Activity, Social Relationships, and Maternal Care in a Bottlenose Dolphin Group under Professional Care
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Activity
3.2. Dyadic Relationships
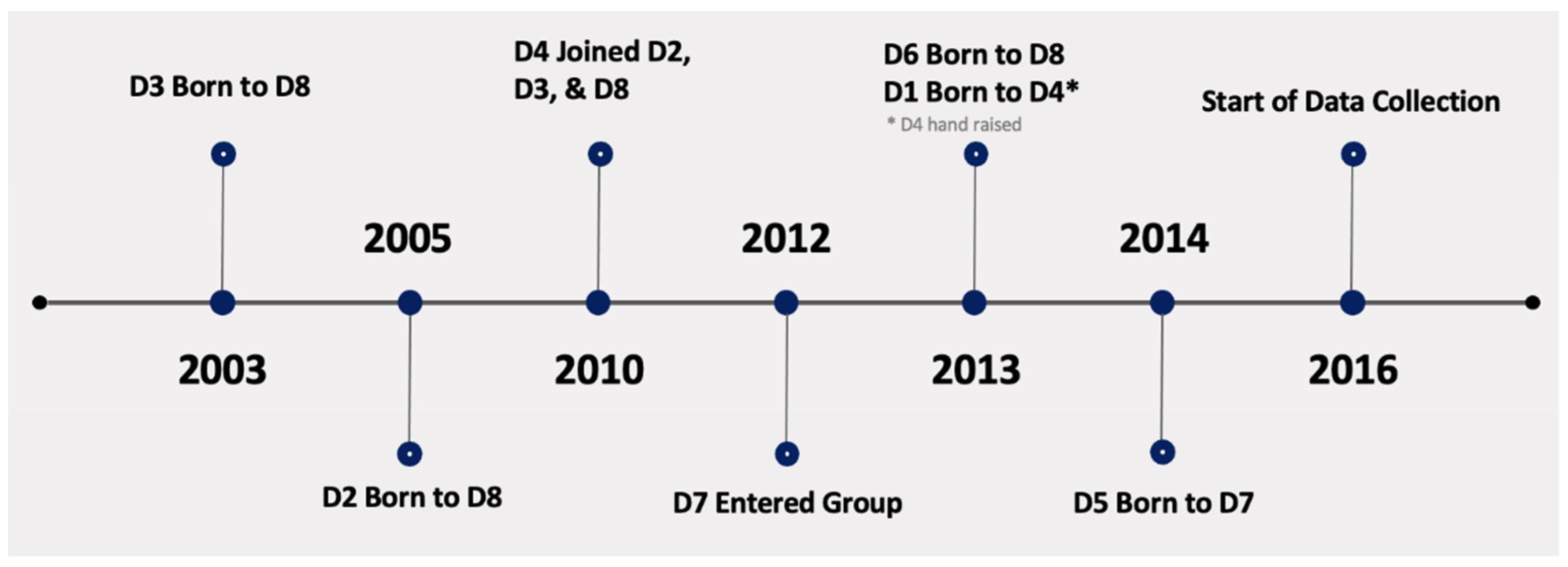
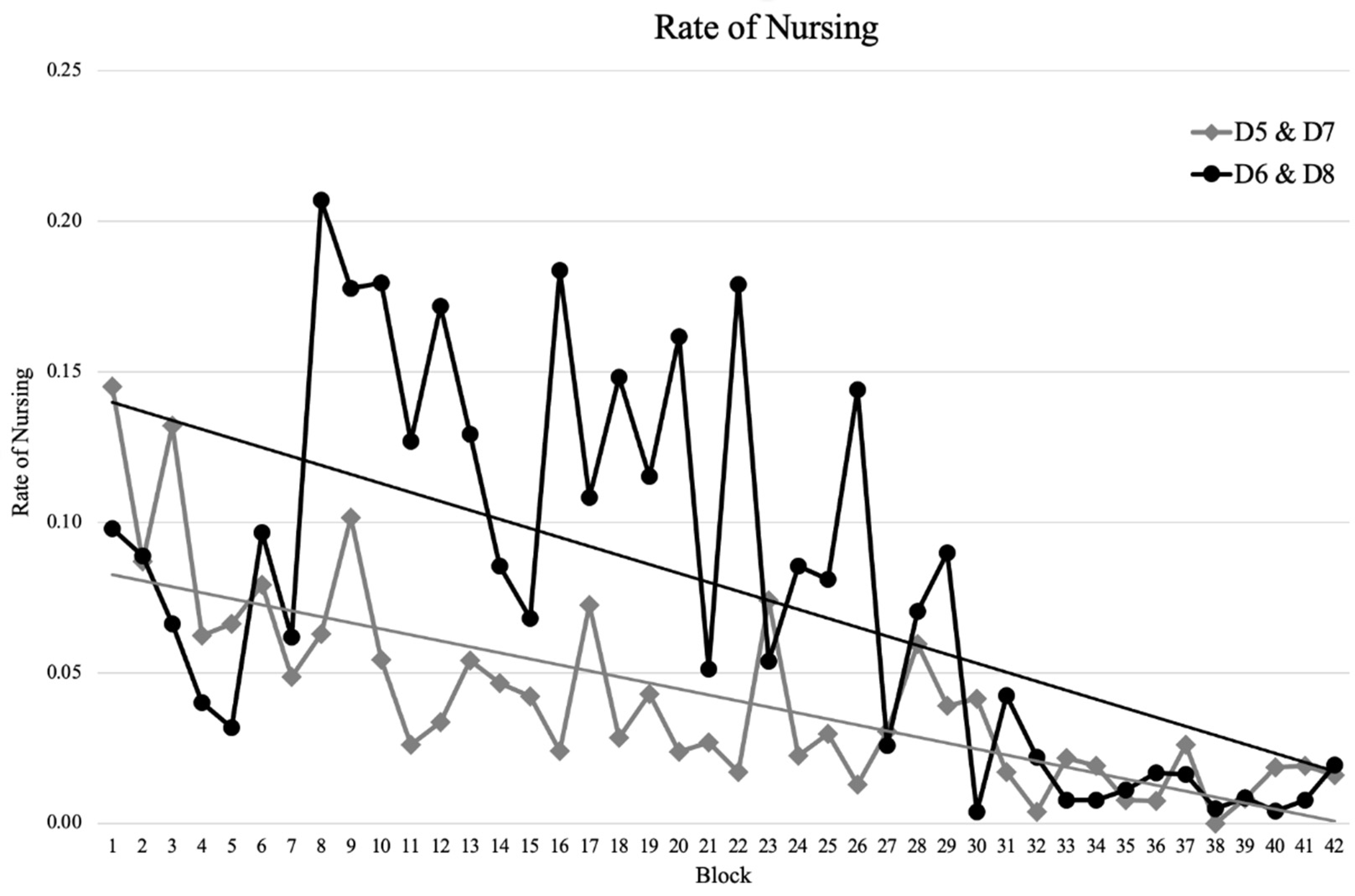
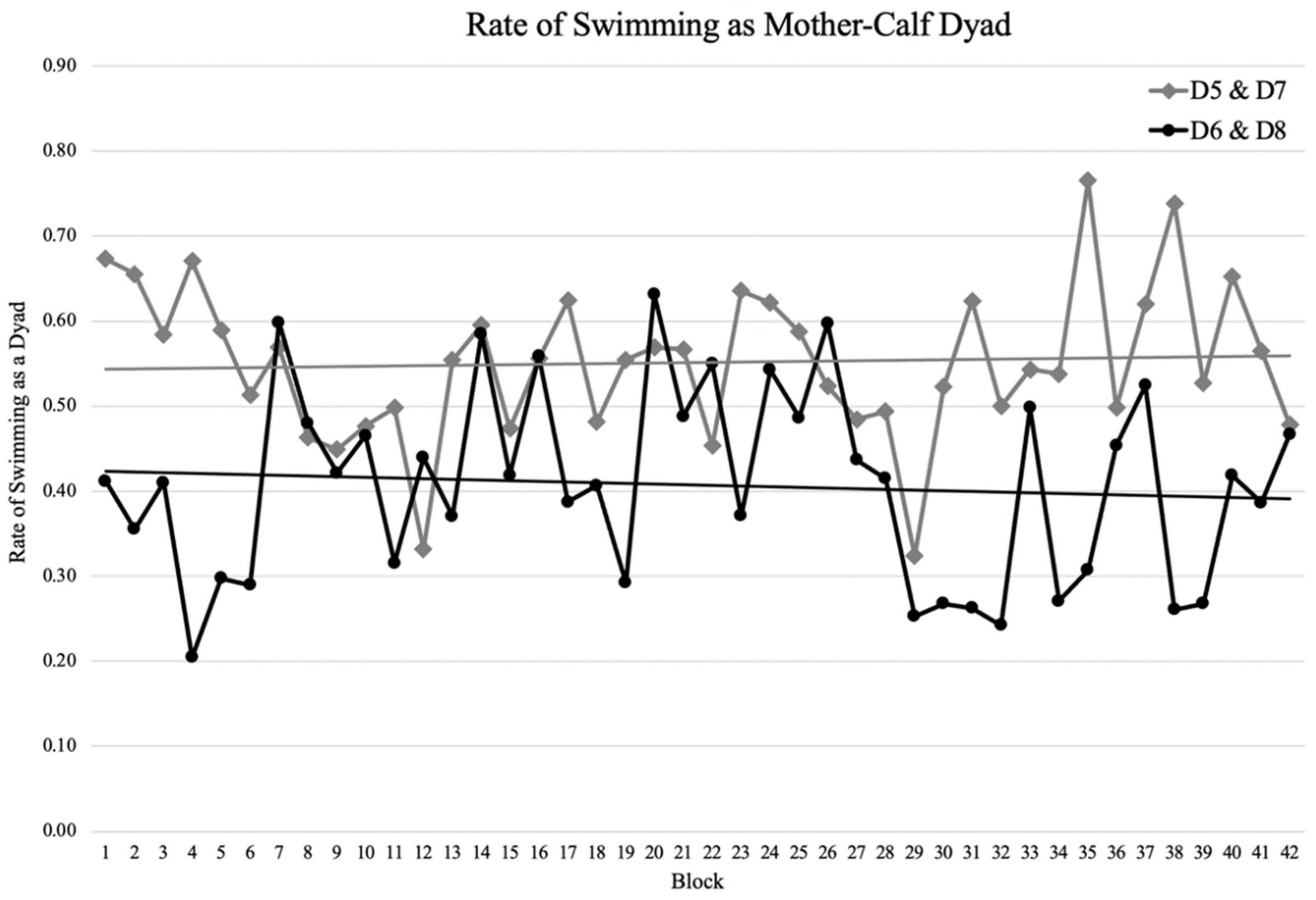
3.3. Maternal Care
4. Discussion
4.1. Activity
4.2. Dyadic Relationships
4.3. Maternal Care
4.4. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Connor, R.C.; Wells, R.S.; Mann, J.; Read, A.J. The bottlenose dolphin: Social relationships in a fission-fusion society. In Cetacean Societies: Field Studies of Dolphins and Whales, 1st ed.; Mann, J., Connor, R., Tyack, P., Whitehead, H., Eds.; The University of Chicago Press: Chicago, IL, USA, 2000; pp. 91–126. [Google Scholar]
- Gowans, S.; Würsig, B.; Karczmarski, L. The Social Structure and Strategies of Delphinids: Predictions Based on an Ecological Framework. Adv. Mar. Biol. 2007, 53, 195–294. [Google Scholar] [CrossRef]
- Bruck, J.N. Decades-long social memory in bottlenose dolphins. Proc. R. Soc. B Boil. Sci. 2013, 280, 20131726. [Google Scholar] [CrossRef] [Green Version]
- Connor, R.C.; Smolker, R.A.; Richards, A.F. Two levels of alliance formation among male bottlenose dolphins (Tursiops sp.). Proc. Natl. Acad. Sci. USA 1992, 89, 987–990. [Google Scholar] [CrossRef] [Green Version]
- Smolker, R.A.; Richards, A.F.; Connor, R.C.; Pepper, J.W. Sex differences in patterns of association among Indian Ocean bottlenose dolphins. Behaviour 1992, 123, 38–69. [Google Scholar] [CrossRef]
- Tyack, P. Whistle repertoires of two bottlenosed dolphins, Tursiops truncatus: Mimicry of signature whistles? Behav. Ecol. Sociobiol. 1986, 18, 251–257. [Google Scholar] [CrossRef]
