Abstract
The anti-inflammatory drug ketoprofen has shown promising results in the field of drug repurposing for the treatment of brain cancer, but currently developed formulations are for invasive administration (intravenous) and have very limited drug strength. Hence, the purpose of this work was to develop an intranasal oil-in-water nanoemulgel, with drug strength maximization, for non-invasive, more effective and safer treatment of glioma. The developed formulations were made of Capryol® 90 (hydrophobic surfactant), Tween® 80 (hydrophilic surfactant), Transcutol® (co-solvent and permeation enhancer), Pluronic® F-127 (surfactant and gelling agent) and ketoprofen. Droplet size, polydispersity index, in vitro drug release and accelerated stability were measured. The results showed that the addition of Pluronic to a preliminary optimized nanoemulsion led to a significant droplet size and PDI reduction (176 to 22 nm and 0.3 to 0.1, respectively). The achieved drug strength was 4 mg/mL, which is more than 50 times higher than ketoprofen’s aqueous solubility. The developed formulations also appeared to have high stability, with instability indexes between 0.130 and 0.265, and a high cumulative drug release percentage, varying between 78 to 93% after 24 h. The formulations also showed a controlled release profile, fitting a Korsmeyer–Peppas kinetic model, with low AIC (43.84 to 54.67) and high R2 (0.9725 to 0.9971) values, depicting non-Fickian diffusion (n between 0.7 and 0.8). Hence, high-drug-strength, high-stability and high-drug-release ketoprofen-loaded nanoemulgels were successfully prepared. Future in vitro cytotoxicity evaluations in glioma cells will assess the true potential of the developed formulations for the treatment of brain cancer.
1. Introduction
A new drug molecule takes more than 10 years to reach the pharmaceutical market and costs more than EUR 1 billion. On the other hand, drug repurposing, which is the redirecting of marketed drugs for therapeutical purposes different from the ones they were first approved for, takes about half of that time and also costs about half as much, making it a much less time-consuming and expensive strategy. Moreover, the fact that the repurposed drug molecule’s characteristics are already known (chemical properties, biodistribution and safety) can be a significant advantage in all stages of pharmaceutical product development. Due to the high potential of this approach, drug repurposing is being performed for several diseases, such as neurological diseases (Alzheimer’s disease and Parkinson’s disease); infectious diseases (bacterial, fungal or viral infections); and oncologic diseases, including colorectal, prostate, ovarian, breast, lung, pancreatic, gastric, skin and brain cancer. Specifically, for cancer treatment, drug repurposing could be the way to find less expensive treatments that also have less side effects [1,2,3,4,5].
Ketoprofen, an anti-inflammatory and analgesic drug, has shown promising results in the field of drug repurposing for the treatment of brain cancer, namely glioma [6,7]. Although the exact mechanisms by which it has an antitumor effect in brain cancer have yet to be elucidated, it is thought to exhibit its anticancer properties due to cyclooxygenase inhibition, caspase activation and cell cycle arrest, which ultimately lead to cancerous cell death [8,9,10,11,12].
Nevertheless, formulations that have already been developed are for invasive administration only (intravenous) and have very limited drug strength [6,7]. Hence, the purpose of this work was to develop an intranasal oil-in-water nanoemulgel, with drug strength maximization, for non-invasive, more effective and safer treatment of glioma.
2. Materials and Methods
Ketoprofen, Tween® 80 (polysorbate 80) and Pluronic® F-127 (poloxamer 407) were purchased from Sigma-Aldrich (Steinheim, Germany). Capryol® 90 (propylene glycol monocaprylate, type II) and Transcutol® (diethylene glycol monoethyl ether) were gift samples from Gattefossé (Lyon, France).
