Variation in Current Density of Aluminum Scrap-Based Propeller Anodization to Increase Surface Hardness †
Abstract
1. Introduction
2. Methods
2.1. Materials
2.2. Anodizing Setup
3. Results and Discussion
3.1. Chemical Composition
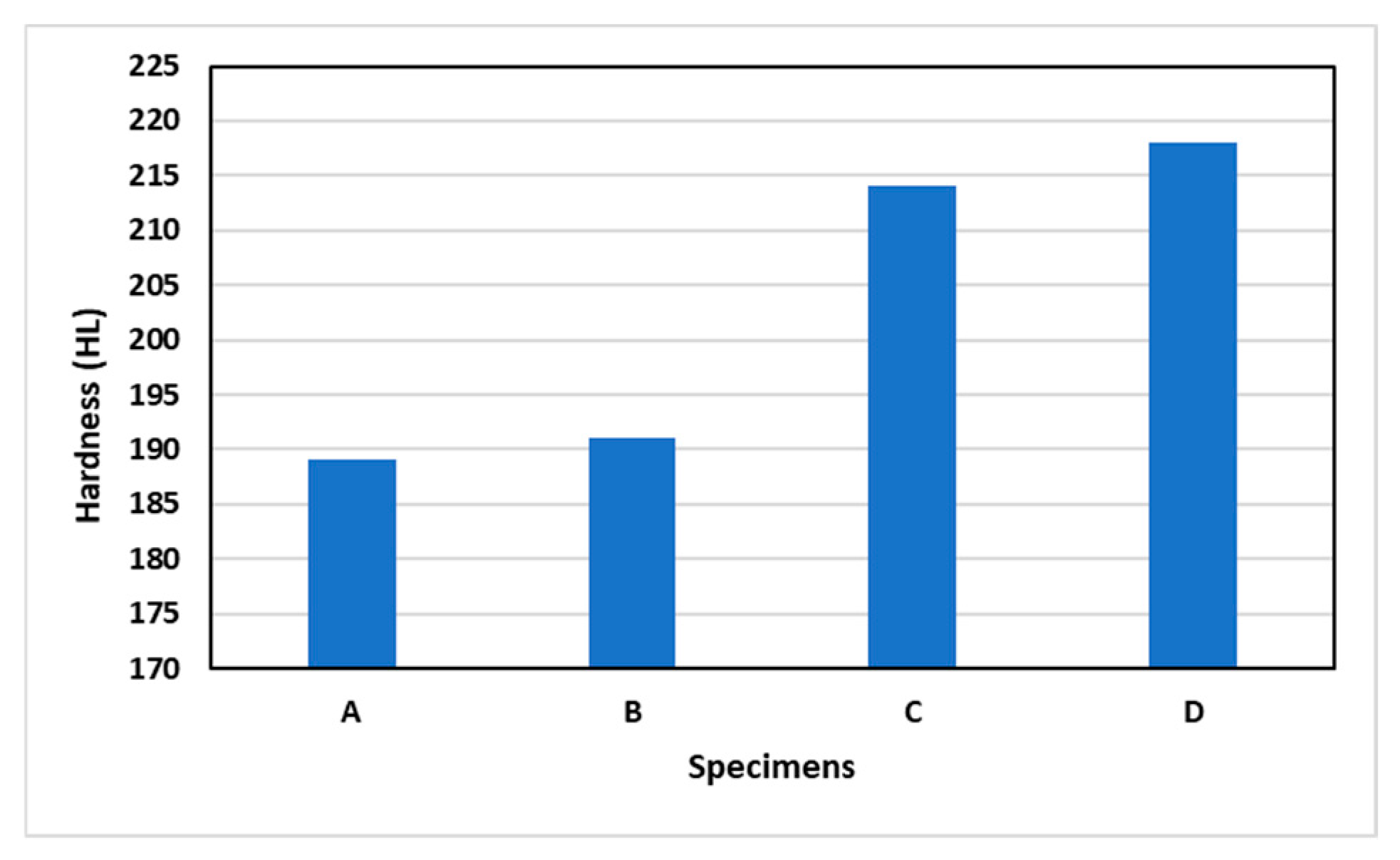
3.2. Hardness Test
4. Conclusions
- The anodizing process can increase the hardness of the surface layer of scrap aluminum material.
- Current density affects the hardness results for aluminum scrap. The greater the current density used, the greater the hardness value of the aluminum scrap.
