In Silico Analysis of Microfluidic Systems for the Purification of Magnetoliposomes †
Abstract
:1. Introduction
2. Results and Discussion
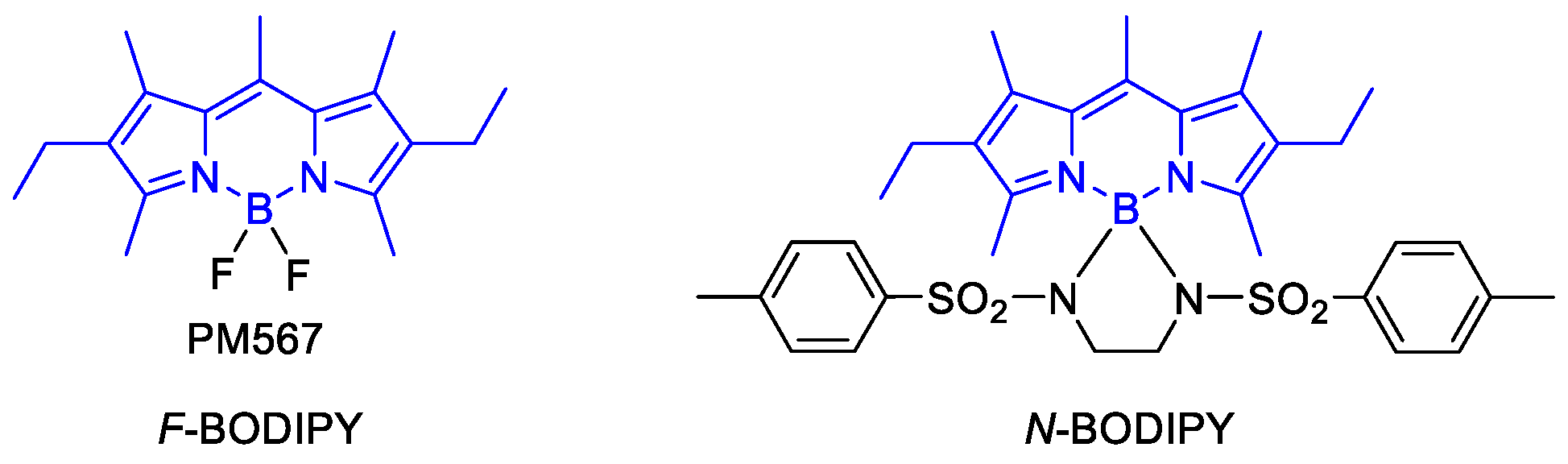
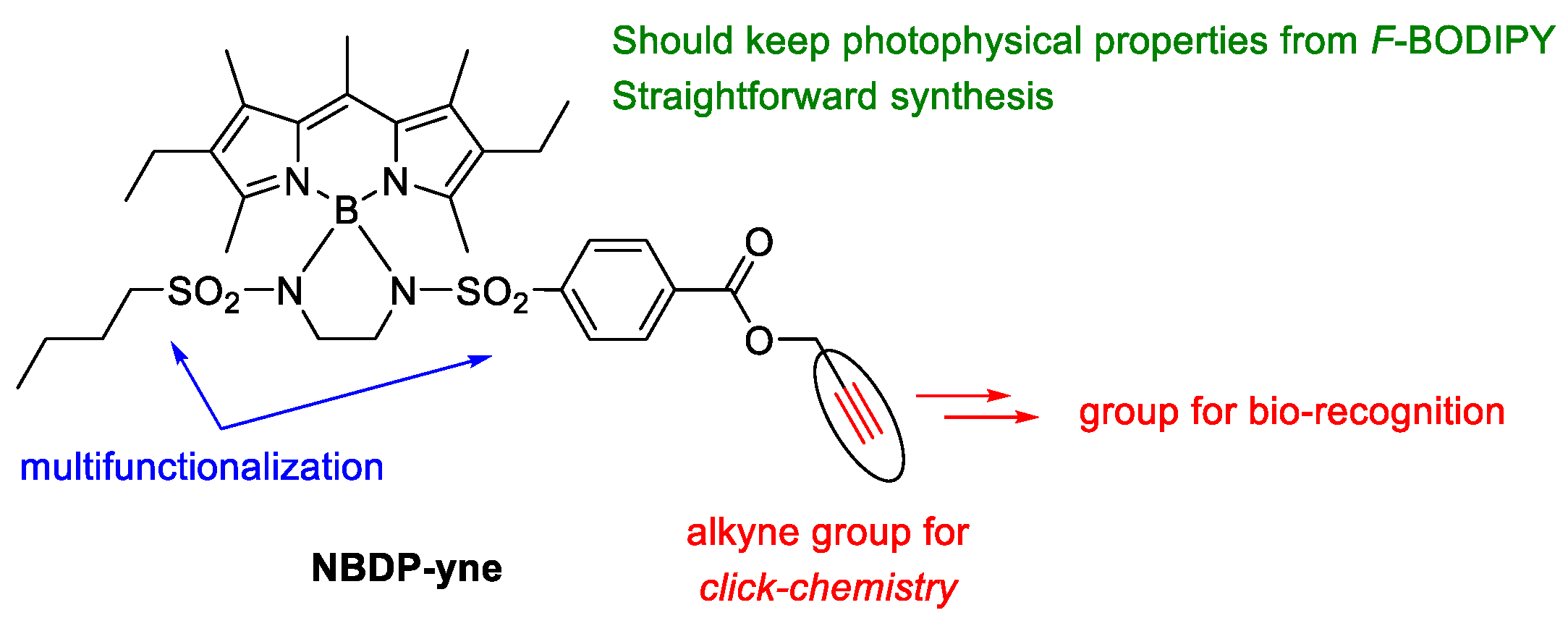
2.1. Design and Synthesis
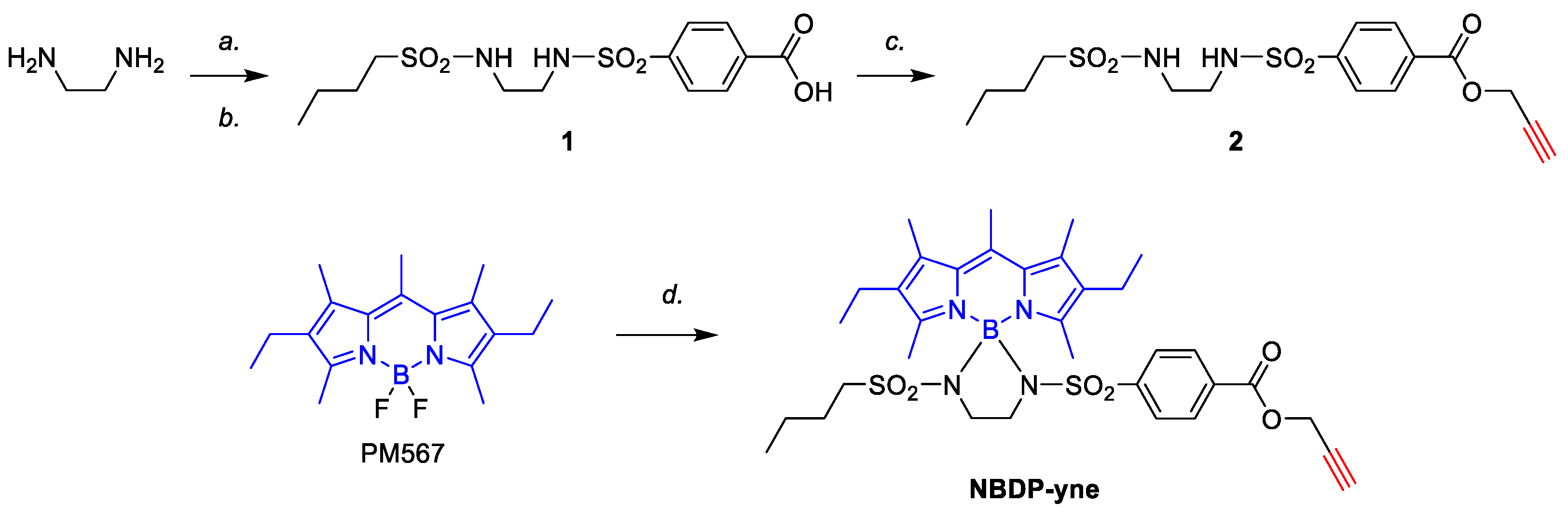
2.1.1. Synthesis of the Alkyne-Based Key Intermediate
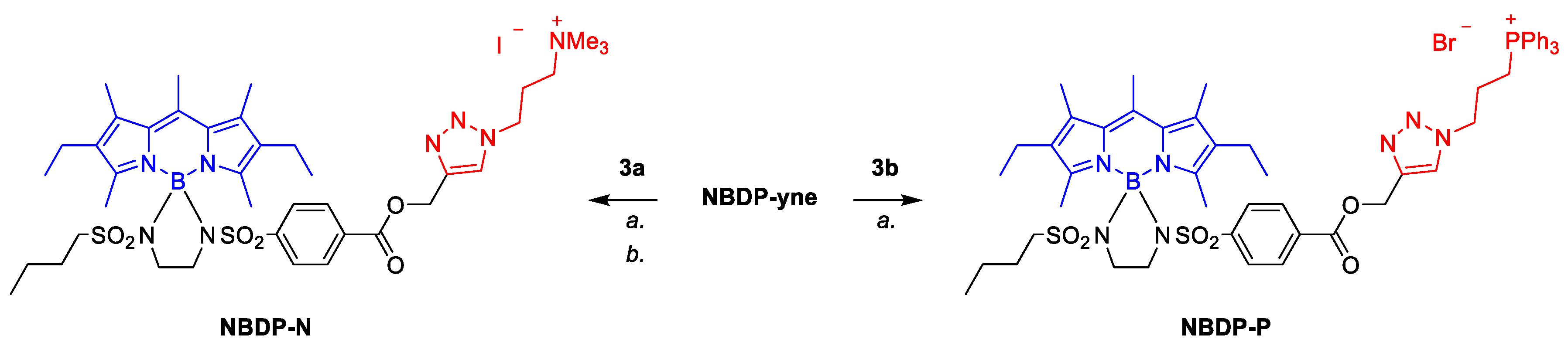
2.1.2. Derivatization of Key Intermediate NBDP-yne Towards Bio-Recognition
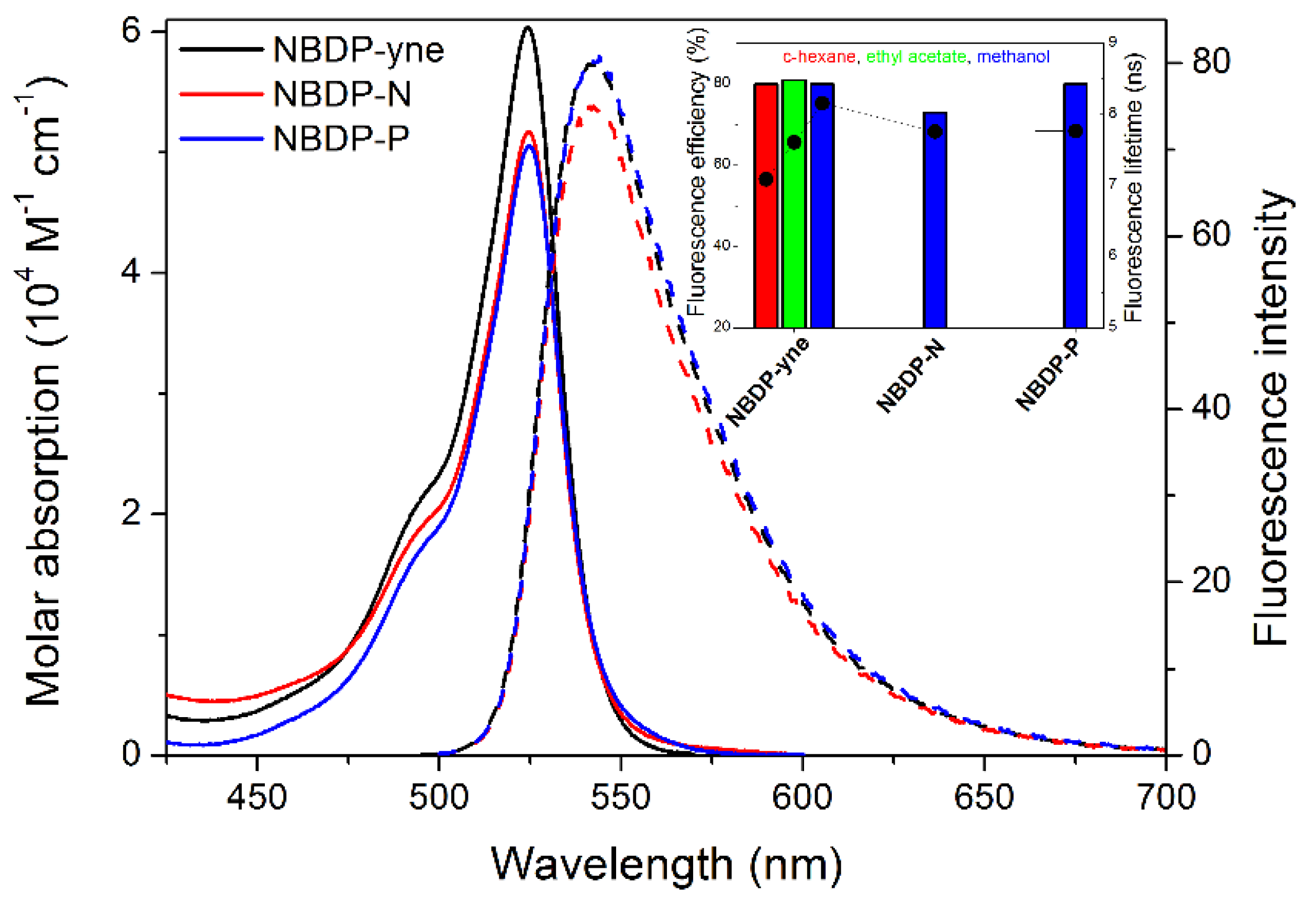
2.2. Spectroscopic Signatures in Solution
2.3. Biological Assessment in Established Pancreatic Adenocarcinoma Cells
2.3.1. Photodynamic Therapy Protocols
2.3.2. N-BODIPYs Internalization in PANC-1 Cells
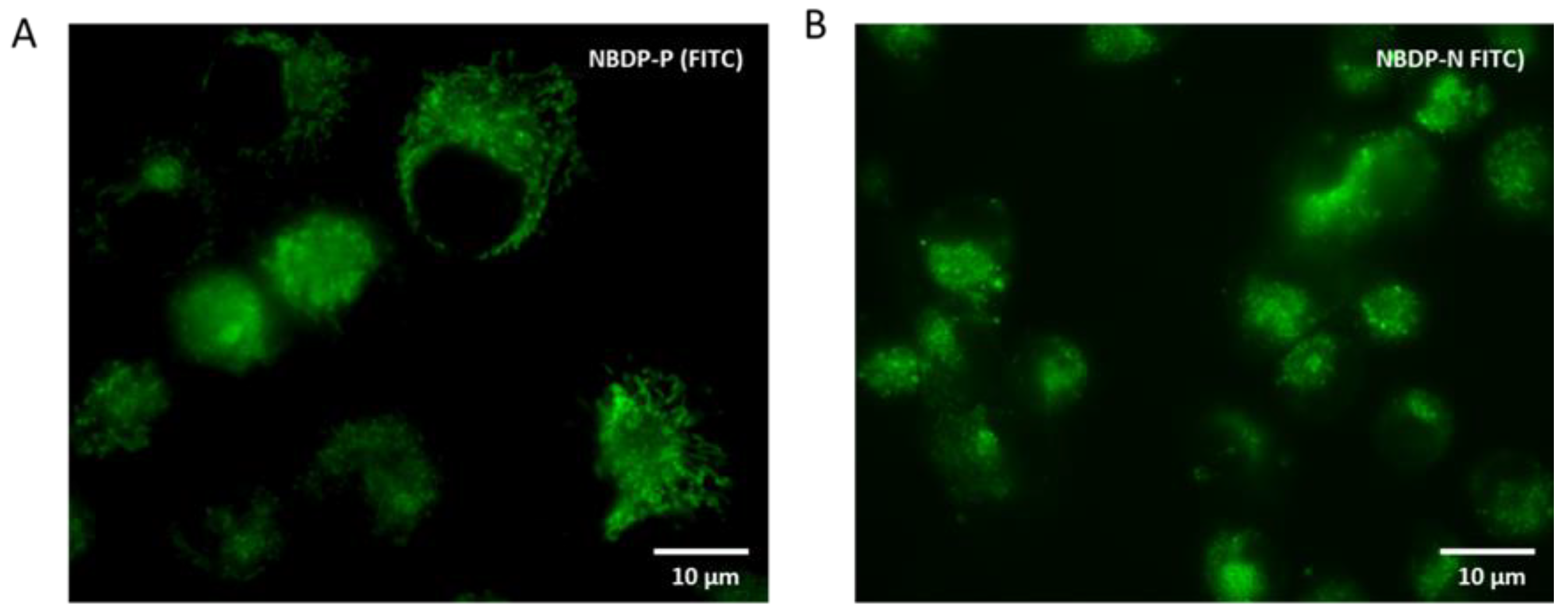
2.3.3. Subcellular Localization
3. Materials and Methods
3.1. Synthetic Procedures
3.2. Photophysical Properties
3.3. Biological Assessment
3.3.1. Cell Culture
3.3.2. Photodynamic Therapy Treatments
3.3.3. Cell Viability Studies
3.3.4. N-BODIPY Cellular Uptake by Flow Cytometry
3.3.5. Subcellular Localization and Accumulation by Fluorescence Microscopy
