Permanent Ad-lib Feeders Decrease the Survival of Wintering Great Tits (Parus major)
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sites and Procedures
2.2. Feeders and Feeding Regimes
2.3. Study Species
2.4. Data Collection
2.5. Survival
2.6. Statistical Analyses
3. Results
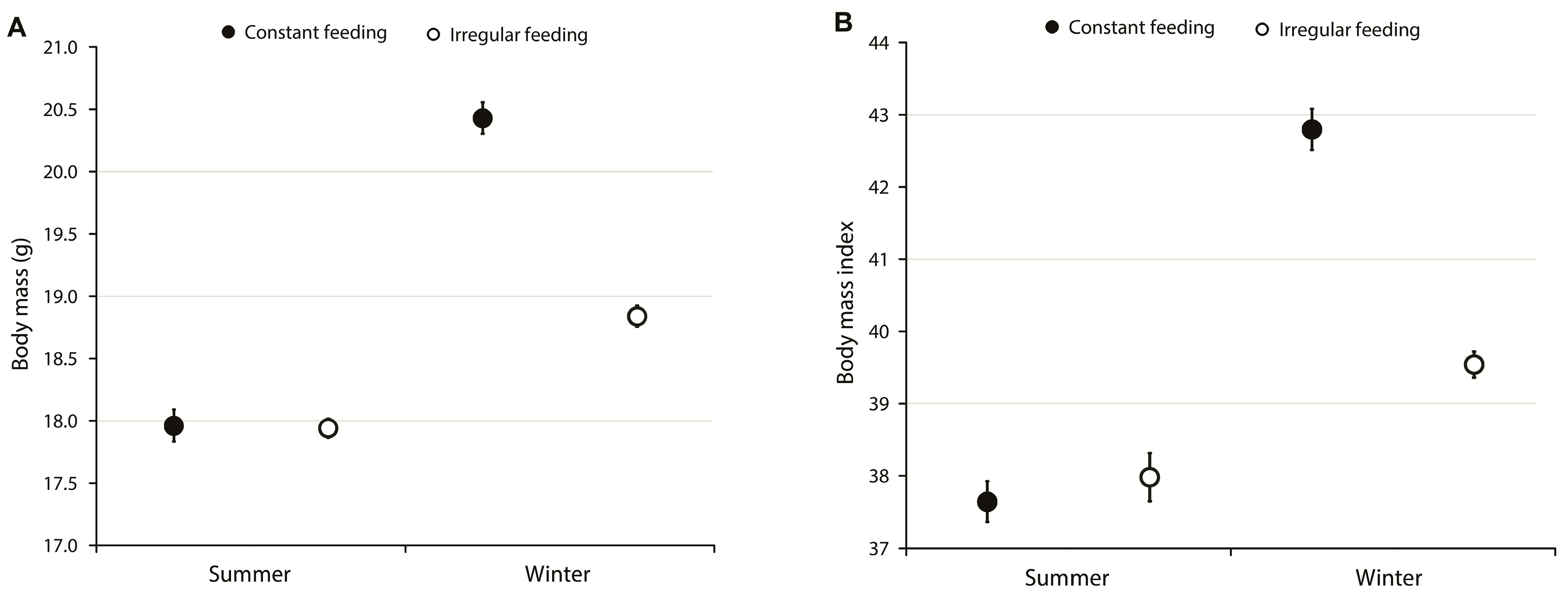
3.1. Body Mass, Body Mass Index, and Survival
3.2. Prey Behavior
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Houston, A.I.; McNamara, J.M. A Theoretical Investigation of the Fat Reserves and Mortality Levels of Small Birds in Winter. Ornis Scand. 1993, 24, 205. [Google Scholar] [CrossRef]
- Brodin, A. Why Do Hoarding Birds Gain Fat in Winter in the Wrong Way? Suggestions from a Dynamic Model. Behav. Ecol. 2000, 11, 27–39. [Google Scholar] [CrossRef]
- Lehikoinen, E. Seasonality of the Daily Weight Cycle in Wintering Passerines and Its Consequences. Ornis Scand. 1987, 18, 216. [Google Scholar] [CrossRef]
- Haftorn, S. Seasonal and Diurnal Body Weight Variations in Titmice, Based on Analyses of Individual Birds. Wilson Bull. 1989, 101, 217–235. [Google Scholar]
- Krams, I.; Cirule, D.; Suraka, V.; Krama, T.; Rantala, M.J.; Ramey, G. Fattening Strategies of Wintering Great Tits Support the Optimal Body Mass Hypothesis under Conditions of Extremely Low Ambient Temperature. Funct. Ecol. 2010, 24, 172–177. [Google Scholar] [CrossRef]
- Krams, I.; Cīrule, D.; Vrublevska, J.; Nord, A.; Rantala, M.J.; Krama, T. Nocturnal Loss of Body Reserves Reveals High Survival Risk for Subordinate Great Tits Wintering at Extremely Low Ambient Temperatures. Oecologia 2013, 172, 339–346. [Google Scholar] [CrossRef]
- Wood, K.A.; Stillman, R.A.; Newth, J.L.; Nuijten, R.J.M.; Hilton, G.M.; Nolet, B.A.; Rees, E.C. Predicting Avian Herbivore Responses to Changing Food Availability and Competition. Ecol. Model. 2021, 441, 109421. [Google Scholar] [CrossRef]
- Lima, S.L. Predation Risk and Unpredictable Feeding Conditions: Determinants of Body Mass in Birds. Ecology 1986, 67, 377–385. [Google Scholar] [CrossRef]
- Zimmer, C.; Boos, M.; Poulin, N.; Gosler, A.; Petit, O.; Robin, J.-P. Evidence of the Trade-Off between Starvation and Predation Risks in Ducks. PLoS ONE 2011, 6, e22352. [Google Scholar] [CrossRef]
- Lilliendahl, K. The Effect of Predator Presence on Body Mass in Captive Greenfinches. Anim. Behav. 1997, 53, 75–81. [Google Scholar] [CrossRef]
- Gentle, L.K.; Gosler, A.G. Fat Reserves and Perceived Predation Risk in the Great Tit, Parus major. Proc. R. Soc. Lond. B 2001, 268, 487–491. [Google Scholar] [CrossRef]
- Gosler, A.G.; Greenwood, J.J.D.; Perrins, C. Predation Risk and the Cost of Being Fat. Nature 1995, 377, 621–623. [Google Scholar] [CrossRef]
- Witter, M.S.; Cuthill, I.C.; Bonser, R.H.C. Experimental Investigations of Mass-Dependent Predation Risk in the European Starling, Sturnus vulgaris. Anim. Behav. 1994, 48, 201–222. [Google Scholar] [CrossRef]