- The hardness value produced on the surface of aluminum scrap (C) with a current density of 0.04 A/cm2 was the highest, namely 218 HL.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sembodo, S.; Anawati, A. Combine effect of temperature and additive ethylene glycol on the characteristics of anodic film formed on aluminum alloy AA7075. In AIP Conference Proceedings, Proceedings of the 3rd International Seminar on Metallurgy and Materials: Exploring New Innovation in Metallurgy and Materials (ISMM2019), Tangerang Selatan, Indonesia, 23–24 October 2019; AIP Publishing: Melville, NY, USA, 2020; Volume 2232, p. 070001. [Google Scholar] [CrossRef]
- Chung, I.C.; Chung, C.K.; Su, Y.K. Effects of anodization parameters on the corrosion resistance of 6061 Al alloy using the Taguchi method. Microsyst. Technol. 2018, 24, 351–359. [Google Scholar] [CrossRef]
- Raabe, D.; Ponge, D.; Uggowitzer, P.J.; Roscher, M.; Paolantonio, M.; Liu, C.; Antrekowitsch, H.; Kozeschnik, E.; Seidmann, D.; Gault, B. Making sustainable aluminum by recycling scrap: The science of ‘dirty’ alloys. Prog. Mater. Sci. 2022, 128, 100947. [Google Scholar] [CrossRef]
- Islami, L.A.; Fathurrahman, N.M.; Sulaiman, Z. Analisis sifat korosi hasil cor terhadap temperatur pembuatan propeller perahu menggunakan metode sand casting. J. Rekayasa Mesin 2023, 18, 385. [Google Scholar] [CrossRef]
- Chung, I.C.; Chung, C.K.; Su, Y.K. Effect of current density and concentration on microstructure and corrosion behavior of 6061 Al alloy in sulfuric acid. Surf. Coat. Technol. 2017, 313, 299–306. [Google Scholar] [CrossRef]
- Shen, X.; Wang, J. Study on mechanical properties of anodized films on 5052 aluminum alloy. Int. J. Electrochem. Sci. 2022, 17, 221022. [Google Scholar] [CrossRef]
- Ren, P.; Zhao, C.; Zuo, Z.; Shui, Y.; Li, D.; Huang, W. Effect of thermal degradation on mechanical properties of cast Al-Si alloys for cylinder heads. Eng. Fract. Mech. 2023, 283, 109200. [Google Scholar] [CrossRef]
- Bulei, C.; Kiss, I.; Todor, M. The chemical composition of post-consumer aluminium scrap—A challenge in aluminium recycling. Acta Polytech. Hung. 2023, 20, 63–76. [Google Scholar] [CrossRef]
- Khan, A.A.; Razin, A.A.; Ahammed, D.S.S.; Kaiser, M.S. Comparison of electrochemical corrosion performance of eutectic Al-Si automotive alloy in deep seawater and 3.5% NaCl solution. Mater. Today Proc. 2023, 82, 241–247. [Google Scholar] [CrossRef]
- Shin, D.H.; Kim, S.J. Effects of hard anodizing on mechanical and electrochemical characteristics of aluminum alloys under tribocorrosion condition. Corros. Sci. 2024, 240, 112503. [Google Scholar] [CrossRef]
- Sano, T.; Wakabayashi, Y.; Asoh, H. Formation of hard anodic films on the 7075-T6 aluminum alloy by anodization in sulfuric acid and ethylene glycol. Surf. Coat. Technol. 2023, 459, 129399. [Google Scholar] [CrossRef]
| Specimen | Current Density (A/cm2) | Code |
|---|---|---|
| 1 | As received | A |
| 2 | 0.030 | B |
| 3 | 0.035 | C |
| 4 | 0.040 | D |
| No | Element | Composition |
|---|---|---|
| 1 | Al | 92.91 |
| 2 | Si | 3.87 |
| 3 | Zn | 0.96 |
| 4 | Fe | 0.97 |
| 5 | Cu | 0.88 |
| 6 | Pb | 0.41 |
| Code | Hardness (HL) | Average Hardness (HL) | ||
|---|---|---|---|---|
| 1 | 2 | 3 | ||
| A | 183 | 212 | 173 | 189 |
| B | 173 | 185 | 214 | 191 |
| C | 212 | 218 | 211 | 214 |
| D | 186 | 240 | 228 | 218 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nayla, R.P.; Bani, P.D.; Udzmatillah, H.; Islami, L.A.; Sellappan, S.N. Variation in Current Density of Aluminum Scrap-Based Propeller Anodization to Increase Surface Hardness. Eng. Proc. 2025, 107, 38. https://doi.org/10.3390/engproc2025107038
Nayla RP, Bani PD, Udzmatillah H, Islami LA, Sellappan SN. Variation in Current Density of Aluminum Scrap-Based Propeller Anodization to Increase Surface Hardness. Engineering Proceedings. 2025; 107(1):38. https://doi.org/10.3390/engproc2025107038
Chicago/Turabian StyleNayla, Rifani Putri, Paulus Dara Bani, Hilmi Udzmatillah, Lazuardi Akmal Islami, and Sivakumar Nallappan Sellappan. 2025. "Variation in Current Density of Aluminum Scrap-Based Propeller Anodization to Increase Surface Hardness" Engineering Proceedings 107, no. 1: 38. https://doi.org/10.3390/engproc2025107038
APA StyleNayla, R. P., Bani, P. D., Udzmatillah, H., Islami, L. A., & Sellappan, S. N. (2025). Variation in Current Density of Aluminum Scrap-Based Propeller Anodization to Increase Surface Hardness. Engineering Proceedings, 107(1), 38. https://doi.org/10.3390/engproc2025107038





