Bottlenose Dolphins Produce Underwater Bubbles Linked to Cognitive Task Engagement but Not Success
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site and Subjects
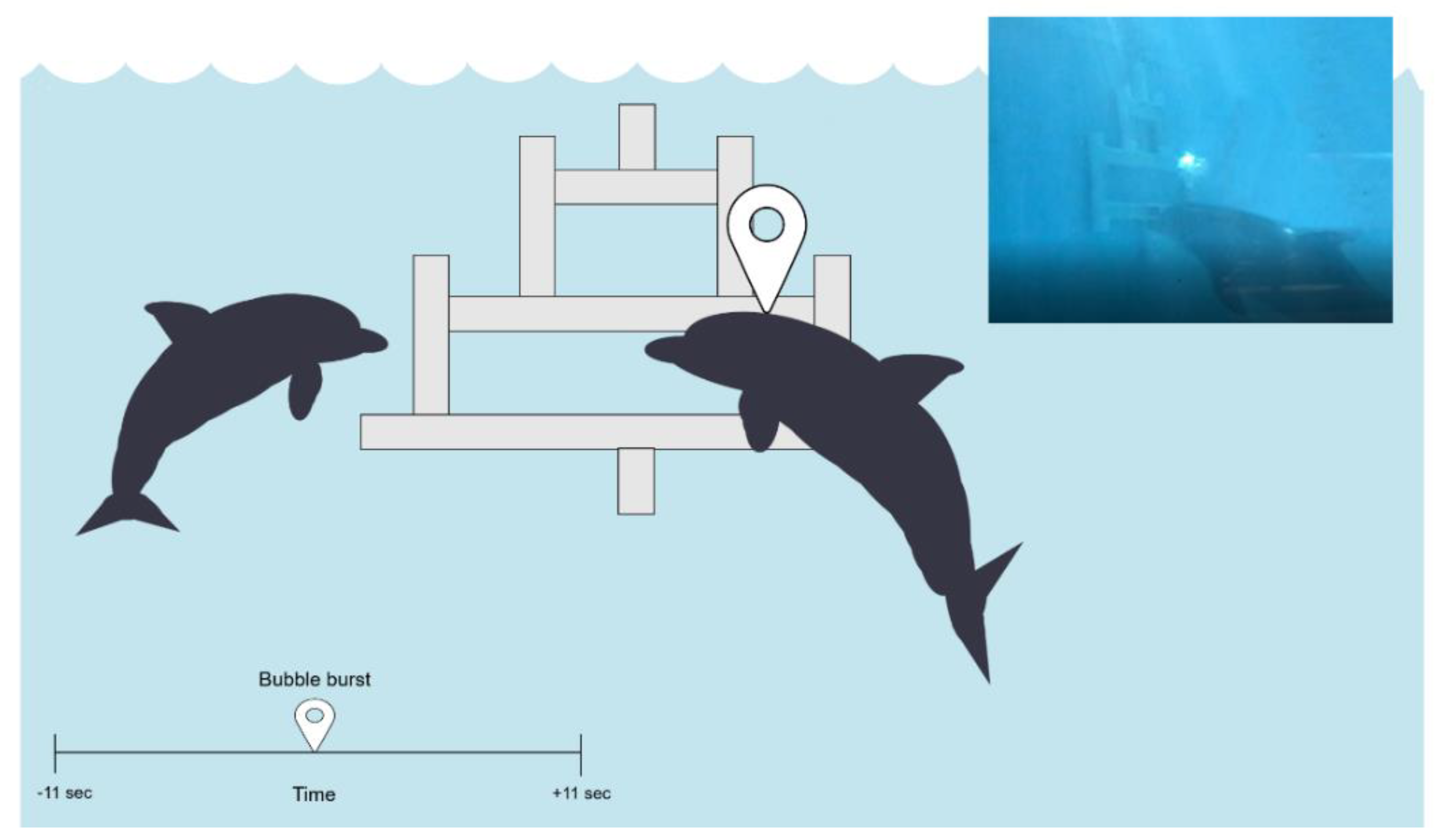
2.2. Task Design and Set-Up
2.3. Behavioural Data
2.3.1. Baseline
2.3.2. Task Trials
2.4. Statistical Analyses
2.4.1. Initial Analyses
2.4.2. THEME Analysis of Behavioural Sequences
2.4.3. Generalised Linear Modelling Task Engagement and Spectating
2.4.4. Burst Production as a Function of Respiration
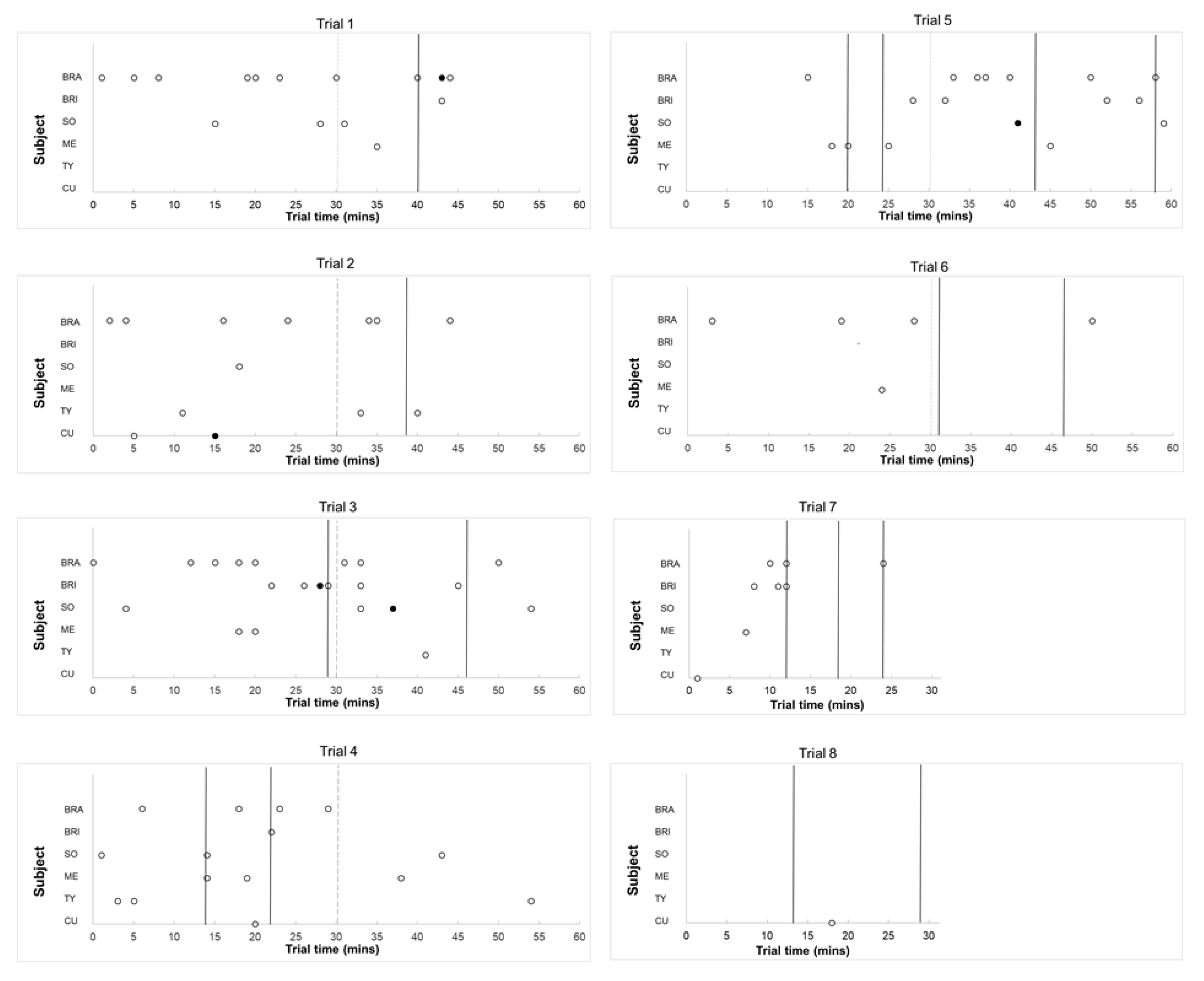
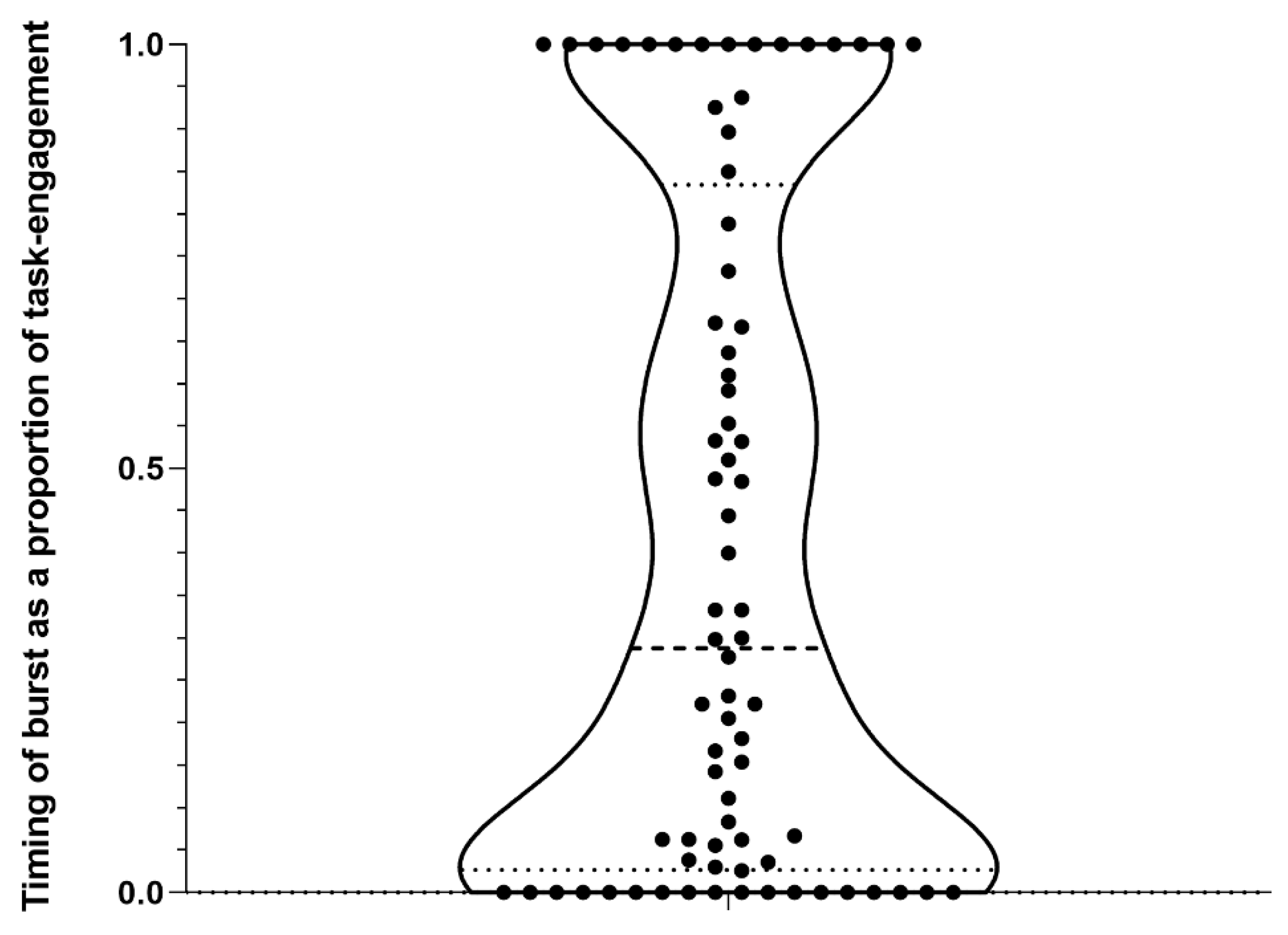
3. Results
4. Discussion
4.1. Burst Production, Task Engagement, and Affective state
4.2. Does Burst Production Have a Social Function?
4.3. Does Burst Production Have a Respiratory Function?
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Herrelko, E.S.; Vick, S.J.; Buchanan-Smith, H.M. Cognitive research in zoo-housed chimpanzees: Influence of personality and impact on welfare. Am. J. Primatol. 2012, 74, 828–840. [Google Scholar] [PubMed]
- Whitehouse, J.; Micheletta, J.; Powell, L.E.; Bordier, C.; Waller, B.M. The impact of cognitive testing on the welfare of group housed primates. PLoS ONE 2013, 8, e78308. [Google Scholar]
- Wagner, K.E.; Hopper, L.M.; Ross, S.R. Asymmetries in the production of self-directed behavior by chimpanzees and gorillas during a computerized cognitive test. Anim. Cogn. 2016, 19, 343–350. [Google Scholar]
- Clark, F.E. Great ape cognition and captive care: Can cognitive challenges enhance well-being? Appl. Anim. Behav. Sci. 2011, 135, 1–12. [Google Scholar]
- Clark, F.E. Cognitive enrichment and welfare: Current approaches and future directions. Anim. Behav. Cogn. 2017, 4, 52–71. [Google Scholar] [CrossRef]
- Zebunke, M.; Puppe, B.; Langbein, J. Effects of cognitive enrichment on behavioural and physiological reactions of pigs. Physiol. Behav. 2013, 118, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Oesterwind, S.; Nürnberg, G.; Puppe, B.; Langbein, J. Impact of structural and cognitive enrichment on the learning performance, behavior and physiology of dwarf goats (Capra aegagrus hircus). Appl. Anim. Behav. Sci. 2016, 177, 34–41. [Google Scholar] [CrossRef]