The preliminary oil-in-water nanoemulsions were prepared by spontaneous emulsification. The first step was adding and mixing different ratios of Capryol 90 (oil and hydrophobic surfactant), Tween 80 (hydrophilic surfactant) and Transcutol (co-solvent, co-surfactant and permeation enhancer), with ratios varying from 1 to 6 parts Capryol, 1 to 6 parts Tween and 3 to 5 parts Transcutol, to make a preconcentrate. After this, the drug was solubilized in the prepared preconcentrate, and only after was the water added and mixed, giving origin to preliminary nanoemulsions. Lead nanoemulsions were transformed into nanoemulgels by adding a Pluronic® F-127 (surfactant and gelling agent) aqueous solution to the preconcentrate, instead of water.
Both preliminary nanoemulsions and final nanoemulgels were characterized for droplet size and polydispersity index (PDI), zeta potential, in vitro drug release and accelerated stability. Droplet size and PDI were measured with dynamic light scattering, and zeta potential was measured with electrophoretic light scattering, using a Zetasizer Nano ZS apparatus (Malvern, United Kingdom).
In vitro drug release assays were performed in Franz diffusion cells (PermeGear, Pennsylvania, United States of America), using synthetic membranes (cellulose dialysis tubing membranes, Sigma-Aldrich, Steinheim, Germany), with temperature being kept at mean nasal temperature (35 °C). The receptor compartment was filled with 5 mL of nasal simulant fluid, pH 6.5 (monobasic sodium phosphate 7 mM, dibasic sodium phosphate 3 mM, potassium chloride 30 mM, sodium chloride 107 mM, calcium chloride 1.5 mM, magnesium chloride 0.75 mM and sodium hydrogen carbonate 5 mM), and 200 μL of the test formulations was placed in the donor compartment on the upper side of the membrane. The experiments were performed under constant stirring, and 300 μL samples were collected at predetermined times (5, 10, 20, 40, 60, 120, 240, 360 and 1440 min), followed by volume replacement with a new buffer solution. Drug sample quantification was performed with absorbance reading at 300 nm in 96-well polystyrene plates (flat bottom, Orange Scientific, Braine-l’Alleud, Belgium) using a microplate reader (xMark spectrophotometer, Bio-Rad, Tokyo, Japan).
