A Polysomnographic and Cluster Analysis of Periodic Limb Movements in Sleep of Restless Legs Syndrome Patients with Psychiatric Conditions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Procedure
2.2. Polysomnography (PSG)
2.2.1. The Apnea–Hypopnea Index (AHI)
2.2.2. The Periodic Limb Movements of Sleep (PLMS) Index
2.3. Statistical Analysis
2.3.1. The Standard Clinical Approach
2.3.2. The Datamining Approach: Cluster Analysis
3. Results
3.1. Clinical Characterization
3.2. First- and Second-Night PSG
3.3. Comparison Across Groups Based on Clinical Cut-Offs of AHI ≥ 5 and PLMS Index ≥15
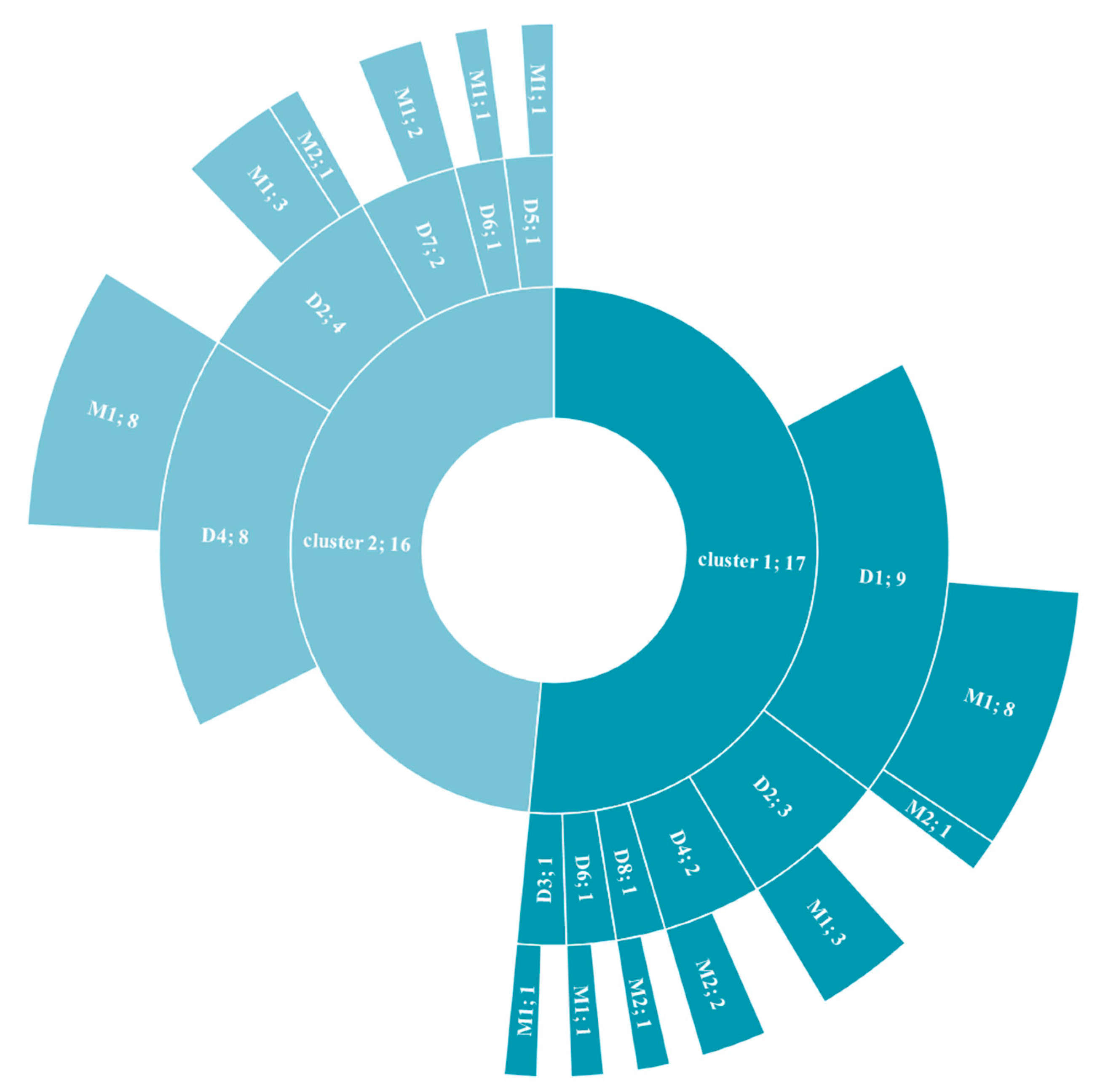
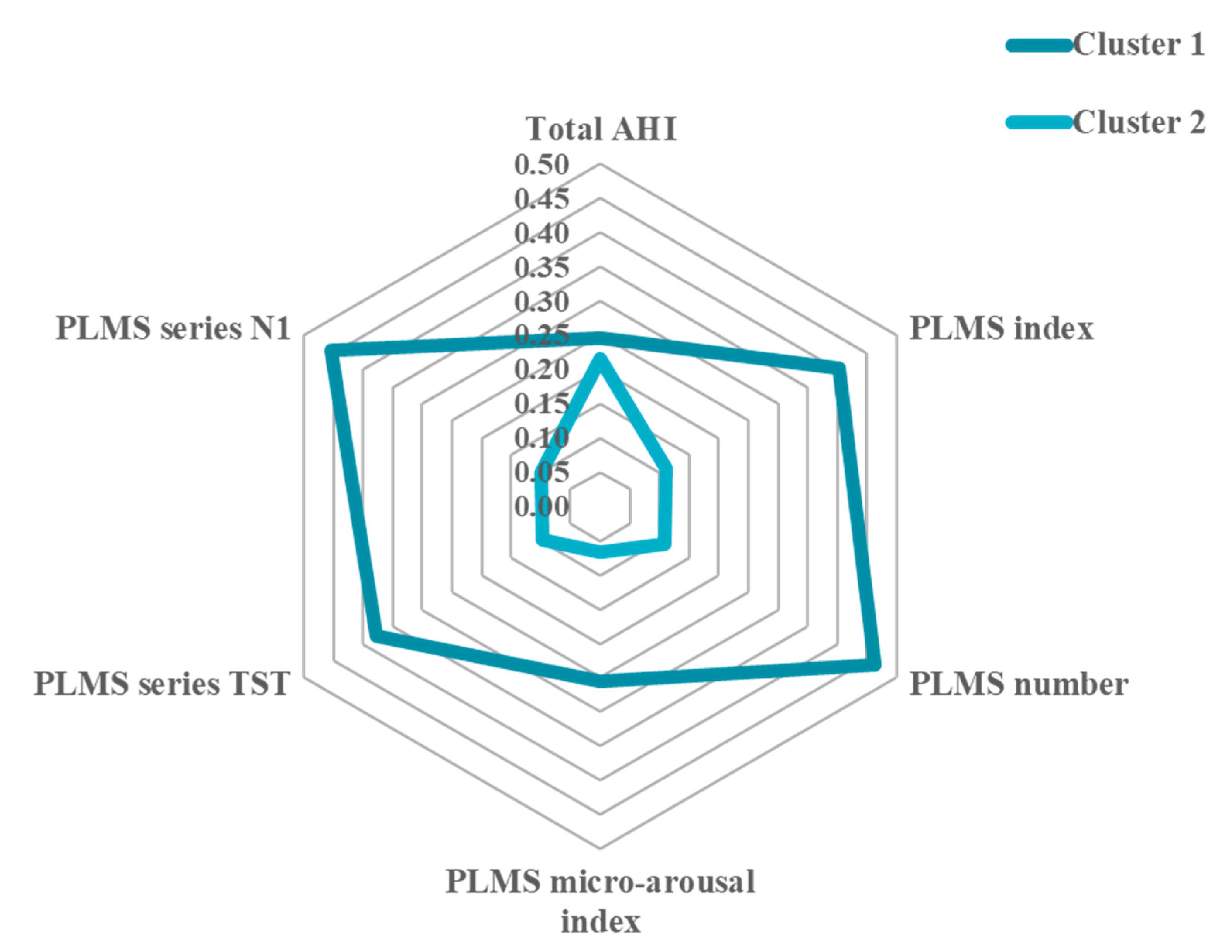
3.4. Cluster Analysis
Sensitivity Analysis
4. Discussion
5. Limitations
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Allen, R.P.; Walters, A.S.; Montplaisir, J.; Hening, W.; Myers, A.; Bell, T.J.; Ferini-Strambi, L. Restless legs syndrome prevalence and impact: REST general population study. Arch. Intern. Med. 2005, 165, 1286–1292. [Google Scholar] [CrossRef] [Green Version]
- Allen, R.P.; Picchietti, D.L.; Garcia-Borreguero, D.; Ondo, W.G.; Walters, A.S.; Winkelman, J.W.; Zucconi, M.; Ferri, R.; Trenkwalder, C.; Lee, H.B. Restless legs syndrome/Willis-Ekbom disease diagnostic criteria: Updated International Restless Legs Syndrome Study Group (IRLSSG) consensus criteria—history, rationale, description, and significance. Sleep Med. 2014, 15, 860–873. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Chen, H.; Liu, Z.Z.; Jia, C.X. Insomnia and psychopathological features associated with restless legs syndrome in chinese adolescents. J. Clin. Psychiatry 2018, 79. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Deng, Q.; Qin, Q.; Vgontzas, A.N.; Basta, M.; Xie, C.; Li, Y. Sleep disorders in Wilson disease: A systematic review and meta-analysis. J. Clin. Sleep Med. 2020, 16, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Montplaisir, J.; Boucher, S.; Poirier, G.; Lavigne, G.; Lapierre, O.; Lesperance, P. Clinical, polysomnographic, and genetic characteristics of restless legs syndrome: A study of 133 patients diagnosed with new standard criteria. Mov. Disord. 1997, 12, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Kang, S.G.; Lee, H.J.; Jung, S.W.; Cho, S.N.; Han, C.; Kim, Y.K.; Kim, S.H.; Lee, M.S.; Joe, S.H.; Jung, I.K.; et al. Characteristics and clinical correlates of restless legs syndrome in schizophrenia. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2007, 31, 1078–1083. [Google Scholar] [CrossRef]
