Highly Sensitive Amperometric Biosensors Based on Oxidases and CuCe Nanoparticles Coupled with Porous Gold †
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Enzymes Isolation and Purification
2.3. Synthesis of Nanoparticles and Estimation of Their Pseudo-Peroxidase Activity
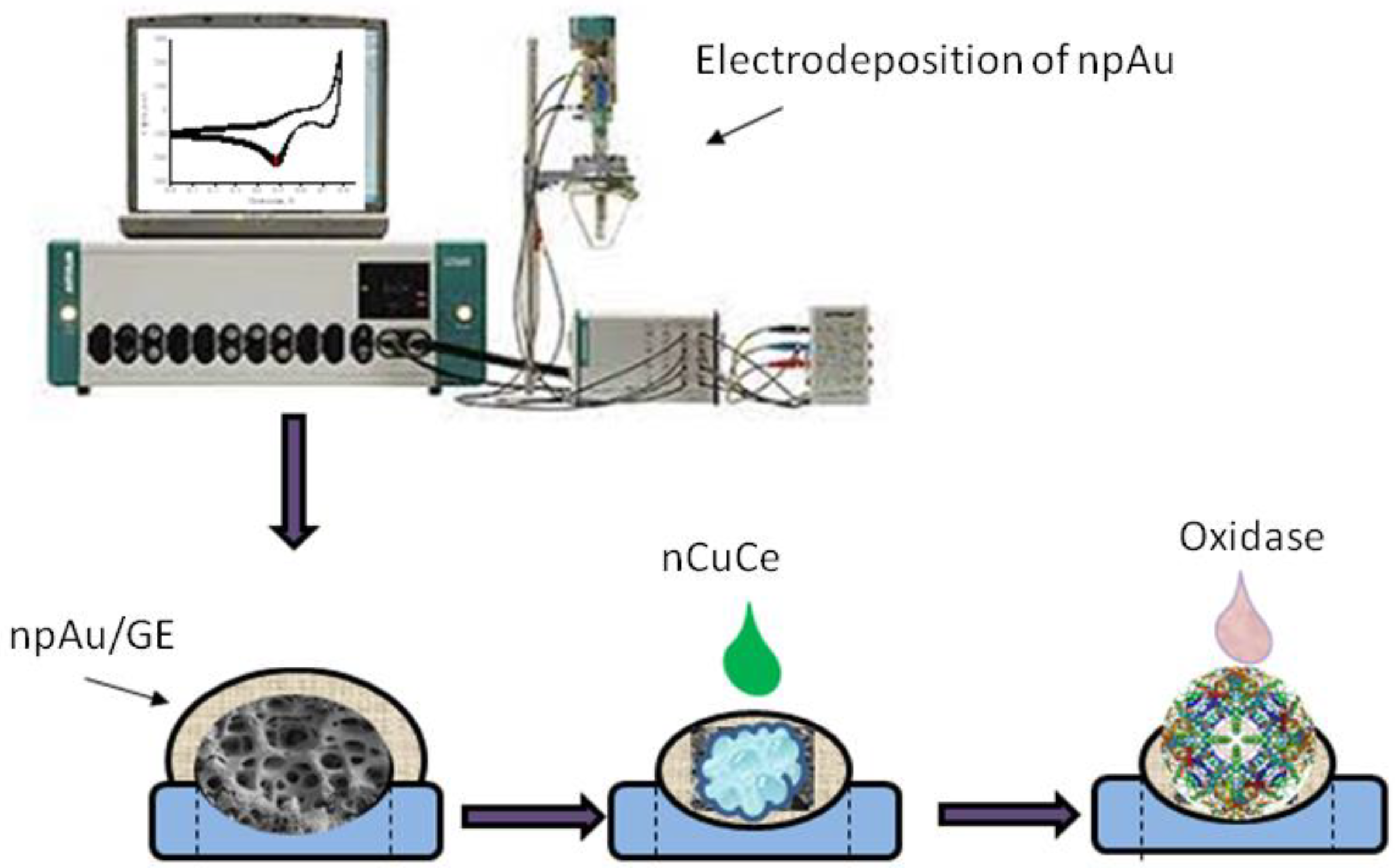
2.4. Apparatus
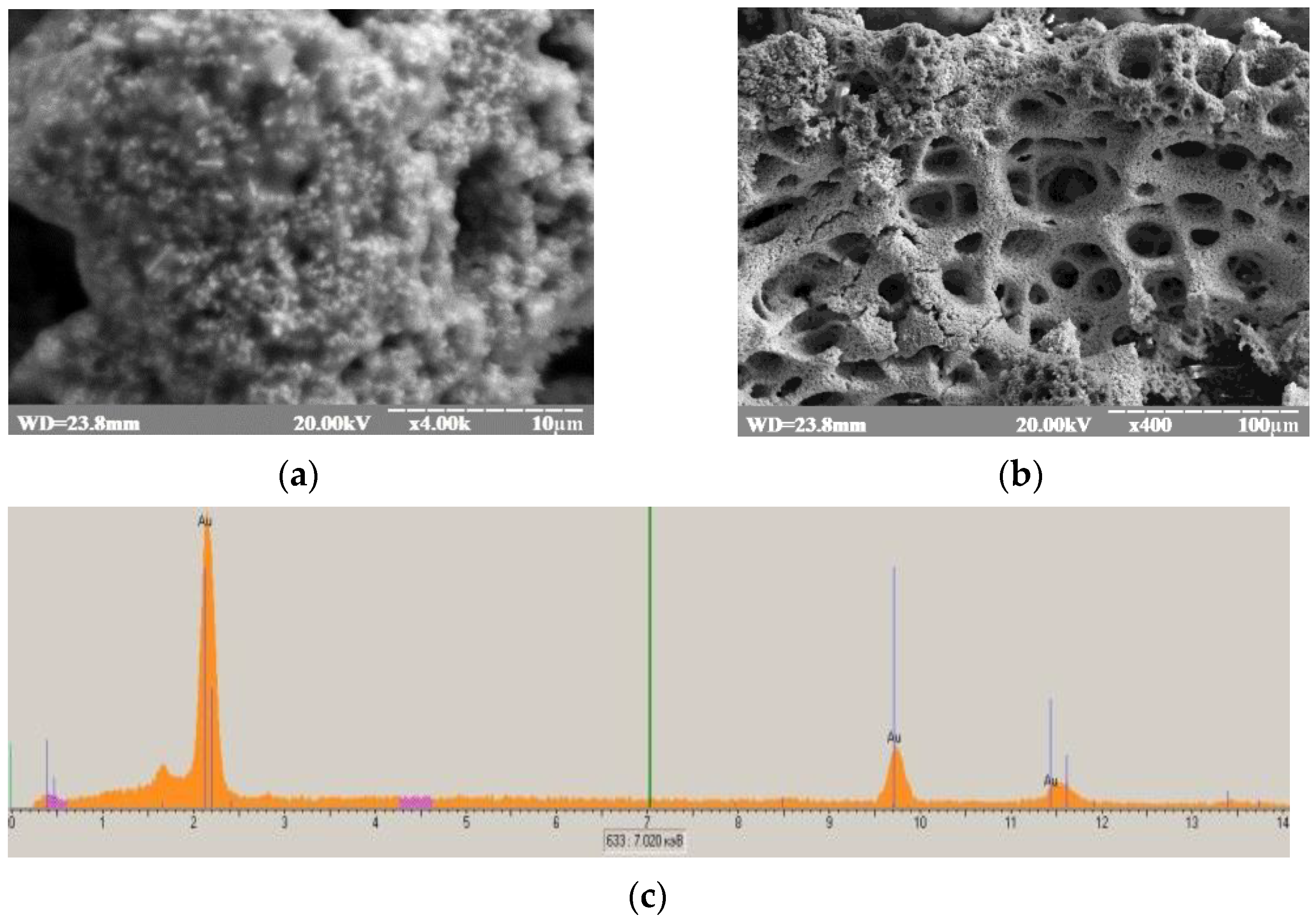
2.5. Electrodeposition of Nanoporous Gold onto Graphite Electrode
2.6. Immobilization of Natural and Artificial Peroxidases onto Electrode
2.7. Immobilization of Oxidases onto the Modified Electrodes
2.8. Measurements and Calculations
3. Results and Discussion
3.1. Development of Oxidase-Based Biosensors Using nCuCe and Porous Gold
3.2. Analytical Characteristics of the Constructed Biosensors
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Naresh, V.; Lee, N. A review on biosensors and recent development of nanostructured materials-enabled biosensors. Sensors 2021, 21, 1109. [Google Scholar] [CrossRef] [PubMed]
- Nayl, A.A.; Abd-Elhamid, A.I.; El-Moghazy, A.Y.; Hussin, M.; Abu-Saied, M.A.; El-Shanshory, A.A.; Solman, H.M.A. The nanomaterials and recent progress in biosensing systems: A review. Trends Environ. Anal. Chem. 2020, 26, e00087. [Google Scholar] [CrossRef]
- Presutti, D.; Agarwal, T.; Zarepour, A.; Celikkin, N.; Hooshmand, S.; Nayak, C.; Ghomi, M.; Zarrabi, A.; Costantini, M.; Behera, B.; et al. Transition Metal Dichalcogenides (TMDC)-Based Nanozymes for Biosensing and Therapeutic Applications. Materials 2022, 15, 337. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Wang, X.; Wang, Q.; Lou, Z.; Li, S.; Zhu, Y.; Qin, L.; Wei, H. Nanomaterials with enzyme-like characteristics (nanozymes): Next-generation artificial enzymes (II). Chem. Soc. Rev. 2019, 48, 1004–1076. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Ren, J.; Qu, X. Nanozymes: Classification, Catalytic Mechanisms, Activity Regulation, and Applications. Chem. Rev. 2019, 119, 4357–4412. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Zhang, A.; Wang, R.; Zhang, Q.; Cui, D. A Review on Metal- and Metal Oxide-Based Nanozymes: Properties, Mechanisms, and Applications. Nano Micro Lett. 2021, 13, 154. [Google Scholar] [CrossRef] [PubMed]
- Stasyuk, N.; Smutok, O.; Demkiv, O.; Prokopiv, T.; Gayda, G.; Nisnevitch, M.; Gonchar, M. Synthesis, Catalytic Properties and Application in Biosensorics of Nanozymes and Electronanocatalysts: A Review. Sensors 2020, 20, 4509. [Google Scholar] [CrossRef] [PubMed]
- Neumann, B.; Wollenberger, U. Electrochemical Biosensors Employing Natural and Artificial Heme Peroxidases on Semiconductors. Sensors 2020, 20, 3692. [Google Scholar] [CrossRef] [PubMed]
- Demkiv, O.; Stasyuk, N.; Serkiz, R.; Gayda, G.; Nisnevitch, M.; Gonchar, M. Peroxidase-Like Metal-Based Nanozymes: Synthesis, Catalytic Properties, and Analytical Application. Appl. Sci. 2021, 11, 777. [Google Scholar] [CrossRef]
- Gayda, G.Z.; Demkiv, O.M.; Gurianov, Y.; Serkiz, R.Y.; Klepach, H.M.; Gonchar, M.V.; Nisnevitch, M. “Green” Prussian Blue Analogues as Peroxidase Mimetics for Amperometric Sensing and Biosensing. Biosensors 2021, 11, 193. [Google Scholar] [CrossRef] [PubMed]