- Hagen, K.; Broom, D.M. Emotional reactions to learning in cattle. Appl. Anim. Behav. Sci. 2004, 85, 203–213. [Google Scholar] [CrossRef]
- McGowan, R.T.; Rehn, T.; Norling, Y.; Keeling, L.J. Positive affect and learning: Exploring the “Eureka Effect” in dogs. Anim. Cogn. 2014, 17, 577–587. [Google Scholar] [CrossRef]
- Coleman, K.; Pierre, P.J. Assessing anxiety in nonhuman primates. ILAR J. 2014, 55, 333–346. [Google Scholar] [CrossRef]
- Elder, C.M.; Menzel, C.R. Dissociation of cortisol and behavioral indicators of stress in an orangutan (Pongo pygmaeus) during a computerized task. Primates 2001, 42, 345–357. [Google Scholar] [CrossRef]
- Leavens, D.A.; Aureli, F.; Hopkins, W.D.; Hyatt, C.W. Effects of cognitive challenge on self-directed behaviors by chimpanzees (Pan troglodytes). Am. J. Primatol. 2001, 55, 1–14. [Google Scholar] [CrossRef]
- Yamanashi, Y.; Matsuzawa, T. Emotional consequences when chimpanzees (Pan troglodytes) face challenges: Individual differences in self-directed behaviours during cognitive tasks. Anim. Welf. 2010, 19, 25–30. [Google Scholar]
- Mendl, M.; Burman, O.H.; Paul, E.S. An integrative and functional framework for the study of animal emotion and mood. Proc. R. Soc. B Biol. Sci. 2010, 277, 2895–2904. [Google Scholar] [CrossRef]
- Paul, E.S.; Sher, S.; Tamietto, M.; Winkielman, P.; Mendl, M.T. Towards a comparative science of emotion: Affect and consciousness in humans and animals. Neurosci. Biobehav. Rev. 2020, 108, 749–770. [Google Scholar] [CrossRef] [PubMed]
- Clark, F.E. Marine mammal cognition and captive care: A proposal for cognitive enrichment in zoos and aquariums. J. Zoo Aquar. Res. 2013, 1, 1–6. [Google Scholar]
- Piscitelli, M.A.; Raverty, S.A.; Lillie, M.A.; Shadwick, R.E. A review of cetacean lung morphology and mechanics. J. Morphol. 2013, 274, 1425–1440. [Google Scholar] [CrossRef]
