Probiotics against Viral Infections: Current Clinical Trials and Future Perspectives
Abstract
1. Introduction
2. Probiotics
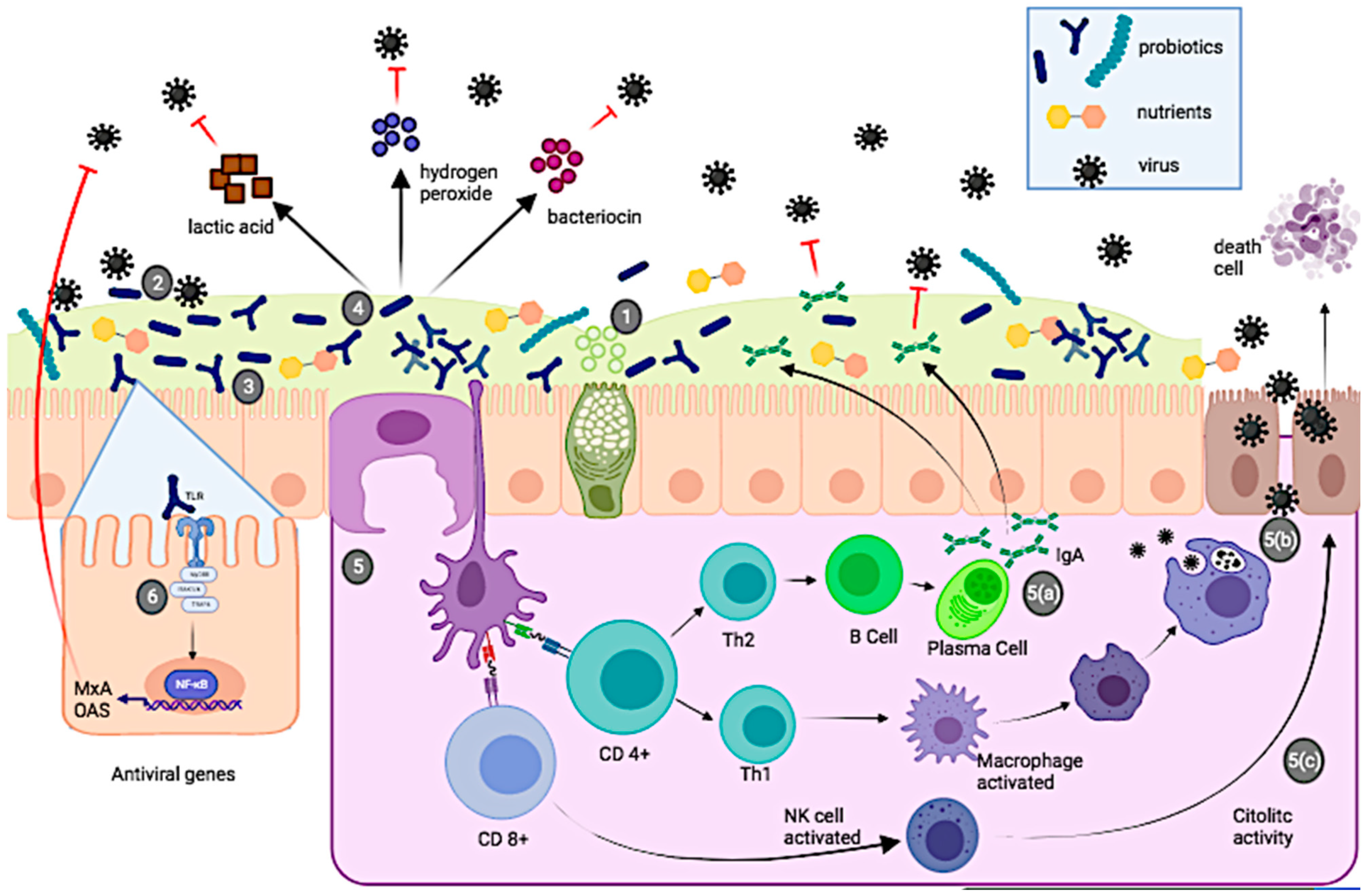
3. Probiotics and Antiviral Effects
4. Probiotics and Respiratory Tract Infections (RTIs)
5. Probiotics and Human Immunodeficiency Virus (HIV)
6. Probiotics and Gastrointestinal Infections
7. Probiotics and Human Papillomavirus (HPV)
8. Probiotics and Hepatic Encephalopathy (HE)
9. Probiotics and Herpes Simplex-2 (HSV-2) Infection
10. Discussion
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Parvez, M.K.; Parveen, S. Evolution and Emergence of Pathogenic Viruses: Past, Present, and Future. Intervirology 2017, 60, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Caminade, C.; McIntyre, K.M.; Jones, A.E. Impact of recent and future climate change on vector-borne diseases. Ann. N. Y. Acad. Sci. 2019, 1436, 157–173. [Google Scholar] [CrossRef] [PubMed]
- Monto, A.S. Vaccines and antiviral drugs in pandemic preparedness. Emerg. Infect. Dis. 2006, 12, 55–60. [Google Scholar] [CrossRef]
- Lopez-Santamarina, A.; Lamas, A.; Del Carmen Mondragon, A.; Cardelle-Cobas, A.; Regal, P.; Rodriguez-Avila, J.A.; Miranda, J.M.; Franco, C.M.; Cepeda, A. Probiotic Effects against Virus Infections: New Weapons for an Old War. Foods 2021, 10, 130. [Google Scholar] [CrossRef]
- Committee on Strategies for Responsible Sharing of Clinical Trial Data; Board on Health Sciences Policy; Institute of Medicine. The clinical trial life cycle and when to share data. In Sharing Clinical Trial Data: Maximizing Benefits; National Academies Press: Washington, DC, USA, 2015. [Google Scholar]
- WHO. Clinical Trial. Available online: https://www.who.int/health-topics/clinical-trials/-tab=tab_1 (accessed on 31 January 2021).
- Clinicaltrials.gov. Clinical Trials. Available online: https://clinicaltrials.gov/ct2/about-studies (accessed on 2 December 2020).
- Plaza-Diaz, J.; Ruiz-Ojeda, F.J.; Gil-Campos, M.; Gil, A. Mechanisms of Action of Probiotics. Adv. Nutr. 2019, 10, S49–S66. [Google Scholar] [CrossRef]
- Bron, P.A.; Kleerebezem, M.; Brummer, R.-J.; Cani, P.D.; Mercenier, A.; MacDonald, T.T.; Garcia-Ródenas, C.L.; Wells, J.M. Can probiotics modulate human disease by impacting intestinal barrier function? Br. J. Nutr. 2017, 117, 93–107. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Favaro-Trindade, C.S.H.; Heinemann, R.J.B.; Pedroso, D.L. Developments in probiotic encapsulation. CAB Rev. 2011, 6, 1–8. [Google Scholar] [CrossRef]
- Chapman, C.M.C.; Gibson, G.R.; Rowland, I. Health benefits of probiotics: Are mixtures more effective than single strains? Eur. J. Nutr. 2011, 50, 1–17. [Google Scholar] [CrossRef]
- Flesch, A.G.T.; Poziomyck, A.K.; Damin, D.C. The therapeutic use of symbiotics. Arq. Bras. De Cir. Dig. ABCD 2014, 27, 206–209. [Google Scholar] [CrossRef]
- Swanson, K.S.; Gibson, G.R.; Hutkins, R.; Reimer, R.A.; Reid, G.; Verbeke, K.; Scott, K.P.; Holscher, H.D.; Azad, M.B.; Delzenne, N.M.; et al. The International Scientific Association for Probiotics and Prebiotics (ISAPP) consensus statement on the definition and scope of synbiotics. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 687–701. [Google Scholar] [CrossRef] [PubMed]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef] [PubMed]
- Olaimat, A.N.; Aolymat, I.; Al-Holy, M.; Ayyash, M.; Abu Ghoush, M.; Al-Nabulsi, A.A.; Osaili, T.; Apostolopoulos, V.; Liu, S.-Q.; Shah, N.P. The potential application of probiotics and prebiotics for the prevention and treatment of COVID-19. NPJ Sci. Food 2020, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.-L.; Shu, C.-C.; Lai, W.-F.; Tzeng, C.-M.; Lai, H.-C.; Lu, C.-C. Investiture of next generation probiotics on amelioration of diseases–Strains do matter. Med. Microecol. 2019, 1–2, 100002. [Google Scholar] [CrossRef]
- WGO. Global Guidelines Probiotics and Prebiotics. Available online: http://www.spg.pt/wp-content/uploads/2015/07/2017-Probiotics-and-Prebiotics.pdf (accessed on 13 December 2020).