Formulation accelerated stability was assessed by photocentrifugation (LUMiSizer, LUM GmbH, Berlin, Germany), with transmission profiles being at a wavelength of 865 nm (CCD-line detector), recorded in 5 s intervals for a total of 1000 profiles in capped disposable polycarbonate cells at 4000 rpm. The formulations’ instability index was determined by the equipment software (LUMiSizer SEPView®, Berlin, Germany), and an adimensional number between 0 and 1 was attributed to each preparation, with 0 meaning no formulation physical destabilization and 1 meaning complete phase separation.
3. Results and Discussion
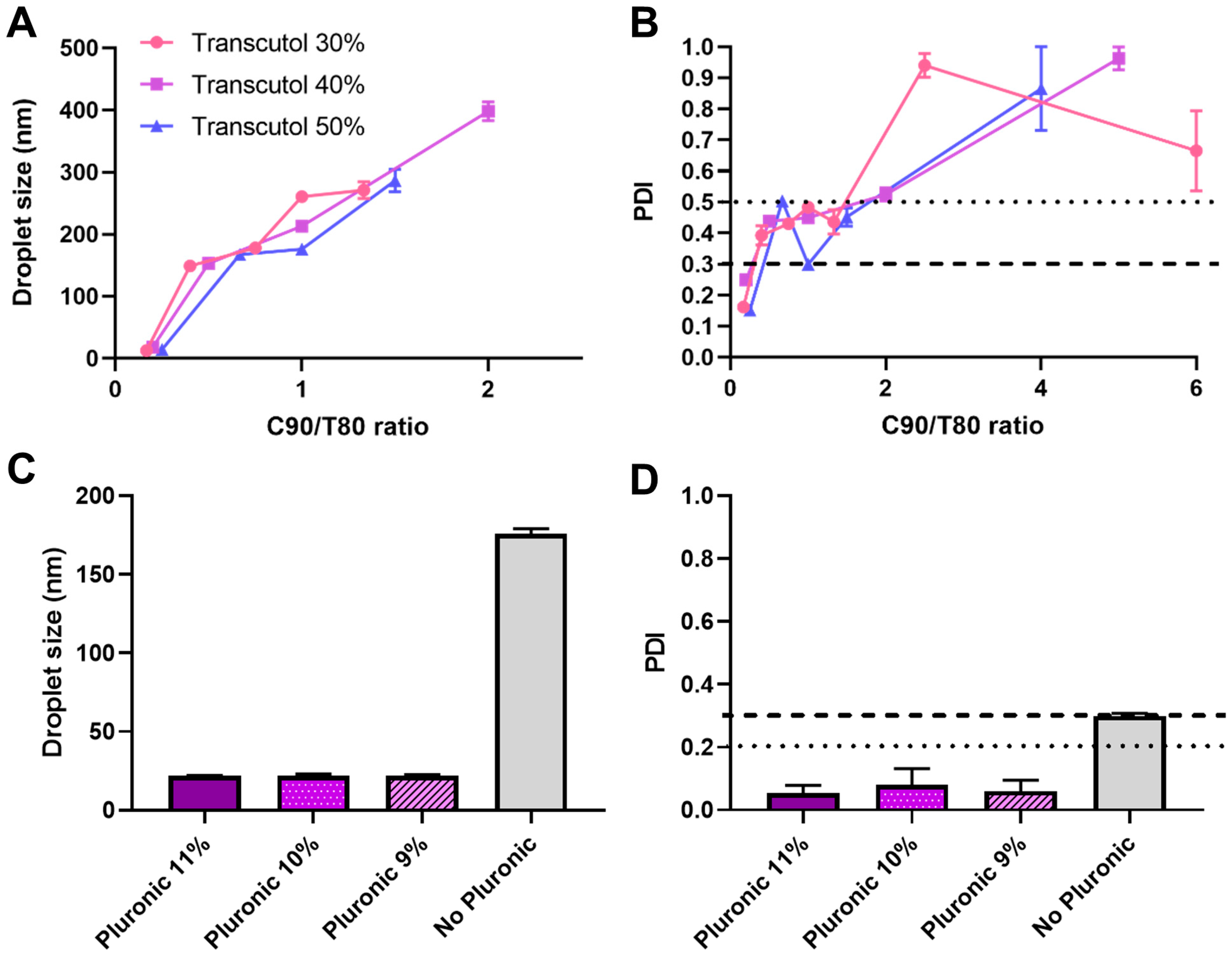
In order to maximize drug strength in the final formulation, preliminary preformulation studies focused on nanoemulsions with a 1:1 preconcentrate-to-water ratio. In these emulsions, after varying the Capryol-to-Tween ratio (and maintaining Transcutol at high amounts in the preconcentrate, namely 30, 40 and 50%, for increased drug solubilization), it was evident that a lower ratio led to smaller droplet size and lower PDI values (Figure 1A,B). These results were expected, since over the years, many studies have proven that a higher hydrophilic surfactant concentration will lead to smaller and more homogeneous nanosized droplets due to more decreased interfacial tension [13,14,15].
Figure 1.
Droplet size (A) and PDI (B) variation with different Capryol 90 (C90) to Tween 80 (T80) ratios, at 30, 40 and 50% Transcutol (percentage in emulsion preconcentrate); Effect of Pluronic addition, at different concentrations, to a preliminary nanoemulsion, in droplet size (C) and PDI (D) values.
Additionally, Pluronic addition to the external phase of a selected preliminary nanoemulsion at various concentrations led to a significant droplet size and PDI reduction (Figure 1C,D), from around 176 to 22 nm and from around 0.3 to 0.1, respectively. This was due to the fact that, aside from being a thermosensitive gelling polymer under the right circumstances, Pluronic F-127 is also a potent surfactant, which led to a further drop in interfacial tension.
