Abstract
We report on the novel electrochemical lactate biosensors based on Prussian blue nanoparticles. The immobilization of lactate oxidase was performed through drop-casting on the sensor surface of a mixture containing enzyme, (3-aminopropyl)triethoxysilane and isopropyl alcohol. The apparent Michaelis constant and inactivation constant were determined (0.29 ± 0.03 mM and 0.042 ± 0.002 min−1, respectively) and compared with values obtained for biosensors based on Prussian blue films. The developed lactate biosensors are not inferior in characteristics to those previously known, while the manufacturing process is less laborious. Obtained values also indicate that lactate biosensors based on Prussian blue nanoparticles and lactate oxidase have sufficient sensitivity and operational stability for analytical application in medical and biological research.
1. Introduction
Prussian blue is a well-known compound in electrochemical catalysis. It is successfully applied in the sensing of hydrogen peroxide [1], amino acids [2] and DNA [3]. Prussian blue nanoparticles are often referred to as “artificial peroxidase” because they have better catalytical properties and operational stability than natural peroxidase enzymes [4]. Artificial peroxidase can be used in oxidase-based biosensors, which are based on oxidation of the substrate accompanied by the forming of hydrogen peroxide as a product. Then, hydrogen peroxide is reduced/oxidized on an electrode, generating an electric current. Prussian blue can catalyze the reduction of hydrogen peroxide, thus decreasing the potential of the electrochemical reaction and enhancing the selectivity of biosensors. Prussian blue was also used in the designing of the glucose biosensors suitable for non-invasive real-time glucose detection in human sweat [5].
Lactate is an important intermediate of many biochemical processes. Accumulation of lactate in the human body can be caused by cancer [6], multiple sclerosis [7] and brain injuries [8], so detection of lactate in biological fluids is useful for biological and medical purposes. Lactate oxidase catalyzes oxidation of lactate to pyruvate and forms hydrogen peroxide, which can be electrochemically detected [9]. Immobilization of the enzyme on the surface of an electrode is commonly performed using polymers with ionic-exchanging properties. (3-aminopropyl)triethoxysilane is one of the monomers used for enzyme immobilization. Polycondensation of this compound leads to the formation of a stable (3-aminopropyl)triethoxysiloxane membrane [10]. Other electrochemical biosensors can be based on different designs, such as using lactate dehydrogenase instead of lactate oxidase, immobilization of the enzyme with another polymer and application of various transducers. Some of the biosensors sensitive to lactate are listed in Table 1.

Table 1.
Examples of described biosensors for lactate detection. Lactate oxidase and lactate dehydrogenase are referred to as LOx and LDH, respectively.
The sensitivity of a biosensor depends on the overall rate of electrochemical and catalytic reactions. Hydrogen peroxide reduction and electron transfer rates are higher than those for an enzyme-catalyzed reaction, so kinetics can be described using the Michaelis–Menten equation [15]. Moreover, the possibility of a biosensor having practical use is determined by its operational stability, which is limited by inactivation of the enzyme. A high operational stability is a prerequisite for continuous real-time detection, which is needed for medical and industrial applications [16]. Inactivation is assumed to be a pseudo-first-order reaction of the enzyme in transforming from an active to an inactive form [17].
In this work the apparent Michaelis constant and inactivation constants were evaluated for lactate biosensors based on Prussian blue and Prussian blue nanoparticles covered with a stabilizing agent–nickel hexacyanoferrate.
2. Materials and Methods
K2HPO4 and KH2PO4 were purchased from Helicon (Russia). KCl and NiCl2∙6H2O were obtained from Chimmed (Moscow, Russia). FeCl3∙6H2O, (NaPO3)6 and K3[Fe(CN)6] were obtained from Sigma Aldrich (Burlington, MA, USA). The 30% H2O2 solution was purchased from Reachim (Moscow, Russia). Isopropyl alcohol was obtained from Plastopolymer (St. Petersburg, Russia). The lactate oxidase enzyme (EC 1.1.3.2) from Pediococcus species (lyophilized powder, activity 72 IU) was purchased from Sorachim (Lausanne, Switzerland). The 40% sodium L-lactic acid solution in water was obtained from Sigma Aldrich (Burlington, MA, USA).