- Komkova, M.A.; Pasquarelli, A.; Andreev, E.A.; Galushin, A.A.; Karyakin, A.A. Prussian Blue modified boron-doped diamond interfaces for advanced H2O2 electrochemical sensors. Electrochim. Acta 2020, 339, 135924. [Google Scholar] [CrossRef]
- Keihan, A.H.; Karimi, R.R.; Sajjadi, S. Wide dynamic range and ultrasensitive detection of hydrogen peroxide based on beneficial role of gold nanoparticles on the electrochemical properties of Prussian blue. J. Electroanal. Chem. 2020, 862, 114001. [Google Scholar] [CrossRef]
- Tripathi, A.; Harris, K.D.; Elias, A.L. High surface area nitrogen-functionalized Ni nanozymes for efficient peroxidase-like catalytic activity. PLoS ONE 2021, 16, e0257777. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.; Zeng, W.; Cai, S.; Li, H.; Ding, J.; Wang, C.; Chen, Y.; Han, N.; Yang, R. Porous Au@Pt nanoparticles with superior peroxidase-like activity for colorimetric detection of spike protein of SARS-CoV-2. J. Colloid Interface Sci. 2021, 604, 113–121. [Google Scholar] [CrossRef]
- Stasyuk, N.; Gayda, G.; Demkiv, O.; Darmohray, L.; Gonchar, M.; Nisnevitch, M. Amperometric biosensors for L-arginine determination based on L-arginine oxidase and peroxidase-like nanozymes. Appl. Sci. 2021, 11, 7024. [Google Scholar] [CrossRef]
- Stine, K.J.; Jefferson, K.; Shulga, O.V. Nanoporous gold for enzyme immobilization. Methods Mol. Biol. 2017, 1504, 37–60. [Google Scholar] [PubMed]