- Metcalfe, N.B.; Ure, S.E. Diurnal Variation in Flight Performance and Hence Potential Predation Risk in Small Birds. Proc. R. Soc. B Biol. Sci. 1995, 261, 395–400. [Google Scholar] [CrossRef]
- Gosler, A.G. Environmental and Social Determinants of Winter Fat Storage in the Great Tit Parus major. J. Anim. Ecol. 1996, 65, 1–17. [Google Scholar] [CrossRef]
- Hake, M. Fattening Strategies in Dominance-Structured Greenfinch (Carduelis chloris) Flocks in Winter. Behav. Ecol. Sociobiol. 1996, 39, 71–76. [Google Scholar] [CrossRef]
- Krams, I. Mass-Dependent Take-off Ability in Wintering Great Tits (Parus major): Comparison of Top-Ranked Adult Males and Subordinate Juvenile Females. Behav. Ecol. Sociobiol. 2002, 51, 345–349. [Google Scholar] [CrossRef]
- van der Veen, I.T.; Lindström, K.M. Escape Flights of Yellowhammers and Greenfinches: More than Just Physics. Anim. Behav. 2000, 59, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Dietz, M.W.; Piersma, T.; Hedenström, A.; Brugge, M. Intraspecific Variation in Avian Pectoral Muscle Mass: Constraints on Maintaining Manoeuvrability with Increasing Body Mass. Funct Ecol. 2007, 21, 317–326. [Google Scholar] [CrossRef]
- van den Hout, P.J.; Mathot, K.J.; Maas, L.R.M.; Piersma, T. Predator Escape Tactics in Birds: Linking Ecology and Aerodynamics. Behav. Ecol. 2010, 21, 16–25. [Google Scholar] [CrossRef]
- Walters, B.T.; Cheng, T.N.N.; Doyle, J.; Guglielmo, C.G.; Clinchy, M.; Zanette, L.Y. Too Important to Tamper with: Predation Risk Affects Body Mass and Escape Behaviour but Not Escape Ability. Funct Ecol 2017, 31, 1405–1417. [Google Scholar] [CrossRef]
- Tryjanowski, P.; Skórka, P.; Sparks, T.H.; Biaduń, W.; Brauze, T.; Hetmański, T.; Martyka, R.; Indykiewicz, P.; Myczko, Ł.; Kunysz, P.; et al. Urban and Rural Habitats Differ in Number and Type of Bird Feeders and in Bird Species Consuming Supplementary Food. Environ. Sci. Pollut. Res. 2015, 22, 15097–15103. [Google Scholar] [CrossRef] [PubMed]
- Tryjanowski, P.; Møller, A.P.; Morelli, F.; Indykiewicz, P.; Zduniak, P.; Myczko, Ł. Food Preferences by Birds Using Bird-Feeders in Winter: A Large-Scale Experiment. Avian. Res. 2018, 9, 16. [Google Scholar] [CrossRef]
- Jokimäki, J.; Kaisanlahti-Jokimäki, M.-L. Residential Areas Support Overwintering Possibilities of Most Bird Species. Annales Zoologici Fennici 2012, 49, 240–256. [Google Scholar] [CrossRef]
- Jokimäki, J.; Suhonen, J. Distribution and Habitat Selection of Wintering Birds in Urban Environments. Landsc. Urban Plan. 1998, 39, 253–263. [Google Scholar] [CrossRef]
- Jones, W.; Kulma, K.; Bensch, S.; Cichoń, M.; Kerimov, A.; Krist, M.; Laaksonen, T.; Moreno, J.; Munclinger, P.; Slater, F.M.; et al. Interspecific Transfer of Parasites Following a Range-Shift in Ficedula Flycatchers. Ecol. Evol. 2018, 8, 12183–12192. [Google Scholar] [CrossRef]