The biosensors were produced using planar screen-printed three-electrode structures with a carbon working electrode with a diameter of 1.8 mm, carbon auxiliary electrode and silver reference electrode. These screen-printed electrodes were obtained from Rusens (Moscow, Russia).
Electrochemical measurements were conducted with potentiostat Palmsens3 (Palm Instruments BV, Houten, The Netherlands). Dynamic light scattering particle size distribution analysis was conducted with Malvern Zetasizer Nano ZS (Malvern Instruments Ltd., Malvern, UK). Spectrophotometrical measurements were conducted with Lambda 950 (Perkin-Elmer, Waltham, MA, USA).
2.1. Synthesis of Prussian Blue Nanoparticles
Synthesis was carried out as described in [4]. The reaction mixture consisted of 75 mM K3[Fe(CN)6] and 75 mM FeCl3 in a solution of 100 mM KCl and 100 mM HCl. Sedimentation was initiated with the addition of a 50 mM H2O2 solution. Mixing and dispersing of the mixture was conducted using ultrasound, followed by centrifugation and separation of the sediment. The sizes of the nanoparticles were measured using the dynamic light scattering method (refraction index value of 1.56 and absorption coefficient value of 0.9). The nanoparticles were stored in a solution of 100 mM KCl and 100 mM HCl. The concentration of the nanoparticles in a suspension was measured using spectrophotometry, with ε700 nm (per PB unit cell) = 4.85 × 104 M−1·cm−1.
2.2. Synthesis of Prussian Blue Nanoparticles with a Nickel Hexacyanoferrate Shell
Synthesis of the stabilizing shell, as described in [18], was carried out on the mixture of 5–10 nM Prussian blue nanoparticles, 0.3–2.1 mM K3[Fe(CN)6] and 0.5–3.5 mM NiCl2∙6H2O in a solution of 100 mM KCl and 100 mM HCl under ultrasonication. Sediment was separated; core–shell nanoparticles were redispersed in a solution of 7 mM (NaPO3)6, 0.5 M KCl and 0.1 M HCl. Further operations are identical to the synthesis of nanoparticles without a coating.
2.3. Preparation of Lactate Biosensors
Screen-printed electrodes were modified with Prussian blue by drop-casting 2 μL of the 6.5 nM nanoparticle suspension on a working surface. Electrodes were air-dried and annealed at a temperature of 100 °C for 1 h. The nanoparticle layer was activated via cyclic voltammetry in a potential range of −0.05 V–0.35 V vs. Ag/AgCl. 2 μL of 1.0 mg·mL−1 lactate oxidase, and a 1.5% vol. (3-aminopropyl)triethoxysilane solution in isopropyl alcohol was dipped on the working surface of the modified electrodes. After this procedure, the biosensors were air-dried and kept at 4 °C in sealed packaging.
2.4. Electrochemical Detection of Lactate
All electrochemical experiments were conducted in the chronoamperometry mode with stirring in a beaker at 25 ± 2 °C. Biosensors were tested in a 0.05 M phosphate buffer and 0.1 M KCl solution at potential 0.0 V vs. Ag/AgCl.