- Saad, N.; Delattre, C.; Urdaci, M.; Schmitter, J.M.; Bressollier, P. An overview of the last advances in probiotic and prebiotic field. LWT -Food Sci. Technol. 2013, 50, 1–16. [Google Scholar] [CrossRef]
- Bron, P.A.; van Baarlen, P.; Kleerebezem, M. Emerging molecular insights into the interaction between probiotics and the host intestinal mucosa. Nat. Rev. Microbiol. 2012, 10, 66–78. [Google Scholar] [CrossRef] [PubMed]
- Terpou, A.; Papadaki, A.; Lappa, I.K.; Kachrimanidou, V.; Bosnea, L.A.; Kopsahelis, N. Probiotics in Food Systems: Significance and Emerging Strategies Towards Improved Viability and Delivery of Enhanced Beneficial Value. Nutrients 2019, 11, 1591. [Google Scholar] [CrossRef]
- Hossain, M.I.; Sadekuzzaman, M.; Ha, S.-D. Probiotics as potential alternative biocontrol agents in the agriculture and food industries: A review. Food Res. Int. 2017, 100, 63–73. [Google Scholar] [CrossRef]
- Dronkers, T.M.G.; Ouwehand, A.C.; Rijkers, G.T. Global analysis of clinical trials with probiotics. Heliyon 2020, 6, e04467. [Google Scholar] [CrossRef]
- FAO; WHO. Guidelines for the Evaluation of Probiotics in Food. Available online: https://www.who.int/foodsafety/fs_management/en/probiotic_guidelines.pdf (accessed on 15 December 2020).
- Sanders, M.E.; Benson, A.; Lebeer, S.; Merenstein, D.J.; Klaenhammer, T.R. Shared mechanisms among probiotic taxa: Implications for general probiotic claims. Curr. Opin. Biotechnol. 2018, 49, 207–216. [Google Scholar] [CrossRef]
- Brüssow, H. Probiotics and prebiotics in clinical tests: An update [version 1; peer review: 2 approved]. F1000Research 2019, 8, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cuestas, M.L.; Minassian, M.L. Virus emergentes y reemergentes: Un nuevo reto para la salud mundial del milenio. Rev. Argent. De Microbiol. 2020, 52, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Roychoudhury, S.; Das, A.; Sengupta, P.; Dutta, S.; Roychoudhury, S.; Choudhury, A.P.; Ahmed, A.B.F.; Bhattacharjee, S.; Slama, P. Viral Pandemics of the Last Four Decades: Pathophysiology, Health Impacts and Perspectives. Int. J. Environ. Res. Public Health 2020, 17, 9411. [Google Scholar] [CrossRef]
- Ginglen, J.G.; Doyle, M.Q. Immunization. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2021. [Google Scholar]
- WHO. Vaccines and Immunization. Available online: https://www.who.int/health-topics/vaccines-and-immunization-tab=tab_1 (accessed on 30 January 2021).
- Kanauchi, O.; Andoh, A.; AbuBakar, S.; Yamamoto, N. Probiotics and Paraprobiotics in Viral Infection: Clinical Application and Effects on the Innate and Acquired Immune Systems. Curr. Pharm. Des. 2018, 24, 710–717. [Google Scholar] [CrossRef]
- Al Kassaa, I. Antiviral Probiotics: A New Concept in Medical Sciences. In New Insights on Antiviral Probiotics: From Research to Applications; Springer International Publishing: Berlin/Heidelberg, Germany, 2017; pp. 1–46. [Google Scholar]
- Tiwari, S.K.; Dicks, L.M.T.; Popov, I.V.; Karaseva, A.; Ermakov, A.M.; Suvorov, A.; Tagg, J.R.; Weeks, R.; Chikindas, M.L. Probiotics at War Against Viruses: What Is Missing From the Picture? Front. Microbiol. 2020, 11, 1877. [Google Scholar] [CrossRef] [PubMed]
- Hardy, H.; Harris, J.; Lyon, E.; Beal, J.; Foey, A.D. Probiotics, Prebiotics and Immunomodulation of Gut Mucosal Defences: Homeostasis and Immunopathology. Nutrients 2013, 5, 1869–1912. [Google Scholar] [CrossRef] [PubMed]
- Shahbazi, R.; Yasavoli-Sharahi, H.; Alsadi, N.; Ismail, N.; Matar, C. Probiotics in Treatment of Viral Respiratory Infections and Neuroinflammatory Disorders. Molecules 2020, 25, 4891. [Google Scholar] [CrossRef]
- Llewellyn, A.; Foey, A. Probiotic Modulation of Innate Cell Pathogen Sensing and Signaling Events. Nutrients 2017, 9, 1156. [Google Scholar] [CrossRef]
- Nakayama, Y.; Moriya, T.; Sakai, F.; Ikeda, N.; Shiozaki, T.; Hosoya, T.; Nakagawa, H.; Miyazaki, T. Oral administration of Lactobacillus gasseri SBT2055 is effective for preventing influenza in mice. Sci. Rep. 2014, 4, 4638. [Google Scholar] [CrossRef]
- Schneider, W.M.; Chevillotte, M.D.; Rice, C.M. Interferon-stimulated genes: A complex web of host defenses. Annu. Rev. Immunol. 2014, 32, 513–545. [Google Scholar] [CrossRef]
- Lehtoranta, L.; Pitkäranta, A.; Korpela, R. Probiotics in respiratory virus infections. Eur. J. Clin. Microbiol. Infect. Dis. 2014, 33, 1289–1302. [Google Scholar] [CrossRef]
- Mahooti, M.; Miri, S.M.; Abdolalipour, E.; Ghaemi, A. The immunomodulatory effects of probiotics on respiratory viral infections: A hint for COVID-19 treatment? Microb. Pathog. 2020, 148, 104452. [Google Scholar] [CrossRef]
- Lee, Y.N.; Youn, H.N.; Kwon, J.H.; Lee, D.H.; Park, J.K.; Yuk, S.S.; Erdene-Ochir, T.O.; Kim, K.T.; Lee, J.B.; Park, S.Y.; et al. Sublingual administration of Lactobacillus rhamnosus affects respiratory immune responses and facilitates protection against influenza virus infection in mice. Antivir. Res. 2013, 98, 284–290. [Google Scholar] [CrossRef]
- Ashraf, R.; Shah, N.P. Immune System Stimulation by Probiotic Microorganisms. Crit. Rev. Food Sci. Nutr. 2014, 54, 938–956. [Google Scholar] [CrossRef]
- Maldonado Galdeano, C.; Cazorla, S.I.; Lemme Dumit, J.M.; Vélez, E.; Perdigón, G. Beneficial Effects of Probiotic Consumption on the Immune System. Ann. Nutr. Metab. 2019, 74, 115–124. [Google Scholar] [CrossRef]
- WHO. WHO Reveals Leading Causes of Death and Disability Worldwide: 2000–2019. Available online: https://www.who.int/news/item/09-12-2020-who-reveals-leading-causes-of-death-and-disability-worldwide-2000-2019 (accessed on 23 January 2021).
