Physico-Chemical Properties of Sewage Sludge Ash and Its Influence on the Chemical Shrinkage of Cement Pastes †
Abstract
1. Introduction
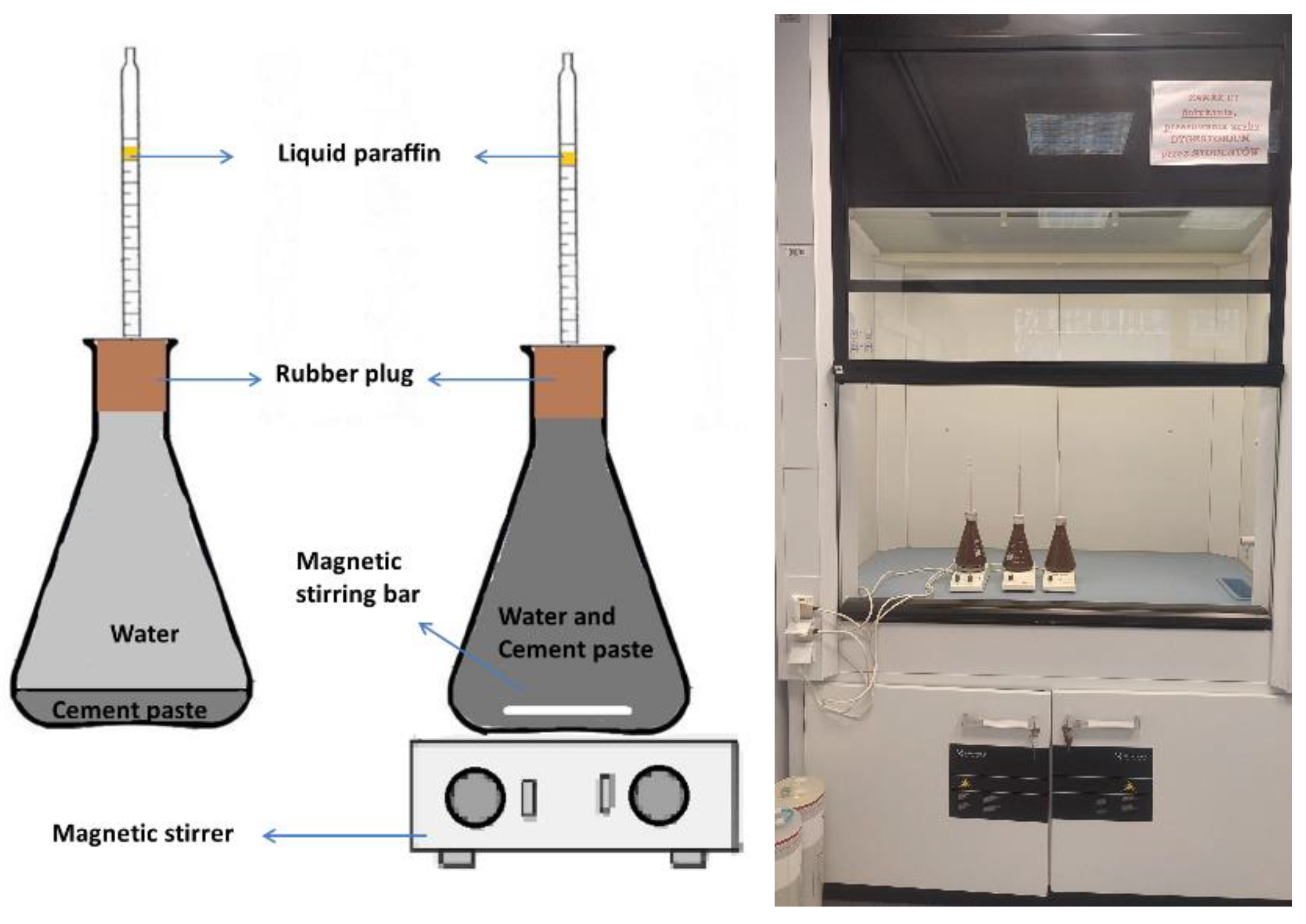
2. Materials and Methodology
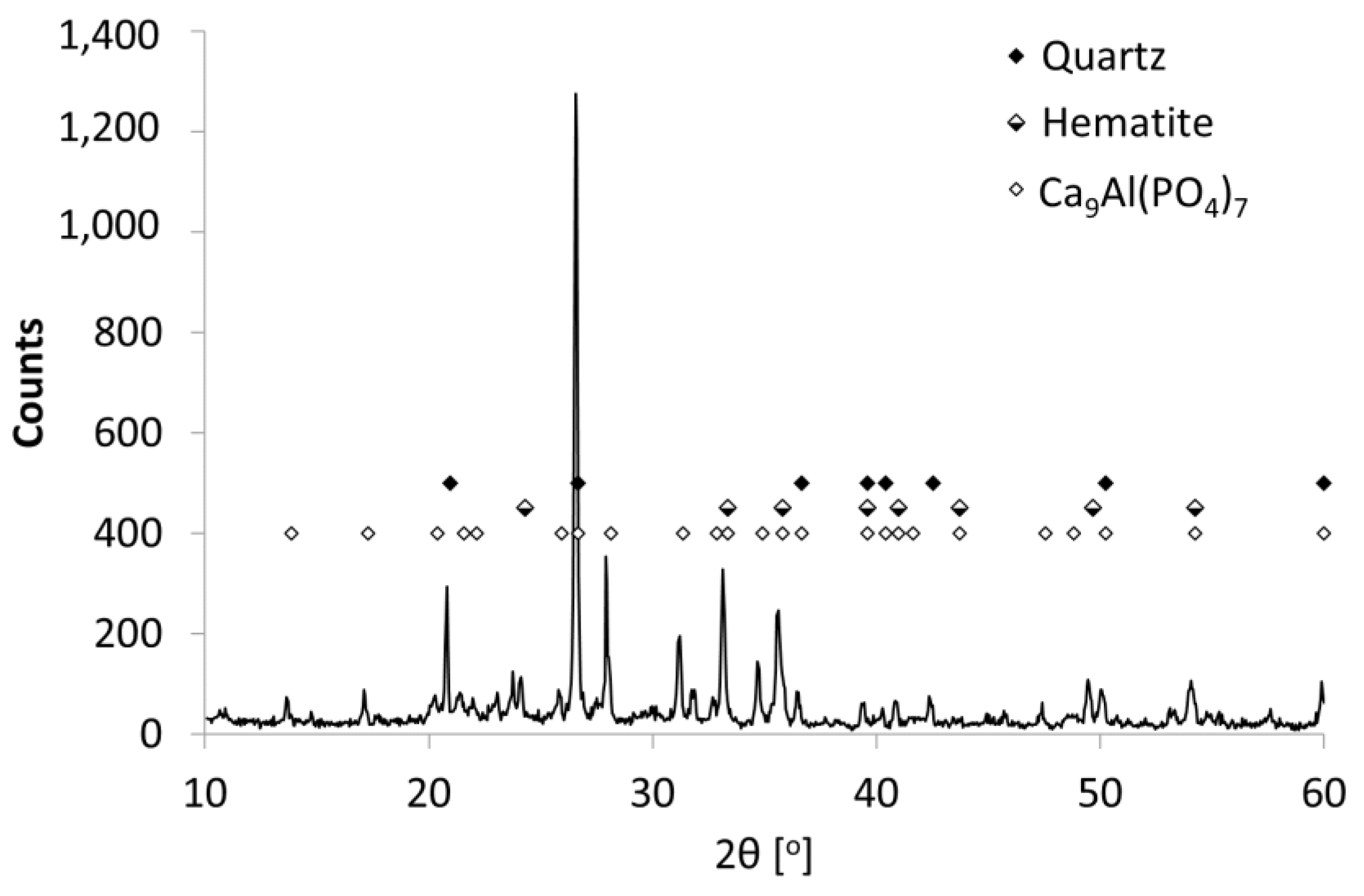
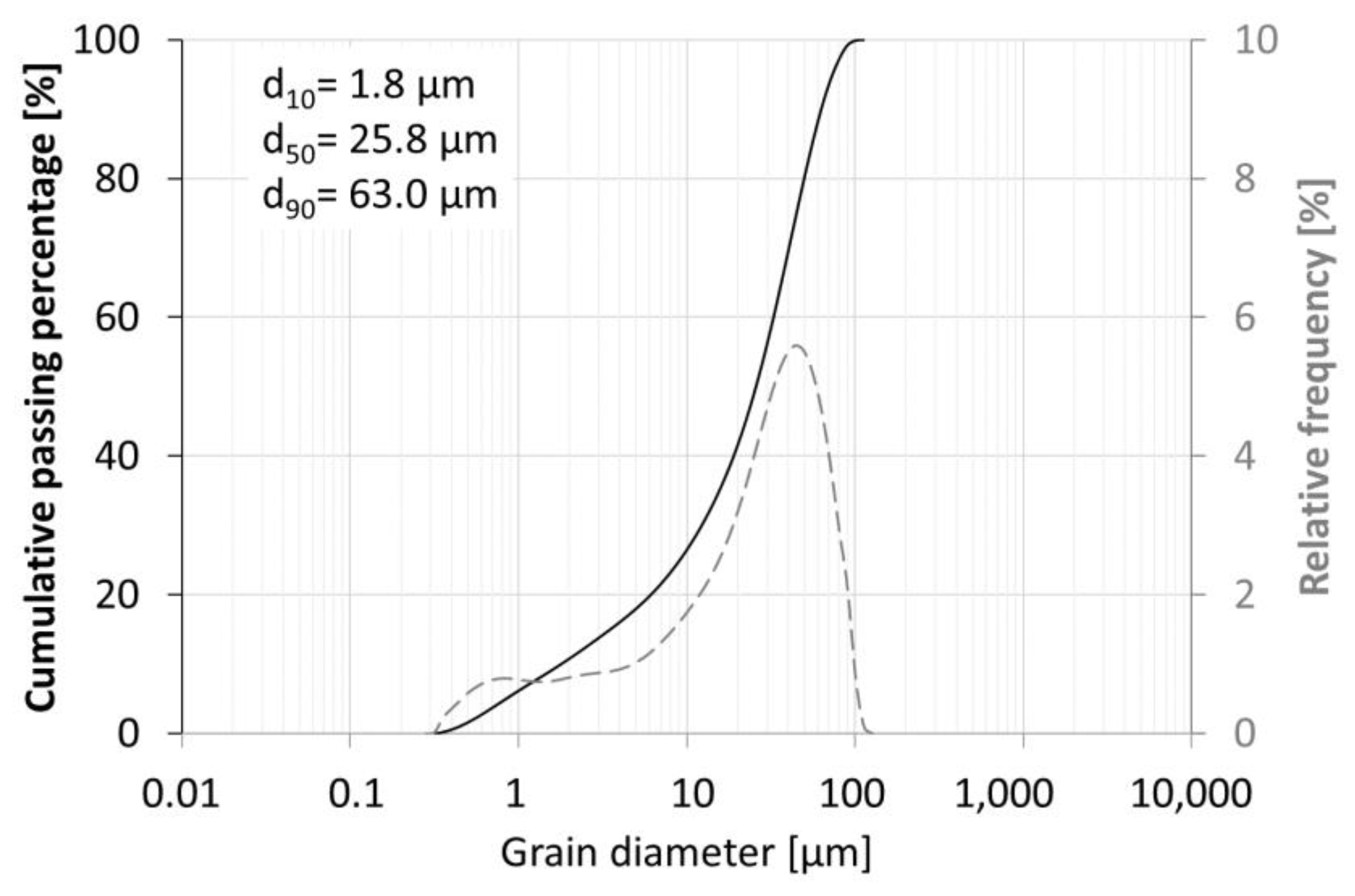
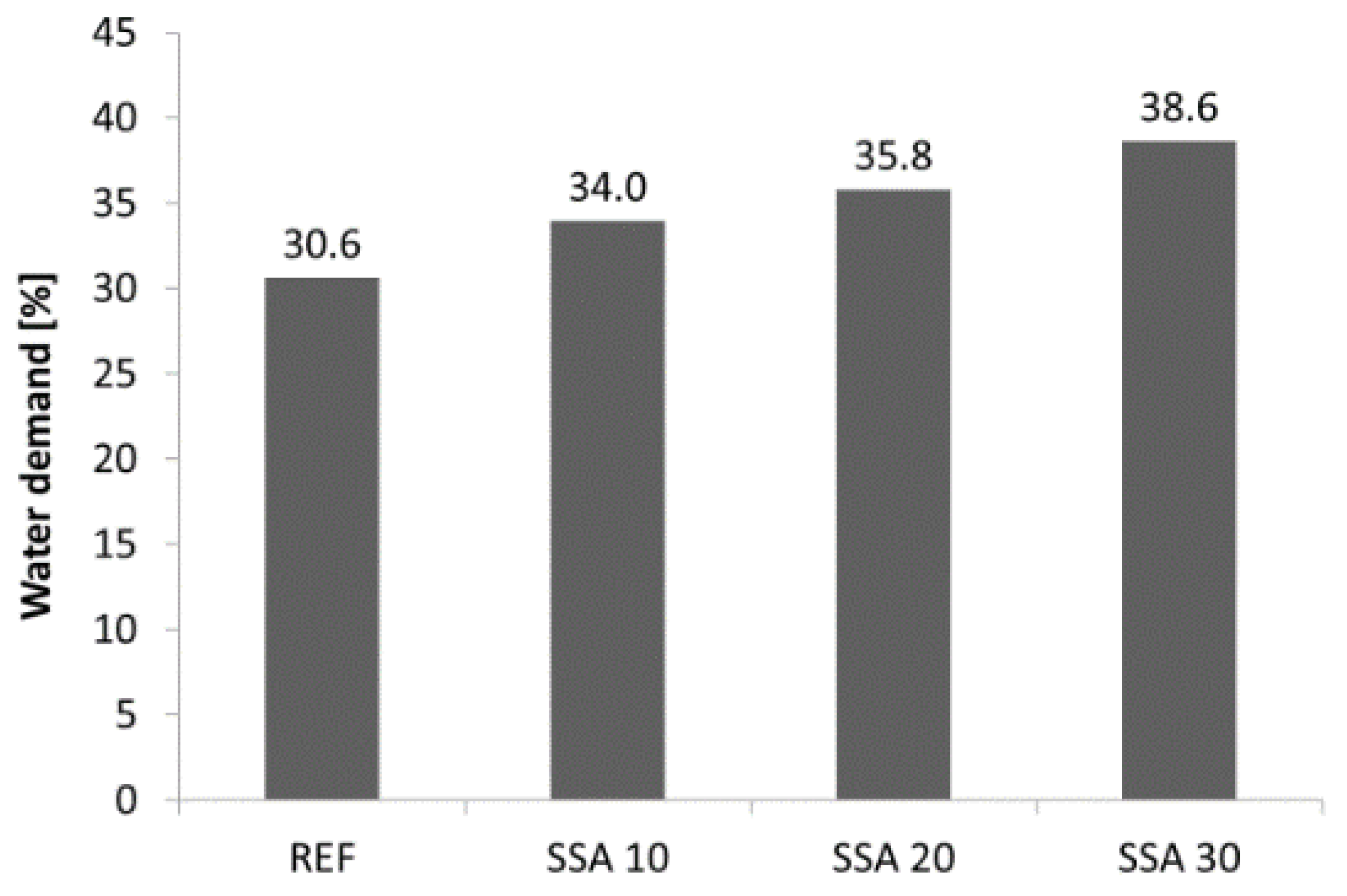
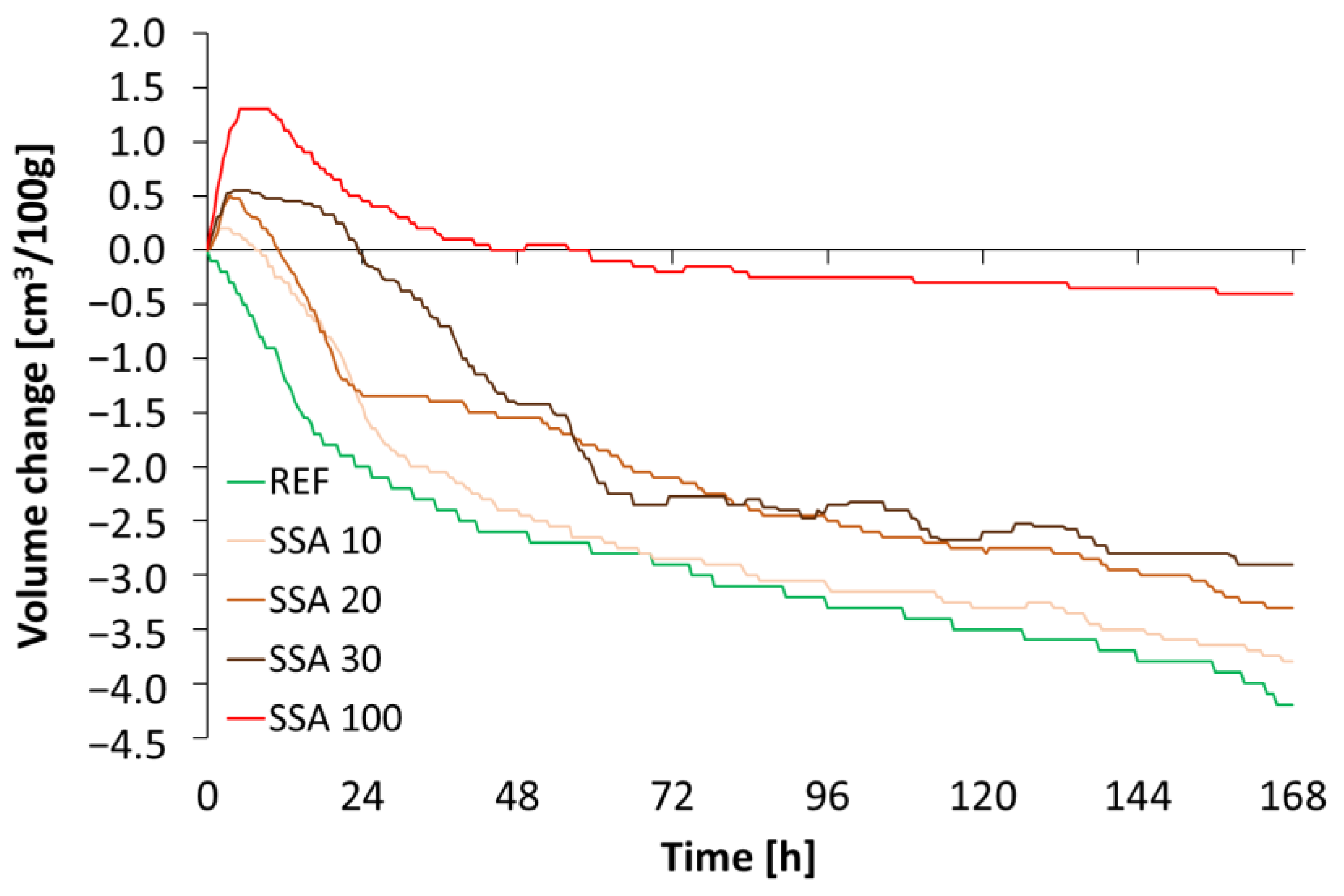
3. Research Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Niu, M.; Li, G.; Wang, Y.; Li, Q.; Han, L.; Song, Z. Comparative study of immobilization and mechanical properties of sulfoaluminate cement and ordinary Portland cement with different heavy metals. Constr. Build. Mater. 2018, 193, 332–343. [Google Scholar] [CrossRef]
- Vyšvařil, M.; Bayer, P. Immobilization of Heavy Metals in Natural Zeolite-blended Cement Pastes. Procedia Eng. 2016, 151, 162–169. [Google Scholar] [CrossRef]