- Wells, R.S.; Scott, M.D.; Irvine, A.B. The Social Structure of Free-Ranging Bottlenose Dolphins. In Current Mammalogy; Springer: Boston, MA, USA, 1987; pp. 247–305. [Google Scholar]
- Wells, R.S. The role of long-term study in understanding the social structure of a bottlenose dolphin community. In Dolphin Societies: Discoveries and Puzzles, 1st ed.; Pryor, K., Norris, K., Eds.; University of California Press: Berkeley, CA, USA, 1991; pp. 199–225. [Google Scholar]
- Kuczaj, S.A.; Makecha, R.; Trone, M.; Paulos, R.D.; Ramos, J.A. Role of peers in cultural innovation and cultural transmission: Evidence from the play of dolphin calves. Int. J. Comp. Psychol. 2006, 31, 223–240. [Google Scholar]
- Mackey, A.; Makecha, R.; Kuczaj, S. The Development of Social Play in Bottlenose Dolphins (Tursiops truncatus). Anim. Behav. Cogn. 2014, 1, 19–35. [Google Scholar] [CrossRef]
- Von Streit, C.; Ganslosser, U.; von Fersen, L. Behavioral development of two captive mother-calf dyads of bottlenose dolphins (Tursiops truncatus) in the calves’ first year. Int. J. Comp. Psychol. 2013, 26, 176–196. [Google Scholar] [CrossRef]
- Walker, R.T.; Miller, L.J.; Ii, S.A.K.; Solangi, M. Seasonal, Diel, and Age Differences in Activity Budgets of a Group of Bottlenose Dolphins (Tursiops truncatus) Under Professional Care. Int. J. Comp. Psychol. 2017, 30, 30. [Google Scholar] [CrossRef]
- Carlstead, K.; Shepherdson, D. Effects of environmental enrichment on reproduction. Zoo Biol. 1994, 13, 447–458. [Google Scholar] [CrossRef]
- McPhee, M.E.; Carlstead, K. The importance of maintaining natural behaviors in captive mammals. In Wild Mammals in Captivity: Principles and Techniques for Zoo Management, 2nd ed.; University of Chicago Press: Chicago, IL, USA, 2010; pp. 303–313. [Google Scholar]
- Rose, P.; Riley, L. The use of Qualitative Behavioural Assessment to zoo welfare measurement and animal husbandry change. J. Zoo Aquar. Res. 2019, 7, 150–171. [Google Scholar]