3. Results and Discussion
3.1. Preparation of Lactate Biosensors
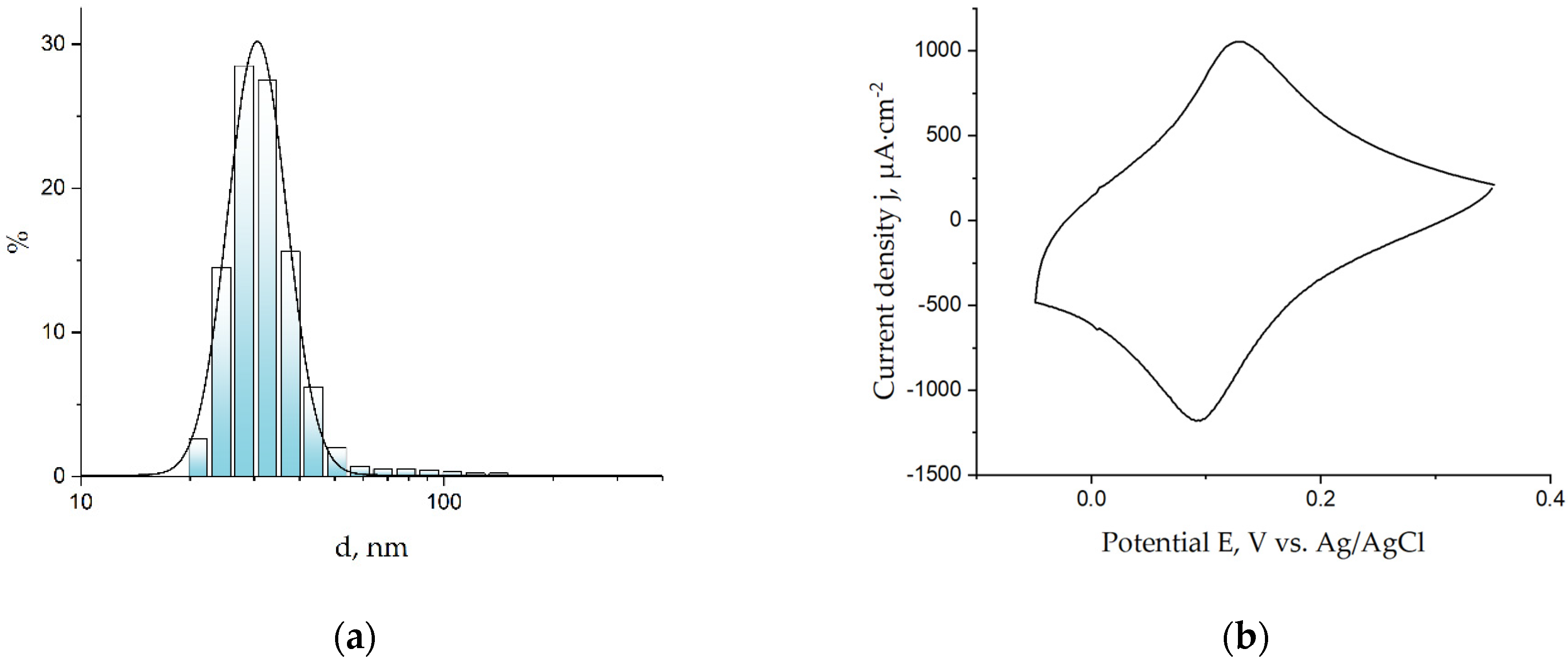
Prussian Blue nanoparticles were prepared via the reduction of the K3[Fe(CN)6] and FeCl3 mixture with hydrogen peroxide under ultrasonication. The size distribution of the nanoparticles was measured with dynamic light scattering. For further steps, nanoparticles with an average diameter of 35 nm (Figure 1a) were used, because this size is within the range in which the electrocatalyst is distributed over the entire surface of the working electrode, according to [19]. Suspension of the nanoparticles was dropped on the surface of the screen-printed electrode, then cyclic voltammogram was registered to activate the coating. A cyclic voltammogram (Figure 1b) of the electrode modified with nanoparticles exhibits a pair of peaks that correspond to the transition from Prussian blue to its reduced form, Prussian White, and vice versa. According to [1], it is needed for the activation of an electroactive coating through the intercalation of K+ cations in the Prussian blue layer. The voltammogram can also be used for a calculation of the Prussian blue concentration on the surface of the biosensor and, consequently, for control of the amount of Prussian blue deposited on the working electrode. According to the voltammogram, the average amount of deposited Prussian blue was 14 nmol·cm−2.