- Soriano, J.B.; Kendrick, P.J.; Paulson, K.R.; Gupta, V.; Abrams, E.M.; Adedoyin, R.A.; Adhikari, T.B.; Advani, S.M.; Agrawal, A.; Ahmadian, E.; et al. Prevalence and attributable health burden of chronic respiratory diseases, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Respir. Med. 2020, 8, 585–596. [Google Scholar] [CrossRef]
- van Doorn, H.R.; Yu, H. 33—Viral Respiratory Infections. In Hunter’s Tropical Medicine and Emerging Infectious Diseases, 10th ed.; Ryan, E.T., Hill, D.R., Solomon, T., Aronson, N.E., Endy, T.P., Eds.; Elsevier: London, UK, 2020; pp. 284–288. [Google Scholar]
- Perk, Y.; Özdil, M. Respiratory syncytial virüs infections in neonates and infants. Turk. Pediatri Ars. 2018, 53, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Pfeiffer, J.K.; Virgin, H.W. Transkingdom control of viral infection and immunity in the mammalian intestine. Science 2016, 351, aad5872. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.; Zhao, S.; Yu, B.; Chen, Y.-M.; Wang, W.; Song, Z.-G.; Hu, Y.; Tao, Z.-W.; Tian, J.-H.; Pei, Y.-Y.; et al. A new coronavirus associated with human respiratory disease in China. Nature 2020, 579, 265–269. [Google Scholar] [CrossRef]
- Lee, T.; Ahn, J.-H.; Park, S.Y.; Kim, G.-H.; Kim, J.; Kim, T.-H.; Nam, I.; Park, C.; Lee, M.-H. Recent Advances in AIV Biosensors Composed of Nanobio Hybrid Material. Micromachines 2018, 9, 651. [Google Scholar] [CrossRef]
- Sundararaman, A.; Ray, M.; Ravindra, P.V.; Halami, P.M. Role of probiotics to combat viral infections with emphasis on COVID-19. Appl. Microbiol. Biotechnol. 2020, 104, 8089–8104. [Google Scholar] [CrossRef]
- Enani, S.; Przemska-Kosicka, A.; Childs, C.E.; Maidens, C.; Dong, H.; Conterno, L.; Tuohy, K.; Todd, S.; Gosney, M.; Yaqoob, P. Impact of ageing and a synbiotic on the immune response to seasonal influenza vaccination; A randomised controlled trial. Clin. Nutr. 2018, 37, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Waki, N.; Matsumoto, M.; Fukui, Y.; Suganuma, H. Effects of probiotic Lactobacillus brevis KB290 on incidence of influenza infection among schoolchildren: An open-label pilot study. Lett. Appl. Microbiol. 2014, 59, 565–571. [Google Scholar] [CrossRef] [PubMed]
- Davidson, L.E.; Fiorino, A.M.; Snydman, D.R.; Hibberd, P.L. Lactobacillus GG as an immune adjuvant for live-attenuated influenza vaccine in healthy adults: A randomized double-blind placebo-controlled trial. Eur. J. Clin. Nutr. 2011, 65, 501–507. [Google Scholar] [CrossRef]
- Sugimura, T.; Takahashi, H.; Jounai, K.; Ohshio, K.; Kanayama, M.; Tazumi, K.; Tanihata, Y.; Miura, Y.; Fujiwara, D.; Yamamoto, N. Effects of oral intake of plasmacytoid dendritic cells-stimulative lactic acid bacterial strain on pathogenesis of influenza-like illness and immunological response to influenza virus. Br. J. Nutr. 2015, 114, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Jespersen, L.; Tarnow, I.; Eskesen, D.; Morberg, C.M.; Michelsen, B.; Bügel, S.; Dragsted, L.O.; Rijkers, G.T.; Calder, P.C. Effect of Lactobacillus paracasei subsp. paracasei, L. casei 431 on immune response to influenza vaccination and upper respiratory tract infections in healthy adult volunteers: A randomized, double-blind, placebo-controlled, parallel-group study. Am. J. Clin. Nutr. 2015, 101, 1188–1196. [Google Scholar] [CrossRef]
- Wang, B.; Hylwka, T.; Smieja, M.; Surrette, M.; Bowdish, D.M.E.; Loeb, M. Probiotics to Prevent Respiratory Infections in Nursing Homes: A Pilot Randomized Controlled Trial. J. Am. Geriatr. Soc. 2018, 66, 1346–1352. [Google Scholar] [CrossRef]
- Fonollá, J.; Gracián, C.; Maldonado-Lobón, J.A.; Romero, C.; Bédmar, A.; Carrillo, J.C.; Martín-Castro, C.; Cabrera, A.L.; García-Curiel, J.M.; Rodríguez, C.; et al. Effects of Lactobacillus coryniformis K8 CECT5711 on the immune response to influenza vaccination and the assessment of common respiratory symptoms in elderly subjects: A randomized controlled trial. Eur. J. Nutr. 2019, 58, 83–90. [Google Scholar] [CrossRef]
- Yamamoto, Y.; Saruta, J.; Takahashi, T.; To, M.; Shimizu, T.; Hayashi, T.; Morozumi, T.; Kubota, N.; Kamata, Y.; Makino, S.; et al. Effect of ingesting yogurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1 on influenza virus-bound salivary IgA in elderly residents of nursing homes: A randomized controlled trial. Acta Odontol. Scand. 2019, 77, 517–524. [Google Scholar] [CrossRef]
- Luoto, R.; Ruuskanen, O.; Waris, M.; Kalliomäki, M.; Salminen, S.; Isolauri, E. Prebiotic and probiotic supplementation prevents rhinovirus infections in preterm infants: A randomized, placebo-controlled trial. J. Allergy Clin. Immunol. 2014, 133, 405–413. [Google Scholar] [CrossRef]
- Clinicaltrials.gov. COVID 19 and Procbiotics in Clinical Trials. Available online: https://clinicaltrials.gov/ct2/results?recrs=&cond=Covid19&term=Probiotics&cntry=&state=&city=&dist= (accessed on 20 June 2021).
- Kumpu, M.; Lehtoranta, L.; Roivainen, M.; Rönkkö, E.; Ziegler, T.; Söderlund-Venermo, M.; Kautiainen, H.; Järvenpää, S.; Kekkonen, R.; Hatakka, K.; et al. The use of the probiotic Lactobacillus rhamnosus GG and viral findings in the nasopharynx of children attending day care. J. Med. Virol. 2013, 85, 1632–1638. [Google Scholar] [CrossRef] [PubMed]
- Lehtoranta, L.; Kalima, K.; He, L.; Lappalainen, M.; Roivainen, M.; Närkiö, M.; Mäkelä, M.; Siitonen, S.; Korpela, R.; Pitkäranta, A. Specific probiotics and virological findings in symptomatic conscripts attending military service in Finland. J. Clin. Virol. 2014, 60, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Tapiovaara, L.; Lehtoranta, L.; Swanljung, E.; Mäkivuokko, H.; Laakso, S.; Roivainen, M.; Korpela, R.; Pitkäranta, A. Lactobacillus rhamnosus GG in the middle ear after randomized, double-blind, placebo-controlled oral administration. Int. J. Pediatric Otorhinolaryngol. 2014, 78, 1637–1641. [Google Scholar] [CrossRef] [PubMed]
- Tapiovaara, L.; Kumpu, M.; Mäkivuokko, H.; Waris, M.; Korpela, R.; Pitkäranta, A.; Winther, B. Human rhinovirus in experimental infection after peroral Lactobacillus rhamnosus GG consumption, a pilot study. Int. Forum Allergy Rhinol. 2016, 6, 848–853. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.B.; Woodfolk, J.A.; Borish, L.; Steinke, J.W.; Patrie, J.T.; Muehling, L.M.; Lahtinen, S.; Lehtinen, M.J. Effect of probiotic on innate inflammatory response and viral shedding in experimental rhinovirus infection—A randomised controlled trial. Benef. Microbes 2017, 8, 207–215. [Google Scholar] [CrossRef]
- Kinoshita, T.; Maruyama, K.; Suyama, K.; Nishijima, M.; Akamatsu, K.; Jogamoto, A.; Katakami, K.; Saito, I. The effects of OLL1073R-1 yogurt intake on influenza incidence and immunological markers among women healthcare workers: A randomized controlled trial. Food Funct. 2019, 10, 8129–8136. [Google Scholar] [CrossRef]
- WHO. VIH/Sida. Available online: https://www.who.int/es/news-room/fact-sheets/detail/hiv-aids (accessed on 30 January 2021).