- Bhattarai, J.K.; Neupane, D.; Nepal, B.; Mikhaylov, V.; Demchenko, A.V.; Stine, K.J. Preparation, Modification, Characterization, and Biosensing Application of Nanoporous Gold Using Electrochemical Techniques. Nanomaterials 2018, 8, 171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klepach, H.M.; Zakalskiy, A.E.; Zakalska, O.M.; Gayda, G.Z.; Smutok, O.V.; Gonchar, M.V. Alcohol Oxidase from the Methylotrophic Yeast Ogataea polymorpha: Isolation, Purification, and Bioanalytical Application. Flavins Flavoproteins 2021, 2280, 231–248. [Google Scholar]
- Stasyuk, N.E.; Smutok, O.V.; Zakalskiy, A.E.; Zakalska, O.M.; Gonchar, M.V. Methylamine-Sensitive Amperometric Biosensor Based on (His)6-Tagged Hansenula polymorpha Methylamine Oxidase Immobilized on the Gold Nanoparticles. Biomed. Res. Int. 2014, 2014, 480498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Demkiv, O.; Stasyuk, N.; Gayda, G.; Gonchar, M. Highly sensitive amperometric sensor based on laccase-mimicking metal-based hybrid nanozymes for adrenaline analysis in pharmaceuticals. Catalysts 2021, 11, 1510. [Google Scholar] [CrossRef]
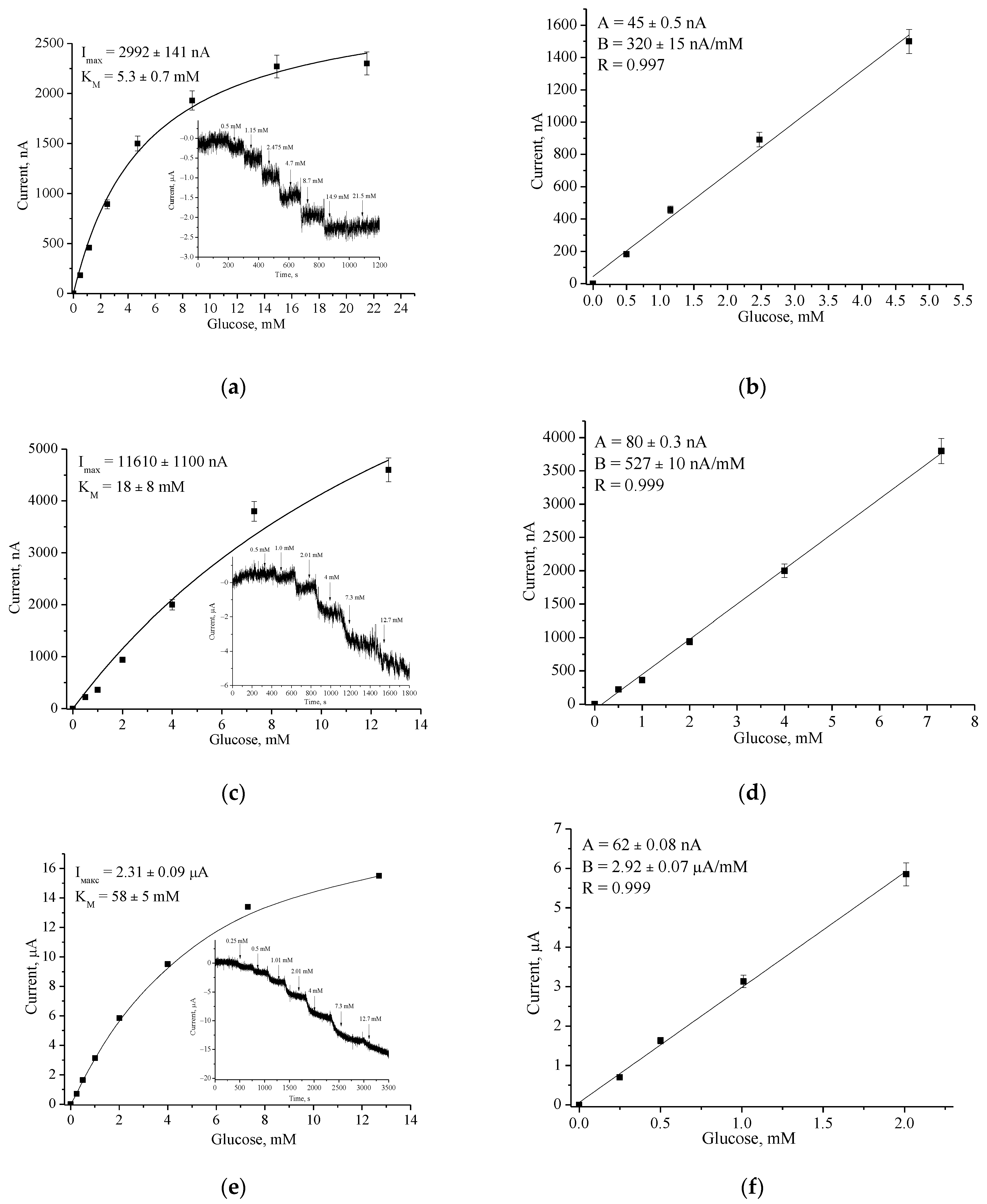
| Bioelectrode | Potential, mV | Sensitivity, A·M−1·m−2 | Linear Range, µM | LOD, µM |
|---|---|---|---|---|
| GO/PO/GE | −50 | 44 | 50–5000 | 150 |
| GO/nCuCe/GE | −50 | 73 | 500–7300 | 150 |
| GO/nCuCe/npAu/GE | −50 | 400 | 25–2000 | 75.7 |
| AMO/PO/GE | −250 | 7 | 200–1700 | 130 |
| AMO/nCuCe/GE | −250 | 35 | 60–1700 | 18 |
| AMO/nCuCe/npAu/GE | −250 | 125 | 60–500 | 18 |
| AO/PO/GE | −50 | 22 | 130–900 | 39 |
| AO/nCuCe/GE | −50 | 32 | 50–2100 | 15 |
| AO/nCuCe/npAu/GE | −50 | 102 | 33–500 | 10 |
| ArgO/PO/GE | −150 | 24 | 75–1150 | 35 |