Figure 1.
(a) Distribution of diameters of Prussian blue nanoparticles in semilogarithmic coordinates; (b) cyclic voltammogram of Prussian blue nanoparticle-modified sensor in the 0.1 M KCl and 0.1 M HCl solution, with 40 mV/s.
Immobilization of lactate oxidase was conducted through the depositing of the mixture containing enzyme, (3-aminopropyl)triethoxysilane and isopropyl alcohol on the sensor surface. Thus, after formation of (3-aminopropyl)triethoxysiloxane, the membrane biosensors were ready for the electrochemical detection of lactate in the chronoamperometric mode.
3.2. Determination of Apparent Michaelis Constant
The apparent Michaelis constant is an important parameter of biosensors, making it possible to compare the catalytical properties of an immobilized enzyme and, consequently, the analytical characteristics of biosensors. Its value depends on the Michaelis constant of the used enzyme, the accessibility of the enzyme in the membrane and the effects of the transducer. Thus, the calculation of the apparent Michaelis constant is useful for the development of biosensors of a certain design.
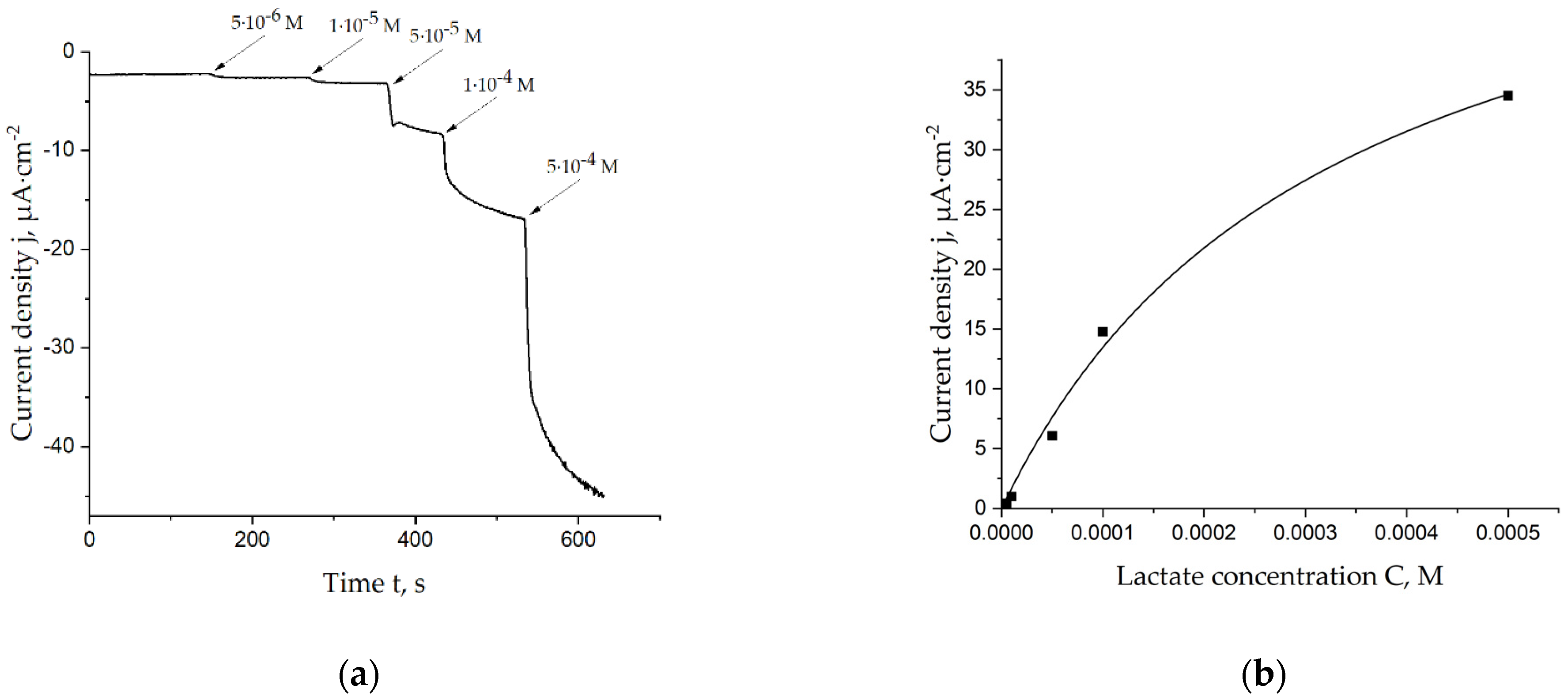
Different concentrations of lactate were added to the cell with the lactate biosensor in a batch mode. Figure 2a demonstrates one of the chronoamperograms which was registered during this procedure. Obtained data were used to plot Michaelis–Menten curves, which were used for the calculation of the apparent Michaelis constant. The corresponding curve is shown in Figure 2b. As seen, the linear range for lactate detection can be estimated to be 5 × 10−6–1 × 10−4 M; sensitivity in this range is 134 ± 12 mA·cm−2·M−1.