- Jung, J.S.; Lee, H.J.; Cho, C.H.; Kang, S.G.; Yoon, H.K.; Park, Y.M.; Moon, J.H.; Yang, H.J.; Song, H.M.; Kim, L. Association between restless legs syndrome and CLOCK and NPAS2 gene polymorphisms in schizophrenia. Chronobiol. Int. 2014, 31, 838–844. [Google Scholar] [CrossRef]
- Picchietti, D.L.; England, S.J.; Walters, A.S.; Willis, K.; Verrico, T. Periodic limb movement disorder and restless legs syndrome in children with attention-deficit hyperactivity disorder. J. Child Neurol. 1998, 13, 588–594. [Google Scholar] [CrossRef] [PubMed]
- Ross, R.J.; Ball, W.A.; Dinges, D.F.; Kribbs, N.B.; Morrison, A.R.; Silver, S.M.; Mulvaney, F.D. Motor dysfunction during sleep in posttraumatic stress disorder. Sleep 1994, 17, 723–732. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Germain, A.; Nielsen, T.A. Sleep pathophysiology in posttraumatic stress disorder and idiopathic nightmare sufferers. Biol. Psychiatry 2003, 54, 1092–1098. [Google Scholar] [CrossRef]
- Lee, T.H.; Yen, T.T.; Chiu, N.Y.; Chang, C.C.; Hsu, W.Y.; Chang, Y.J.; Chang, T.G. Depression is differently associated with sleep measurement in obstructive sleep apnea, restless leg syndrome and periodic limb movement disorder. Psychiatry Res. 2019, 273, 37–41. [Google Scholar] [CrossRef]
- Zak, R.; Fisher, B.; Couvadelli, B.V.; Moss, N.M.; Walters, A.S. Preliminary study of the prevalence of restless legs syndrome in adults with attention deficit hyperactivity disorder. Percept. Mot. Ski. 2009, 108, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.B.; Hening, W.A.; Allen, R.P.; Kalaydjian, A.E.; Earley, C.J.; Eaton, W.W.; Lyketsos, C.G. Restless legs syndrome is associated with DSM-IV major depressive disorder and panic disorder in the community. J. Neuropsychiatry Clin. Neurosci. 2008, 20, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, S.; Na, M.; Winkelman, J.W.; Ba, D.; Liu, C.F.; Liu, G.; Gao, X. Association of restless legs syndrome with risk of suicide and self-harm. JAMA Netw. Open 2019, 2, e199966. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rubí, G.P. Restless legs syndrome from the perspective of psychiatry. Rev. Med. Chile 2018, 146, 1041–1049. [Google Scholar] [CrossRef]
- Mackie, S.; Winkelman, J.W. Restless legs syndrome and psychiatric disorders. Sleep Med. Clin. 2015, 10, 351–357. [Google Scholar] [CrossRef]
- Flick, U. Introducing Research Methodology: A Beginner’s Guide to Doing a Research Project, 2nd ed.; SAGE Publications Ltd.: Thousand Oaks, CA, USA, 2015. [Google Scholar]
- Leclair-Visonneau, L.; Vecchierini, M.F.; Schröder, C.; Charley Monaca, C. French Consensus: How to diagnose restless legs syndrome. Rev. Neurol. 2018, 174, 508–514. [Google Scholar] [CrossRef]