| ArgO/nCuCe/GE | −150 | 113 | 50–2250 | 15 |
| ArgO/nCeCu/npAu/GE | −150 | 200 | 100–500 | 33 |
| Laccase/GE | +200 | 2300 | 8–160 | 2 |
| Laccase/nCuCe/GE | +200 | 5055 | 3–40 | 1.5 |
| Laccase/nCuCe/npAu/GE | +200 | 9280 | 2–40 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stasyuk, N.; Demkiv, O.; Gayda, G.; Serkiz, R.; Zakalskiy, A.; Zakalska, O.; Klepach, H.; Al-Maali, G.; Bisko, N.; Gonchar, M. Highly Sensitive Amperometric Biosensors Based on Oxidases and CuCe Nanoparticles Coupled with Porous Gold . Eng. Proc. 2022, 16, 3. https://doi.org/10.3390/IECB2022-12251
Stasyuk N, Demkiv O, Gayda G, Serkiz R, Zakalskiy A, Zakalska O, Klepach H, Al-Maali G, Bisko N, Gonchar M. Highly Sensitive Amperometric Biosensors Based on Oxidases and CuCe Nanoparticles Coupled with Porous Gold . Engineering Proceedings. 2022; 16(1):3. https://doi.org/10.3390/IECB2022-12251
Chicago/Turabian StyleStasyuk, Nataliya, Olha Demkiv, Galina Gayda, Roman Serkiz, Andriy Zakalskiy, Oksana Zakalska, Halyna Klepach, Galeb Al-Maali, Nina Bisko, and Mykhailo Gonchar. 2022. "Highly Sensitive Amperometric Biosensors Based on Oxidases and CuCe Nanoparticles Coupled with Porous Gold " Engineering Proceedings 16, no. 1: 3. https://doi.org/10.3390/IECB2022-12251
APA StyleStasyuk, N., Demkiv, O., Gayda, G., Serkiz, R., Zakalskiy, A., Zakalska, O., Klepach, H., Al-Maali, G., Bisko, N., & Gonchar, M. (2022). Highly Sensitive Amperometric Biosensors Based on Oxidases and CuCe Nanoparticles Coupled with Porous Gold . Engineering Proceedings, 16(1), 3. https://doi.org/10.3390/IECB2022-12251






