Evaluation of Thiosulfate for Gold Recovery from Pressure Oxidation Residues †
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Experimental Conditions
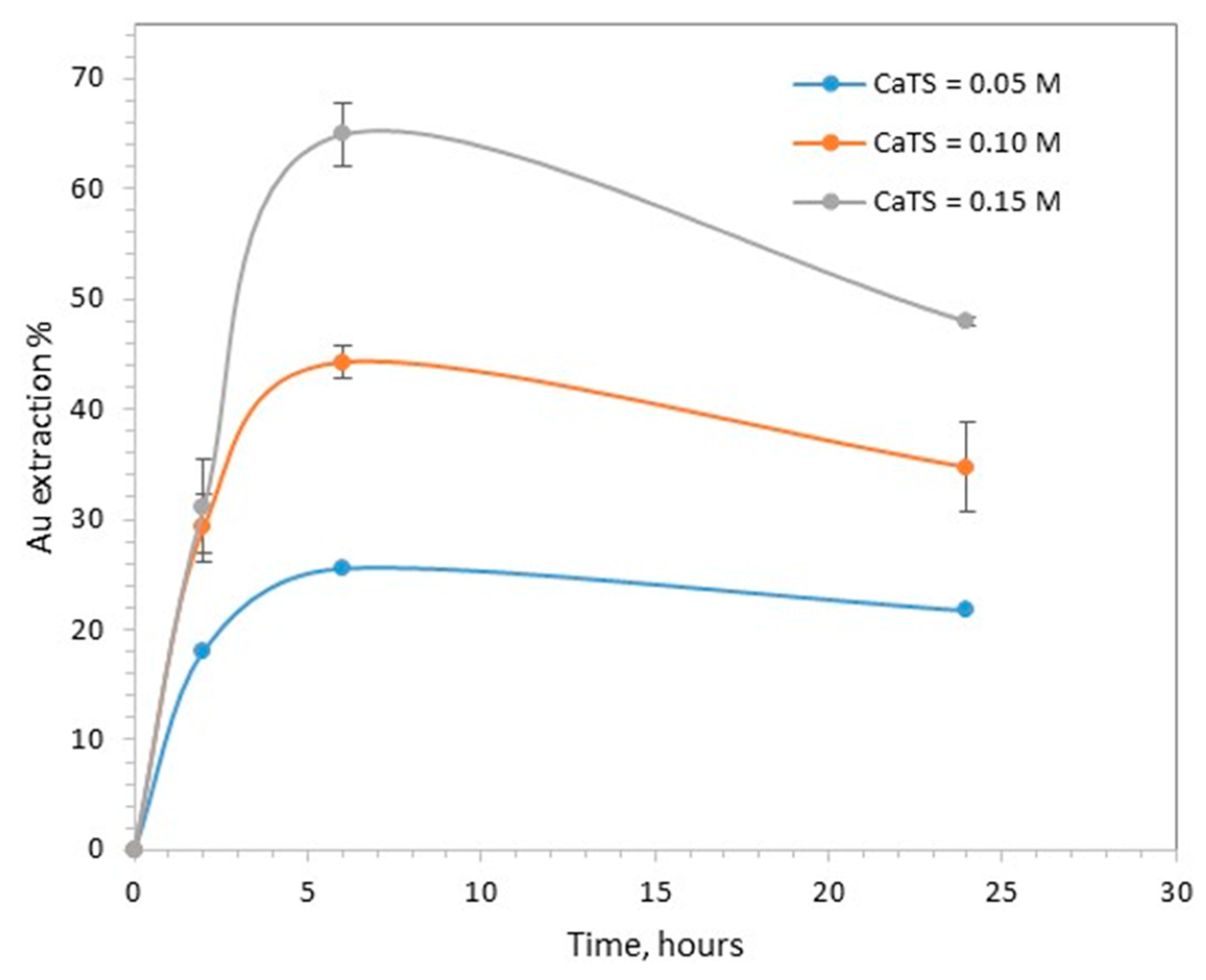
- The initial concentration of CaTS, 0.05, 0.10 and 0.15 M;