- De Moura Lima, A. Production and Perception of Acoustic Signals in Captive Bottlenose Dolphins (Tursiops truncatus): Con-textual Use of Social Signals and Recognition of Artificial Labels. Ph.D. Thesis, Université Rennes 1, Rennes, France, 16 December 2019. Available online: https://tel.archives-ouvertes.fr/tel-02412889 (accessed on 24 March 2021).
- Saayman, G.S.; Tayler, C.K.; Bower, D. Diurnal Activity Cycles in Captive and Free-Ranging Indian Ocean Bottlenose Dolphins (Tursiops Aduncus Ehrenburg). Behaviour 1973, 44, 212–233. [Google Scholar] [CrossRef]
- Serres, A.; Delfour, F. Environmental changes and anthropogenic factors modulate social play in captive bottlenose dolphins (Tursiops truncatus). Zoo Biol. 2017, 36, 99–111. [Google Scholar] [CrossRef]
- Serres, A.; Hao, Y.; Wang, D. Body Contacts and Social Interactions in Captive Odontocetes Are Influenced by the Context: An Implication for Welfare Assessment. Animals 2020, 10, 924. [Google Scholar] [CrossRef]
- Shorter, K.A.; Shao, Y.; Ojeda, L.; Barton, K.; Rocho-Levine, J.; Van Der Hoop, J.; Moore, M. A day in the life of a dolphin: Using bio-logging tags for improved animal health and well-being. Mar. Mammal Sci. 2017, 33, 785–802. [Google Scholar] [CrossRef]
- Clark, F.E. Marine mammal cognition and captive care: A proposal for cognitive enrichment in zoos and aquariums. J. Zoo Aquar. Res. 2013, 1, 1–6. [Google Scholar]
- Held, S.D.; Špinka, M. Animal play and animal welfare. Anim. Behav. 2011, 81, 891–899. [Google Scholar] [CrossRef]
- Hill, H.M.; Dietrich, S.; Yeater, D.; McKinnon, M.; Miller, M.; Aibel, S.; Dove, A. Developing a catalog of socio-sexual behaviors of beluga whales (Delphinapterus leucas). Anim. Behav. Cog. 2015, 2, 105–123. [Google Scholar] [CrossRef]