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Prasad, P.N. Introduction to Biophotonics; Wiley-Interscience: New York, NY, USA, 2003. [Google Scholar]
- Lovell, J.F.; Liu, T.W.B.; Chen, J.; Zheng, G. Activatable photosensitizers for imaging and therapy. Chem. Rev. 2010, 110, 2839–2857. [Google Scholar] [CrossRef] [PubMed]
- van Straten, D.; Mashayekhi, V.; de Bruijn, H.S.; Oliveira, S.; Robinson, D.J. Oncologic Photodynamic Therapy: Basic Principles, Current Clinical Status and Future Directions. Cancers 2017, 9, 19. [Google Scholar] [CrossRef] [PubMed]
- Hambling, M.R.; Huang, Y. Imaging in Photodynamic Therapy (Series in Cellular and Clinical Imaging); CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Ferreira dos Santos, A.; Quieroz de Almeida, D.R.; Ferreira Terra, L.; Baptista, M.S.; Labriola, L. Photodynamic therapy in cancer treatment—An update review. J. Cancer Metastasis Treat 2019, 5, 25. [Google Scholar] [CrossRef]
- Acedo, P.; Stockert, J.C.; Cañete, M.; Villanueva, A. Two combined photosensitizers: A goal for more effective photodynamic therapy of cancer. Cell Death Dis. 2014, 5, e1122. [Google Scholar] [CrossRef] [PubMed]