- Chen, Q.Y.; Tyrer, M.; Hills, C.D.; Yang, X.M.; Carey, P. Immobilisation of heavy metal in cement-based solidification/stabilisation: A review. Waste Manag. 2009, 29, 390–403. [Google Scholar] [CrossRef] [PubMed]
- Belebchouche, C.; Moussaceb, K.; Tahakourt, A.; Aït-Mokhtar, A. Parameters controlling the release of hazardous waste (Ni2+, Pb2+ and Cr3+) solidified/stabilized by cement-CEM I. Mater. Struct. 2015, 48, 2323–2338. [Google Scholar] [CrossRef]
- Pawelska-Mazur, M.; Kaszynska, M. Mechanical Performance and Environmental Assessment of Sustainable Concrete Reinforced with Recycled End-of-Life Tyre Fibres. Materials 2021, 14, 256. [Google Scholar] [CrossRef]
- Skibicki, S.; Pułtorak, M.; Kaszyńska, M.; Hoffmann, M.; Ekiert, E.; Sibera, D. The effect of using recycled PET aggregates on mechanical and durability properties of 3D printed mortar. Constr. Build. Mater. 2022, 335, 127443. [Google Scholar] [CrossRef]
- Giergiczny, Z.; Król, A. Immobilization of heavy metals (Pb, Cu, Cr, Zn, Cd, Mn) in the mineral additions containing concrete composites. J. Hazard. Mater. 2008, 160, 247–255. [Google Scholar] [CrossRef]
- Lynn, C.J.; Dhir, R.K.; Ghataora, G.S.; West, R.P. Sewage sludge ash characteristics and potential for use in concrete. Constr. Build. Mater. 2015, 98, 767–779. [Google Scholar] [CrossRef]
- Chang, Z.; Long, G.; Zhou, J.L.; Ma, C. Valorization of sewage sludge in the fabrication of construction and building materials: A review. Resour. Conserv. Recycl. 2020, 154, 104606. [Google Scholar] [CrossRef]