- Iber, C. American Academy of Sleep. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications; American Academy of Sleep Medicine: Westchester, IL, USA, 2007. [Google Scholar]
- Ferri, R.; Fulda, S.; Allen, R.P.; Zucconi, M.; Bruni, O.; Chokroverty, S.; Ferini-Strambi, L.; Frauscher, B.; Garcia-Borreguero, D.; Hirshkowitz, M.; et al. World Association of Sleep Medicine (WASM) 2016 standards for recording and scoring leg movements in polysomnograms developed by a joint task force from the International and the European Restless Legs Syndrome Study Groups (IRLSSG and EURLSSG). Sleep Med. 2016, 26, 86–95. [Google Scholar] [CrossRef]
- Berry, R.B.; Budhiraja, R.; Gottlieb, D.J.; Gozal, D.; Iber, C.; Kapur, V.K.; Marcus, C.L.; Mehra, R.; Parthasarathy, S.; Quan, S.F.; et al. Rules for scoring respiratory events in sleep: Update of the 2007 AASM manual for the scoring of sleep and associated events. Deliberations of the sleep apnea definitions task force of the American Academy of Sleep Medicine. J. Clin. Sleep Med. 2012, 8, 597–619. [Google Scholar] [CrossRef] [Green Version]
- Sateia, M.J. International classification of sleep disorders: Highlights and modifications. Chest 2014, 146, 1387–1394. [Google Scholar] [CrossRef]
- Berry, R.B.; Brooks, R.; Gamaldo, C.E.; Harding, S.M.; Marcus, C.L.; Vaughn, B.V. The AASM Manual for the Scoring of Sleep and Associated Events: Rules, Terminology and Technical Specifications; American Academy of Sleep Medicine: Darien, IL, USA, 2012. [Google Scholar]
- Ferri, R.; Rundo, F.; Zucconi, M.; Manconi, M.; Aricò, D.; Bruni, O.; Ferini-Strambi, L.; Fulda, S. Putting the periodicity back into the periodic leg movement index: An alternative data-driven algorithm for the computation of this index during sleep and wakefulness. Sleep Med. 2015, 16, 1229–1235. [Google Scholar] [CrossRef]
- Hennig, C.; Meila, M.; Murtagh, F.; Rocci, R. Handbook of Cluster Analysis, 1st ed.; Henning, C., Meila, M., Murtagh, F., Rocci, R., Eds.; Chapman and Hall/CRC: London, UK, 2020. [Google Scholar]
- Hornyak, M.; Feige, B.; Voderholzer, U.; Philipsen, A.; Riemann, D. Polysomnography findings in patients with restless legs syndrome and in healthy controls: A comparative observational study. Sleep 2007, 30, 861–865. [Google Scholar] [CrossRef] [Green Version]
- Aksu, M.; Demirci, S.; Bara-Jimenez, W. Correlation between putative indicators of primary restless legs syndrome severity. Sleep Med. 2007, 8, 84–89. [Google Scholar] [CrossRef]
