Seasonal Differences in Expression of Neuropeptide Y (NPY) in Visual Centers of Spotted Munia (Lonchura punctulata)
Abstract
:Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Animal Housing and Experiment
2.2. Measurement of Body Mass and Food Intake
2.3. Tissue Preparation
2.3.1. Immunohistochemistry
2.3.2. Microscopy and Photography
2.3.3. NPY-ir Cell Counting
2.3.4. NPY-ir Fiber Area
2.4. Statistics
3. Results
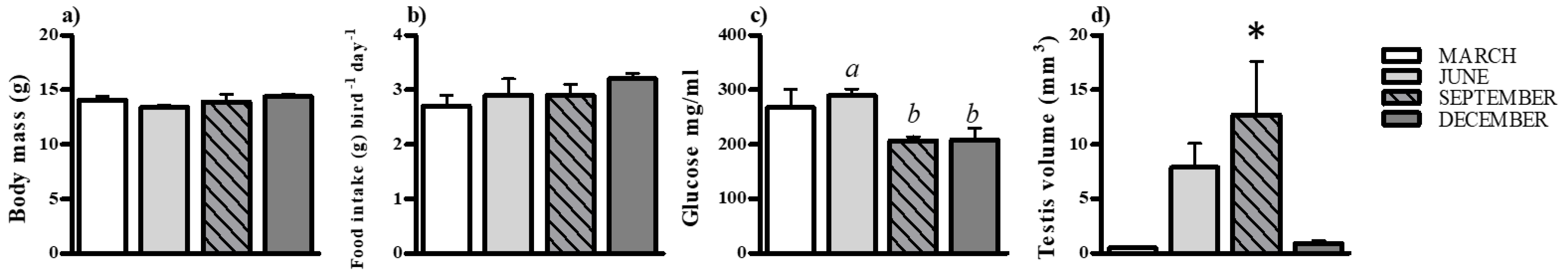
3.1. Effect on Physiology
3.2. Effect on Neuropeptide Y (NPY) Immunoreactivity
4. Discussion
4.1. Effect of Different Seasons on Physiology
4.2. Effect of Different Seasons on Expression of NPY
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Corning, A. Birds, Bees, and Human Visual Perception; Radiant Vision Systems: Redmond, WA, USA, 2020. [Google Scholar]
- de L. Brooke, M.; Hanley, S.; Laughlin, S.B. The scaling of eye size with body mass in birds. Proc. R. Soc. B 1999, 266, 1417. [Google Scholar]
- Bons, N.; Jallageas, M.; Assenmacher, I. Influence des recepteurs retiniens et extra-retiniens dans la stimulation testiculaire de la Caille par les “jours longs”. J. Physiol. 1975, 71, 265–266. [Google Scholar]
- Shields, K.M.; Yamamoto, J.T.; Millam, J.R. Reproductive behavior and LH levels of cockatiels (Nymphicus hollandicus) associated with photostimulation, nest-box presentation, and degree of mate access. Horm. Behav. 1989, 23, 68–82. [Google Scholar] [CrossRef]
- Endler, J.A.; Gaburro, J.; Kelley, L.A. Visual effects in great bowerbird sexual displays and their implications for signal design. Proc. R. Soc. B 2014, 281, 20140235. [Google Scholar] [CrossRef] [Green Version]
- Bennett, A.T.D.; Cuthill, I.C.; Partridge, J.C.; Maier, E.J. Ultraviolet vision and mate choice in zebra finch. Nature 1996, 380, 433–435. [Google Scholar]
- Bennett, A.T.D.; Cuthill, I.C.; Partridge, J.C.; Lunau, K. Ultraviolet plumage colors predict mate preferences in starlings. Proc. Natl. Acad. Sci. USA 1997, 94, 8618–8621. [Google Scholar]
- Lall, G.S.; Biello, S.M. Attenuation of phase shifts to light by activity or neuropeptide Y: A time course study. Brain Res. 2002, 957, 109–116. [Google Scholar]
- Söderberg, C.; Pieribone, V.A.; Dahlstrand, J.; Brodin, L.; Larhammar, D. Neuropeptide role of both peptide YY and neuropeptide Y in vertebrates suggested by abundant expression of their mRNAs in a cyclostome brain. J. Neurosci. Res. 1994, 37, 633–640. [Google Scholar] [CrossRef]
- Gaikwad, A.; Biju, K.C.; Saha, S.G.; Subhedar, N. Neuropeptide Y in the olfactory system, forebrain and pituitary of the teleost, Clarias batrachus. J. Chem. Neuroanat. 2004, 27, 55–70. [Google Scholar] [CrossRef]
- Danger, J.M.; Guy, J.; Benyamina, M.; Je’gou, S.; Leboulenger, F.; Cote’, J.; Tonon, M.C.; Pelletier, G.; Vaudry, H. Localization and identification of neuropeptide Y (NPY)-like immunoreactivity in the frog brain. Peptides 1985, 6, 1225–1233. [Google Scholar] [CrossRef]
- Medina, L.; Martì, E.; Artero, C.; Fasolo, A.; Puelles, L. Distribution of neuropeptide Y-like immunoreactivity in the brain of the lizard Gallotia galotti. J. Comp. Neurol. 1992, 319, 387–405. [Google Scholar] [CrossRef] [PubMed]