- Garcés, P.; Carrión, M.P.; García-Alcocel, E.; Payá, J.; Monzó, J.; Borrachero, M.V. Mechanical and physical properties of cement blended with sewage sludge ash. Waste Manag. 2008, 28, 2495–2502. [Google Scholar] [CrossRef]
- Payá, J.; Monzó, J.; Borrachero, M.V.; Morenilla, J.J.; Bonilla, M.; Calderón, P. Some Strategies for Reusing Residues From Waste Water Treatment Plants: Preparation of Binding Materials. In International RILEM Conference on the Use of Recycled Materials in Building and Structures; RILEM Publications SARL: Champs-sur-Marne, France, 2004; pp. 814–823, Print-ISBN: 2-912143-52-7. [Google Scholar]
- Cyr, M.; Coutand, M.; Clastres, P. Technological and environmental behavior of sewage sludge ash (SSA) in cement-based materials. Cem. Concr. Res. 2007, 37, 1278–1289. [Google Scholar] [CrossRef]
- Zdeb, T.; Tracz, T.; Adamczyk, M. Physical, mechanical properties and durability of cement mortars containing fly ash from the sewage sludge incineration process. J. Clean. Prod. 2022, 345, 131055. [Google Scholar] [CrossRef]
- Rutkowska, G.; Wichowski, P.; Fronczyk, J.; Franus, M.; Chalecki, M. Use of fly ashes from municipal sewage sludge combustion in production of ash concretes. Constr. Build. Mater. 2018, 188, 874–883. [Google Scholar] [CrossRef]
- Kurdowski, W. Deformation of the Paste. In Cement and Concrete Chemistry, 1st ed.; Springer: Dordrecht, The Netherlands; Heidelberg, Germany, 2014. [Google Scholar] [CrossRef]
- Yodsudjai, W.; Wang, K. Chemical shrinkage behavior of pastes made with different types of cements. Constr. Build. Mater. 2013, 40, 854–862. [Google Scholar] [CrossRef]
- Zhang, T.; Gao, P.; Luo, R.; Guo, Y.; Wei, J.; Yu, Q. Measurement of chemical shrinkage of cement paste: Comparison study of ASTM C 1608 and an improved method. Constr. Build. Mater. 2013, 48, 662–669. [Google Scholar] [CrossRef]
- ASTM C1608-07; Standard Test Method for Chemical Shrinkage of Hydraulic Cement Paste. 2012. Available online: https://www.astm.org/c1608-07.html (accessed on 21 November 2022).
- PN EN 196-3; Metody badania cementu, Część 3. Oznaczanie czasów wiązania stałości objętości. 2006. Available online: https://sklep.pkn.pl/pn-en-196-3-2016-12p.html (accessed on 15 November 2022).
- PN EN 450-1:2012; Popiół lotny do betonu—Część 1: Definicje, specyfikacje i kryteria zgodności. 2012. Available online: https://sklep.pkn.pl/pn-en-450-1-2012p.html (accessed on 15 November 2022).
- Internet System of Legal Acts. Available online: https://isap.sejm.gov.pl/isap.nsf/DocDetails.xsp?id=wdu20130000021 (accessed on 6 November 2022).