- Xiaoyuan, C.; Stephen, W. Cancer Theranostics; Elsevier: Amsterdam, The Netherlands, 2014. [Google Scholar]
- Li, B.; Zhao, M.; Zhang, F. Rational Design of Near-Infrared-II Organic Molecular Dyes for Bioimaging and Biosensing. ACS Mater. Lett. 2020, 2, 905–917. [Google Scholar] [CrossRef]
- Kowada, T.; Maeda, H.; Kikuchi, K. BODIPY-based probes for the fluorescence imaging of biomolecules in living cells. Chem. Soc. Rev. 2015, 44, 4953–4972. [Google Scholar] [CrossRef]
- Kamkaew, A.; Lim, S.H.; Lee, H.B.; Kiew, L.V.; Chung, L.Y.; Burgess, K. BODIPY dyes in photodynamic therapy. Chem. Soc. Rev. 2013, 42, 77–88. [Google Scholar] [CrossRef]
- Prieto-Montero, R.; Prieto-Castañeda, A.; Sola-Llano, R.; Agarrabeitia, A.R.; García-Fresnadillo, D.; López-Arbeloa, I.; Villanueva, A.; Ortiz, M.J.; de la Moya, S.; Martínez-Martínez, V. Exploring BODIPY Derivatives as Singlet Oxygen Photosensitizers for PDT. Photochem. Photobiol. 2020, 96, 458–477. [Google Scholar] [CrossRef]
- Bañuelos, J. BODIPY Dye, the Most Versatile Fluorophore Ever? Chem. Rec. 2016, 16, 335–348. [Google Scholar] [CrossRef]