- Moreno, K.; Macgregor, R. Bubble trails, bursts, rings, and more: A review of multiple bubble types produced by cetaceans. Anim. Behav. Cogn. 2019, 6, 105–126. [Google Scholar] [CrossRef]
- Wiley, D.; Ware, C.; Bocconcelli, A.; Cholewaik, D.; Friedlaender, A.; Thompson, M.; Weinrich, M. Underwater components of humpback whale bubble-net feeding behaviour. Behaviour 2011, 148, 575–602. [Google Scholar]
- Pace, D.S. Fluke-made bubble rings as toys in bottlenose dolphin calves (Tursiops truncatus). Aquat. Mamm. 2000, 26, 57–64. [Google Scholar]
- Paulos, R.D.; Trone, M.; Kuczaj, S.A., II. Play in wild and captive cetaceans. Int. J. Comp. Psychol. 2010, 23, 701–722. [Google Scholar]
- Dudzinski, K.M. Contact behavior and signal exchange in Atlantic spotted dolphins (Stenella frontalis). Aquat. Mamm. 1998, 24, 129–142. [Google Scholar]
- Hill, H.M.; Kahn, M.S.; Brilliott, L.J.; Roberts, B.M.; Gutierrez, C.; Artz, S. Beluga (Delphinapterus leucas) Bubble bursts: Surprise, protection, or play? Int. J. Comp. Psychol. 2011, 24, 235–243. [Google Scholar]
- Pryor, K.W. Non-acoustic communication in small cetaceans: Glance, touch, position, gesture, and bubbles. In Sensory Abilities of Cetaceans; Thomas, J.A., Kastelein, R.A., Eds.; Springer: Boston, MA, USA, 1990; pp. 537–544. [Google Scholar]
- Delfour, F.; Marten, K. Mirror image processing in three marine mammal species: Killer whales (Orcinus orca), false killer whales (Pseudorca crassidens) and California sea lions (Zalophus californianus). Behav. Process. 2001, 53, 181–190. [Google Scholar] [CrossRef]
- Moreno, K. Cetacean Exhalation: An Examination of Bottlenose Dolphin (Tursiops Truncatus) Use of Three Bubble Production Types through Associated Behaviors. Ph.D. Thesis, University of Southern Mississippi, Hattiesburg, MS, USA, 2017. [Google Scholar]
- Clark, F.E.; Davies, S.L.; Madigan, A.W.; Warner, A.J.; Kuczaj, S.A. Cognitive enrichment for bottlenose dolphins (Tursiops truncatus): Evaluation of a novel underwater maze device. Zoo Biol. 2013, 32, 608–619. [Google Scholar] [CrossRef] [PubMed]