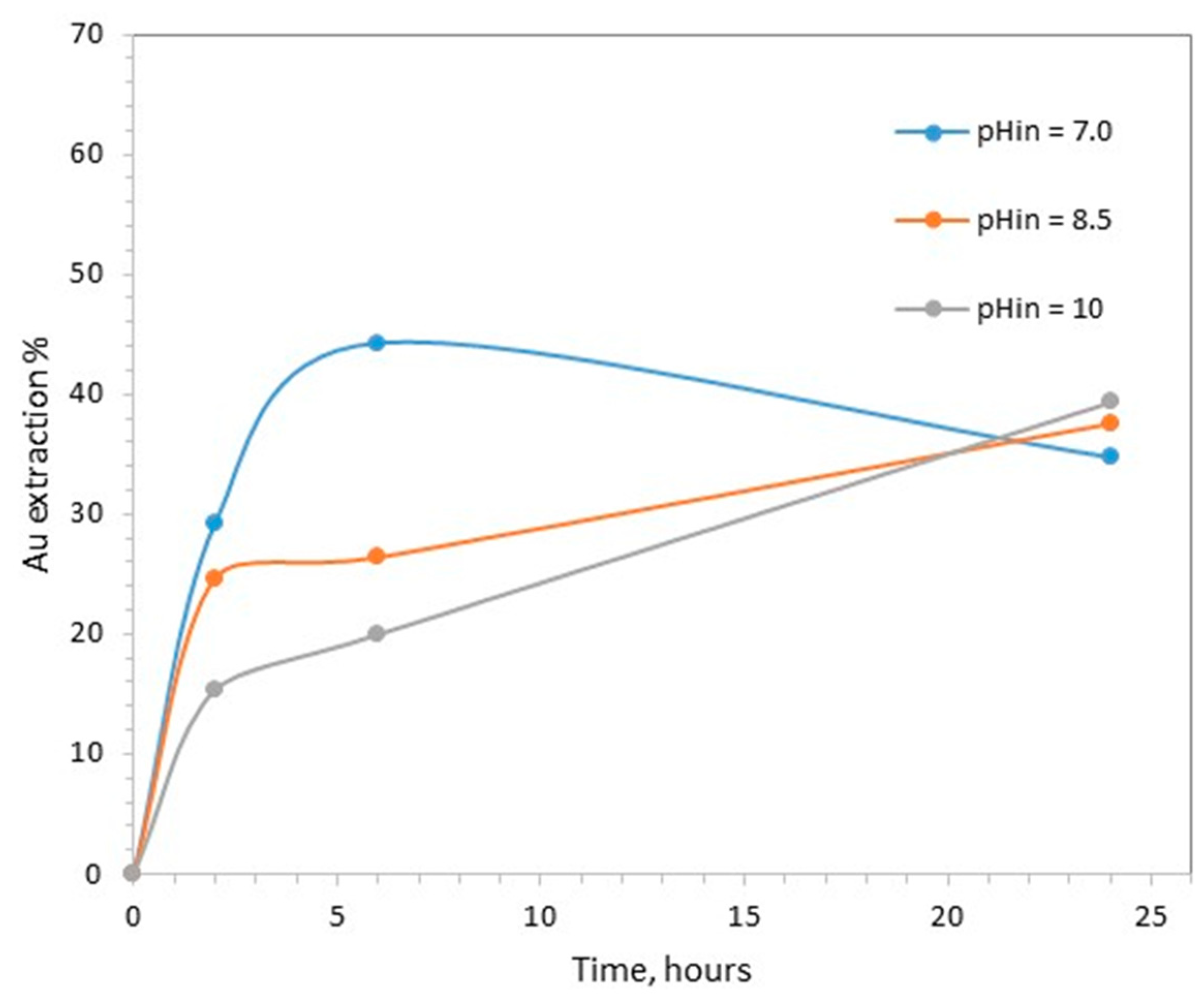
- The initial pH, 7, 8.5 and 10;
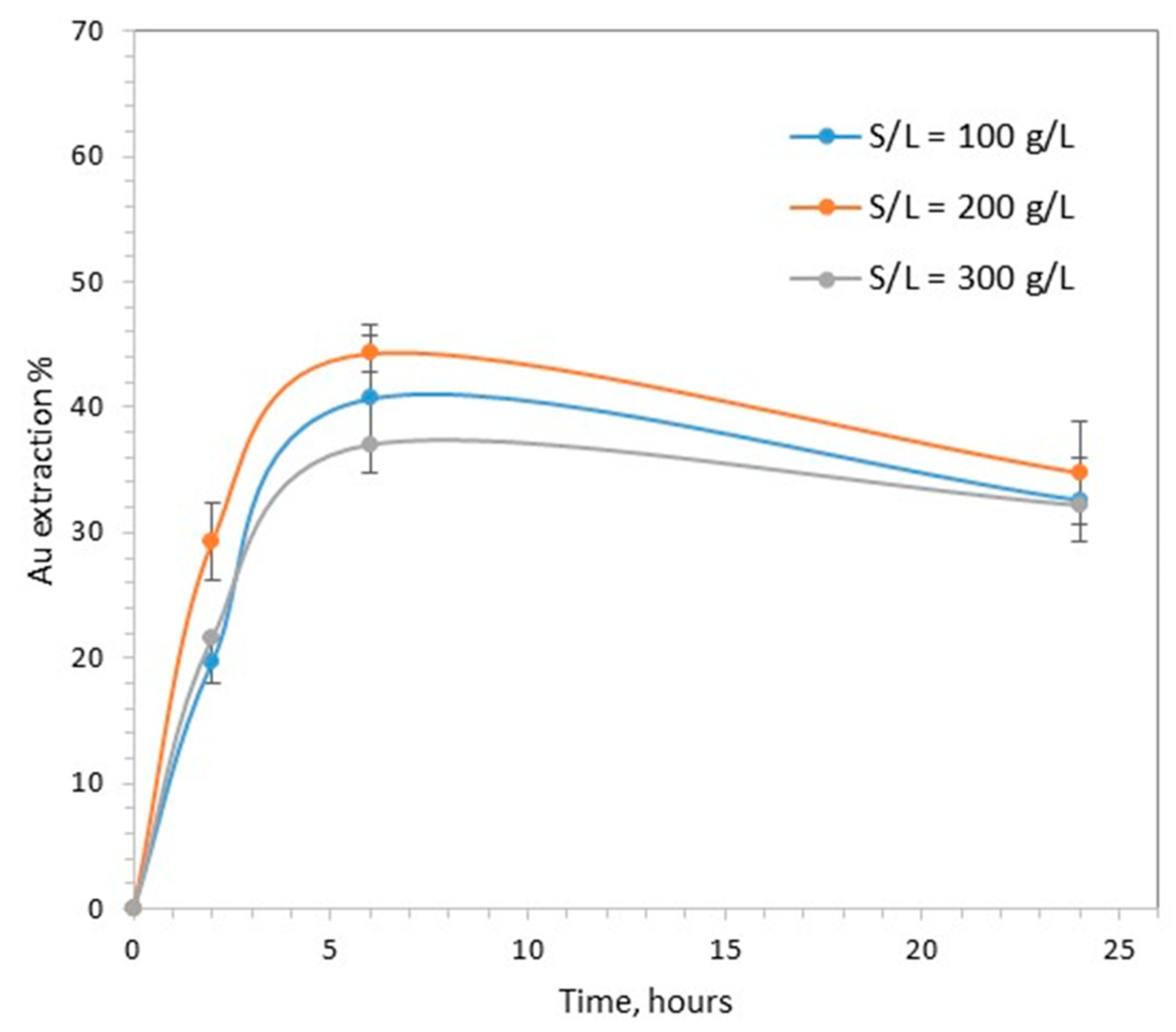