- Hill, H.; Guarino, S.; Crandall, S.; Lenhart, E.; Dietrich, S. Young belugas diversify adult beluga (Delphinapterus leucas) behavior. Anim. Behav. Cog. 2015, 2, 267–284. [Google Scholar] [CrossRef]
- Eskelinen, H.C.; Borger-Turner, J.L.; Ii, S.A.K. Observations of a Paternal Male with Bottlenose Dolphin Calf (Tursiops truncatus): A Case Study. Int. J. Comp. Psychol. 2017, 30, 30. [Google Scholar] [CrossRef]
- Samuels, A.; Gifford, T. A Quantitative Assessment of Dominance Relations among Bottlenose Dolphins. Mar. Mammal Sci. 1997, 13, 70–99. [Google Scholar] [CrossRef]
- Mann, J.; Connor, R.C.; Barre, L.M.; Heithaus, M.R. Female reproductive success in bottlenose dolphins (Tursiops sp.): Life history, habitat, provisioning, and group-size effects. Behav. Ecol. 2000, 11, 210–219. [Google Scholar] [CrossRef] [Green Version]
- Wells, R.S.; Scott, M.D. Bottlenose dolphin Tursiops truncatus (montagu, 1821). Handb. Mar. Mamm. 1999, 6, 137–182. [Google Scholar]
- Gubbins, C.; McOwan, B.; Lynn, S.K.; Hooper, S.; Reiss, D. Mother-Infant Spatial Relations in Captive Bottlenose Dolphins, Tursiops Truncatus. Mar. Mammal Sci. 1999, 15, 751–765. [Google Scholar] [CrossRef]
- Hill, H.M.; Greer, T.; Solangi, M.; Kuczaj, S.A. All mothers are not the same: Maternal styles in bottlenose dolphins (Tursiops truncatus). Int. J. Comp. Psychol. 2007, 20, 35–54. [Google Scholar]
- Mann, J.; Smuts, B.B. Natal attraction: Allomaternal care and mother–infant separations in wild bottlenose dolphins. Anim. Behav. 1998, 55, 1097–1113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reid, K.; Mann, J.; Weiner, J.R.; Hecker, N. Infant development in two aquarium bottlenose dolphins. Zoo Biol. 1995, 14, 135–147. [Google Scholar] [CrossRef]
- McBride, A.F.; Kritzler, H. Observations on Pregnancy, Parturition, and Postnatal Behavior in the Bottlenose Dolphin. J. Mammal. 1951, 32, 251–266. [Google Scholar] [CrossRef]