- Loudet, A.; Burgess, K. BODIPY Dyes and Their Derivatives: Syntheses and Spectroscopic Properties. Chem. Rev. 2007, 107, 4891–4932. [Google Scholar] [CrossRef]
- Clarke, R.G.; Hall, M.J. Chapter Three—Recent developments in the synthesis of the BODIPY dyes. In Advances in Heterocyclic Chemistry; Hall, M.J., Ed.; Academic Press: Cambridge, MA, USA, 2019; Volume 128, pp. 181–261. [Google Scholar]
- Zhao, J.; Xu, K.; Yang, W.; Wang, Z.; Zhong, F. The triplet excited state of Bodipy: Formation, modulation and application. Chem. Soc. Rev. 2015, 44, 8904–8939. [Google Scholar] [CrossRef] [PubMed]
- Alamudi, S.H.; Satapathy, R.; Kim, J.; Su, D.; Ren, H.; Das, R.; Hu, L.; Alvarado-Martínez, E.; Lee, J.Y.; Hoppmann, C.; et al. Development of background-free tame fluorescent probes for intracellular live cell imaging. Nat. Commun. 2016, 7, 11964. [Google Scholar] [CrossRef] [PubMed]
- Fan, G.; Yang, L.; Chen, Z. Water-soluble BODIPY and aza-BODIPY dyes: Synthetic progress and applications. Front. Chem. Sci. Eng. 2014, 8, 405–417. [Google Scholar] [CrossRef]