- Boswell, T.; Millam, J.R.; Li, Q.; Dunn, I.C. Cellular localization of neuropeptide Y mRNA and peptide in the brain of the Japanese quail and domestic chicken. Cell Tissue Res. 1998, 193, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Allen, Y.S.; Adrian, T.E.; Allen, J.M.; Tatemoto, K.; Crow, T.J.; Bloom, S.R.; Polak, J.M. Neuropeptide Y distribution in the rat brain. Science 1983, 221, 877–879. [Google Scholar] [CrossRef] [PubMed]
- Fraley, G.S.; Kuenzel, W.J. Precocious puberty in chicks (Gallus domesticus) induced by central injection of neuropeptide Y. Life Sci. 1993, 52, 1649–1656. [Google Scholar] [CrossRef]
- Fiore, M.; Clayton, N.S.; Pistillo, L.; Angelucci, F.; Alleva, E.; Aloe, L. Song behavior, NGF level and NPY distribution in the brain of adult male zebra finches. Behav. Brain. Res. 1999, 101, 85–92. [Google Scholar] [CrossRef]
- Mercer, R.E.; Chee, M.J.; Colmers, W.F. The role of NPY in hypothalamic mediated food intake. Front. Neuroendocrinol. 2011, 32, 398–415. [Google Scholar] [CrossRef]
- Wallman, J.; McKenna, O.C.; Burns, S.; Velez, J.; Weinstein, B. Relation of the accessory optic system and pretectum to optokinetic responses in chickens. In Progress in oculomotor research; Fuchs, A., Becker, W., Eds.; Elsevier: North Holland, NY, USA, 1981; pp. 435–442. [Google Scholar]
- Mishra, I.; Agarwal, N.; Rani, S.; Kumar, V. Scotostimulation of reproductive neural pathways and gonadal maturation are not correlated with hypothalamic expression of deiodinases in subtropical spotted munia. J. Neuroendocrinol. 2018, 30, e12627. [Google Scholar] [CrossRef]
- Surbhi; Rastogi, A.; Malik, S.; Rani, S.; Kumar, V. Changes in brain peptides associated with reproduction and energy homeostasis in photosensitive and photorefractory migratory redheaded buntings. Gen.Com. Endocrinol. 2016, 230–231, 67–75. [Google Scholar] [CrossRef]
- Rastogi, A.; Kumari, Y.; Rani, S.; Kumar, V. Neural correlates of migration: Activation of hypothalamic clock(s) in and out of migratory state in the black headed bunting (Emberiza melanocephala). PLoS ONE 2013, 8, e70065. [Google Scholar] [CrossRef] [Green Version]
- Thorsell, A.; Heilig, M. Diverse functions of neuropeptide Y revealed using genetically modified animals. Neuropeptides 2002, 36, 182–193. [Google Scholar] [CrossRef] [PubMed]
- Decressac, M.; Barker, R.A. Neuropeptide Y and its role in CNS disease and repair. Exp. Neurol. 2012, 238, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Gotzsche, C.R.; Woldbye, D.P. The role of NPY in learning and memory. Neuropeptides 2016, 55, 79–89. [Google Scholar] [CrossRef] [PubMed]
- Reichmann, F.; Holzer, P. Neuropeptide Y: A stressful review. Neuropeptides 2016, 55, 99–109. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berman, N.E.; Fredrickson, E. Morphology and laminar distribution of neuropeptide Y immunoreactive neurons in the human striate cortex. Synapse 1992, 11, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Van Reeth, O.; Goldman, S.; Schiffmann, S.; Verstappen, A.; Pelletier, G.; Vaudry, H.; Vanderhaeghen, J.J. Distribution of neuropeptide Y immunoreactivity in human visual cortex and underlying white matter. Peptides 1987, 8, 1107–1117. [Google Scholar] [CrossRef]