- Reikvam, D.H.; Meyer-Myklestad, M.H.; Trøseid, M.; Stiksrud, B. Probiotics to manage inflammation in HIV infection. Curr. Opin. Infect. Dis. 2020, 33, 34–43. [Google Scholar] [CrossRef]
- Yang, O.O.; Kelesidis, T.; Cordova, R.; Khanlou, H. Immunomodulation of Antiretroviral Drug-Suppressed Chronic HIV-1 Infection in an Oral Probiotic Double-Blind Placebo-Controlled Trial. AIDS Res. Hum. Retrovir. 2014, 30, 988–995. [Google Scholar] [CrossRef]
- Vujkovic-Cvijin, I.; Somsouk, M. HIV and the Gut Microbiota: Composition, Consequences, and Avenues for Amelioration. Curr. HIV/AIDS Rep. 2019, 16, 204–213. [Google Scholar] [CrossRef]
- Lu, W.; Feng, Y.; Jing, F.; Han, Y.; Lyu, N.; Liu, F.; Li, J.; Song, X.; Xie, J.; Qiu, Z.; et al. Association Between Gut Microbiota and CD4 Recovery in HIV-1 Infected Patients. Front. Microbiol. 2018, 9, 1451. [Google Scholar] [CrossRef]
- Dillon, S.M.; Lee, E.J.; Kotter, C.V.; Austin, G.L.; Dong, Z.; Hecht, D.K.; Gianella, S.; Siewe, B.; Smith, D.M.; Landay, A.L.; et al. An altered intestinal mucosal microbiome in HIV-1 infection is associated with mucosal and systemic immune activation and endotoxemia. Mucosal Immunol. 2014, 7, 983–994. [Google Scholar] [CrossRef] [PubMed]
- Gori, A.; Tincati, C.; Rizzardini, G.; Torti, C.; Quirino, T.; Haarman, M.; Amor, K.B.; van Schaik, J.; Vriesema, A.; Knol, J.; et al. Early Impairment of Gut Function and Gut Flora Supporting a Role for Alteration of Gastrointestinal Mucosa in Human Immunodeficiency Virus Pathogenesis. J. Clin. Microbiol. 2008, 46, 757–758. [Google Scholar] [CrossRef] [PubMed]
- Schunter, M.; Chu, H.; Hayes, T.L.; McConnell, D.; Crawford, S.S.; Luciw, P.A.; Bengmark, S.; Asmuth, D.M.; Brown, J.; Bevins, C.L.; et al. Randomized pilot trial of a synbiotic dietary supplement in chronic HIV-1 infection. BMC Complement. Altern. Med. 2012, 12, 84. [Google Scholar] [CrossRef]
- D’Angelo, C.; Reale, M.; Costantini, E. Microbiota and Probiotics in Health and HIV Infection. Nutrients 2017, 9, 615. [Google Scholar] [CrossRef]
- Rajasuriar, R.; Wright, E.; Lewin, S.R. Impact of antiretroviral therapy (ART) timing on chronic immune activation/inflammation and end-organ damage. Curr. Opin. HIV AIDS 2015, 10, 35. [Google Scholar] [CrossRef] [PubMed]
- Ishizaki, A.; Bi, X.; Nguyen, L.V.; Matsuda, K.; Pham, H.V.; Phan, C.T.T.; Khu, D.T.K.; Ichimura, H. Effects of Short-Term Probiotic Ingestion on Immune Profiles and Microbial Translocation among HIV-1-Infected Vietnamese Children. Int. J. Mol. Sci. 2017, 18, 2185. [Google Scholar] [CrossRef] [PubMed]
- Villar-García, J.; Hernández, J.J.; Güerri-Fernández, R.; González, A.; Lerma, E.; Guelar, A.; Saenz, D.; Sorlí, L.; Montero, M.; Horcajada, J.P.; et al. Effect of Probiotics (Saccharomyces boulardii) on Microbial Translocation and Inflammation in HIV-Treated Patients: A Double-Blind, Randomized, Placebo-Controlled Trial. JAIDS J. Acquir. Immune Defic. Syndr. 2015, 68, 256–263. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Villar, S.; de Lagarde, M.; Vázquez-Castellanos, J.; Vallejo, A.; Bernadino, J.I.; Madrid, N.; Matarranz, M.; Díaz-Santiago, A.; Gutiérrez, C.; Cabello, A.; et al. Effects of Immunonutrition in Advanced Human Immunodeficiency Virus Disease: A Randomized Placebo-controlled Clinical Trial (Promaltia Study). Clin. Infect. Dis. 2018, 68, 120–130. [Google Scholar] [CrossRef] [PubMed]
- d’Ettorre, G.; Rossi, G.; Scagnolari, C.; Andreotti, M.; Giustini, N.; Serafino, S.; Schietroma, I.; Scheri, G.C.; Fard, S.N.; Trinchieri, V.; et al. Probiotic supplementation promotes a reduction in T-cell activation, an increase in Th17 frequencies, and a recovery of intestinal epithelium integrity and mitochondrial morphology in ART-treated HIV-1-positive patients. Immun. Inflamm. Dis. 2017, 5, 244–260. [Google Scholar] [CrossRef]
- Ceccarelli, G.; Fratino, M.; Selvaggi, C.; Giustini, N.; Serafino, S.; Schietroma, I.; Corano Scheri, G.; Pavone, P.; Passavanti, G.; Alunni Fegatelli, D.; et al. A pilot study on the effects of probiotic supplementation on neuropsychological performance and microRNA-29a-c levels in antiretroviral-treated HIV-1-infected patients. Brain Behav. 2017, 7, e00756. [Google Scholar] [CrossRef]
- d’Ettorre, G.; Ceccarelli, G.; Giustini, N.; Serafino, S.; Calantone, N.; De Girolamo, G.; Bianchi, L.; Bellelli, V.; Ascoli-Bartoli, T.; Marcellini, S.; et al. Probiotics Reduce Inflammation in Antiretroviral Treated, HIV-Infected Individuals: Results of the “Probio-HIV” Clinical Trial. PLoS ONE 2015, 10, e0137200. [Google Scholar] [CrossRef]
- Hemsworth, J.C.; Hekmat, S.; Reid, G. Micronutrient supplemented probiotic yogurt for HIV-infected adults taking HAART in London, Canada. Gut Microbes 2012, 3, 414–419. [Google Scholar] [CrossRef] [PubMed]
- Gautam, N.; Dayal, R.; Agarwal, D.; Kumar, R.; Singh, T.P.; Hussain, T.; Singh, S.P. Role of Multivitamins, Micronutrients and Probiotics Supplementation in Management of HIV Infected Children. Indian J. Pediatrics 2014, 81, 1315–1320. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.J.; Walmsley, S.L.; Raboud, J.M.; Kovacs, C.; Coburn, B.; Rousseau, R.; Reinhard, R.; Rosenes, R.; Kaul, R. Can Probiotics Reduce Inflammation and Enhance Gut Immune Health in People Living with HIV: Study Designs for the Probiotic Visbiome for Inflammation and Translocation (PROOV IT) Pilot Trials. HIV Clin. Trials 2016, 17, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Shin, D.Y.; Yi, D.Y.; Jo, S.; Lee, Y.M.; Kim, J.-H.; Kim, W.; Park, M.r.; Yoon, S.m.; Kim, Y.; Yang, S.; et al. Effect of a new Lactobacillus plantarum product, LRCC5310, on clinical symptoms and virus reduction in children with rotaviral enteritis. Medicine 2020, 99, e22192. [Google Scholar] [CrossRef]
- Graves, N.S. Acute Gastroenteritis. Prim. Care: Clin. Off. Pract. 2013, 40, 727–741. [Google Scholar] [CrossRef]
- Stuempfig, N.D.; Seroy, J. Viral Gastroenteritis. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2021. [Google Scholar]
- Crawford, S.E.; Ramani, S.; Tate, J.E.; Parashar, U.D.; Svensson, L.; Hagbom, M.; Franco, M.A.; Greenberg, H.B.; O’Ryan, M.; Kang, G.; et al. Rotavirus infection. Nat. Rev. Dis. Primers 2017, 3, 17083. [Google Scholar] [CrossRef]
- Nagata, S.; Asahara, T.; Ohta, T.; Yamada, T.; Kondo, S.; Bian, L.; Wang, C.; Yamashiro, Y.; Nomoto, K. Effect of the continuous intake of probiotic-fermented milk containing Lactobacillus casei strain Shirota on fever in a mass outbreak of norovirus gastroenteritis and the faecal microflora in a health service facility for the aged. Br. J. Nutr. 2011, 106, 549–556. [Google Scholar] [CrossRef]
- Robilotti, E.; Deresinski, S.; Pinsky Benjamin, A. Norovirus. Clin. Microbiol. Rev. 2015, 28, 134–164. [Google Scholar] [CrossRef]