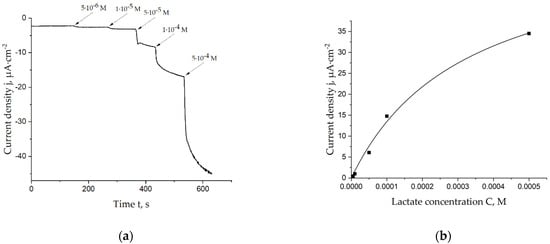
Figure 2.
(a) Chronoamperogram registered on the lactate biosensor during additions of lactate in a 0.05 M PBS, 0.1 M KCl solution, pH = 6.0, E = 0.0 V vs. Ag/AgCl; (b) Calibration curve of the lactate biosensor, which was used for calculation of the apparent Michaelis constant.
The mean apparent Michaelis constant is 0.29 ± 0.03 mM. It is equal to the constant of the Prussian blue film-based biosensors and less than the constants of previously known lactate biosensors, which are presented in Table 1. However, this parameter can be optimized by testing other membranes, buffer systems and shapes of Prussian blue nanoparticles.
Thus, manufactured biosensors are not inferior in terms of sensitivity to other lactate biosensors, including Prussian blue film-based ones. Similar values of the apparent Michaelis constant are significant for the simplified approaches to the development of the Prussian blue-based biosensors. Mass production of Prussian blue biosensors is easier with the use of nanoparticles because of the opportunity to prepare large amounts of nanoparticle suspension and apply them using automatic methods.
3.3. Determination of Inactivation Constant
The inactivation constant is important for practical use of the biosensors in online chemical analysis. Calculation of this parameter makes it possible to determine if the biosensor has sufficient operational stability for continuous detection of the chemical substance, or if further improvement of the sensor design is required.