- Hendry, S.H.; Jones, E.G.; Emson, P.C. Morphology, distribution, and synaptic relations of somatostatin- and neuropeptide Y-immunoreactive neurons in rat and monkey neocortex. J. Neurosci. 1984, 4, 2497–2517. [Google Scholar] [CrossRef] [Green Version]
- Kuljis, R.O.; Rakic, P. Neuropeptide Y-containing neurons are situated predominantly outside cytochrome oxidase puffs in macaque visual cortex. Vis. Neurosci. 1989, 2, 57–62. [Google Scholar] [CrossRef]
- Kuljis, R.O.; Rakic, P. Distribution of neuropeptide Y-containing perikarya and axons in various neocortical areas in the macaque monkey. J. Comp. Neurol. 1989, 280, 383–392. [Google Scholar] [CrossRef]
- Kuljis, R.O.; Rakic, P. Multiple types of neuropeptide Y-containing neurons in primate neocortex. J. Comp. Neurol. 1989, 280, 393–409. [Google Scholar] [CrossRef]
- Hogan, D.; Berman, N.E. The development of neuropeptide Y immunoreactive neurons in cat visual cortical areas. Brain Res. Dev. Brain Res. 1992, 67, 343–369. [Google Scholar] [CrossRef]
- Wahle, P.; Meyer, G.; Albus, K. Localization of NPY-immunoreactivity in the cat’s visual cortex. Exp. Brain Res. 1986, 61, 364–374. [Google Scholar] [CrossRef]
- Wahle, P.; Meyer, G. Morphology and quantitative changes of transient NPY-ir neuronal populations during early postnatal development of the cat visual cortex. J. Comp. Neurol. 1987, 261, 165–192. [Google Scholar] [PubMed]
- Papadopoulos, G.C.; Parnavelas, J.G.; Cavanagh, M.E. Extensive co-existence of neuropeptides in the rat visual cortex. Brain Res. 1987, 420, 95–99. [Google Scholar] [CrossRef]
- Gonchar, Y.; Wang, Q.; Burkhalter, A. Multiple distinct subtypes of GABAergic neurons in mouse visual cortex identified by triple immunostaining. Front. Neuroanat. 2008, 28, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Montesano, A.; Baumgart, M.; Avallone, L.; Castaldo, L.; Lucini, C.; Tozzini, E.T.; Cellerino, A.; D’Angelo, L.; de Girolamo, P. Age-related central regulation of orexin and NPY in the short-lived African killifish Nothobranchius furzeri. J. Comp. Neurol. 2019, 527, 1508–1526. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Yamamoto, T.; Nakagawa, M.; Uemura, H. Neuropeptide Y-immunoreactive neuronal system and colocalization with FMRFamide in the optic lobe and peduncle complex of the octopus (Octopus vulgaris). Cell Tissue Res. 2002, 307, 255–264. [Google Scholar] [PubMed]
- Bennis, M.; Ba m’hamed, S.; Rio, J.P.; Le Cren, D.; Reperant, J.; Ward, R. The distribution of NPY-like immunoreactivity in the chameleon brain. Anat. Embryol. 2001, 203, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Chapman, A.M.; Debski, E.A. Neuropeptide Y immunoreactivity of a projection from the lateral thalamic nucleus to the optic tectum of the leopard frog. Vis. Neurosci. 1995, 12, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Aoki, C.; Pickel, V.M. Neuropeptide Y in cortex and striatum. Ultrastructural distribution and coexistence with classical neurotransmitters and neuropeptides. Ann. N. Y. Acad. Sci. 1990, 611, 186–205. [Google Scholar]