- Bodio, E.; Goze, C. Investigation of B-F substitution on BODIPY and aza-BODIPY dyes: Development of B-O and B-C BODIPYs. Dyes Pigments 2019, 160, 700–710. [Google Scholar] [CrossRef]
- Ray, C.; Díaz-Casado, L.; Avellanal-Zaballa, E.; Bañuelos, J.; Cerdán, L.; García-Moreno, I.; Moreno, F.; Maroto, B.L.; López-Arbeloa, Í.; de la Moya, S. N-BODIPYs Come into Play: Smart Dyes for Photonic Materials. Chem. Eur. J. 2017, 23, 9383–9390. [Google Scholar] [CrossRef]
- Kim, E.; Koo, H. Biomedical applications of copper-free click chemistry: In vitro, in vivo, and ex vivo. Chem. Sci. 2019, 10, 7835–7851. [Google Scholar] [CrossRef]
- Gao, T.; He, H.; Huang, R.; Zheng, M.; Wang, F.-F.; Hu, Y.-J.; Jiang, F.-L.; Liu, Y. BODIPY-based fluorescent probes for mitochondria-targeted cell imaging with superior brightness, low cytotoxicity and high photostability. Dyes Pigments 2017, 141, 530–535. [Google Scholar] [CrossRef]
- Alamudi, S.H.; Chang, Y.-T. Advances in the design of cell-permeable fluorescent probes for applications in live cell imaging. Chem. Commun. 2018, 54, 13641–13653. [Google Scholar] [CrossRef]