- Mann, J. Individual differences in bottlenose dolphin infants. Fam. Sys. 1997, 4, 34–48. [Google Scholar]
- Howells, E.M.; Reif, J.S.; Bechdel, S.E.; Murdoch, M.E.; Bossart, G.D.; McCulloch, S.D.; Mazzoil, M.S. A Novel Case of Non-Offspring Adoption in a Free-Ranging Atlantic Bottlenose Dolphin (Tursiops truncatus) Inhabiting the Indian River La-goon, Florida. Aqua. Mamm. 2009, 35, 43–47. [Google Scholar] [CrossRef]
- Kogi, K.; Hishii, T.; Imamura, A.; Iwatani, T.; Dudzinski, K.M. Demographic Parameters of Indo-Pacific Bottlenose Dolphins (Tursiops aduncus) Around Mikura Island, Japan. Mar. Mammal Sci. 2004, 20, 510–526. [Google Scholar] [CrossRef]
- Dudzinski, K. Communication and Behavior in the Atlantic Spotted Dolphins (Stenella frontalis): Relationships between Vocal and Behavioral Activities. Ph.D. Thesis, Texas A&M University, College Station, TX, USA, August 1996. Available online: https://www.researchgate.net/profile/Kathleen-Dudzinski/publication/35768063_Communication_and_behavior_in_the_Atlan-tic_spotted_dolphins_Stenella_frontalis_relationships_between_vocal_and_behavioral_activities/links/5421ca190cf26120b7a00a4d/Communication-and-behavior-in-the-Atlantic-spotted-dolphins-Stenella-frontalis-relationships-between-vocal-and-behavioral-activities.pdf (accessed on 24 March 2021).
- Harvey, B.N. The Nature of Social Relationships in Bottlenose Dolphins (Tursiops truncatus): Associations and the Role of Afiliative, Agonistic, and Socio-Sexual Behaviors. Master’s Thesis, University of Southern Mississippi, Hattiesburg, MS, USA, 2015. Available online: https://aquila.usm.edu/cgi/viewcontent.cgi?referer=https://scholar.google.com/&httpsredir=1&article=1131&context=masters_theses (accessed on 24 March 2021).