- Garcia-Borreguero, D.; Larrosa, O.; de la Llave, Y.; Granizo, J.J.; Allen, R. Correlation between rating scales and sleep laboratory measurements in restless legs syndrome. Sleep Med. 2004, 5, 561–565. [Google Scholar] [CrossRef]
- Dennis, E.A.-O.; Baron, D.; Bartnik-Olson, B.; Caeyenberghs, K.; Esopenko, C.; Hillary, F.G.; Kenney, K.; Koerte, I.A.-O.; Lin, A.P.; Mayer, A.A.-O.; et al. ENIGMA brain injury: Framework, challenges, and opportunities. Hum. Brain Mapp. 2020. [Google Scholar] [CrossRef]
- Smoller, J.W.; Andreassen, O.A.; Edenberg, H.J.; Faraone, S.V.; Glatt, S.J.; Kendler, K.S. Psychiatric genetics and the structure of psychopathology. Mol. Psychiatry 2019, 24, 409–420. [Google Scholar] [CrossRef] [PubMed]
- Thorpy, M.J. Classification of sleep disorders. Neurotherapeutics 2012, 9, 687–701. [Google Scholar] [CrossRef] [PubMed]
- Winkelmann, J.; Prager, M.; Lieb, R.; Pfister, H.; Spiegel, B.; Wittchen, H.U.; Holsboer, F.; Trenkwalder, C.; Ströhle, A. Anxietas tibiarum. Depression and anxiety disorders in patients with restless legs syndrome. J. Neurol. 2005, 252, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, O.; Şengül, Y.; Şengül, H.S.; Parlakkaya, F.B.; Öztürk, A. Investigation of alexithymia and levels of anxiety and depression among patients with restless legs syndrome. Neuropsychiatr. Dis. Treat. 2018, 14, 2207–2214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Angriman, M.; Cortese, S.; Bruni, O. Somatic and neuropsychiatric comorbidities in pediatric restless legs syndrome: A systematic review of the literature. Sleep Med. Rev. 2017, 34, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Japaridze, G.; Kasradze, S.; Maisuradze, L.; Popp, R.; Wetter, T. The Restless legs syndrome. Georgian Med. News 2018, 74–81. [Google Scholar]
- Cuellar, N.G. The psychopharmacological management of RLS in psychiatric conditions: A review of the literature. J. Am. Psychiatr. Nurses Assoc. 2012, 18, 214–225. [Google Scholar] [CrossRef]
- Patatanian, E.; Claborn, M.K. Drug-induced restless legs syndrome. Ann. Pharmacother. 2018, 52, 662–672. [Google Scholar] [CrossRef]
- Perez-Lloret, S.; Rey, M.V.; Bondon-Guitton, E.; Rascol, O.; Montastruc, A.J. Drugs associated with restless legs syndrome: A case/noncase study in the French Pharmacovigilance Database. J. Clin. Psychopharmacol. 2012, 32, 824–827. [Google Scholar] [CrossRef] [PubMed]
- Romero-Peralta, S.; Cano-Pumarega, I.; Garcia-Malo, C.; Agudelo Ramos, L.; Garcia-Borreguero, D. Treating restless legs syndrome in the context of sleep disordered breathing comorbidity. Eur. Respir. Rev. 2019, 28. [Google Scholar] [CrossRef]