- Clark, C.M.; Clark, R.M.; Hoyle, J.A.; Dickson, T.C. Pathogenic or protective? Neuropeptide Y in amyotrophic lateral sclerosis. J. Neurochem. 2020, 156, 273–289. [Google Scholar]
- Duarte-Neves, J.; Gonçalves, N.; Cunha-Santos, J.; Simões, A.T.; den Dunnen, W.F.; Hirai, H.; Kügler, S.; Cavadas, C.; Pereira de Almeida, L. Neuropeptide Y mitigates neuropathology and motor deficits in mouse models of Machado-Joseph disease. Hum. Mol. Genet. 2015, 24, 5451–5463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klüver, H.; and Barrera, E. A method for the combined staining of cells and fibres in the nervous system. J. Neuropathol. Exp. Neurol. 1953, 12, 400–403. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Goguen, D.M.; Guido, M.E.; Rusak, B. Melatonin does not influence the expression of c-fos in the suprachiasmatic nucleus of rats and hamsters. Mol. Brain Res. 1997, 52, 242–248. [Google Scholar] [CrossRef]
- Singh, D.; Kumari, Y.; Rastogi, A.; Rani, S.; and Kumar, V. Neuropeptide Y mRNA and peptide in the night-migratory redheaded bunting brain. Cell. Tissue Res. 2013, 354, 551–562. [Google Scholar] [CrossRef]
- Rastogi, A.; Kumari, Y.; Rani, S.; Kumar, V. Phase inversion of neural activity in the olfactory and visual systems of a night-migratory bird during migration. Eur. J. Neurosci. 2011, 34, 99–109. [Google Scholar] [CrossRef]
- Stokes, T.M.; Leonard, C.M.; Nottebohm, F. The telencephalon, diencephalon, and mesencephalon of the canary, Serinus canaria, in stereotaxic coordinates, J. Comp. Neurol. 1974, 156, 337–374. [Google Scholar] [CrossRef]
- Kuenzel, W.J.; Masson, M.A. Stereotaxic Atlas of the Brain of the Chick (Gallus domesticus); Johns Hopkins University Press: Baltimore, MD, USA, 1988. [Google Scholar]
- Reiner, A.; David, J.P.; Bruce, L.L.; Butler, A.B.; Csillag, A.; Kuenzel, W.; Medina, L.A.; Paxinos, G.; Shimizu, T.; Striedter, G.; et al. Revised Nomenclature for Avian Telencephalon and Some Related Brainstem Nuclei. J. Comp. Neurol. 2004, 473, 377–414. [Google Scholar] [CrossRef]
- Gentner, T.Q.; Hulse, S.H.; Duffy, D.; Ball, G.F. Response biases in auditory forebrain regions of female songbirds following exposure to sexually relevant variation in male song. J. Neurobiol. 2001, 46, 48–58. [Google Scholar] [CrossRef]
- Shimizu, T.; Bowers, A.N.; Budzynski, C.A.; Kahn, M.C.; Bingman, V.P. What does a pigeon (Columba livia) brain look like during homing: Selective examination of ZENK expression. Behav. Neurosci. 2004, 118, 845–851. [Google Scholar] [CrossRef]
- Patzke, N.; Manns, M.; Güntürkün, O.; Ioalè, P.; and Gagliardo, A. Navigation-induced ZENK expression in the olfactory system of pigeons (Columba livia). Eur. J. Neurosci. 2010, 31, 2062–2072. [Google Scholar] [CrossRef]