- Pieścik-Lech, M.; Shamir, R.; Guarino, A.; Szajewska, H. Review article: The management of acute gastroenteritis in children. Aliment. Pharmacol. Ther. 2013, 37, 289–303. [Google Scholar] [CrossRef]
- Machado, K. Uso de probióticos en el tratamiento y la prevención de diarrea aguda en niños. Arch. De Pediatría Del Urug. 2020, 91, 35–45. [Google Scholar]
- Sniffen, J.C.; McFarland, L.V.; Evans, C.T.; Goldstein, E.J.C. Choosing an appropriate probiotic product for your patient: An evidence-based practical guide. PLoS ONE 2018, 13, e0209205. [Google Scholar] [CrossRef]
- Grandy, G.; Medina, M.; Soria, R.; Terán, C.G.; Araya, M. Probiotics in the treatment of acute rotavirus diarrhoea. A randomized, double-blind, controlled trial using two different probiotic preparations in Bolivian children. BMC Infect. Dis. 2010, 10, 253. [Google Scholar] [CrossRef]
- Sindhu, K.N.C.; Sowmyanarayanan, T.V.; Paul, A.; Babji, S.; Ajjampur, S.S.R.; Priyadarshini, S.; Sarkar, R.; Balasubramanian, K.A.; Wanke, C.A.; Ward, H.D.; et al. Immune Response and Intestinal Permeability in Children With Acute Gastroenteritis Treated With Lactobacillus rhamnosus GG: A Randomized, Double-Blind, Placebo-Controlled Trial. Clin. Infect. Dis. 2014, 58, 1107–1115. [Google Scholar] [CrossRef]
- Lee, D.K.; Park, J.E.; Kim, M.J.; Seo, J.G.; Lee, J.H.; Ha, N.J. Probiotic bacteria, B. longum and L. acidophilus inhibit infection by rotavirus in vitro and decrease the duration of diarrhea in pediatric patients. Clin. Res. Hepatol. Gastroenterol. 2015, 39, 237–244. [Google Scholar] [CrossRef]
- Huang, Y.-F.; Liu, P.-Y.; Chen, Y.-Y.; Nong, B.-R.; Huang, I.F.; Hsieh, K.-S.; Chen, K.-T. Three-Combination Probiotics Therapy in Children With Salmonella and Rotavirus Gastroenteritis. J. Clin. Gastroenterol. 2014, 48, 37–42. [Google Scholar] [CrossRef]
- Freedman, S.B.; Xie, J.; Nettel-Aguirre, A.; Pang, X.-L.; Chui, L.; Williamson-Urquhart, S.; Schnadower, D.; Schuh, S.; Sherman, P.M.; Lee, B.E.; et al. A randomized trial evaluating virus-specific effects of a combination probiotic in children with acute gastroenteritis. Nat. Commun. 2020, 11, 2533. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Gupta, P.K.; Das, R.R. Efficacy and Safety of Saccharomyces boulardii in Acute Rotavirus Diarrhea: Double Blind Randomized Controlled Trial from a Developing Country. J. Trop. Pediatrics 2016, 62, 464–470. [Google Scholar] [CrossRef][Green Version]
- Martins, A.E.S.; Lucena-Silva, N.; Garcia, R.G.; Welkovic, S.; Barboza, A.; Menezes, M.L.B.; Maruza, M.; Tenório, T.; Ximenes, R.A.A. Prevalence of human papillomavirus infection, distribution of viral types and risk factors in cervical samples from human immunodeficiency virus-positive women attending three human immunodeficiency virus-acquired immune deficiency syndrome reference centres in northeastern Brazil. Mem. Do Inst. Oswaldo Cruz 2014, 109, 738–747. [Google Scholar] [CrossRef][Green Version]
- WHO. Human Papillomavirus (HPV) and Cervical Cancer. Available online: https://www.who.int/es/news-room/fact-sheets/detail/human-papillomavirus-(hpv)-and-cervical-cancer (accessed on 10 January 2021).
- D’Alessandro, P.; Arduino, B.; Borgo, M.; Saccone, G.; Venturella, R.; Di Cello, A.; Zullo, F. Loop Electrosurgical Excision Procedure versus Cryotherapy in the Treatment of Cervical Intraepithelial Neoplasia: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Gynecol Minim Invasive Ther. 2018, 7, 145–151. [Google Scholar] [CrossRef]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Human Papillomaviruses. Available online: https://www.ncbi.nlm.nih.gov/books/NBK321760/ (accessed on 10 February 2021).
- Palma, E.; Recine, N.; Domenici, L.; Giorgini, M.; Pierangeli, A.; Panici, P.B. Long-term Lactobacillus rhamnosus BMX 54 application to restore a balanced vaginal ecosystem: A promising solution against HPV-infection. BMC Infect. Dis. 2018, 18, 13. [Google Scholar] [CrossRef] [PubMed]
- Ou, Y.-C.; Fu, H.-C.; Tseng, C.-W.; Wu, C.-H.; Tsai, C.-C.; Lin, H. The influence of probiotics on genital high-risk human papilloma virus clearance and quality of cervical smear: A randomized placebo-controlled trial. BMC Women’s Health 2019, 19, 103. [Google Scholar] [CrossRef] [PubMed]
- Verhoeven, V.; Renard, N.; Makar, A.; Royen, P.V.; Bogers, J.-P.; Lardon, F.; Peeters, M.; Baay, M. Probiotics enhance the clearance of human papillomavirus-related cervical lesions: A prospective controlled pilot study. Eur. J. Cancer Prev. 2013, 22, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Recine, N.; Palma, E.; Domenici, L.; Giorgini, M.; Imperiale, L.; Sassu, C.; Musella, A.; Marchetti, C.; Muzii, L.; Benedetti Panici, P. Restoring vaginal microbiota: Biological control of bacterial vaginosis. A prospective case–control study using Lactobacillus rhamnosus BMX 54 as adjuvant treatment against bacterial vaginosis. Arch. Gynecol. Obstet. 2016, 293, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Ferenci, P. Hepatic encephalopathy. Gastroenterol. Rep. 2017, 5, 138–147. [Google Scholar] [CrossRef]
- Xia, X.; Chen, J.; Xia, J.; Wang, B.; Liu, H.; Yang, L.; Wang, Y.; Ling, Z. Role of probiotics in the treatment of minimal hepatic encephalopathy in patients with HBV-induced liver cirrhosis. J. Int. Med. Res. 2018, 46, 3596–3604. [Google Scholar] [CrossRef]
- Bleibel, W.; Al-Osaimi, A. Hepatic encephalopathy. Saudi J. Gastroenterol. 2012, 18, 301–309. [Google Scholar] [CrossRef]
- Kang, Y.; Cai, Y. Gut microbiota and hepatitis-B-virus-induced chronic liver disease: Implications for faecal microbiota transplantation therapy. J. Hosp. Infect. 2017, 96, 342–348. [Google Scholar] [CrossRef]
- Mancini, A.; Campagna, F.; Amodio, P.; Tuohy, K.M. Gut:Liver:Brain axis: The microbial challenge in the hepatic encephalopathy. Food Funct. 2018, 9, 1373–1388. [Google Scholar] [CrossRef] [PubMed]
- Patidar, K.R.; Bajaj, J.S. Antibiotics for the treatment of hepatic encephalopathy. Metab. Brain Dis. 2013, 28, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Malaguarnera, M.; Gargante, M.P.; Malaguarnera, G.; Salmeri, M.; Mastrojeni, S.; Rampello, L.; Pennisi, G.; Volti, G.L.; Galvano, F. Bifidobacterium combined with fructo-oligosaccharide versus lactulose in the treatment of patients with hepatic encephalopathy. Eur. J. Gastroenterol. Hepatol. 2010, 22, 199–206. [Google Scholar] [CrossRef]
- Lee, D.K.; Kang, J.Y.; Shin, H.S.; Park, I.H.; Ha, N.J. Antiviral activity of Bifidobacterium adolescentis SPM0212 against Hepatitis B virus. Arch. Pharmacal Res. 2013, 36, 1525–1532. [Google Scholar] [CrossRef]
- Parra-Sánchez, M. Úlceras genitales por virus herpes simplex. Enferm. Infecc. Y Microbiol. Clínica 2019, 37, 260–264. [Google Scholar] [CrossRef] [PubMed]
- WHO. Herpes simplex virus. Available online: https://www.who.int/news-room/fact-sheets/detail/herpes-simplex-virus (accessed on 20 January 2021).