- Haidet, K.K.; Tate, J.; Divirgilio-Thomas, D.; Kolanowski, A.; Happ, M.B. Methods to improve reliability of video-recorded behavioral data. Res. Nurs. Health 2009, 32, 465–474. [Google Scholar] [CrossRef]
- Lauderdale, L.K.; Miller, L.J. Common bottlenose dolphin (Tursiops truncatus) problem solving strategies in response to a novel interactive apparatus. Behav. Proc. 2019, 169, 103990. [Google Scholar] [CrossRef] [PubMed]
- Lauderdale, L.K.; Miller, L.J. Efficacy of an interactive apparatus as environmental enrichment for common bottlenose dolphins (Tursiops truncatus). Anim. Welf 2020, 29, 379–386. [Google Scholar] [CrossRef]
- Cairns, S.J.; Schwager, S.J. A comparison of association indices. Anim. Behav. 1987, 35, 1454–1469. [Google Scholar] [CrossRef] [Green Version]
- Dugard, P.; File, P.; Todman, J.; Todman, J.B. Single-Case and Small-n Experimental Designs: A Practical Guide to Randomization Tests, 2nd ed.; Routledge: London, UK, 2012. [Google Scholar]
- Kratochwill, T.R. Single Subject Research: Strategies for Evaluating Change, 1st ed.; Academic Press: Cambridge, MA, USA, 2013. [Google Scholar]
- Ruiz, C.U.; Sánchez, A.; Maldonado, F.G. Social and individual behavior of a group of bottlenose dolphins (Tursiops truncatus) in open and closed facilities. Vet. Mex. 2009, 40, 381–387. [Google Scholar]
- Paulos, R.D.; Trone, M.; Kuczaj, S.A. Play in wild and captive cetaceans. Int. J. Comp. Psychol. 2010, 23, 701–722. [Google Scholar]
- Würsig, B.E.; Würsig, M.E. Behavior and ecology of the bottlenose dolphin, Tursiops truncatus, in the South Atlantic. Fish. Bull. 1979, 77, 399–412. [Google Scholar]
- Cockcroft, V.G.; Ross, G.J.; Peddemors, V.M. Bottlenose dolphin Tursiops truncatus distribution in Natal’s coastal waters. Sou. Afri. J. Mar. Sci. 1990, 9, 1–10. [Google Scholar] [CrossRef]
- Caldwell, D.K.; Caldwell, M.C. The World of the Bottlenose Dolphin, 1st ed.; Lippencott: Philadelphia, PA, USA, 1972. [Google Scholar]
- Clegg, I.; Van Elk, C.; Delfour, F. Applying welfare science to bottlenose dolphins (Tursiops truncatus). Anim. Welf. 2017, 26, 165–176. [Google Scholar] [CrossRef] [Green Version]
- Mann, J.; Smuts, B. Behavioral development in wild bottlenose dolphin newborns (Tursiops sp.). Behaviour 1999, 136, 529–566. [Google Scholar]
- Gibson, Q.A.; Mann, J. The size, composition and function of wild bottlenose dolphin (Tursiops sp.) mother–calf groups in Shark Bay, Australia. Anim. Behav. 2008, 76, 389–405. [Google Scholar] [CrossRef]
- Stanton, M.A.; Gibson, Q.A.; Mann, J. When mum’s away: A study of mother and calf ego networks during separations in wild bottlenose dolphins (Tursiops sp.). Anim. Behav. 2011, 82, 405–412. [Google Scholar] [CrossRef]
- Krzyszczyk, E.; Patterson, E.M.; Stanton, M.A.; Mann, J. The transition to independence: Sex differences in social and behavioural development of wild bottlenose dolphins. Anim. Behav. 2017, 129, 43–59. [Google Scholar] [CrossRef]
- Hamilton, W.D. The genetical evolution of social behavior. J. Theor. Biol. 1964, 7, 17–52. [Google Scholar] [CrossRef]