- Tkaczewska, E. Wpływ jonów fosforanowych PO43- na proces hydratacji cementu. Cem. Wapno Bet. 2012, 17/79, 401–408. [Google Scholar]
- Cau Dit Coumes, C.; Courtois, S. Cementation of a low-level radioactive waste of complex chemistry: Investigation of the combined action of borate, chloride, sulfate and phosphate on cement hydration using response surface methodology. Cem. Concr. Res. 2003, 33, 305–316. [Google Scholar] [CrossRef]
- Bénard, P.; Garrault, S.; Nonat, A.; Cau-Dit-Coumes, C. Hydration process and rheological properties of cement pastes modified by orthophosphate addition. J. Eur. Ceram. Soc. 2005, 25, 1877–1883. [Google Scholar] [CrossRef]
- Bouasker, M.; Mounanga, P.; Turcry, P.; Loukili, A.; Khelidj, A. Chemical shrinkage of cement pastes and mortars at very early age: Effect of limestone filler and granular inclusions. Cem. Concr. Compos. 2008, 30, 13–22. [Google Scholar] [CrossRef]
- Wang, J.; Cheng, Y.; Yuan, L.; Xu, D.; Du, P.; Hou, P.; Zhou, Z.; Cheng, X.; Liu, S.; Wang, Y. Effect of nano-silica on chemical and volume shrinkage of cement-based composites. Constr. Build. Mater. 2020, 247, 118529. [Google Scholar] [CrossRef]
- Park, B.; Choi, Y.C. Effects of fineness and chemical activators on the hydration and physical properties of high-volume fly-ash cement pastes. J. Build. Eng. 2022, 51, 104274. [Google Scholar] [CrossRef]
- Gao, Y.-L.; Zhou, S.-Q. Influence of ultra-fine fly ash on hydration shrinkage of cement paste. J. Cent. South Univ. Technol. 2005, 12, 596–600. [Google Scholar] [CrossRef]
- Jang, J.-H.; Dempsey, B.A.; Burgos, W.D. Solubility of Hematite Revisited: Effects of Hydration. Environ. Sci. Technol. 2007, 41, 7303–7308. [Google Scholar] [CrossRef] [PubMed]
- Langmuir, D. Particle size effect on the reaction goethite = hematite + water; correction. Am. J. Sci. 1972, 272, 972. [Google Scholar] [CrossRef]
- Czarnecki, L.; Emmons, P.H. Mechanizmy destrukcji. In Naprawa i ochrona konstrukcji betonowych, 1st ed.; Polski Cement: Kraków, Poland, 2002; ISBN 83-91300-6-4. [Google Scholar]
- Mancini, A.; Lothenbach, B.; Geng, G.; Grolimund, D.; Sanchez, D.; Fakra, S.; Dähn, R.; Wehrli, B.; Wieland, E. Iron speciation in blast furnace slag cements. Cem. Concr. Res. 2021, 140, 106287. [Google Scholar] [CrossRef]

| Component [%] | SiO2 | Al2O3 | Fe2O3 | CaO | Na2Oe | SO3 | P2O5 | MgO | Cl− | LOI |
|---|---|---|---|---|---|---|---|---|---|---|
| SSA | 29.9 | 9.6 | 22.5 | 13.5 | 1.0 | 0.1 | 18.0 | - | 0.15 | 0.3 |
| Cement | 19.8 | 4.9 | 2.7 | 64.6 | 0.48 | 2.9 | - | 1.0 | 0.024 | 2.9 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Adamczyk, M.; Zdeb, T.; Tracz, T. Physico-Chemical Properties of Sewage Sludge Ash and Its Influence on the Chemical Shrinkage of Cement Pastes. Mater. Proc. 2023, 13, 26. https://doi.org/10.3390/materproc2023013026
Adamczyk M, Zdeb T, Tracz T. Physico-Chemical Properties of Sewage Sludge Ash and Its Influence on the Chemical Shrinkage of Cement Pastes. Materials Proceedings. 2023; 13(1):26. https://doi.org/10.3390/materproc2023013026
Chicago/Turabian StyleAdamczyk, Marcin, Tomasz Zdeb, and Tomasz Tracz. 2023. "Physico-Chemical Properties of Sewage Sludge Ash and Its Influence on the Chemical Shrinkage of Cement Pastes" Materials Proceedings 13, no. 1: 26. https://doi.org/10.3390/materproc2023013026
APA StyleAdamczyk, M., Zdeb, T., & Tracz, T. (2023). Physico-Chemical Properties of Sewage Sludge Ash and Its Influence on the Chemical Shrinkage of Cement Pastes. Materials Proceedings, 13(1), 26. https://doi.org/10.3390/materproc2023013026