- Bodsworth, N.; Fife, K.; Koltun, W.; Tyring, S.; Abudalu, M.; Prichard, M.; Hamed, K. Single-day famciclovir for the treatment of genital herpes: Follow-up results of time to next recurrence and assessment of antiviral resistance. Curr. Med. Res. Opin. 2009, 25, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Mousavi, E.; Makvandi, M.; Teimoori, A.; Ataei, A.; Ghafari, S.; Samarbaf-Zadeh, A. Antiviral effects of Lactobacillus crispatus against HSV-2 in mammalian cell lines. J. Chin. Med. Assoc. 2018, 81, 262–267. [Google Scholar] [CrossRef]
- Mohseni, A.H.; Taghinezhad-S, S.; Keyvani, H.; Ghobadi, N. Comparison of Acyclovir and Multistrain Lactobacillus brevis in Women with Recurrent Genital Herpes Infections: A Double-Blind, Randomized, Controlled Study. Probiotics Antimicrob. Proteins 2018, 10, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Harper, A.; Vijayakumar, V.; Ouwehand, A.C.; Ter Haar, J.; Obis, D.; Espadaler, J.; Binda, S.; Desiraju, S.; Day, R. Viral Infections, the Microbiome, and Probiotics. Front. Cell. Infect. Microbiol. 2021, 10, 925. [Google Scholar] [CrossRef]
- Kurian, S.J.; Unnikrishnan, M.K.; Miraj, S.S.; Bagchi, D.; Banerjee, M.; Reddy, B.S.; Rodrigues, G.S.; Manu, M.K.; Saravu, K.; Mukhopadhyay, C.; et al. Probiotics in Prevention and Treatment of COVID-19: Current Perspective and Future Prospects. Arch. Med. Res. 2021, 52, 582–594. [Google Scholar] [CrossRef]
- Santacroce, L.; Inchingolo, F.; Topi, S.; Del Prete, R.; Di Cosola, M.; Charitos, I.A.; Montagnani, M. Potential beneficial role of probiotics on the outcome of COVID-19 patients: An evolving perspective. Diabetes Metab. Syndr. Clin. Res. Rev. 2021, 15, 295–301. [Google Scholar] [CrossRef]
- Hao, Q.; Dong, B.R.; Wu, T. Probiotics for preventing acute upper respiratory tract infections. Cochrane Database Syst. Rev. 2015, 2, Cd006895. [Google Scholar] [CrossRef]
- Xu, K.; Cai, H.; Shen, Y.; Ni, Q.; Chen, Y.; Hu, S.; Li, J.; Wang, H.; Yu, L.; Huang, H.; et al. Management of corona virus disease-19 (COVID-19): The Zhejiang experience. Zhejiang Da Xue Xue Bao Yi Xue Ban 2020, 49, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Suez, J.; Zmora, N.; Segal, E.; Elinav, E. The pros, cons, and many unknowns of probiotics. Nat. Med. 2019, 25, 716–729. [Google Scholar] [CrossRef]
- Koirala, S.; Anal, A.K. Probiotics-based foods and beverages as future foods and their overall safety and regulatory claims. Future Foods 2021, 3, 100013. [Google Scholar] [CrossRef]
- de Simone, C. The Unregulated Probiotic Market. Clin. Gastroenterol. Hepatol. 2019, 17, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Phavichitr, N.; Puwdee, P.; Tantibhaedhyangkul, R. Cost-benefit analysis of the probiotic treatment of children hospitalized for acute diarrhea in Bangkok, Thailand. Southeast Asian J. Trop. Med. Public Health 2013, 44, 1065–1071. [Google Scholar]
- Lau, V.I.; Rochwerg, B.; Xie, F.; Johnstone, J.; Basmaji, J.; Balakumaran, J.; Iansavichene, A.; Cook, D.J. Probiotics in hospitalized adult patients: A systematic review of economic evaluations. Can. J. Anaesth. 2020, 67, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Coria, D.; Canto-Losa, J.; Carrillo-Vázquez, D.; Carbajal-Morelos, L.; Estrada-León, R.; Corona-Rodarte, E. Lactobacillus gasseri liver abscess and bacteremia: A case report. BMC Infect. Dis. 2021, 21, 518. [Google Scholar] [CrossRef]
- Boyle, R.J.; Robins-Browne, R.M.; Tang, M.L. Probiotic use in clinical practice: What are the risks? Am. J. Clin. Nutr. 2006, 83, 1256–1264. [Google Scholar] [CrossRef]
- Salminen, M.K.; Rautelin, H.; Tynkkynen, S.; Poussa, T.; Saxelin, M.; Valtonen, V.; Järvinen, A. Lactobacillus bacteremia, clinical significance, and patient outcome, with special focus on probiotic L. rhamnosus GG. Clin. Infect. Dis. 2004, 38, 62–69. [Google Scholar] [CrossRef]
| Condition | Virus | Population (Age Range) | Probiotic & Dose | Route | Duration | Type of Study | Results | Ref. |
|---|---|---|---|---|---|---|---|---|
| Healthy adults | - Influenza virus H1N1 and B strains and H3N2 | Adults (18–49 years) | - LGG (1 × 1010 CFU/twice daily) | Oral | 28 days | Randomized double-blind placebo-controlled | - Similar protection rates against the vaccine H1N1 and B strains in subjects receiving LGG and placebo - For H3N2 strain, 84% receiving LGG vs. 55% receiving placebo had a protective titer after vaccination (p = 0.048) | [54] |
| Respiratory tract infection | - Rhinovirus - Syncytal virus - Parainfluenza virus - Enterovirus - Influenza A virus (H1N1) - Human bocavirus Adenovirus - Influenza A virus H3N2 | Children (2–6 years) | - LGG (1 × 108 CFU/twice daily) | Oral | 196 days | Randomized, double-blinded and placebo-controlled parallel group | Reduces respiratory symptoms compared to the placebo (p values no reported) without reducing the number of respiratory viruses | [62] |
| Respiratory tract infection | Rhinovirus | Premature newborns (1–3 days) | - 30 days: LGG + ATCC 5310 (1 × 109 CFU) plus 1 × 600 mg of mix PDX (1:1) - 31–20 days: LGG+ ATCC 5310 (2 × 109 CFU) plus 2 × 600 mg of mix PDX (1:1) | Oral | 60 days | Randomized, double-blind, placebo-controlled | - The incidence of respiratory tract infections was significantly lower in infants receiving prebiotics (rate ratio “RR”, 0.24; 95% CI, 0.12–0.49, p < 0.001) or probiotics RR, 0.50; 95% CI, 0.28–0.90, p = 0.022) compared to placebo - No significant differences were found in terms of viral RNA load during infection, duration of excretion and severity of rhinovirus infections. | [60] |
| Respiratory tract infection. | Rhinovirus Enterovirus | Adults (18–28 years) | - LGG (1 × 109 CFU) - B. animalis spp. lactis BB-12 (2 × 109 CFU) | Oral | 150 days | Randomized, double-blind, placebo-controlled | Decrease in the presence of picornavirus after 3 months (p = 0.0069) in the probiotic group than placebo; however, the appearance of virus in the asymptomatic population was not reduced | [63] |
| Otitis media | Rhinovirus Enterovirus | Children (1–5 years) | - LGG (8–9 × 109 CFU) | Oral | 21 days | Randomized, double-blind, placebo-controlled | No reduction in the presence of rhinoviruses and enteroviruses in children with otitis media | [64] |
| Influenza infection | Influenza A virus | Children (6–12 years) | L. brevis KB290 (1 × 109 CFU) | Oral | 40 days | Open-label and parallel-group | Reduces the incidence of influenza in schoolchildren treatment probiotic than no treatment (15·7 vs. 23.9, p < 0·001); the effect was especially evident in subjects not vaccinated against influenza. | [53] |
| Influenza Symptoms | Influenza | Adults (30–59 years) | L. lactis spp. lactis JCM5805 (1 × 1111 CFU) | Oral | 70 days | Randomized double-blind, placebo-controlled | Inhibits symptom incidence days of cough (p = 0.015) and feverishness (p < 0.009), development by transcriptional upregulation of the IFN-α gene and IFN-stimulated antiviral factor ISG15 (interferon-stimulated gene 15) (p = 0·019), compared to placebo. No final decrease in viral RNA was observed. | [55] |
| Respiratory infections | Influenza | Adults (18–60 years) | - L. paracasei spp. paracasei (1 × 109 CFU) - L. casei 431 (1 × 109 CFU) | Oral | 42 days | Randomized, double-blinded and placebo-controlled parallel group | No effect on the components of the immune response to influenza vaccination was observed, but the duration of upper respiratory symptoms was reduced (p < 0.0059). | [56] |