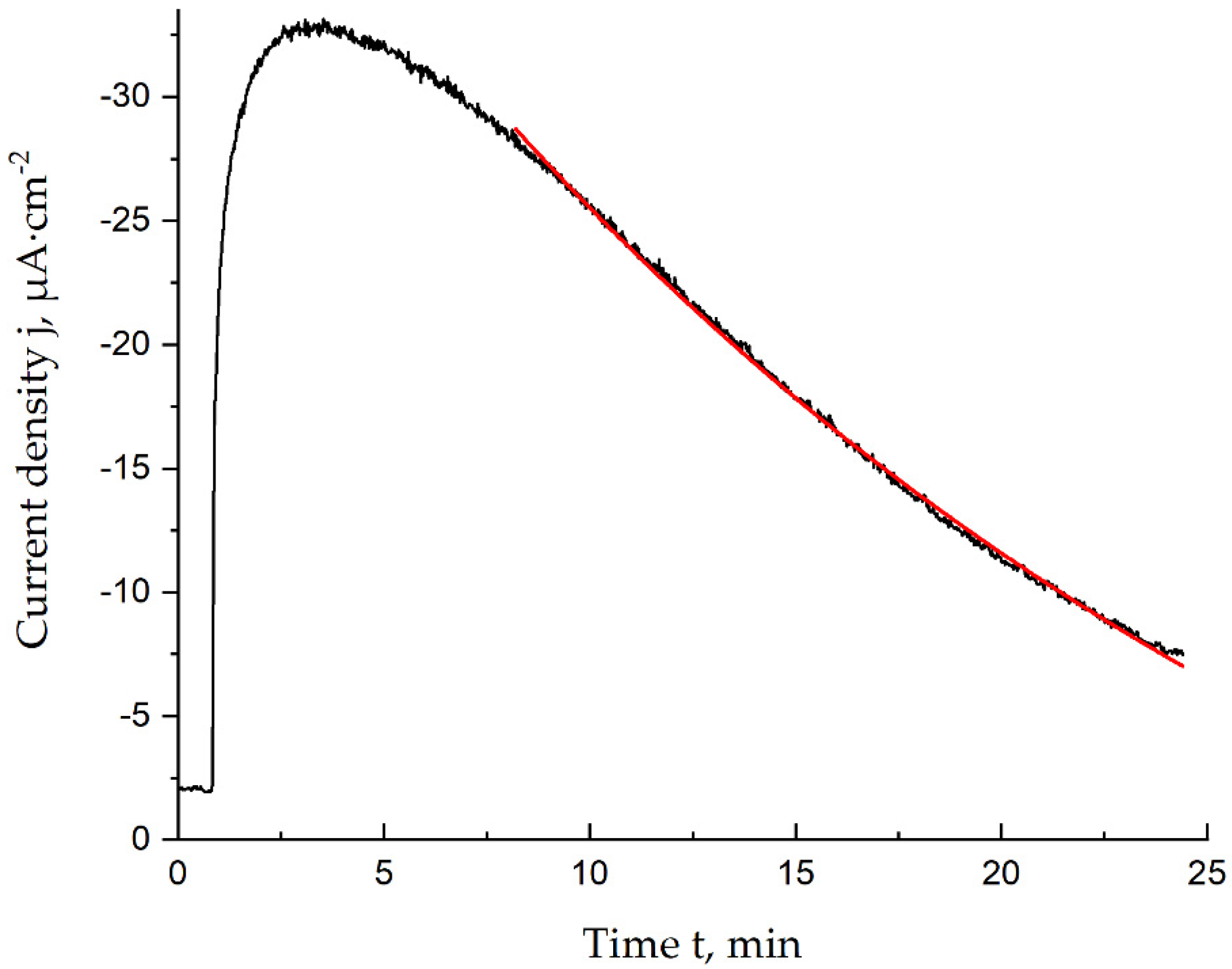
The stability of the lactate biosensors was evaluated using the inactivation constant. Calculation of this parameter was based on the assumption that the inactivation is a pseudo-first-order reaction. Use of the semi-logarithmic chronoamperograms registered during the 1.0 mM lactate solution addition (Figure 3) makes it possible to calculate the inactivation constant as the slope of the inactivation curve.
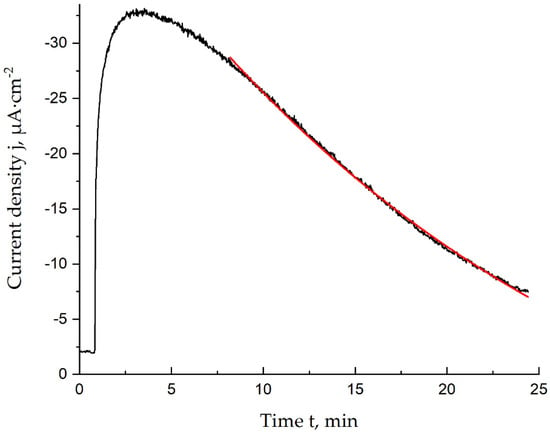
Figure 3.