- Lee, M.H.; Han, J.H.; Lee, J.-H.; Choi, H.G.; Kang, C.; Kim, J.S. Mitochondrial Thioredoxin-Responding Off-On Fluorescent Probe. J. Am. Chem. Soc. 2012, 134, 17314–17319. [Google Scholar] [CrossRef]
- Robinson, K.M.; Janes, M.S.; Pehar, M.; Monette, J.S.; Ross, M.F.; Hagen, T.M.; Murphy, M.P.; Beckman, J.S. Selective fluorescent imaging of superoxide in vivo using ethidium-based probes. Proc. Natl. Acad. Sci. USA 2006, 103, 15038–15043. [Google Scholar] [CrossRef]
- St John-Campbell, S.; White, A.J.P.; Bull, J.A. Single Operation Palladium Catalysed C(sp3)-H Functionalisation of Tertiary Aldehydes: Investigations into Transient Imine Directing Groups. Chem. Sci. 2017, 8, 4840–4847. [Google Scholar] [CrossRef] [PubMed]
- Dash, J.; Waller, Z.A.E.; Pantoş, G.D.; Balasubramanian, S. Synthesis and Binding Studies of Novel Diethynyl-Pyridine Amides with Genomic Promoter DNA G-Quadruplexes. Chem. Eur. J. 2011, 17, 4571–4581. [Google Scholar] [CrossRef] [PubMed]