- Sakharkar, A.J.; Singru, P.S.; Sarkar, K.; Subhedar, N. Neuropeptide Y in the forebrain of the adult male cichlid fish Oreochromis mossambicus: Distribution, effects of castration and testosterone replacement. J. Comp. Neurol. 2005, 489, 148–165. [Google Scholar] [CrossRef] [PubMed]
- Bhatt, D.; Lakhera, C.P.; Saklani-Chandola, A. Effect of artificially simulated annual photocycles on testicular cycle of the spotted munia Lonchura punctulata. Indian J. Exp. Biol. 1987, 24, 747–749. [Google Scholar]
- Remage-Healey, L.; Romero, L.M. Daily and seasonal variation in response to stress in captive starlings (Sturnus vulgaris): Glucose. Gen. Comp. Endocrinol. 2000, 119, 60–68. [Google Scholar] [CrossRef]
- Buttemer, W.A.; Addison, B.A.; Klasing, K.C. The energy cost of feather replacement is not intrinsically inefficient. Can. J. Zool. 2019, 98, 2. [Google Scholar] [CrossRef] [Green Version]
- Braun, E.J.; Sweazea, K.L. Glucose regulation in birds. Comp. Bio. Physiol. Part B 2008, 151, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Beuchat, C.A.; Chong, C.R. Hyperglycemia in hummingbirds and its consequences for hemoglobin glycation. Comp. Biochem. Physiol. A. 1998, 120, 409–416. [Google Scholar] [CrossRef]
- Renner, R.; Elcombe, A.M. Metabolic effects of feeding “carbohydrate-free” diets to chicks. J. Nutr. 1967, 93, 31–36. [Google Scholar] [CrossRef]
- Tinker, D.A.; Brosnan, J.T.; Herzberg, G.R. Interorgan metabolism of amino acids, glucose, lactate, glycerol and uric acid in the domestic fowl (Gallus domesticus). Biochem. J. 1986, 240, 829–836. [Google Scholar] [CrossRef] [Green Version]
- Adeghate, E. Pattern of distribution of neuropeptides in the camel lacrimal gland. Neuropeptides. 1996, 30, 566–571. [Google Scholar] [CrossRef]
- Cavallotti, C.; Artico, M.; Pescosolido, N.; Tranquilli Leali, F.M.; Pacella, E. Distribution of peptidergic nerve fibres in the guinea pig trabecular meshwork. Anat. Histol. Embryol. 2000, 29, 387–391. [Google Scholar] [CrossRef]
- Jones, M.A.; Marfurt, C.F. Peptidergic innervation of the rat cornea. Exp. Eye Res. 1998, 66, 421–435. [Google Scholar] [CrossRef]
- Lambiase, A.; Micera, A.; Sacchetti, M.; Cortes, M.; Mantelli, F.; Bonini, S. Alterations of tear neuromediators in dry eye disease. Arch. Ophthalmol. 2011, 129, 981–986. [Google Scholar] [CrossRef]
- Nilsson, S.F. Neuropeptide Y (NPY): A vasoconstrictor in the eye, brain and other tissues in the rabbit. Acta Physiol. Scand. 1991, 141, 455–467. [Google Scholar] [CrossRef]
- Paulsen, F.; Hallmann, U.; Paulsen, J.; Thale, A. Innervation of the cavernous body of the human efferent tear ducts and function in tear outflow mechanism. J. Anat. 2000, 197, 177–187. [Google Scholar] [CrossRef] [Green Version]
- Rittig, M.G.; Licht, K.; Funk, R.H. Innervation of the ciliary process vasculature and epithelium by nerve fibers containing catecholamines and neuropeptide Y. Ophthalmic Res. 1993, 25, 108–118. [Google Scholar] [CrossRef] [PubMed]
- Selbach, J.M.; Gottanka, J.; Wittmann, M.; Lutjen-Drecoll, E. Efferent and afferent innervation of primate trabecular meshwork and scleral spur. Investig. Ophthalmol. Vis. Sci. 2000, 41, 2184–2191. [Google Scholar]