- The solid to liquid ratio, 10, 20 and 30% w/v;
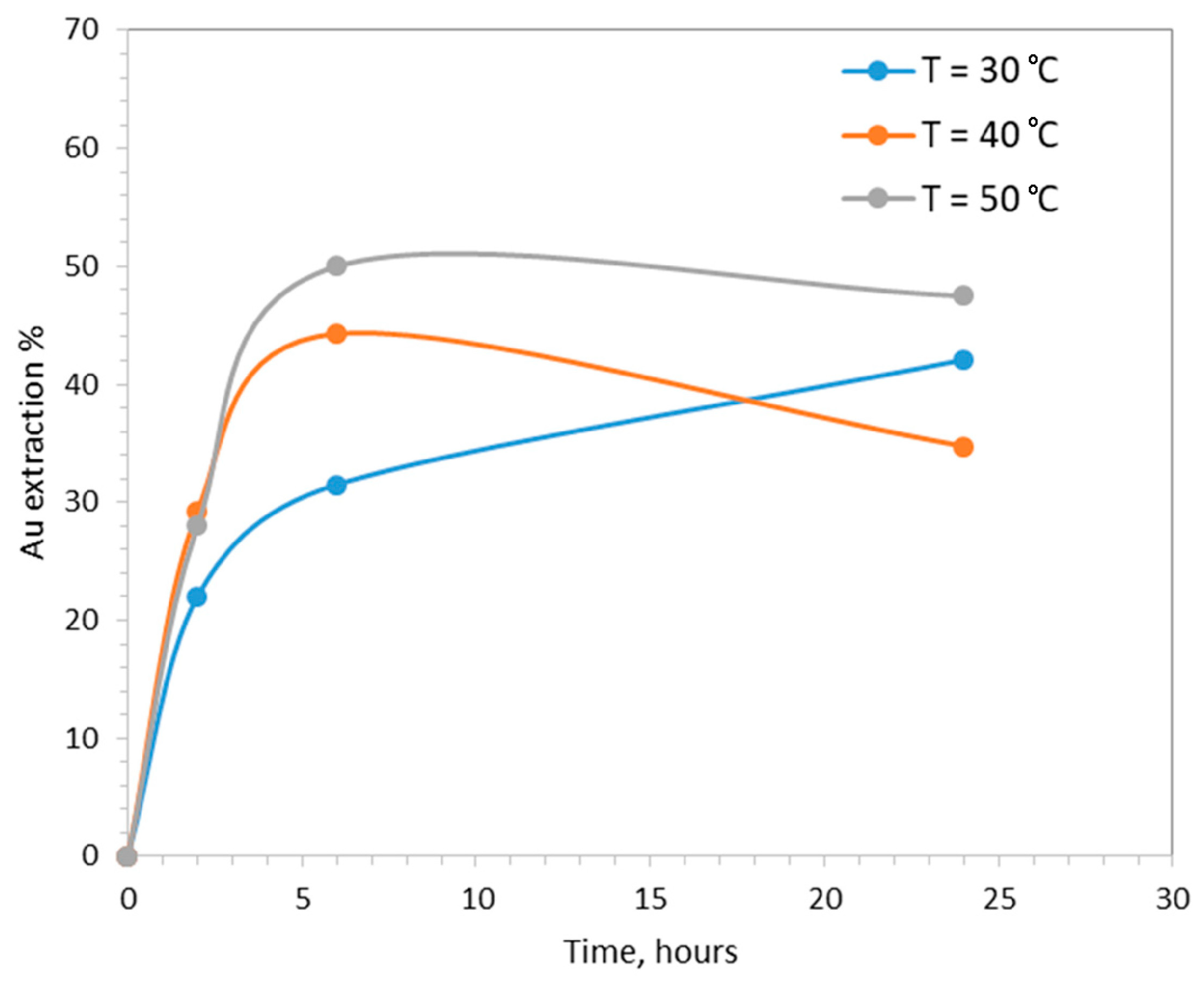
- The operating temperature, 30, 40 and 50 °C.