- Orros, M.E.; Fellowes, M.D.E. Wild Bird Feeding in an Urban Area: Intensity, Economics and Numbers of Individuals Supported. Acta Ornithol. 2015, 50, 43–58. [Google Scholar] [CrossRef]
- Jones, D.N.; James Reynolds, S. Feeding Birds in Our Towns and Cities: A Global Research Opportunity. J. Avian Biol. 2008, 39, 265–271. [Google Scholar] [CrossRef]
- Cox, D.T.C.; Gaston, K.J. Human–Nature Interactions and the Consequences and Drivers of Provisioning Wildlife. Phil. Trans. R. Soc. B 2018, 373, 20170092. [Google Scholar] [CrossRef]
- Shutt, J.D.; Trivedi, U.H.; Nicholls, J.A. Faecal Metabarcoding Reveals Pervasive Long-Distance Impacts of Garden Bird Feeding. Proc. R. Soc. B. 2021, 288, 20210480. [Google Scholar] [CrossRef]
- Broggi, J.; Watson, H.; Nilsson, J.; Nilsson, J. Carry-over Effects on Reproduction in Food-supplemented Wintering Great Tits. J. Avian Biol. 2022, 2022, e02969. [Google Scholar] [CrossRef]
- Broggi, J.; Hohtola, E.; Koivula, K. Winter Feeding Influences the Cost of Living in Boreal Passerines. Ibis 2021, 163, 260–267. [Google Scholar] [CrossRef]
- Malpass, J.S.; Rodewald, A.D.; Matthews, S.N. Species-Dependent Effects of Bird Feeders on Nest Predators and Nest Survival of Urban American Robins and Northern Cardinals. Condor Ornithol. Appl. 2017, 119, 1–16. [Google Scholar] [CrossRef]
- Lawson, B.; Robinson, R.A.; Toms, M.P.; Risely, K.; MacDonald, S.; Cunningham, A.A. Health Hazards to Wild Birds and Risk Factors Associated with Anthropogenic Food Provisioning. Phil. Trans. R. Soc. B 2018, 373, 20170091. [Google Scholar] [CrossRef]
- Shutt, J.D.; Lees, A.C. Killing with Kindness: Does Widespread Generalised Provisioning of Wildlife Help or Hinder Biodiversity Conservation Efforts? Biol. Conserv. 2021, 261, 109295. [Google Scholar] [CrossRef]
- Pravosudov, V.V. A Dynamic Model of Short-Term Energy Management in Small Food-Caching and Non-Caching Birds. Behav. Ecol. 2001, 12, 207–218. [Google Scholar] [CrossRef]
- Pravosudov, V.V.; Lucas, J.R. The Effect of Social Dominance on Fattening and Food-Caching Behaviour in Carolina Chickadees, Poecile carolinensis. Anim. Behav. 2000, 60, 483–493. [Google Scholar] [CrossRef]
- Rytkönen, S.; Krams, I. Does Foraging Behaviour Explain the Poor Breeding Success of Great Tits Parus major in Northern Europe? J. Avian Biol. 2003, 34, 288–297. [Google Scholar] [CrossRef]
- Brūmelis, G.; Dauškane, I.; Elferts, D.; Strode, L.; Krama, T.; Krams, I. Estimates of Tree Canopy Closure and Basal Area as Proxies for Tree Crown Volume at a Stand Scale. Forests 2020, 11, 1180. [Google Scholar] [CrossRef]
- Rendenieks, Z.; Nikodemus, O.; Brūmelis, G. Dynamics in Forest Patterns during Times of Forest Policy Changes in Latvia. Eur. J. For. Res. 2015, 134, 819–832. [Google Scholar] [CrossRef]
- Krams, I. Dominance-Specific Vigilance in the Great Tit. J. Avian Biol. 1998, 29, 55. [Google Scholar] [CrossRef]
- Krams, I. Rank-Dependent Fattening Strategies of Willow Tit Parus montanus and Crested Tit P. cristatus Mixed Flock Members. Ornis Fenn. 1998, 75, 19–26. [Google Scholar]
