SARS-CoV-2. Med. Hypotheses 2020, 144, 110263. [Google Scholar] [CrossRef] [PubMed]
- Cheng, M.H.; Zhang, S.; Porritt, R.A.; Noval Rivas, M.; Paschold, L.; Willscher, E.; Binder, M.; Arditi, M.; Bahar, I. Superantigenic character of an insert unique to SARS-CoV-2 spike supported by skewed TCR repertoire in patients with hyperinflammation. Proc. Natl. Acad. Sci. USA 2020, 117, 25254–25262. [Google Scholar] [CrossRef] [PubMed]
- Rowley, A.H.; Shulman, S.T.; Arditi, M. Immune pathogenesis of COVID-19–related multisystem inflammatory syndrome in children. J. Clin. Investig. 2020, 130, 5619–5621. [Google Scholar] [CrossRef] [PubMed]
- Thiriard, A.; Meyer, B.; Eberhardt, C.S.; Loevy, N.; Grazioli, S.; Adouan, W.; Fontannaz, P.; Marechal, F.; L’Huillier, A.G.; Siegrist, C.-A.; et al. Antibody response in children with multisystem inflammatory syndrome related to COVID-19 (MIS-C) compared to children with uncomplicated COVID-19. Front. Immunol. 2023, 14, 1107156. [Google Scholar] [CrossRef] [PubMed]
- Jain, E.; Donowitz, J.R.; Aarons, E.; Marshall, B.C.; Miller, M.P. Multisystem inflammatory syndrome in children after SARS-CoV-2 vaccination. Emerg. Infect. Dis. 2022, 28, 990. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, S.; El-Kalliny, M.; Kotby, A.; El-Ganzoury, M.; Fouda, E.; Ibrahim, H. Treatment of MIS-C in Children and Adolescents. Curr. Pediatr. Rep. 2022, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Giacobbe, D.R.; Di Bella, S.; Lovecchio, A.; Ball, L.; De Maria, A.; Vena, A.; Bruzzone, B.; Icardi, G.; Pelosi, P.; Luzzati, R.; et al. Herpes Simplex Virus 1 (HSV-1) Reactivation in Critically Ill COVID-19 Patients: A Brief Narrative Review. Infect. Dis. Ther. 2022, 11, 1779–1791. [Google Scholar] [CrossRef] [PubMed]
- McLane, L.M.; Abdel-Hakeem, M.S.; Wherry, E.J. CD8 T cell exhaustion during chronic viral infection and cancer. Annu. Rev. Immunol. 2019, 37, 457–495. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J. Corticosteroid effects on cell signaling. Eur. Respir. J. 2006, 27, 413–426. [Google Scholar] [CrossRef] [PubMed]
- Karampinis, E.; Goudouras, G.; Ntavari, N.; Bogdanos, D.P.; Roussaki-Schulze, A.V.; Zafiriou, E. Serum vitamin D levels can be predictive of psoriasis flares up after COVID-19 vaccination: A retrospective case control study. Front. Med. 2023, 10, 1203426. [Google Scholar] [CrossRef] [PubMed]
- Alshiyab, D.M.; Al-Qarqaz, F.A.; Alhaje, E.; Mayou, J.A.; Jaradat, S.; Asaad, A.; Muhaidat, J.M.; Khader, Y.; Alsheyab, S.M.; Oweis, A.O.; et al. Skin Manifestations Among Patients Admitted with COVID-19: A Cross-Sectional Study at a University-Based Tertiary Hospital in Jordan. Clin. Cosmet. Investig. Dermatol. 2023, 16, 1331–1340. [Google Scholar] [CrossRef] [PubMed]


| Vitals and Laboratory Results | At Hospitalization | After Treatment | At HSV-1 Detection |
|---|---|---|---|
| Vitals | |||
| Heart Rate | 142 | 99 | 122 |
| Respiratory Rate | 42 | 34 | 26 |
| Temperature (F°) | 104.6 | 99.3 | 102.2 |
| Lab (normal range) | |||
| Ferritin (36–84) | 259 | 361 | 568 |
| C-reactive protein (<3) | 119.2 | 89.6 | 53.1 |
| Platelets (145–370 × 103) | 199 | 121 | 799 |
| WBCs (5.5–15.5 × 103) | 17 | 9.8 | 45.4 |
| Estimated sedimentation rate (2–34) | 48 | 3 | 25 |
| Alanine transaminase (0–34) | 299 | 62 | 31 |
| Aspartate aminotransferase (17–50) | 265 | 60 | 33 |
| Albumin (3.3–4.9) | 2.1 | 2.4 | 3.1 |
| Pro-BNP (≤124) | 17,990.00 | 15,790.00 | 8478.00 |
| D-dimer (215–780) | 5781.00 | 5668 | 2709.00 |
| Bilirubin (≤1) | 4.3 | 1.8 | 1.1 |
| MIS-C Features | Kawasaki Disease Features | HSV-1 Features |
|---|---|---|
| Fever; Gastrointestinal symptoms; maculo-papular rash; Extremity edema; Mucous membrane changes; Myocardial involvement with dilated coronary arteries without aneurysms and elevated pro-BNP; Hepatic involvement with elevated ALT, AST, and bilirubin | Recorded fever for 3 days (highest of 102.2 F); Morbilliform rash involving hands, feet, trunk, and extremities; Acral edema | Ulcers with crusting on upper and lower lip, vermillion border, and oral commissure |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hodson, E.L.; Salem, I.; Bradley, K.E.; Okorie, C.L.; Marka, A.; Abraham, N.D.; Pace, N.C.; Dagrosa, A.T.; Ratts, R.C.; Mann, J.A. Post-COVID Kawasaki-like Multisystem Inflammatory Syndrome Complicated by Herpes Simplex Virus-1 in a Two-Year-Old Child. Dermato 2023, 3, 224-231. https://doi.org/10.3390/dermato3030017
Hodson EL, Salem I, Bradley KE, Okorie CL, Marka A, Abraham ND, Pace NC, Dagrosa AT, Ratts RC, Mann JA. Post-COVID Kawasaki-like Multisystem Inflammatory Syndrome Complicated by Herpes Simplex Virus-1 in a Two-Year-Old Child. Dermato. 2023; 3(3):224-231. https://doi.org/10.3390/dermato3030017
Chicago/Turabian StyleHodson, Emma L., Iman Salem, Katherine E. Bradley, Chiamaka L. Okorie, Arthur Marka, Nigel D. Abraham, Nicole C. Pace, Alicia T. Dagrosa, Ryan C. Ratts, and Julianne A. Mann. 2023. "Post-COVID Kawasaki-like Multisystem Inflammatory Syndrome Complicated by Herpes Simplex Virus-1 in a Two-Year-Old Child" Dermato 3, no. 3: 224-231. https://doi.org/10.3390/dermato3030017
APA StyleHodson, E. L., Salem, I., Bradley, K. E., Okorie, C. L., Marka, A., Abraham, N. D., Pace, N. C., Dagrosa, A. T., Ratts, R. C., & Mann, J. A. (2023). Post-COVID Kawasaki-like Multisystem Inflammatory Syndrome Complicated by Herpes Simplex Virus-1 in a Two-Year-Old Child. Dermato, 3(3), 224-231. https://doi.org/10.3390/dermato3030017