- Bateson, M.; Martin, P. Measuring Behaviour: An Introductory Guide; Cambridge University Press: Cambridge, UK, 2021. [Google Scholar]
- Holobinko, A.; Waring, G.H. Conflict and reconciliation behavior trends of the bottlenose dolphin (Tursiops truncatus). Zoo Biol. 2010, 29, 567–585. [Google Scholar] [CrossRef] [PubMed]
- Magnusson, M.S.; Burgoon, J.K.; Casarrubea, M. Discovering Hidden Temporal Patterns in Behavior and Interaction; Springer: New York, NY, USA, 2016. [Google Scholar]
- Magnusson, M.S. T-pattern detection and analysis (TPA) with THEMETM: A mixed methods approach. Front. Psychol. 2020, 10, 2663. [Google Scholar] [CrossRef]
- Fish, F.E.; Hui, C.A. Dolphin swimming—A review. Mammal Rev. 1991, 21, 181–195. [Google Scholar] [CrossRef]
- Videler, J.; Kamermans, P. Differences between upstroke and downstroke in swimming dolphins. J. Exp. Biol. 1985, 119, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Serres, A.; Delfour, F. Social behaviors modulate bottlenose dolphins (Tursiops truncatus) breathing rate. Anim. Behav. Cogn. 2019, 6, 127–140. [Google Scholar] [CrossRef]
- Kuczaj, S.; Lacinak, T.; Fad, O.; Trone, M.; Solangi, M.; Ramos, J. Keeping environmental enrichment enriching. Int. J. Comp. Psychol. 2002, 15, 127–137. [Google Scholar]
- Ellsworth, P.C. Appraisal theory: Old and new questions. Emot. Rev. 2013, 5, 125–131. [Google Scholar] [CrossRef]
- Moors, A.; Ellsworth, P.C.; Scherer, K.R.; Frijda, N.H. Appraisal theories of emotion: State of the art and future development. Emot. Rev. 2013, 5, 119–124. [Google Scholar] [CrossRef]
- McCowan, B.; Marino, L.; Vance, E.; Walke, L.; Reiss, D. Bubble ring play of bottlenose dolphins (Tursiops truncatus): Implications for cognition. J. Comp. Psychol. 2000, 114, 98–106. [Google Scholar] [CrossRef][Green Version]
- Lilley, M.K.; de Vere, A.J.; Yeater, D.B.; Kuczaj, S.A., II. Characterizing curiosity-related behavior in bottlenose (Tursiops truncatus) and rough-toothed (Steno bredanensis) dolphins. Int. J. Comp. Psychol. 2018, 31, 1–22. [Google Scholar] [CrossRef]