- Selbach, J.M.; Schonfelder, U.; Funk, R.H. Arteriovenous anastomoses of the episcleral vasculature in the rabbit and rat eye. J. Glaucoma. 1998, 7, 50–57. [Google Scholar] [CrossRef]
- Stone, R.A.; Laties, A.M.; Emson, P.C. Neuropeptide Y and the ocular innervations of rat, guinea pig, cat and monkey. Neuroscience 1986, 17, 1207–1216. [Google Scholar] [CrossRef]
- Trivino, A.; de Hoz, R.; Rojas, B.; Salazar, J.J.; Ramirez, A.I.; Ramirez, J.M. NPY and TH innervation in human choroidal whole-mounts. Histol. Histopathol. 2005, 20, 393–402. [Google Scholar]
- Wetzel, R.K.; Eldred, W.D. Specialized neuropeptide Y- and glucagon-like immunoreactive amacrine cells in the peripheral retina of the turtle. Vis. Neurosci. 1997, 14, 867–877. [Google Scholar] [CrossRef] [PubMed]
- Yamaji, K.; Yoshitomi, T.; Usui, S. Action of biologically active peptides on monkey iris sphincter and dilator muscles. Exp. Eye Res. 2005, 80, 815–820. [Google Scholar] [CrossRef] [PubMed]
- Larhammar, D.; Blomqvist, A.G.; Yee, F.; Jazin, E.; Yoo, H.; Wahlested, C. Cloning and functional expression of a human neuropeptide Y/peptide YY receptor of the Y1 type. J. Biol. Chem. 1992, 267, 10935–10938. [Google Scholar] [CrossRef]
- Bruun, A.; Tornqvist, K.; Ehinger, B. Neuropeptide Y (NPY) immunoreactive neurons in the retina of different species. Histochemistry 1986, 86, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.J.; D’Angelo, I.; Lee, E.J.; Chun, M.H.; Brecha, N.C. Distribution and synaptic connectivity of neuropeptide Y-immunoreactive amacrine cells in the rat retina. J. Comp. Neurol. 2002, 446, 219–234. [Google Scholar] [CrossRef] [PubMed]
- Sinclair, J.R.; Nirenberg, S. Characterization of neuropeptide Y-expressing cells in the mouse retina using immunohistochemical and transgenic techniques. J. Comp. Neurol. 2001, 432, 296–306. [Google Scholar] [CrossRef]
- Ferriero, D.M.; Sagar, S.M. Development of neuropeptide Y-immunoreactive neurons in the rat retina. Brain Res. Dev. Brain Res. 1989, 48, 19–26. [Google Scholar] [CrossRef]
- Hiscock, J.; Straznicky, C. Neuropeptide Y- and substance P-like immunoreactive amacrine cells in the retina of the developing Xenopus laevis. Brain Res. Dev. Brain Res. 1990, 54, 105–113. [Google Scholar] [CrossRef]
- Hutsler, J.J.; Chalupa, L.M. Development of neuropeptide Y immunoreactive amacrine and ganglion cells in the pre- and postnatal cat retina. J. Comp. Neurol. 1995, 361, 152–164. [Google Scholar] [CrossRef]
- Jen, P.Y.; Li, W.W.; Yew, D.T. Immunohistochemical localization of neuropeptide Y and somatostatin in human fetal retina. Neuroscience 1994, 60, 727–735. [Google Scholar] [CrossRef]
- Jotwani, G.; Itoh, K.; Wadhwa, S. Immunohistochemical localization of tyrosine hydroxylase, substance P, neuropeptide-Y and leucine-enkephalin in developing human retinal amacrine cells. Brain Res. Dev. Brain Res. 1994, 77, 285–289. [Google Scholar] [CrossRef]