- Kushida, C.A.; Allen, R.P.; Atkinson, M.J. Modeling the causal relationships between symptoms associated with restless legs syndrome and the patient-reported impact of RLS. Sleep Med. 2004, 5, 485–488. [Google Scholar] [CrossRef] [PubMed]
- Martikainen, M.H.; Ng, Y.S.; Gorman, G.S.; Alston, C.L.; Blakely, E.L.; Schaefer, A.M.; Chinnery, P.F.; Burn, D.J.; Taylor, R.W.; McFarland, R.; et al. Clinical, genetic, and radiological features of extrapyramidal movement disorders in mitochondrial disease. JAMA Neurol. 2016, 73, 668–674. [Google Scholar] [CrossRef] [Green Version]
- Roche, S.; Jacquesson, J.M.; Destee, A.; Defebvre, L.; Derambure, P.; Monaca, C. Sleep and vigilance in corticobasal degeneration: A descriptive study. Neurophysiol. Clin. 2007, 37, 261–264. [Google Scholar] [CrossRef]
- Pauletto, G.; Belgrado, E.; Marinig, R.; Bergonzi, P. Sleep disorders and extrapyramidal diseases: An historical review. Sleep Med. 2004, 5, 163–167. [Google Scholar] [CrossRef]
- Velázquez-Pérez, J.M.; Marsal-Alonso, C. Drug-induced movement disorders. Rev. Neurol. 2009, 48, S57–S60. [Google Scholar]
- Hutka, P.; Krivosova, M.; Muchova, Z.; Tonhajzerova, I.; Hamrakova, A.; Mlyncekova, Z.; Mokry, J.; Ondrejka, I. Association of sleep architecture and physiology with depressive disorder and antidepressants treatment. Inter. J. Molecular Sci. 2021, 22. [Google Scholar] [CrossRef] [PubMed]
| Total (n = 43) | PLMS Index < 15 (n = 27) | PLMS Index ≥15 (n = 16) | Mann–Whitney U Test | p-Value | |
|---|---|---|---|---|---|
| Age, years | 57.3 ± 15.6 | 55.2 ± 15.0 | 61.0 ± 16.3 | 1.26 | 0.210 |
| Male/female,n | 27/16 | 16/11 | 11/5 | χ2(1) = 0.39 | 0.534 |
| Total sleep time, min. | 377.7 ± 100.0 | 378.6 ± 107.1 | 376.2 ± 90.1 | 0.5 | 0.598 |
| Sleep efficiency, % | 75.4 ± 14.6 | 73.5 ± 12.9 | 78.8 ± 17.1 | 1.7 | 0.100 |
| Sleep quality, % | 16.2 ± 8.4 | 17.5 ± 9.0 | 13.9 ± 7.0 | −1.2 | 0.238 |
| Sleep latency, min. | 23.5 ± 24.0 | 23.7 ±19.7 | 23.3 ± 30.6 | −0.9 | 0.346 |
| REM sleep latency, min. | 131.7 ± 86.5 | 137.6 ± 79.4 | 121.9 ± 99.4 | −1.0 | 0.327 |
| Wake after sleep onset, min. | 81.8 ± 58.9 | 94.3 ± 61.1 | 60.8 ± 49.8 | −2.0 | 0.042 |
| Arousals, n | 34.4 ± 21.8 | 37.1 ± 24.6 | 29.9 ± 15.6 | −1.0 | 0.315 |
| Stage N1, % | 22.3 ± 12.4 | 22.2 ± 13.9 | 22.5 ± 9.8 | 0.6 | 0.563 |
| Stage N2, % | 61.6 ± 12.1 | 60.3 ± 12.6 | 63.7 ± 11.5 | 0.8 | 0.429 |
| Stage N3, % | 3.3 ± 4.9 | 4.0 ± 5.0 | 2.1 ± 4.6 | −1.7 | 0.089 |
| REM, % | 12.8 ± 5.3 | 13.4 ± 6.0 | 11.8 ± 8.8 | 1.3 | 0.210 |
| Total AHI, n/hr | 18.1 ± 17.2 | 18.0 ± 18.8 (n = 22) | 18.3 ± 14.3 (n = 12) | 0.5 | 0.652 |
| Snore index,/hr | 22.2 ± 60.8 | 5.8 ± 18.9 (n = 22) | 49.5 ± 95.1 (n = 12) | 2.0 | 0.051 |