- Knight, J.D.; Sauer, S.J.; Coltart, D.M. Asymmetric Total Synthesis of the Antimalarial Drug (+)-Mefloquine Hydrochloride via Chiral N-Amino Cyclic Carbamate Hydrazones. Org. Lett. 2011, 13, 3118–3121. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres, C.E.; Aranguren, A.; Reyes, L.H.; Osma, J.F.; Cruz, J.C. In Silico Analysis of Microfluidic Systems for the Purification of Magnetoliposomes. Mater. Proc. 2021, 4, 90. https://doi.org/10.3390/IOCN2020-07797
Torres CE, Aranguren A, Reyes LH, Osma JF, Cruz JC. In Silico Analysis of Microfluidic Systems for the Purification of Magnetoliposomes. Materials Proceedings. 2021; 4(1):90. https://doi.org/10.3390/IOCN2020-07797
Chicago/Turabian StyleTorres, Carlos E., Andres Aranguren, Luis H. Reyes, Johann F. Osma, and Juan C. Cruz. 2021. "In Silico Analysis of Microfluidic Systems for the Purification of Magnetoliposomes" Materials Proceedings 4, no. 1: 90. https://doi.org/10.3390/IOCN2020-07797
APA StyleTorres, C. E., Aranguren, A., Reyes, L. H., Osma, J. F., & Cruz, J. C. (2021). In Silico Analysis of Microfluidic Systems for the Purification of Magnetoliposomes. Materials Proceedings, 4(1), 90. https://doi.org/10.3390/IOCN2020-07797