- Krams, I.; Cīrule, D.; Krama, T.; Vrublevska, J. Extremely Low Ambient Temperature Affects Haematological Parameters and Body Condition in Wintering Great Tits (Parus major). J. Ornithol. 2011, 152, 889–895. [Google Scholar] [CrossRef]
- Ekman, J.B.; Lilliendahl, K. Using Priority to Food Access: Fattening Strategies in Dominance-Structured Willow Tit (Parus montanus) Flocks. Behav. Ecol. 1993, 4, 232–238. [Google Scholar] [CrossRef]
- McNamara, J.M.; Houston, A.I. The Value of Fat Reserves and the Tradeoff between Starvation and Predation: There’s a Special Providence in the Fall of a Sparrow Hamlet Act V Sc Ii. Acta Biotheor 1990, 38, 37–61. [Google Scholar] [CrossRef] [PubMed]
- Pravosudov, V.V.; Grubb, T.C.; Doherty, P.F.; Bronson, C.L.; Pravosudova, E.V.; Dolby, A.S. Social Dominance and Energy Reserves in Wintering Woodland Birds. Condor 1999, 101, 880–884. [Google Scholar] [CrossRef]
- Krams, I.A.; Luoto, S.; Krama, T.; Krams, R.; Sieving, K.; Trakimas, G.; Elferts, D.; Rantala, M.J.; Goodale, E. Egalitarian Mixed-Species Bird Groups Enhance Winter Survival of Subordinate Group Members but Only in High-Quality Forests. Sci. Rep. 2020, 10, 4005. [Google Scholar] [CrossRef] [PubMed]
- Krama, T.; Krams, R.; Cīrule, D.; Moore, F.R.; Rantala, M.J.; Krams, I.A. Intensity of Haemosporidian Infection of Parids Positively Correlates with Proximity to Water Bodies, but Negatively with Host Survival. J. Ornithol. 2015, 156, 1075–1084. [Google Scholar] [CrossRef]
- Kullberg, C.; Fransson, T.; Jakobsson, S. Impaired Predator Evasion in Fat Blackcaps (Sylvia atricapilla). Proc. R. Soc. B Biol. Sci. 1996, 263, 1671–1675. [Google Scholar]
- Veasey, J.S.; Metcalfe, N.B.; Houston, D.C. A Reassessment of the Effect of Body Mass upon Flight Speed and Predation Risk in Birds. Anim. Behav. 1998, 56, 883–889. [Google Scholar] [CrossRef] [PubMed]
- Sargisson, R.J.; McLean, I.G.; Brown, G.S.; White, K.G. Seasonal Variation in Pigeon Body Weight and Delayed Matching-to-Sample Performance. J. Exp. Anal. Behav. 2007, 88, 395–404. [Google Scholar] [CrossRef] [PubMed]
- McNamara, J.M.; Ekman, J.; Houston, A.J. The Effect of Thermoregulatory Substitution on Optimal Energy Reserves of Small Birds in Winter. Oikos 2004, 105, 192–196. [Google Scholar] [CrossRef]
- Sandell, M.; Smith, H.G. Dominance, Prior Occupancy, and Winter Residency in the Great Tit (Parus major). Behav. Ecol. Sociobiol. 1991, 29, 147–152. [Google Scholar] [CrossRef]
- Krams, I. Length of Feeding Day and Body Weight of Great Tits in a Single- and a Two-Predator Environment. Behav. Ecol. Sociobiol. 2000, 48, 147–153. [Google Scholar] [CrossRef]
- Gosler, A.G. Strategy and Constraint in the Winter Fattening Response to Temperature in the Great Tit Parus major. J. Anim. Ecol. 2002, 71, 771–779. [Google Scholar] [CrossRef]