- Pope, T.R. Reproductive success increases with degree of kinship in cooperative coalitions of female red howler monkeys (Alouatta seniculus). Behav. Ecol. Socio. Biol. 2000, 48, 253–267. [Google Scholar] [CrossRef]
- Silk, J.B. Kin Selection in Primate Groups. Int. J. Primatol. 2002, 23, 849–875. [Google Scholar] [CrossRef]
- Duffield, D.; Wells, R.S. The combined application of chromosome, protein and molecular data for the investigation of social unit structure and dynamics in Tursiops truncatus. Rep. Int. Whal. Comm. 1991, 13, 155–169. [Google Scholar]
- Möller, L.M.; Beheregaray, L.B.; Allen, S.; Harcourt, R.G. Association patterns and kinship in female Indo-Pacific bottlenose dolphins (Tursiops aduncus) of southeastern Australia. Behav. Ecol. Sociobiol. 2006, 61, 109–117. [Google Scholar] [CrossRef]
- Gibson, Q.A.; Mann, J. Early social development in wild bottlenose dolphins: Sex differences, individual variation and maternal influence. Anim. Behav. 2008, 76, 375–387. [Google Scholar] [CrossRef]
- Eastcott, A.; Dickinson, T. Underwater observations of the suckling and social behavior of a newborn bottlenosed dolphin (Tusiops truncatus). Aqua. Mamm. 1987, 13, 51–56. [Google Scholar]
- Clegg, I.L.K.; Rödel, H.G.; Cellier, M.; Vink, D.; Michaud, I.; Mercera, B.; Böye, M.; Hausberger, M.; Lemasson, A.; Delfour, F. Schedule of human-controlled periods structures bottlenose dolphin (Tursiops truncatus) behavior in their free-time. J. Comp. Psychol. 2017, 131, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Serres, A.; Robeck, T.; Deng, X.; Steinman, K.; Hao, Y.; Wang, D. Social, Reproductive and Contextual Influences on Fecal Glucocorticoid Metabolites in Captive Yangtze Finless Porpoises (Neophocaena asiaeorientalis asiaeorientalis) and Bottlenose Dolphins (Tursiops truncatus). J. Zool. Bot. Gard. 2020, 1, 24–41. [Google Scholar] [CrossRef]
- Clegg, I.L.; Rödel, H.G.; Delfour, F. Bottlenose dolphins engaging in more social affiliative behaviour judge ambiguous cues more optimistically. Behav. Brain Res. 2017, 322, 115–122. [Google Scholar] [CrossRef] [PubMed]

| Dolphin | Age | Sex | Sub-Group |
|---|---|---|---|
| D1 | 2 | Male | A |
| D2 | 11 | Female | A |
| D3 | 13 | Female | A |
| D4 | 14 | Female | A |
| D5 | 1 | Female | B |
| D6 | 2 | Male | B |
| D7 | 30 | Female | B |
| D8 | 34 | Female | B |
| Dolphin | Solo Swim | Social Swim |
|---|---|---|
| D1 | 67.63 | 32.36 |
| D2 | 67.78 | 32.22 |
| D3 | 60.36 | 39.65 |
| D4 | 64.65 | 35.35 |
| D5 | 24.84 | 75.15 |
| D6 | 22.45 | 77.56 |
| D7 | 31.25 | 68.75 |
| D8 | 31.86 | 68.13 |
| Dolphin | Social Active | Social Agonistic | Social Sexual | Solitary Active |
|---|---|---|---|---|
| D1 | 1.05 | 0.02 | 0.07 | 0.49 |
| D2 | 0.47 | 0.01 | 0.01 | 0.26 |
| D3 | 0.30 | 0.01 | 0.01 | 0.25 |
| D4 | 0.47 | 0.01 | 0.01 | 0.25 |
| D5 | 0.57 | 0.01 | 0.03 | 0.33 |
| D6 | 1.14 | 0.02 | 0.06 | 0.27 |
| D7 | 0.25 | 0.01 | 0.01 | 0.16 |
| D8 | 0.45 | <0.01 | 0.03 | 0.30 |
| Predictor | Response | Beta | Std. Error | Lower 95% CI | Upper 95% CI | p Value |
|---|---|---|---|---|---|---|
| Solo Swim | (Intercept) | 0.324 | 0.072 | 0.182 | 0.465 | <0.001 |
| Early Morning | - | - | - | - | - | |