| O2 saturation nadir, % | 93.2 ± 1.6 | 93.1 ± 1.7 (n = 22) | 93.4 ± 1.6 (n = 12) | −0.1 | 0.900 |
| PLMS index, n/h | 17.8 ± 32.5 | 5.4 ± 4.9 | 38.8 ± 46.6 | 5.4 | 0.0000 |
| PLMS, n | 111.7 ± 258.0 | 33.4 ± 35.2 | 243.8 ± 393.3 | 4.6 | 0.0000 |
| PLMS micro-arousal index, n/h | 2.3 ± 3.2 | 0.8 ± 1.0 | 4.7 ± 4.1 | 3.7 | 0.0002 |
| PLMS average interval, s | 31.6 ± 15.1 | 30.2 ± 17.9 | 33.8 ± 8.5 | −0.2 | 0.880 |
| PLMW index, n/h | 2.5 ± 8.4 | 0.5 ± 1.4 | 5.9 ± 13.1 | 2.1 | 0.036 |
| PLMW, n | 2.8 ± 10.8 | 0.7 ± 2.0 | 6.4 ± 17.3 | 2.0 | 0.051 |
| PLMW average interval, s | 8.1 ± 19.1 | 3.3 ± 12.2 | 16.3 ± 25.6 | 2.3 | 0.021 |
| PLMS series TST,% | 10.9 ± 14.7 | 4.2 ± 4.4 | 22.3 ± 18.8 | 4.7 | 0.0000 |
| PLMS series N1,% | 10.5 ± 13.8 | 4.2 ± 4.9 | 21.2 ± 17.4 | 4.7 | 0.0000 |
| PLMS series N2,% | 11.9 ± 16.6 | 4.8 ± 5.7 | 23.9 ± 21.7 | 4.2 | 0.0000 |
| PLMS series N3,% | 3.4 ± 12.1 | 1.8 ± 6.1 | 6.3 ± 18.2 | 0.3 | 0.804 |
| PLMS series REM,% | 3.6 ± 9.8 | 1.1 ± 4.2 | 7.8 ± 14.4 | −0.7 | 0.506 |
| Available AHI First Night Versus Second Night | Mean ± sd | Wilcoxon Matched Pairs Test, p-Value |
|---|---|---|
| all; n = 13 versus n = 13 | 21.6 ± 20.3 versus 24.4 ± 17.7 | p-value = 0.46 |
| excluding those that discontinued CPAP (n = 2) but including those that continued CPAP (n = 3); n = 10 versus n = 10, | 23.9 ± 22.8 versus 21.8 ± 18.5 | p-value = 0.38 |
| all non-CPAP users; n = 8 versus n = 8 | 26.6 ± 24.9 versus 24.7 ± 19.7 | p-value = 0.54 |
| 1 Subjects Without SDB and PLMS (n = 6) | 2 Subjects With Only SDB (n = 16) | 3 Subjects With Only PLMS (n = 2) | 4 Subjects With SDB and PLMS (n = 10) | Kruskall–Wallis Test H (3, n = 34) | p-Value | Group Comparisons | |
|---|---|---|---|---|---|---|---|
| Age, years | 48.0 ± 17.7 | 59.4 ± 13.2 | 61.5 ± 30.4 | 61.9 ± 15.5 | 3.2 | 0.3590 | |
| Male/female, n | 1/5 | 13/3 | 2/0 | 7/3 | χ2(3) = 9.5 | 0.0234 | |
| Total sleep time, min | 428.2 ± 55.1 | 366.8 ± 129.2 | 365.5 ± 94.0 | 384.7 ±99.6 | 3.6 | 0.3057 | |
| Sleep efficiency, % | 85.8 ± 5.7 | 70.4 ± 13.2 | 74.3 ± 29.7 | 79.5 ± 17.1 | 6.3 | 0.0961 | |
| Sleep quality, % | 24.3 ± 9.3 | 16.1 ± 8.4 | 16.9 ± 12.2 | 12.9 ± 5.8 | 5.4 | 0.1421 | |
| Sleep latency, min. | 15.4 ± 8.7 | 27.2 ± 24.3 | 44.0 ± 46.1 | 19.8 ± 27.6 | 1.8 | 0.6151 | |
| Sleep latency REM, min | 112.0 ± 49.3 | 135.0 ± 91.2 | 176.8 ± 175.7 | 114.3 ± 110.0 | 0.8 | 0.8385 | |
| Wake after sleep onset, min | 42.2 ± 26.0 | 100.2 ± 60.7 | 88.3 ± 112.8 | 53.3 ± 46.3 | 7.4 | 0.0614 | |
| Arousals, n/h | 22.3 ± 9.3 | 42.9 ± 30 | 25.5 ± 20.5 | 30.5 ± 17.8 | 4.6 | 0.2025 | |
| Stage N1, % | 12.0 ± 5.9 | 26.7 ± 15.9 | 9.9 ± 5.4 | 24.4 ± 10.0 | 10.9 | 0.0122 | |