Chronoamperogram (black) and approximation result (red) in the time range corresponding to the inactivation process, 1.0 mM lactate, 0.05 M PBS and 0.1 M KCl, pH = 6.0, E = 0.0 V vs. Ag/AgCl.
Thus, the calculated inactivation constant for lactate biosensors modified with Prussian blue nanoparticles was found to be 0.042 ± 0.002 min−1. The operational stability can also be evaluated using the response decrease curve, with biosensors retaining 90% of their initial response for 6 ± 1 min. This operational stability is relatively small, but it can be enhanced with the synthesis of a coating to be applied on the Prussian blue nanoparticles.
Use of Prussian blue nanoparticles coated with nickel hexacyanoferrate for lactate biosensors allowed us to achieve between 2–10-fold-lower values for the inactivation constants. This fact indicates that the low stability of the biosensors based on Prussian blue nanoparticles is due to transducer degradation rather than enzyme inactivation. As shown in Table 2, inactivation constants for biosensors based on core–shell nanozymes are dependent on the size and amount of the stabilizing coating. Therefore, operational stability is also better for lactate biosensors with higher amounts of stabilizing agent (nickel hexacyanoferrate). It should be noted that biosensors with stabilized nanoparticles have the same linear range as those with Prussian blue nanoparticles.

Table 2.
Inactivation constants for biosensors based on core–shell technology.
The obtained values indicate that inactivation can be reduced by the use of this type of core–shell nanozyme, but the synthetic parameters need optimizing to maximize the stability of the biosensors.
Author Contributions
V.P.—investigation, original draft preparation, visualization; E.D.—conceptualization, methodology, review and editing; A.K.—supervision, project administration, funding acquisition. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Russian Science Foundation (RSF), grant number 19-13-00131 (https://rscf.ru/en/project/19-13-00131/ (accessed on 6 February 2023)).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Karyakin, A.A. Prussian blue and its analogues: Electrochemistry and analytical applications. Electroanal. Int. J. Devoted Fundam. Pract. Asp. Electroanal. 2001, 13, 813–819. [Google Scholar] [CrossRef]
- Suprun, E.V.; Karpova, E.V.; Radko, S.P.; Karyakin, A.A. Advanced electrochemical detection of amino acids and proteins through flow injection analysis and catalytic oxidation on Prussian Blue. Electrochim. Acta 2020, 331, 135289. [Google Scholar] [CrossRef]
- Suprun, E.V.; Karpova, E.V.; Khmeleva, S.A.; Radko, S.P.; Karyakin, A.A. Advanced electrochemical detection of nitrogenous bases, synthetic oligonucleotides, and single-stranded DNA through flow injection analysis and catalytic oxidation on Prussian Blue. Electrochim. Acta 2021, 378, 138119. [Google Scholar] [CrossRef]
- Komkova, M.A.; Karyakina, E.E.; Karyakin, A.A. Catalytically synthesized prussian blue nanoparticles defeating natural enzyme peroxidase. J. Am. Chem. Soc. 2018, 140, 11302–11307. [Google Scholar] [CrossRef] [PubMed]
- Karpova, E.V.; Shcherbacheva, E.V.; Galushin, A.A.; Vokhmyanina, D.V.; Karyakina, E.E.; Karyakin, A.A. Noninvasive diabetes monitoring through continuous analysis of sweat using flow-through glucose biosensor. Anal. Chem. 2019, 91, 3778–3783. [Google Scholar] [CrossRef] [PubMed]
- Hirschhaeuser, F.; Sattler, U.G.; Mueller-Klieser, W. Lactate: A metabolic key player in cancer. Cancer Res. 2011, 71, 6921–6925. [Google Scholar] [CrossRef] [PubMed]