| Late Morning | 0.140 | 0.019 | 0.103 | 0.177 | <0.001 * | |
| Afternoon | 0.245 | 0.024 | 0.197 | 0.293 | <0.001 * | |
| Social Swim | (Intercept) | 0.676 | 0.072 | 0.535 | 0.818 | <0.001 |
| Early Morning | - | - | - | - | - | |
| Late Morning | −0.140 | 0.019 | −0.177 | −0.103 | <0.001 * | |
| Afternoon | −0.250 | 0.025 | −0.299 | −0.201 | <0.001 * | |
| Social | (Intercept) | 0.559 | 0.106 | 0.351 | 0.767 | <0.001 |
| Active | Early Morning | - | - | - | - | - |
| Late Morning | 0.019 | 0.026 | −0.032 | 0.070 | 0.470 | |
| Afternoon | −0.050 | 0.018 | −0.085 | −0.015 | 0.006 * | |
| Social | (Intercept) | 0.006 | 0.003 | 0.001 | 0.011 | 0.011 |
| Agonistic | Early Morning | - | - | - | - | - |
| Late Morning | 0.004 | 0.003 | −0.002 | 0.010 | 0.216 | |
| Afternoon | 0.006 | 0.003 | 0.001 | 0.011 | 0.011 * | |
| Social | (Intercept) | 0.043 | 0.014 | 0.015 | 0.070 | 0.003 |
| Sexual | Early Morning | - | - | - | - | - |
| Late Morning | −0.013 | 0.008 | −0.028 | 0.003 | 0.112 | |
| Afternoon | −0.028 | 0.010 | −0.048 | −0.007 | 0.007 * | |
| Solitary | (Intercept) | 0.155 | 0.018 | 0.120 | 0.190 | <0.001 |
| Active | Early Morning | - | - | - | - | - |
| Late Morning | 0.150 | 0.025 | 0.102 | 0.198 | <0.001 * | |
| Afternoon | 0.224 | 0.042 | 0.141 | 0.306 | <0.001 * |
| Dyad | SRI Phase 1 and 2 | SRI Phase 2 and 3 | Dyad | SRI Phase 1 and 2 | SRI Phase 2 and 3 |
|---|---|---|---|---|---|
| D1/D2 | −0.01 | −0.02 | D3/D5 | 0.03 | −0.03 |
| D1/D3 | 0.01 | 0.00 | D3/D6 | −0.04 | −0.01 |
| D1/D4 | 0.03 | 0.02 | D3/D7 | 0.03 | −0.02 |
| D1/D5 | 0.04 | −0.03 | D3/D8 | −0.06 | −0.03 |
| D1/D6 | −0.04 | −0.02 | D4/D5 | 0.02 | −0.02 |
| D1/D7 | 0.01 | −0.01 | D4/D6 | −0.01 | −0.04 |
| D1/D8 | 0.01 | 0.01 | D4/D7 | 0.02 | −0.03 |
| D2/D3 | 0.03 | −0.05 | D4/D8 | −0.14 | −0.09 |
| D2/D4 | 0.00 | −0.05 | D5/D6 | 0.05 | 0.01 |
| D2/D5 | 0.01 | −0.01 | D5/D7 | 0.00 | 0.08 |
| D2/D6 | −0.03 | 0.03 | D5/D8 | 0.04 | −0.04 |
| D2/D7 | 0.01 | −0.01 | D6/D7 | 0.04 | −0.03 |
| D2/D8 | 0.01 | 0.04 | D6/D8 | 0.20 | 0.00 |
| D3/D4 | 0.06 | −0.14 | D7/D8 | 0.06 | −0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lauderdale, L.K.; Miller, L.J. Activity, Social Relationships, and Maternal Care in a Bottlenose Dolphin Group under Professional Care. J. Zool. Bot. Gard. 2021, 2, 273-286. https://doi.org/10.3390/jzbg2020019
Lauderdale LK, Miller LJ. Activity, Social Relationships, and Maternal Care in a Bottlenose Dolphin Group under Professional Care. Journal of Zoological and Botanical Gardens. 2021; 2(2):273-286. https://doi.org/10.3390/jzbg2020019
Chicago/Turabian StyleLauderdale, Lisa K., and Lance J. Miller. 2021. "Activity, Social Relationships, and Maternal Care in a Bottlenose Dolphin Group under Professional Care" Journal of Zoological and Botanical Gardens 2, no. 2: 273-286. https://doi.org/10.3390/jzbg2020019
APA StyleLauderdale, L. K., & Miller, L. J. (2021). Activity, Social Relationships, and Maternal Care in a Bottlenose Dolphin Group under Professional Care. Journal of Zoological and Botanical Gardens, 2(2), 273-286. https://doi.org/10.3390/jzbg2020019