- Dudzinski, K.M. Communication and Behavior in the Atlantic Spotted Dolphins (Stenella frontalis): Relationship between Vocal and Behavioral Activities. Ph.D. Thesis, Texas A&M University, College Station, TX, USA, 1996. [Google Scholar]
- Shane, S.H. Behavior and ecology of the bottlenose dolphin at Sanibel Island, Florida. In The Bottlenose Dolphin; Leatherwood, S., Reeves, R.R., Eds.; Academic Press: San Diego, CA, USA, 1990; pp. 245–265. [Google Scholar]
- Herzing, D.L. Vocalizations and associated underwater behavior of free-ranging Atlantic spotted dolphins, Stenella frontalis and bottlenose dolphins, Tursiops truncatus. Aquat. Mamm. 1996, 22, 61–79. [Google Scholar]
- Bowles, A.E.; Anderson, R.C. Behavioral responses and habituation of pinnipeds and small cetaceans to novel objects and simulated fishing gear with and without a pinger. Aquat. Mamm. 2012, 38, 161. [Google Scholar] [CrossRef]
- Ridgway, S.; Dibble, D.S.; Van Alstyne, K.; Price, D. On doing two things at once: Dolphin brain and nose coordinate sonar clicks, buzzes and emotional squeals with social sounds during fish capture. J. Exp. Biol. 2015, 218, 3987–3995. [Google Scholar] [CrossRef] [PubMed]
- Dibble, D.S.; Van Alstyne, K.R.; Ridgway, S. Dolphins signal success by producing a victory squeal. International. J. Comp. Psychol. 2016, 29. [Google Scholar] [CrossRef]
- King, S.L.; Guarino, E.; Donegan, K.; McMullen, C.; Jaakkola, K. Evidence that bottlenose dolphins can communicate with vocal signals to solve a cooperative task. R. Soc. Open Sci. 2021, 8, 202073. [Google Scholar] [CrossRef]
- Kuczaj, S.A.; Winship, K.A.; Eskelinen, H.C. Can bottlenose dolphins (Tursiops truncatus) cooperate when solving a novel task? Anim. Cogn. 2015, 18, 543–550. [Google Scholar] [CrossRef]
- Matrai, E.; Kwok, S.T.; Boos, M.; Pogány, Á. Cognitive enrichment device provides evidence for intersexual differences in collaborative actions in Indo-Pacific bottlenose dolphins (Tursiops aduncus). Anim. Cogn. 2021, 1–11. [Google Scholar] [CrossRef]
- Jaakkola, K.; Guarino, E.; Donegan, K.; King, S.L. Bottlenose dolphins can understand their partner’s role in a cooperative task. Proc. R. Soc. B Biol. Sci. 2018, 285, 20180948. [Google Scholar] [CrossRef] [PubMed]