Nevertheless, even when attempting to increase Pluronic F-127 concentration in the formulations up to levels where it is known to gellify around room temperature [16], we were not able to produce true nanoemulgels (fluid consistency even under heating up to 40 °C). Therefore, the amount of preconcentrate in the final formulation was decreased, and the amount of Pluronic solution was increased, until nanoemulgels that had good droplet size and PDI and were gels at mean nasal temperature (35 °C) and fluid under refrigeration (4 °C) were obtained. After several attempts, it was not possible to make formulations that were fluid under refrigeration and at room temperature (20 or 25 °C) and gels at mean nasal temperature (35 °C), but it was possible to develop nanoemulgels that were fluid under refrigeration and gels both at room temperature and mean nasal temperature. Hence, it would be possible to administer these formulations immediately after refrigeration, and when reaching the nasal cavity, they would form a gel quickly, hence decreasing mucociliary clearance and increasing nasal retention time, giving more time for the drug to be absorbed.
Three final formulations were obtained, with a preconcentrate-to-Pluronic F-127 solution proportion of 1:9 or 2:8 and with Pluronic F-127 either at 8, 10 or 11% (w/w%). Drug incorporation into these formulations did not significantly change their characteristics (droplet size and PDI), and the final achieved drug strength was 4 mg/mL, which was at least more than 50 times higher than ketoprofen’s aqueous solubility [17,18]. The zeta potential values were essentially neutral (between −10 and +10 mV) [19,20], which was also expected since all used excipients were also neutral (uncharged).
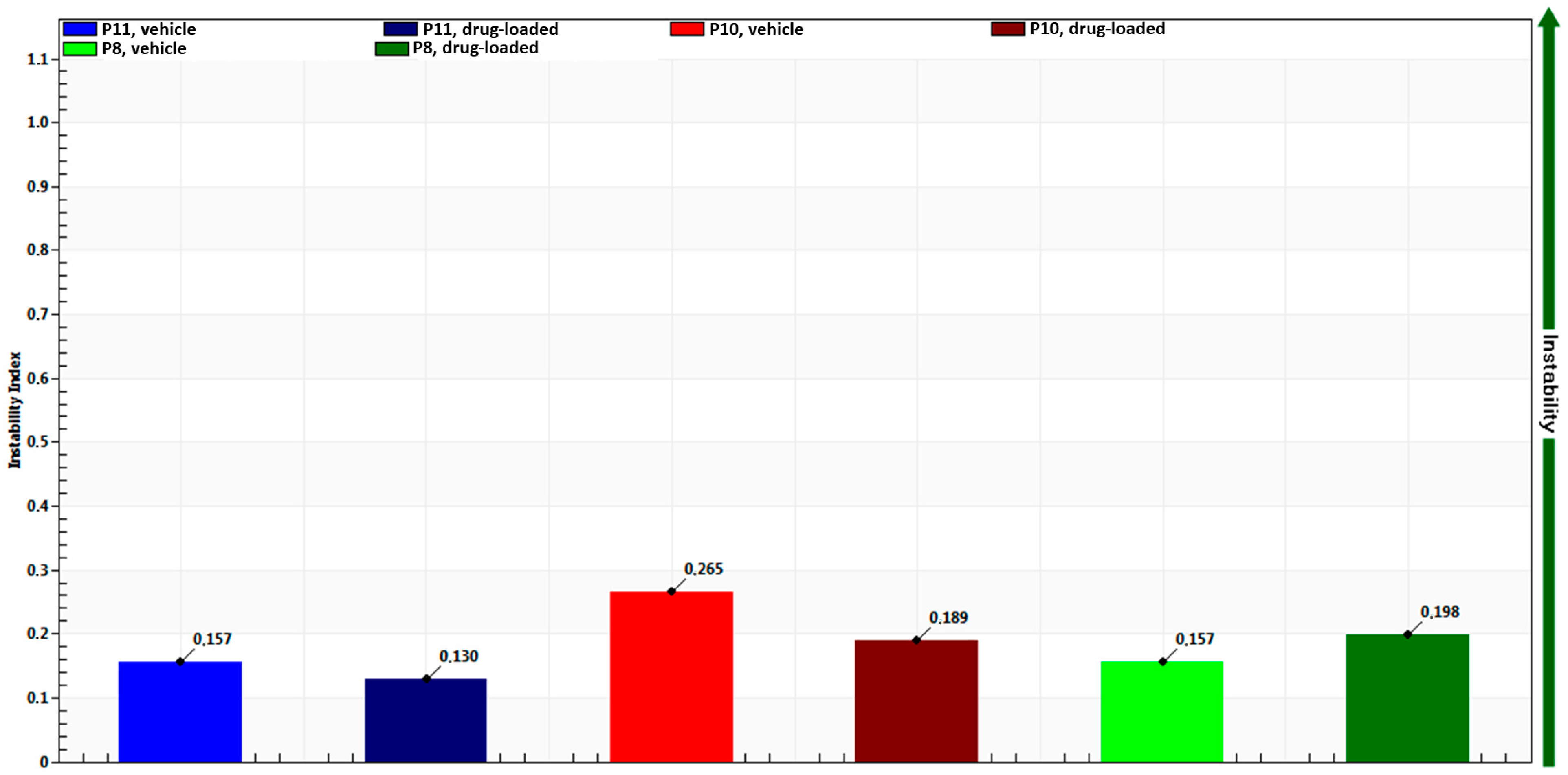
Additionally, the developed nanoemulgels appeared to have good physical stability, with instability indexes between 0.130 and 0.265 in accelerated stability studies (Figure 2). Drug incorporation into the formulations did not seem to have a significant effect on formulation stability (vehicle- vs. drug-loaded preparations).
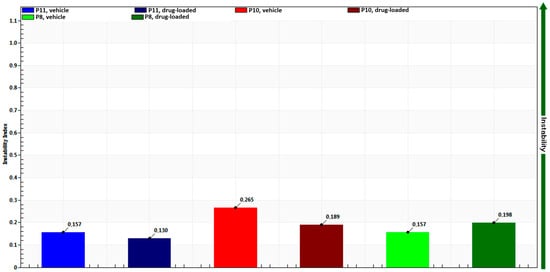
Figure 2.
Instability indexes in accelerated stability studies of nanoemulgels with 11 (P11), 10 (P10) or 8% (P8) Pluronic, encapsulating ketoprofen (drug-loaded) or not (vehicle).
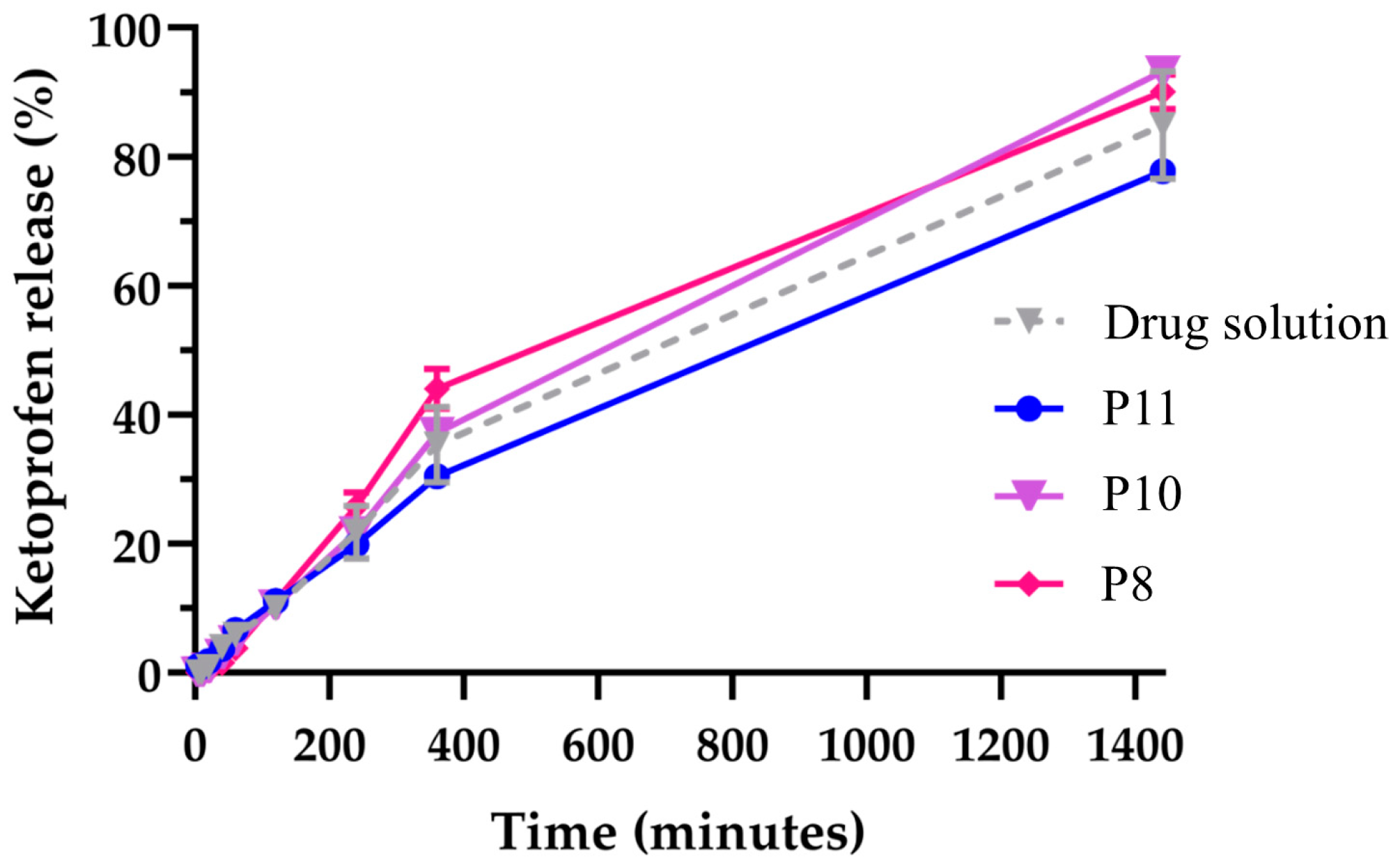
The selected formulations were evaluated for in vitro drug release, and all led to high cumulative drug release percentages after 24 h, varying between 78 and 93%, and being comparable to the