| Stage N2, % | 63.7 ± 7.3 | 57.2 ± 14.8 | 73.3 ± 6.9 | 62.8 ± 11.1 | 3.1 | 0.3712 | |
| Stage N3, % | 8.3 ± 6.5 | 2.8 ± 4.0 | 3.9 ± 5.6 | 1.1 ± 3.1 | 7.9 | 0.0473 | |
| REM,% | 16.0 ± 5.3 | 13.3 ± 6.7 | 12.9 ± 6.6 | 11.7 ± 3.8 | 2.6 | 0.4499 | |
| Total AHI, n/h | 2.2 ±1.2 | 23.9 ± 19.0 | 1.9 ± 1.8 | 21.6 ± 13.3 | 17.9 | 0.0005 | 1 < 2, 1 < 4 |
| Snore index,/h | 0.5 ± 1.1 | 7.7 ± 22.0 | 0.4 ± 0.5 | 59.3 ± 102.0 | 10.8 | 0.0127 | |
| O2 saturation nadir, % | 94.7 ± 1.2 | 92.6 ± 1.5 | 95.6 ± 0.1 | 92.9 ± 1.4 | 12.4 | 0.0062 | |
| PLMS index, n/h | 2.3 ± 3.7 | 6.1 ± 4.5 | 33.9 ± 21.3 | 46.1 ± 58.1 | 24.3 | 0.0000 | 1 < 3, 1 < 4, 2 < 4 |
| PLMS, n | 15.3 ± 23.2 | 36.9 ± 35.5 | 189.5 ± 77.1 | 298.5 ± 497.5 | 17.0 | 0.0007 | 1 < 4, 2 < 4 |
| PLMS micro-arousal index, n/h | 0.3 ± 0.5 | 0.9 ± 1.0 | 0.3 ± 0.4 | 5.4 ± 3.8 | 6.6 | 0.0009 | 1 < 4, 2 < 4 |
| PLMS average interval, s | 17.4 ± 19.8 | 36.5 ± 15.7 | 35.3 ± 10.3 | 30.6 ± 7.3 | 7.6 | 0.0542 | |
| PLMW index, n/h | 1.3 ± 2.8 | 0.3 ± 0.7 | 1.1 ± 1.5 | 8.9 ± 16.1 | 4.6 | 0.1996 | |
| PLMW, n | 1.7 ± 3.6 | 0.5 ± 1.3 | 4.5 ± 6.4 | 9.0 ± 21.6 | 4.0 | 0.2583 | |
| PLMW average interval, s | 5.8 ± 14.3 | 3.4 ± 13.6 | 19.2 ± 27.2 | 13.5 ± 18.8 | 4.6 | 0.2028 | |
| PLMS series TST, % | 2.1 ± 3.3 | 4.8 ± 4.6 | 29.7 ± 12.2 | 23.9 ± 22.8 | 18.9 | 0.0003 | 1 < 3, 1 < 4, 2 < 4 |
| PLMS series N1,% | 1.9 ± 3.1 | 3.9 ± 3.7 | 16.0 ± 5.5 | 24.1 ± 20.5 | 22.2 | 0.0001 | 1 < 4, 2 < 4 |
| PLMS series N2,% | 2.7 ± 4.3 | 5.7 ± 6.4 | 35.4 ± 16.3 | 25.0 ± 26.0 | 15.2 | 0.0017 | |
| PLMS series N3,% | 3.9 ± 9.4 | 1.5 ± 5.7 | 16.6 ± 23.4 | 6.7 ± 21.3 | 5.7 | 0.1281 | |
| PLMS series REM,% | 0 | 1.7 ± 5.4 | 2.6 ± 3.6 | 9.5 ± 17.6 | 7.9 | 0.0487 |
| Total AHI, n/h | PLMS Index, n/h | PLMS, n | PLMS Micro-Arousal Index, n/h | PLMS Series TST, % | PLMS Series N1,% | |
|---|---|---|---|---|---|---|
| Cluster 1 (n = 17) | 19.05 ± 16.74 | 20.14 ± 16.00 | 113.38 ± 88.34 | 2.85 ± 3.16 | 14.43 ± 11.52 | 13.04 ± 8.28 |
| Cluster 2 (n = 16) | 16.78 ± 18.55 | 5.60 ± 5.43 | 26.44 ± 25.35 | 0.74 ± 1.23 | 3.69 ± 3.54 | 2.84 ± 3.25 |
| F(2,31)= | 0.14 | 11.90 | 14.36 | 6.27 | 12.75 | 21.20 |
| p-value | 0.715 | 0.002 | 0.001 | 0.018 | 0.001 | 0.000 |
| power | - | 91.6% | 95.6% | 67.7% | 93.2% | 99.4% |
| Leg Movements | Cluster 1 (Mean ± sd) | Cluster 2 (Mean ± sd) | Kolmogorov–Smirnov Test, p-Value |
|---|---|---|---|
| Periodicity onset-to-onset | 0.21 ± 0.09 | 0.21 ± 0.13 | p-value > 0.10 |
| Periodicity end-to-onset | 0.21 ± 0.09 | 0.21 ± 0.13 | p-value > 0.10 |
| PLMS Respiratory related index per WASM old * | 0.55 ± 0.58 | 0.12 ± 0.17 | p-value > 0.10 |