- Ekman, J. Ecology of Non-Breeding Social Systems of Parus. Wilson Bull. 1989, 101, 263–288. [Google Scholar]
- Jokimäki, J.; Suhonen, J.; Kaisanlahti-Jokimäki, M.-L. Differential Long-Term Population Responses of Two Closely Related Human-Associated Sparrow Species with Respect to Urbanization. Birds 2021, 2, 230–249. [Google Scholar] [CrossRef]
- Auman, H.J.; Meathrel, C.E.; Richardson, A. Supersize Me: Does Anthropogenic Food Change the Body Condition of Silver Gulls? A Comparison Between Urbanized and Remote, Non-Urbanized Areas. Waterbirds 2008, 31, 122–126. [Google Scholar] [CrossRef]
- Risi, T.C.; Sumasgutner, P.; Cunningham, S.J. Anthropogenic Food Availability and Body Mass Maintenance in Urban Red-Winged Starlings Onychognathus morio. Ostrich 2021, 92, 16–25. [Google Scholar] [CrossRef]
- Hosseinian, S.A.; Hasanzadeh, F. Impact of High Dietary Energy on Obesity and Oxidative Stress in Domestic Pigeons. Vet. Med. Sci. 2021, 7, 1391–1399. [Google Scholar] [CrossRef]
- Hinde, R.A. The Behavior of the Great Tit (Parus major), and Some Other Related Species; E. J. Brill: Leiden, The Netherlands, 1952. [Google Scholar]
- Grabowska-Zhang, A.M.; Sheldon, B.C.; Hinde, C.A. Long-Term Familiarity Promotes Joining in Neighbour Nest Defence. Biol. Lett. 2012, 8, 544–546. [Google Scholar] [CrossRef] [PubMed]
- Giles, E.D.; Jackman, M.R.; MacLean, P.S. Modeling Diet-Induced Obesity with Obesity-Prone Rats: Implications for Studies in Females. Front. Nutr. 2016, 3, 50. [Google Scholar] [CrossRef]
- Lin, X.; Li, H. Obesity: Epidemiology, Pathophysiology, and Therapeutics. Front. Endocrinol. 2021, 12, 706978. [Google Scholar] [CrossRef]
- Cherian, G. Metabolic and Cardiovascular Diseases in Poultry: Role of Dietary Lipids. Poult. Sci. 2007, 86, 1012–1016. [Google Scholar] [CrossRef]
- Coogan, S.C.P.; Raubenheimer, D.; Zantis, S.P.; Machovsky-Capuska, G.E. Multidimensional Nutritional Ecology and Urban Birds. Ecosphere 2018, 9. [Google Scholar] [CrossRef]
- Bandyopadhyay, S. Systemic Clinical and Metabolic Diseases. In Pet Bird Diseases and Care; Springer: Singapore, 2017; pp. 167–252. ISBN 978-981-10-3673-6. [Google Scholar]
- Harakeh, S.M.; Khan, I.; Kumosani, T.; Barbour, E.; Almasaudi, S.B.; Bahijri, S.M.; Alfadul, S.M.; Ajabnoor, G.M.A.; Azhar, E.I. Gut Microbiota: A Contributing Factor to Obesity. Front. Cell. Infect. Microbiol. 2016, 6, 95. [Google Scholar] [CrossRef]
- Powell-Wiley, T.M.; Poirier, P.; Burke, L.E.; Després, J.-P.; Gordon-Larsen, P.; Lavie, C.J.; Lear, S.A.; Ndumele, C.E.; Neeland, I.J.; Sanders, P.; et al. Obesity and Cardiovascular Disease: A Scientific Statement From the American Heart Association. Circulation 2021, 143, e984–e1010. [Google Scholar] [CrossRef] [PubMed]
- Dantzer, R.; Kelley, K.W. Twenty Years of Research on Cytokine-Induced Sickness Behavior. Brain Behav. Immun. 2007, 21, 153–160. [Google Scholar] [CrossRef]