positive control, a drug solution (Figure 3). The formulations also showed a controlled release profile, fitting a Korsmeyer–Peppas kinetic model, with low AIC (43.84 to 54.67) and high R2 (0.9725 to 0.9971) values, depicting non-Fickian diffusion (n between 0.7 and 0.8), which was indicative that the drug was released from the developed formulations in a combination of both diffusion from within the oil-phase droplets and slow erosion of the droplets themselves, which is in accordance with previous studies [21].
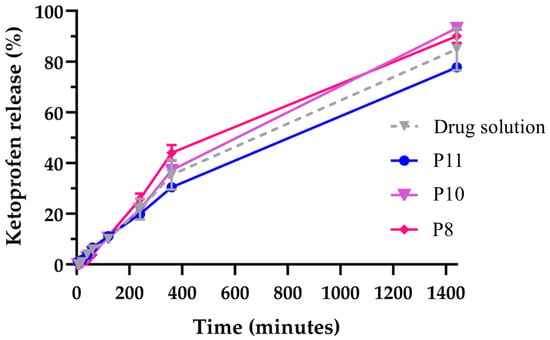
Figure 3.
In vitro drug release percentage (cumulative release) of nanoemulgels P11 (11% Pluronic), P10 (10% Pluronic) and P8 (8% Pluronic), containing Capryol 90, Tween 80, Transcutol and ketoprofen, and positive control (drug solution, with ketoprofen dissolved in Transcutol) for 24 h (1440 min).
4. Conclusions and Future Prospects
High-drug-strength, high-stability and high-drug-release ketoprofen-loaded oil-in-water nanoemulgels were successfully prepared for intranasal delivery for the treatment of glioma. Future studies will include the assessment of in vitro cytotoxicity in glioma cells to further assess the potential of these developed formulations as new technological strategies for the treatment of brain cancer.