| PLMS Respiratory related index per WASM new * | 0.03 ± 0.04 | 0.01 ± 0.01 | p-value < 0.05 |
| PLMS Respiratory related index per AASM * | 0.66 ± 0.75 | 0.12 ± 0.17 | p-value < 0.025 |
| LM index per WASM old * | 1.69 ± 2.16 | 0.51 ± 0.73 | p-value > 0.10 |
| LM index per WASM new * | 4.89 ± 5.19 | 1.88 ± 2.78 | p-value > 0.10 |
| LM index per AASM * | 1.84 ± 2.29 | 0.54 ± 0.82 | p-value > 0.10 |
| LMs in N1 | 88.86 ± 76.43 | 44.75 ± 38.83 | p-value > 0.10 |
| LMs in N2 | 27.69 ± 23.96 | 12.56 ± 11.32 | p-value > 0.10 |
| LMs in N3 | 23.63 ± 25.96 | 6.28 ± 9.5 | p-value > 0.10 |
| LMs in REM | 18.94 ± 14.97 | 8.4 ± 10.94 | p-value < 0.005 |
| Final Sample (n = 33) Compared to | AHI of 17.9 ± 17.4 | PLMS Index of 13.1 ± 14.0 |
|---|---|---|
| sample excluding the subjects using CPAP (n = 28) | 18.7 ± 18.8; p-value = 0.86 | 13.1 ± 14.4; p-value = 1 |
| sample excluding subjects with CPAP and somatic conditions (n = 26) | 19.8 ± 19.0; p-value = 0.69 | 11.2 ± 12.8; p-value = 0.59 |
| sample excluding subjects with CPAP, somatic conditions and medications (n = 24) | 19.0 ± 19.7; p-value = 0.83 | 10.5 ± 12.7; p-value = 0.48 |
| sample excluding subjects on known TCA, SSRI, SARI, TeCA (n = 28) | 18.7 ± 18.4; p-value = 0.86 | 11.3 ± 12.8; p-value = 0.60 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Walters, A.S.; Brunelin, J.; Catoire, S.; Suaud-Chagny, M.-F.; Spruyt, K. A Polysomnographic and Cluster Analysis of Periodic Limb Movements in Sleep of Restless Legs Syndrome Patients with Psychiatric Conditions. Psychiatry Int. 2021, 2, 250-264. https://doi.org/10.3390/psychiatryint2030019
Walters AS, Brunelin J, Catoire S, Suaud-Chagny M-F, Spruyt K. A Polysomnographic and Cluster Analysis of Periodic Limb Movements in Sleep of Restless Legs Syndrome Patients with Psychiatric Conditions. Psychiatry International. 2021; 2(3):250-264. https://doi.org/10.3390/psychiatryint2030019
Chicago/Turabian StyleWalters, Arthur S., Jérôme Brunelin, Sébastien Catoire, Marie-Françoise Suaud-Chagny, and Karen Spruyt. 2021. "A Polysomnographic and Cluster Analysis of Periodic Limb Movements in Sleep of Restless Legs Syndrome Patients with Psychiatric Conditions" Psychiatry International 2, no. 3: 250-264. https://doi.org/10.3390/psychiatryint2030019
APA StyleWalters, A. S., Brunelin, J., Catoire, S., Suaud-Chagny, M.-F., & Spruyt, K. (2021). A Polysomnographic and Cluster Analysis of Periodic Limb Movements in Sleep of Restless Legs Syndrome Patients with Psychiatric Conditions. Psychiatry International, 2(3), 250-264. https://doi.org/10.3390/psychiatryint2030019