- Rantala, M.J.; Luoto, S.; Borráz-León, J.I.; Krams, I. Bipolar Disorder: An Evolutionary Psychoneuroimmunological Approach. Neurosci. Biobehav. Rev. 2021, 122, 28–37. [Google Scholar] [CrossRef]
- Kelley, K.W.; Bluthé, R.-M.; Dantzer, R.; Zhou, J.-H.; Shen, W.-H.; Johnson, R.W.; Broussard, S.R. Cytokine-Induced Sickness Behavior. Brain Behav. Immun. 2003, 17, 112–118. [Google Scholar] [CrossRef]
- Buehler, D.M.; Tieleman, B.I.; Piersma, T. Indices of Immune Function Are Lower in Red Knots (Calidris canutus) Recovering Protein Than in Those Storing Fat during Stopover in Delaware Bay. Auk 2010, 127, 394–401. [Google Scholar] [CrossRef]
- Matson, K.D.; Horrocks, N.P.C.; Tieleman, B.I.; Haase, E. Intense Flight and Endotoxin Injection Elicit Similar Effects on Leukocyte Distributions but Dissimilar Effects on Plasma-Based Immunological Indices in Pigeons. J. Exp. Biol. 2012, 215 Pt 21, 3734–3741. [Google Scholar] [CrossRef]
- Eikenaar, C.; Hegemann, A.; Packmor, F.; Kleudgen, I.; Isaksson, C. Not Just Fuel: Energy Stores Are Correlated with Immune Function and Oxidative Damage in a Long-Distance Migrant. Curr. Zool. 2020, 66, 21–28. [Google Scholar] [CrossRef]
- Eikenaar, C.; Hessler, S.; Hegemann, A. Migrating Birds Rapidly Increase Constitutive Immune Function during Stopover. R. Soc. Open Sci. 2020, 7, 192031. [Google Scholar] [CrossRef] [PubMed]
- Lima, S.L. Stress and Decision Making under the Risk of Predation: Recent Developments from Behavioral, Reproductive, and Ecological Perspectives. In Advances in the Study of Behavior; Elsevier: Amsterdam, The Netherlands, 1998; Volume 27, pp. 215–290. ISBN 978-0-12-004527-3. [Google Scholar]
- Krams, R.; Krama, T.; Brūmelis, G.; Elferts, D.; Strode, L.; Dauškane, I.; Luoto, S.; Šmits, A.; Krams, I.A. Ecological Traps: Evidence of a Fitness Cost in a Cavity-Nesting Bird. Oecologia 2021, 196, 735–745. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Krama, T.; Krams, R.; Popovs, S.; Trakimas, G.; Rantala, M.J.; Freeberg, T.M.; Krams, I.A. Permanent Ad-lib Feeders Decrease the Survival of Wintering Great Tits (Parus major). Birds 2023, 4, 225-235. https://doi.org/10.3390/birds4020019
Krama T, Krams R, Popovs S, Trakimas G, Rantala MJ, Freeberg TM, Krams IA. Permanent Ad-lib Feeders Decrease the Survival of Wintering Great Tits (Parus major). Birds. 2023; 4(2):225-235. https://doi.org/10.3390/birds4020019
Chicago/Turabian StyleKrama, Tatjana, Ronalds Krams, Sergejs Popovs, Giedrius Trakimas, Markus J. Rantala, Todd M. Freeberg, and Indrikis A. Krams. 2023. "Permanent Ad-lib Feeders Decrease the Survival of Wintering Great Tits (Parus major)" Birds 4, no. 2: 225-235. https://doi.org/10.3390/birds4020019
APA StyleKrama, T., Krams, R., Popovs, S., Trakimas, G., Rantala, M. J., Freeberg, T. M., & Krams, I. A. (2023). Permanent Ad-lib Feeders Decrease the Survival of Wintering Great Tits (Parus major). Birds, 4(2), 225-235. https://doi.org/10.3390/birds4020019