- Amorini, A.M.; Nociti, V.; Petzold, A.; Gasperini, C.; Quartuccio, E.; Lazzarino, G.; Tavazzi, B.; Di Pietro, V.; Belli, A.; Signoretti, S.; et al. Serum lactate as a novel potential biomarker in multiple sclerosis. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 2014, 1842, 1137–1143. [Google Scholar] [CrossRef] [PubMed]
- Kawamata, T.; Katayama, Y.; Hovda, D.A.; Yoshino, A.; Becker, D.P. Lactate accumulation following concussive brain injury: The role of ionic fluxes induced by excitatory amino acids. Brain Res. 1995, 674, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Rathee, K.; Dhull, V.; Dhull, R.; Singh, S. Biosensors based on electrochemical lactate detection: A comprehensive review. Biochem. Biophys. Rep. 2016, 5, 35–54. [Google Scholar] [CrossRef] [PubMed]
- Vashist, S.K.; Lam, E.; Hrapovic, S.; Male, K.B.; Luong, J.H. Immobilization of antibodies and enzymes on 3-aminopropyltriethoxysilane-functionalized bioanalytical platforms for biosensors and diagnostics. Chem. Rev. 2014, 114, 11083–11130. [Google Scholar] [CrossRef] [PubMed]
- Iwuoha, E.I.; Rock, A.; Smyth, M.R. Amperometric l-lactate biosensors: 1. Lactic acid sensing electrode containing lactate oxidase in a composite poly-l-lysine matrix. Electroanal. Int. J. Devoted Fundam. Pract. Asp. Electroanal. 1999, 11, 367–373. [Google Scholar]
- Nesakumar, N.; Sethuraman, S.; Krishnan, U.M.; Rayappan, J.B.B. Fabrication of lactate biosensor based on lactate dehydrogenase immobilized on cerium oxide nanoparticles. J. Colloid Interface Sci. 2013, 410, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhanen, Z.; Xiaolin, Z.; Deyao, Q.; Liu, Y.; Yu, T.; Deng, J. A phenazine methosulphate-mediated sensor sensitive to lactate based on entrapment of lactate oxidase and horseradish peroxidase in composite membrane of poly (vinyl alcohol) and regenerated silk fibroin. Electrochim. Acta 1997, 42, 349–355. [Google Scholar] [CrossRef]
- Vokhmyanina, D.V.; Andreeva, K.D.; Komkova, M.A.; Karyakina, E.E.; Karyakin, A.A. ‘Artificial peroxidase’nanozyme–enzyme based lactate biosensor. Talanta 2020, 208, 120393. [Google Scholar] [CrossRef] [PubMed]
- Kirthiga, O.M.; Rajendran, L. Approximate analytical solution for non-linear reaction diffusion equations in a mono-enzymatic biosensor involving Michaelis–Menten kinetics. J. Electroanal. Chem. 2015, 751, 119–127. [Google Scholar] [CrossRef]
- Gouda, M.D.; Kumar, M.A.; Thakur, M.S.; Karanth, N.G. Enhancement of operational stability of an enzyme biosensor for glucose and sucrose using protein based stabilizing agents. Biosens. Bioelectron. 2002, 17, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Baynton, K.J.; Bewtra, J.K.; Biswas, N.; Taylor, K.E. Inactivation of horseradish peroxidase by phenol and hydrogen peroxide: A kinetic investigation. Biochim. Et Biophys. Acta (BBA)-Protein Struct. Mol. Enzymol. 1994, 1206, 272–278. [Google Scholar] [CrossRef]
- Karpova, E.V.; Shcherbacheva, E.V.; Komkova, M.A.; Eliseev, A.A.; Karyakin, A.A. Core–Shell Nanozymes “Artificial Peroxidase”: Stability with Superior Catalytic Properties. J. Phys. Chem. Lett. 2021, 12, 5547–5551. [Google Scholar] [CrossRef] [PubMed]
- Daboss, E.V.; Shcherbacheva, E.V.; Karyakin, A.A. Simultaneous Noninvasive Monitoring of Diabetes and Hypoxia Using Core-Shell Nanozyme–Oxidase Enzyme Biosensors. Sens. Actuators B Chem. 2023, 380, 133337. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).