| Rotavirus Infection | Rotavirus | Adults (18–65 years) | - LGG (1 × 109 CFU) | Oral | 42 days | Randomized, controlled, pilot trial | No significant differences in viral load were shown | [65] |
| Rotavirus Infection | Rotavirus | Adults (18–60 years) | - B. animalis spp. Bl-04 (2 × 109 CFU) | Oral | 5 days | Randomized controlled trial | Decreases rhinovirus replication in nasal secretions (p = 0.03) and reduce virus titer in nasal lavage (p = 0.04). However, not influence on the inflammatory response to rhinovirus infection | [66] |
| Elderly in nursing homes | Influenza | Elderly (≥65 years) | - LGG (1 × 1012 CFU) | Oral | 182 days | Randomized, double-blind, placebo-controlled | Reduces the risk of influenza and other viral respiratory infections by up to 35%, but no significant difference was reported | [57] |
| Aging | Influenza | Adults (60–85 years) | B. longum infantis CCUG 52,486 (1 × 109 CFU) plus gluco-oligosaccharides 8 g/day | Oral | 42 days | Randomized, double-blind, placebo-controlled | Vaccination increased numbers of IgA memory (p < 0.01), IgG memory (p < 0.001) and total IgG B cells (p < 0.001) in young subjects, but not shows same effects in older subjects | [52] |
| Elderly in nursing homes | Influenza | Elderly (≥65 years) | L. coryniformis K8 CECT5711 (3 × 109 CFU) | Oral | 14 days | Randomized, double-blind, placebo-controlled | Increases immune response to flu vaccine (p = 0.036) and decreases symptoms associated with respiratory infections (p = 0.007) compared to placebo | [58] |
| Health workers | Influenza | Adults (20–71 years) | - L. delbrueckii spp. bulgaricus OLL1073R-1, S. thermophilus (1.1 × 109 CFU) | Oral | 112 days | Randomized controlled | Probiotic intake shows a significant preventive effect against influenza or NK cell activity. However, increase in the production of IFN-γ (2.69 to 6.21 pg/mL, p = 0.03) | [67] |
| Elderly in nursing homes | Influenza | Elderly (mean 87.35 years) | - L. delbrueckii spp. bulgaricus OLL1073R-1 (1.1 × 108 CFU) | Oral | 84 days | Randomized, double-blind, parallel-group | Prevents infection caused by influenza A virus subtype H3N2 and increasing IgA (p = 0.04) and H3N2-bound IgA (p = 0.001) levels in saliva | [59] |
| Condition | Population (Age Range) | Probiotic & Dose | Route | Duration | Type of Study | Results | Ref. |
|---|---|---|---|---|---|---|---|
| HIV Infection | Adults (mean 48 years) | - L. rhamnosus CAN-1 (1 × 109 CFU) plus micronutrients (vitamin A- 1500 IU, vitamin E- 5.7 IU, niacinamide- 3.8 mg, vitamin B1- 0.3 mg, vitamin- B12 0.6 μg, vitamin B6- 0.3 mg, vitamin C- 21 mg, Fe- 3.3 mg, Se-13.8 μg, Zc-2.4 mg DHA (omega-3 fatty acid from fish oil) 13 mg, and EPA (omega-3 fatty acid from fish oil) 19 mg). | Oral | 30 days | Randomized, double-blind, three-period cross-over controlled | Increases immune function and CD 4+ lymphocyte count, with micronutrient alone 41 cells/µL, probiotic + micronutrient +19 cells/µL and probiotic alone—7 cells/µL, in HIV-positive individuals. However not shows significant difference. | [84] |
| Chronic HIV infection | Women (mean 47.5 years) | - P. pentosaceus (1 × 1010 CFU) - L. mesenteroides (1 × 1010 CFU) - L. paracasei spp. paracasei (1 × 1010 CFU) - L. plantarum (1 × 1010 CFU) | Oral | 28 days | Randomized, placebo-controlled | Increases the levels of probiotic species (L. plantarum p = 0.001 and P. pentosaceus p = 0.036) in the gut during chronic HIV-1 infection. However, plasma CD14 and C-reactive protein levels were not affected during treatment | [75] |
| Chronic HIV infection | Adults (37–72 years) | - B. coagulans GBI-30, 6086 (2 × 1012 CFU) | Oral | 90 days | Double-blind placebo-controlled | Increases the percentage of CD4+ T cells (p = 0.018), and shown inflammation decrease by correlation between D-dimer with CRP and sCD14 with tumor necrosis factor (TNF)-α. | [70] |
| HIV infection | Children (≤15 years) | - L. sporogens (2.5 × 1010 CFU) plus micronutrients (Cu- 25 μg, Zn- 5 mg, Se- 10 μg, I- 38μg, vitamin A-1250 IU, vitamin B1 and B2- 0.75 mg, vitamin B6- 0.5 mg, vitamin B5- 1.25 mg, vitamin B12- 0.5 μg, vitamin D- 100 IU, and vitamin E- 2.5 IU) | Oral | 90 days | Randomized control study | Increases CD4+ T cells compared to control group (p = 0.0022). In addition, micronutrient supplementation shows a significant delay (p = 0.049), in the progression of the advanced stage of the disease, according to WHO clinical staging | [85] |
| HIV infection | Adults (18–80 years) | - S. salivarius (2.0 × 1011 CFU) - B. breve (9.3 × 1010 CFU) - B. infantis (9.3 × 1010 CFU) - B. longum (9.3 × 1010 CFU) - L. acidophilus (2.0 × 109 CFU) - L. plantarum (2.2 × 1011 CFU) - L. casei (2.2 × 1011 CFU) - L. delbrueckii (3.0 × 1011 CFU) - S. faecium (3.0 × 107 CFU) | Oral | 336 days | Unspecified | Provides a specific benefit in HIV-infected patients during antiretroviral treatment, reduced immune activation on CD4 T-lymphocytes. Probiotics intake reducing systemic inflammation (CRP plasma levels, p = 0.006) | [83] |
| HIV infection | Adults (≥18 years) | Visbiome®: L. acidophilus, L. plantarum, L. paracasei, L. delbrueckii, B. breve, B. infantis, B. longum, and S. thermophilus (4.5 × 1011 CFU/packet) | Oral | 168 days | Prospective, double-blinded, randomized, placebo-controlled, multicenter pilot studies | Reduces inflammation and improves gut immune health; moreover, it was safe and tolerated by HIV patients. | [86] |
| Neuropsychological performance in HIV-infected patients | Adults (≥18 years) | Vivomixx®; Visbiome®: L. plantarum, S. thermophilus, B. breve, L. paracasei, L. delbrueckii spp. bulgaricus, L. acidophilus, B. longum, and B. infantis (4.5 × 1011 CFU) | Oral | 180 days | Longitudinal, nonrandomized designed, single-arm, pilot study | Improvement of neurological cognitive functions, such as abstract reasoning and short-term (p = 0.0058) and long-term memory (p = 0.0019). However, no direct effect on viral load was observed | [82] |
| HIV infection | Adults (≥18 years) | - L. plantarum, S. thermophilus, B. breve, L. paracasei, L. delbrueckii spp. bulgaricus, L. acidophilus, B. longum, and B. infantis (1.8 × 1012 CFU) | Oral | 180 days | Longitudinal pilot study | - Increase significantly Bifidobacteria spp. Compared to their basal level (p = 0.019). - Reduces the frequency of CD8+ lymphocytes (not shows significant difference) and increases the percentage of Th17 cell subsets (p = 0.059). - Restore the physical and immunological integrity of the intestinal mucosal barrier in HIV patients | [81] |
| HIV infection | Children (9 months–5.8 years) | - L. casei Shirota (6.5 × 109 CFU) | Oral | 56 days | Nonrandomized, open-labeled, prospective study | Significantly increase in CD4+ cells count (p < 0.01), especially Th17 (p < 0.05), and decrease in CD8+ cells (27.5% to 13.2%, p < 0.001). However, it was not associated with an increase in plasma HIV load or serious adverse events | [78] |
| HIV infection | Adults (≥18 years) | - S. boulardii (250 g) | Oral | 336 days | Pilot Multicenter randomized, placebo-controlled, double-blind | Did not improve the number of circulating T cells, inflammation or immune activation | [80] |