Author Contributions
Conceptualization, P.C.P., A.C.P.-S. and F.V.; methodology, P.C.P. and A.C.P.-S.; validation, P.C.P. and A.C.P.-S.; formal analysis, P.C.P. and A.C.P.-S.; investigation, P.C.P. and M.C.; resources, A.C.P.-S. and F.V.; data curation, P.C.P. and M.C.; writing—original draft preparation, P.C.P.; writing—review and editing, P.C.P., A.C.P.-S. and F.V.; project administration, A.C.P.-S. and F.V.; funding acquisition, A.C.P.-S. and F.V. All authors have read and agreed to the published version of the manuscript.
Funding
REQUIMTE was funded by UIDB/50006/2020 and UIDP/50006/2020 national funds.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data are contained within the paper.
Acknowledgments
The authors thank Gattefossé for their kindness in sending us free samples of the required excipients.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Hua, Y.; Dai, X.; Xu, Y.; Xing, G.; Liu, H.; Lu, T.; Chen, Y.; Zhang, Y. Drug Repositioning: Progress and Challenges in Drug Discovery for Various Diseases. Eur. J. Med. Chem. 2022, 234, 114239. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.S.; Prasad, A.; Pradeep, S.; Dharmashekar, C.; Achar, R.R.; Ekaterina, S.; Victor, S.; Amachawadi, R.G.; Prasad, S.K.; Pruthvish, R.; et al. Everything Old Is New Again: Drug Repurposing Approach for Non-Small Cell Lung Cancer Targeting MAPK Signaling Pathway. Front. Oncol. 2021, 11, 741326. [Google Scholar] [CrossRef] [PubMed]
- Turabi, K.S.; Deshmukh, A.; Paul, S.; Swami, D.; Siddiqui, S.; Kumar, U.; Naikar, S.; Devarajan, S.; Basu, S.; Paul, M.K.; et al. Drug Repurposing—An Emerging Strategy in Cancer Therapeutics. Naunyn-Schmiedeberg Arch. Pharmacol. 2022, 395, 1139–1158. [Google Scholar] [CrossRef]
- Fu, L.; Jin, W.; Zhang, J.; Zhu, L.; Lu, J.; Zhen, Y.; Zhang, L.; Ouyang, L.; Liu, B.; Yu, H. Repurposing Non-Oncology Small-Molecule Drugs to Improve Cancer Therapy: Current Situation and Future Directions. Acta Pharm. Sin. B 2022, 12, 532–557. [Google Scholar] [CrossRef] [PubMed]
- Lyne, S.B.; Yamini, B. An Alternative Pipeline for Glioblastoma Therapeutics: A Systematic Review of Drug Repurposing in Glioblastoma. Cancers 2021, 13, 1953. [Google Scholar] [CrossRef]
- Ferreira, L.M.; Cervi, V.F.; Gehrcke, M.; da Silveira, E.F.; Azambuja, J.H.; Braganhol, E.; Sari, M.H.M.; Zborowski, V.A.; Nogueira, C.W.; Cruz, L. Ketoprofen-Loaded Pomegranate Seed Oil Nanoemulsion Stabilized by Pullulan: Selective Antiglioma Formulation for Intravenous Administration. Colloids Surf. B Biointerfaces 2015, 130, 272–277. [Google Scholar] [CrossRef] [PubMed]
- da Silveira, E.F.; Chassot, J.M.; Teixeira, F.C.; Azambuja, J.H.; Debom, G.; Beira, F.T.; Del Pino, F.A.B.; Lourenço, A.; Horn, A.P.; Cruz, L.; et al. Ketoprofen-Loaded Polymeric Nanocapsules Selectively Inhibit Cancer Cell Growth in Vitro and in Preclinical Model of Glioblastoma Multiforme. Investig. New Drugs 2013, 31, 1424–1435. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.; Javeed, A.; Ashraf, M.; Al-Zaubai, N.; Stewart, A.; Mukhtar, M.M. Non-Steroidal Anti-Inflammatory Drugs, Tumour Immunity and Immunotherapy. Pharmacol. Res. 2012, 66, 7–18. [Google Scholar] [CrossRef]