- Mathieu, M.; Tagliafierro, G.; Bruzzone, F.; Vallarino, M. Neuropeptide tyrosine-like immunoreactive system in the brain, olfactory organ and retina of the zebrafish, Danio rerio, during development. Brain Res. Dev. Brain Res. 2002, 139, 255–265. [Google Scholar] [CrossRef]
- Negishi, K.; Wagner, H.J. Differentiation of photoreceptors, glia, and neurons in the retina of the cichlid fish Aequidens pulcher; an immunocytochemical study. Brain Res. Dev. Brain Res. 1995, 89, 87–102. [Google Scholar] [CrossRef]
- Prada Oliveira, J.A.; Verastegui Escolano, C.; Gomez Luy, C.; Collantes Ruiz, J. Ontogenic attendance of neuropeptides in the embryo chicken retina. Histol. Histopathol. 2003, 18, 1013–1026. [Google Scholar]
- D’Angelo, I.; Brecha, N.C. Y2 receptor expression and inhibition of voltage dependent Ca2þ influx into rod bipolar cell terminals. Neuroscience 2004, 125, 1039–1049. [Google Scholar] [CrossRef] [PubMed]
- Morin, L.P.; Allen, C.N. The circadian visual system. Brain Res. Rev. 2006, 51, 102–127. [Google Scholar] [CrossRef]
- Benowitz, L.I.; Karten, H.J. Organization of the tectofugal visual pathway in the pigeon: A retrograde transport study. J. Comp. Neurol. 1976, 167, 503–520. [Google Scholar] [CrossRef]
- Granda, A.M.; Yazulla, S. The spectral sensitivity of single units in the nucleus rotundus of Pigeon, Columba livia. J. Gen. Physiol. 1971, 57, 363–384. [Google Scholar] [CrossRef] [Green Version]
- Beck, B.; Pourié, G. Ghrelin. Neuropeptide Y, and other feeding-regulatory peptides active in the hippocampus: Role in learning and memory. Nutr. Rev. 2013, 71, 541–561. [Google Scholar] [CrossRef] [PubMed]
- Kuenzel, W.J. Neuroanatomical substrates involved in the control of food intake. Poult. Sci. 1989, 68, 926–937. [Google Scholar] [CrossRef]
- Krützfeldt, N.O.; Wild, J.M. Definition and novel connections of the entopallium in the pigeon (Columba livia). J. Comp. Neurol. 2005, 490, 40–56. [Google Scholar] [CrossRef]
- Gioanni, H.; Palacios, A.; Sansonetti, A.; Varela, F. Role of the nucleus geniculatus lateralis ventralis (GLv) in the optokinetic reflex: A lesion study in the pigeon. Exp. Brain Res. 1991, 86, 601–607. [Google Scholar] [CrossRef] [PubMed]
- Kuenzel, W.J.; Kang, S.W.; Zhou, Z.J. Exploring avian deep-brain photoreceptors and their role in activating the neuroendocrine regulation of gonadal development. Poult. Sci. 2015, 94, 786–798. [Google Scholar] [CrossRef] [PubMed]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naseem, A.; Vaish, V.; Rani, S.; Malik, S. Seasonal Differences in Expression of Neuropeptide Y (NPY) in Visual Centers of Spotted Munia (Lonchura punctulata). Birds 2022, 3, 245-259. https://doi.org/10.3390/birds3030016
Naseem A, Vaish V, Rani S, Malik S. Seasonal Differences in Expression of Neuropeptide Y (NPY) in Visual Centers of Spotted Munia (Lonchura punctulata). Birds. 2022; 3(3):245-259. https://doi.org/10.3390/birds3030016
Chicago/Turabian StyleNaseem, Asma, Vaibhav Vaish, Sangeeta Rani, and Shalie Malik. 2022. "Seasonal Differences in Expression of Neuropeptide Y (NPY) in Visual Centers of Spotted Munia (Lonchura punctulata)" Birds 3, no. 3: 245-259. https://doi.org/10.3390/birds3030016