- Clark, F.E. Space to choose: Network analysis of social preferences in a captive chimpanzee community, and implications for management. Am. J. Primatol. 2011, 73, 748–757. [Google Scholar] [CrossRef] [PubMed]
- Wood, F.G. Underwater Sound Production and Concurrent Behavior of Captive Porpoises, Tursiops truncatus and Stenella plagiodon. Bull. Mar. Sci. Gulf Caribb. 1953, 3, 120–133. [Google Scholar]
- Van der Woude, S.E. Bottlenose dolphins (Tursiops truncatus) moan as low in frequency as baleen whales. J. Acoust. Soc. Am. 2009, 126, 1552. [Google Scholar] [CrossRef]
- Boissy, A.; Manteuffel, G.; Jensen, M.B.; Moe, R.O.; Spruijt, B.; Keeling, L.; Winckler, C.; Forkman, B.; Dimitrov, I.; Langbein, J.; et al. Assessment of positive emotions in animals to improve their welfare. Physiol. Behav. 2007, 92, 375–397. [Google Scholar] [CrossRef] [PubMed]
| Behaviour | Description 1 |
|---|---|
| Bubble production: | |
| Bubble burst | A large singular bubble, shaped like a mushroom cloud, produced from the blowhole. |
| Bubble scrap | A thin ribbon-like bubble, produced from the blowhole, which is immediately eaten by the producer. |
| Task engagement: | |
| Approach | Movement within one body length (approx. 2.5 m) of the task, with the body aligned towards the task (i.e., not passively swimming past). |
| Observe | Stationary or slowly moving within one body length of the task while paying close visual attention to the task. Head may be moving around. |
| Contact | Physical contact with the task. Includes push, pull, bite, slide ball (with rostrum), gentle touch. |
| Play with ball | Playful interactions with the (gelatine) task ball. Includes throwing ball around in water, mouthing, chasing and tussling. |
| Task spectating: | |
| Vigilance of task user | Close visual inspection of a dolphin engaged with the task, within one body length. |
| Vigilance of experimenter | Close visual attention to experimenters or trainers above water. |