- Goradel, N.; Najafi, M.; Salehi, E.; Farhood, B.; Mortezaee, K. Cyclooxygenase-2 in Cancer: A Review. J. Cell. Physiol. 2019, 234, 5683–5699. [Google Scholar] [CrossRef]
- Pu, D.; Yin, L.; Huang, L.; Qin, C.; Zhou, Y.; Wu, Q.; Li, Y.; Zhou, Q.; Li, L. Cyclooxygenase-2 Inhibitor: A Potential Combination Strategy With Immunotherapy in Cancer. Front. Oncol. 2021, 11, 637504. [Google Scholar] [CrossRef] [PubMed]
- Patra, I.; Naser, R.H.; Hussam, F.; Hameed, N.M.; Kadhim, M.M.; Ahmad, I.; Awadh, S.A.; Hamad, D.A.; Parra, R.M.R.; Mustafa, Y.F. Ketoprofen Suppresses Triple Negative Breast Cancer Cell Growth by Inducing Apoptosis and Inhibiting Autophagy. Mol. Biol. Rep. 2023, 50, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, Q.; Li, L.; Chen, Y.; Cui, J.; Liu, M.; Zhang, N.; Liu, Z.; Han, J.; Wang, Z. Ketoprofen and Loxoprofen Platinum(IV) Complexes Displaying Antimetastatic Activities by Inducing DNA Damage, Inflammation Suppression, and Enhanced Immune Response. J. Med. Chem. 2021, 64, 17920–17935. [Google Scholar] [CrossRef] [PubMed]
- Sarheed, O.; Dibi, M.; Ramesh, K.V.R.N.S. Studies on the Effect of Oil and Surfactant on the Formation of Alginate-Based O/W Lidocaine Nanocarriers Using Nanoemulsion Template. Pharmaceutics 2020, 12, 1223. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Eral, H.B.; Hatton, T.A.; Doyle, P.S. Nanoemulsions: Formation, Properties and Applications. Soft Matter 2016, 12, 2826–2841. [Google Scholar] [CrossRef] [PubMed]
- Handa, M.; Ujjwal, R.R.; Vasdev, N.; Flora, S.J.S.; Shukla, R. Optimization of Surfactant- and Cosurfactant-Aided Pine Oil Nanoemulsions by Isothermal Low-Energy Methods for Anticholinesterase Activity. ACS Omega 2021, 6, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Pires, P.C.; Santos, L.T.; Rodrigues, M.; Alves, G.; Santos, A.O. Intranasal Fosphenytoin: The Promise of Phosphate Esters in Nose-to-Brain Delivery of Poorly Soluble Drugs. Int. J. Pharm. 2021, 592, 120040. [Google Scholar] [CrossRef] [PubMed]
- Ketoprofen. Available online: https://go.drugbank.com/drugs/DB01009 (accessed on 15 August 2023).
- National Center for Biotechnology Information PubChem Compound Summary for CID 3825, Ketoprofen. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/Ketoprofen (accessed on 15 August 2023).
- Barhoum, A.; García-Betancourt, M.L.; Rahier, H.; Van Assche, G. Physicochemical Characterization of Nanomaterials: Polymorph, Composition, Wettability, and Thermal Stability. In Emerging Applications of Nanoparticles and Architecture Nanostructures; Elsevier: Amsterdam, The Netherlands, 2018; pp. 255–278. [Google Scholar]
- Clogston, J.D.; Patri, A.K. Zeta Potential Measurement. In Methods in Molecular Biology; Springer: Berlin/Heidelberg, Germany, 2011; Volume 697, pp. 63–70. [Google Scholar]
- Laxmi, M.; Bhardwaj, A.; Mehta, S.; Mehta, A. Development and Characterization of Nanoemulsion as Carrier for the Enhancement of Bioavailability of Artemether. Artif. Cells Nanomed. Biotechnol. 2015, 43, 334–344. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).