2.3. Experimental Procedure
2.4. Sampling and Analyses
3. Results and Discussion
3.1. Effect of Initial pH on Gold Leaching
3.2. Effect of CaTS Concentration
3.3. The Effect of Solid to Liquid Ratio
3.4. The Effect of Temperature
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aylmore, M.G. Chapter 27—Alternative Lixiviants to Cyanide for Leaching Gold Ores. In Gold Ore Processing, 2nd ed.; Adams, M.D., Ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 447–484. [Google Scholar]
- Senanayake, G. Gold leaching in non-cyanide lixiviant systems: Critical issues on fundamentals and applications. Miner. Eng. 2004, 17, 785–801. [Google Scholar] [CrossRef]
- Aylmore, M.G. Alternative lixiviants to cyanide for leaching gold ores. In Developments in Mineral Processing, 1st ed.; Adams, M.D., Wills, B.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2005; pp. 501–539. [Google Scholar]
- Zhang, Y. Current status on leaching precious metals from waste printed circuit boards. Procedia Environ. Sci. 2012, 16, 560–568. [Google Scholar] [CrossRef]
- Wan, R.Y. Importance of solution chemistry for thiosulfate leaching of gold. In Proceedings of the World Gold ’97, Singapore, 1–3 September 1997; The Society for Mining, Metallurgy and Exploration, Inc.: Littleton, CO, USA, 1997; pp. 159–162. [Google Scholar]
- Zhang, H.; Jeffrey, M. A study of pyrite catalysed oxidation of thiosulfate. In Hydrometallurgy Proceedings of the Sixth International Symposium; Society for Mining, Metallurgy, and Exploration (SME): Phoenix, AZ, USA, 2008. [Google Scholar]
- Zhang, H.; Dai, X.; Breuer, P. Factors affecting gold leaching in thiosulfate-O2 solutions. In Proceedings of the ALTA 2013, Perth, Australia, 30–31 May 2013; ALTA Metallurgical Services Publications: Perth, Australia, 2013. [Google Scholar]
- Daenzer, R.; Dreiseinger, D.; Choi, Y. Role of polythionates on the stability gold in the leaching of double refractory ores in the calcium thiosulfate-air leaching system. In ALTA 2016 Gold Pm Proceedings; ALTA Metallurgical Services Publications: Perth, Australia, 2016; pp. 282–298. [Google Scholar]
- Zhu, Y.; Liang, X.; Zhang, M. Thiosulfate leaching of gold: A review. J. Hazard. Mater. 2017, 336, 174–184. [Google Scholar]
- Yildiz, O.; Erdogan, B.M.; Ozkan, G. Thiosulfate leaching of gold from pyrite: The effect of solution pH and redox potential. Miner. Eng. 2018, 124, 145–152. [Google Scholar]
- Heidari, M.; Pourabbas, A. The role of thiosulfate oxidation on gold leaching from a refractory gold ore. Hydrometallurgy 2018, 179, 17–24. [Google Scholar]
| No | S/L, % | T, °C | pH | CaTS, M |
|---|---|---|---|---|
| 1 | 20 | 40 | 7 | 0.1 |
| 2 | 20 | 40 | 8.5 | 0.1 |
| 3 | 20 | 40 | 10 | 0.1 |
| 4 | 20 | 40 | 7 | 0.05 |
| 5 | 20 | 40 | 7 | 0.15 |
| 6 | 20 | 30 | 7 | 0.1 |
| 7 | 20 | 50 | 7 | 0.1 |
| 8 | 10 | 40 | 7 | 0.1 |
| 9 | 30 | 40 | 7 | 0.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mystrioti, C.; Kousta, K.; Papassiopi, N.; Adam, K.; Taxiarchou, M.; Paspaliaris, I. Evaluation of Thiosulfate for Gold Recovery from Pressure Oxidation Residues. Mater. Proc. 2023, 15, 87. https://doi.org/10.3390/materproc2023015087
Mystrioti C, Kousta K, Papassiopi N, Adam K, Taxiarchou M, Paspaliaris I. Evaluation of Thiosulfate for Gold Recovery from Pressure Oxidation Residues. Materials Proceedings. 2023; 15(1):87. https://doi.org/10.3390/materproc2023015087
Chicago/Turabian StyleMystrioti, Christiana, Konstantina Kousta, Nymphodora Papassiopi, Katerina Adam, Maria Taxiarchou, and Ioannis Paspaliaris. 2023. "Evaluation of Thiosulfate for Gold Recovery from Pressure Oxidation Residues" Materials Proceedings 15, no. 1: 87. https://doi.org/10.3390/materproc2023015087
APA StyleMystrioti, C., Kousta, K., Papassiopi, N., Adam, K., Taxiarchou, M., & Paspaliaris, I. (2023). Evaluation of Thiosulfate for Gold Recovery from Pressure Oxidation Residues. Materials Proceedings, 15(1), 87. https://doi.org/10.3390/materproc2023015087