| Condition | Agent | Population (Age Range) | Probiotics & Dose | Route | Duration | Type of Study | Results | Ref. |
|---|---|---|---|---|---|---|---|---|
| Acut ediarrhea | Rotavirus | Children (1–23 months) | - One probiotic: S. boulardii (4 × 1010 CFU) - Mix of probiotics: L. acidophilus (6.6 × 107 CFU), L. rhamnosus (3.6 × 107 CFU), B. longum (8.7 × 106 CFU) and S. boulardii (1.3 × 107 CFU) | Oral | 5 days | Randomized double-blind controlled | - S boulardii diminished diarrhea in (58 vs. 84.5 h, p = 0.04) and fever (18 vs. 67 h, p = 0.0042) compared with the control. No decrease in rotavirus load when S. boluardii was used. - Mixed probiotic administrations decrease the duration of vomiting (0 vs. 42.5 h, p = 0.041) compared to oral rehydration solution | [96] |
| Gastroenteritis | Norovirus | Elderly (mean 84 years) | - L. casei Shirota (4 × 1010 CFU) | Oral | 90 days | Open study | - Contributes positively to the relief of fever (p < 0.05), caused by norovirus. - Restored the intestinal microbiota, significantly increased levels of Bifidobacterium (p < 0.01) and L. casei subgroup (p < 0.01). However, it does not provide protection against viral gastroenteritis | [91] |
| Acute Gastroenteritis | Rotavirus | Children (6 months–5 years) | - LGG (1 × 1010 CFU) | Oral | 28 days | Randomized, double-blind, placebo-controlled | - Improved intestinal permeability in children with rotavirus (p = 0.027) - Increased IgG levels response in children with rotavirus diarrhea (456 vs. 2215 EU, p = 0.003) - Reduce episodes of diarrhea in children with rotavirus gastroenteritis (25% vs. 46%, p = 0.048), but not showed in Cryptosporidium gastroenteritis. | [97] |
| Gastroenteritis | Rotavirus | Children (3 months–14 years) | - C. butyricum (2.0 × 107 CFU) - E. faecalis (3.17 × 108 CFU) - B. mesentericus (1.1 × 107 CFU) | Oral | 7 days | Single-center, open-label, randomized, controlled trial | Reduction in the duration of diarrhea in children (p < 0.0001). In pregnancy, no decrease in viral load after probiotic treatment was observed | [99] |
| Gastroenteritis | Rotavirus | Children (3 months–7 years) | - B. longum, B. lactis, L. acidophilus, L. rhamnosus, L. plantarum, P. pentosaceus (1.1 × 109 CFU/g; 1 × 108 CFU/strain) | Oral | 7 days | Randomized, double-blind | Decreases the duration of diarrhea, compared with a placebo (6.1 vs. 7.2, p = 0.030), without any adverse effect. B. longum and L. acidophilus inhibited rotavirus infection in vitro | [98] |
| Acute diarrhea | Rotavirus | Children (3 months–5 years) | S. boulardii (500 mg/day) | Oral | 5 days | Randomized and controlled | Probiotics intake significantly decrease to diarrhea (60 vs. 89 h; 95% CI: −41.2 to −16.8) and the duration of hospitalization (74 vs. 91 h; 95% CI: −33.46 to −0.54), compared with a control group. However, fever, vomiting and viral load were not reduced | [101] |
| Gastroenteritis acute | Rotavirus | Children (3–48 months) | - L. rhamnosus R0011 and L. helveticus R0052 (4.0 × 109 CFU) (95:5) | Not specified | 5 days | Multi-center, double-blind trial, randomized | No beneficial effects were shown. Administration did accelerate clearance of rhinovirus associated with viral gastroenteritis | [100] |
| Gastroenteritis | Rotavirus | Children (14–40 months) | L. plantarum LRCC5310 (dose: not specified) | Not specified | Not specified | Inhibits rotavirus growth, reduces virus titer (p < 0.001), and improves gastroenteritis symptoms such as diarrhea and Vesikari score | [87] |
| Condition | Population (Age Range) | Probiotic & Dose | Route | Duration | Type of Study | Results | Ref. |
|---|---|---|---|---|---|---|---|
| Cervical lesion | Women (mean 31.75 years) | L. casei Shirota (1 × 1010 CFU/day) | Oral | 6-month | Prospective controlled pilot | Reduces twice as likely to clear cytological abnormalities compared than control (60 vs. 31%, p = 0.05) | [108] |
| HPV-infection | Women (>18 years) | Lactobacillus rhamnosus BMX54 (1 × 104 CFU/tablet) | Vaginal | Short-term 3 months Long term 9 months | Randomized, pilot study | Short-term restore the vaginal microbiota generating a state of bacterial balance. The patients who used probiotics long-term, decrease in HPV clearance (31.2% vs. 11.6%, p = 0.044) and reduce HPV-related cytologic abnormalities compared with short-term (79.4% vs. 37.5%, p = 0.041) | [106] |
| Genital high-risk by HPV-infection | Women (30–65 years) | Lactobacillus rhamnosus GR-1 Lactobacillus reuteri RC-14 (5.4 × 109 CFU) | Oral | Not specified | Randomized, double-blinded, placebo-controlled trial | Reduces genital risk in women with high-risk HPV (p = 0.006); however, no significant differences were found in the HPV clearance rate | [107] |
| Hepatic encephalopathy (HE) | Adults (unspecified age) | Bifidobacterium plus fructo-oligosaccharide (FOS) | Oral | 60 days | Unspecified | Reduces significant ammonia levels (50.2 vs. 61.4 mmol/L, p < 0.001) compared with lactulose treatment and an improvement in traceability tests (p < 0.05), symbol digit modalities (p < 0.001) and block design (p < 0.001) | [116] |
| Minimal hepatic encephalopathy (MHE) | Adults (unspecified age) | C. butyricum (1.0 × 107 CFU) B. infantis (1 × 106 CFU) | Oral | 3 months | Unspecified | - Improvement in psychometric tests, the digit symbol test and the number connection test - Modified the diversity of the intestinal microbiota, finding an increase in Clostridium cluster I and Bifidobacterium (p < 0.05) and decrease Enterococcus and Enterobacteriaceae (p < 0.05) - Shows lows ammonia levels in the probiotics group than control group (76.4 vs. 152.0 μmol/mL, p = 0.032) | [111] |
| Genital Herpes Infections | Women (17–57 years) | Lactobacillus brevis CD2 Lactobacillus brevis KB290 Lactobacillus brevis SBC8803 (2 × 109 CFU/capsule) | Oral | 6 months | Randomized double-blind controlled trial | -Probiotic decrease lesion healing time in comparison with aciclovir-treatment (6.5 vs. 5.2 days, p = 0.06). - Suppress recurrent herpes virus infection (p = 0.03). - Moreover, probiotic therapy has no side effects (headache, nausea, diarrhea and abdominal pain). | [122] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reyes-Castillo, P.A.; González-Vázquez, R.; Torres-Maravilla, E.; Tello, M.; Bermúdez-Humarán, L.G.; Mayorga-Reyes, L. Probiotics against Viral Infections: Current Clinical Trials and Future Perspectives. Immuno 2021, 1, 468-498. https://doi.org/10.3390/immuno1040034
Reyes-Castillo PA, González-Vázquez R, Torres-Maravilla E, Tello M, Bermúdez-Humarán LG, Mayorga-Reyes L. Probiotics against Viral Infections: Current Clinical Trials and Future Perspectives. Immuno. 2021; 1(4):468-498. https://doi.org/10.3390/immuno1040034
Chicago/Turabian StyleReyes-Castillo, Pedro A., Raquel González-Vázquez, Edgar Torres-Maravilla, Mario Tello, Luis G. Bermúdez-Humarán, and Lino Mayorga-Reyes. 2021. "Probiotics against Viral Infections: Current Clinical Trials and Future Perspectives" Immuno 1, no. 4: 468-498. https://doi.org/10.3390/immuno1040034
APA StyleReyes-Castillo, P. A., González-Vázquez, R., Torres-Maravilla, E., Tello, M., Bermúdez-Humarán, L. G., & Mayorga-Reyes, L. (2021). Probiotics against Viral Infections: Current Clinical Trials and Future Perspectives. Immuno, 1(4), 468-498. https://doi.org/10.3390/immuno1040034