| Possible frustration behaviour: | |
| Jaw-clap | Rapid opening and closing of the jaw. |
| Tail-beat | Strong vertical movement of the tail flukes. |
| Subject (Age in Years) | Rearing History | Burst Frequency | Success Frequency | Total Task Engagement (min) | Total Bout Frequency | Median Bout Duration (s) |
|---|---|---|---|---|---|---|
| Brandy (12) | c | 47 | 12 | 134 | 205 | 26 (40) |
| Brisby (18) | c | 17 | 5 | 92 | 141 | 26 (37) |
| Sonny (12) | c | 17 | 0 | 51 | 93 | 21.5 (41.8) |
| Merlin (26) | w | 12 | 0 | 20 | 43 | 22 (26.5) |
| Ty (22) | c | 7 | 0 | 36 | 91 | 18 (21.5) |
| Cupid (10) | w | 6 | 0 | 27 | 65 | 16 (35) |
| Total | 106 | 17 | 360 | 638 |
| Explanatory Variable | Regression Coefficient | Standard Error | p-Value |
|---|---|---|---|
| Proportion of interval 1 spent task-engaged | 0.66 | 0.16 | <0.001 |
| Frequency spectators | 0.54 | 0.09 | <0.001 |
| Frequency successes | 0.13 | 0.07 | 0.061 |
| Frequency new ball loads | −0.51 | 17.51 | 0.977 |
| Frequency ball location changes | −0.11 | 0.10 | 0.254 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alexander, E.; Abrahams, M.; Clark, F.E. Bottlenose Dolphins Produce Underwater Bubbles Linked to Cognitive Task Engagement but Not Success. J. Zool. Bot. Gard. 2021, 2, 287-299. https://doi.org/10.3390/jzbg2020020
Alexander E, Abrahams M, Clark FE. Bottlenose Dolphins Produce Underwater Bubbles Linked to Cognitive Task Engagement but Not Success. Journal of Zoological and Botanical Gardens. 2021; 2(2):287-299. https://doi.org/10.3390/jzbg2020020
Chicago/Turabian StyleAlexander, Elena, Mark Abrahams, and Fay E. Clark. 2021. "Bottlenose Dolphins Produce Underwater Bubbles Linked to Cognitive Task Engagement but Not Success" Journal of Zoological and Botanical Gardens 2, no. 2: 287-299. https://doi.org/10.3390/jzbg2020020
APA StyleAlexander, E., Abrahams, M., & Clark, F. E. (2021). Bottlenose Dolphins Produce Underwater Bubbles Linked to Cognitive Task Engagement but Not Success. Journal of Zoological and Botanical Gardens, 2(2), 287-299. https://doi.org/10.3390/jzbg2020020







