Optimized Spectrophotometry Method for Starch Quantification
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Instrumentation
2.3. Sample Preparation
2.3.1. Amylose Iodine Reagent
2.3.2. Starch Preparation
2.3.3. Bioaerosol Preparation
3. Results and Discussion
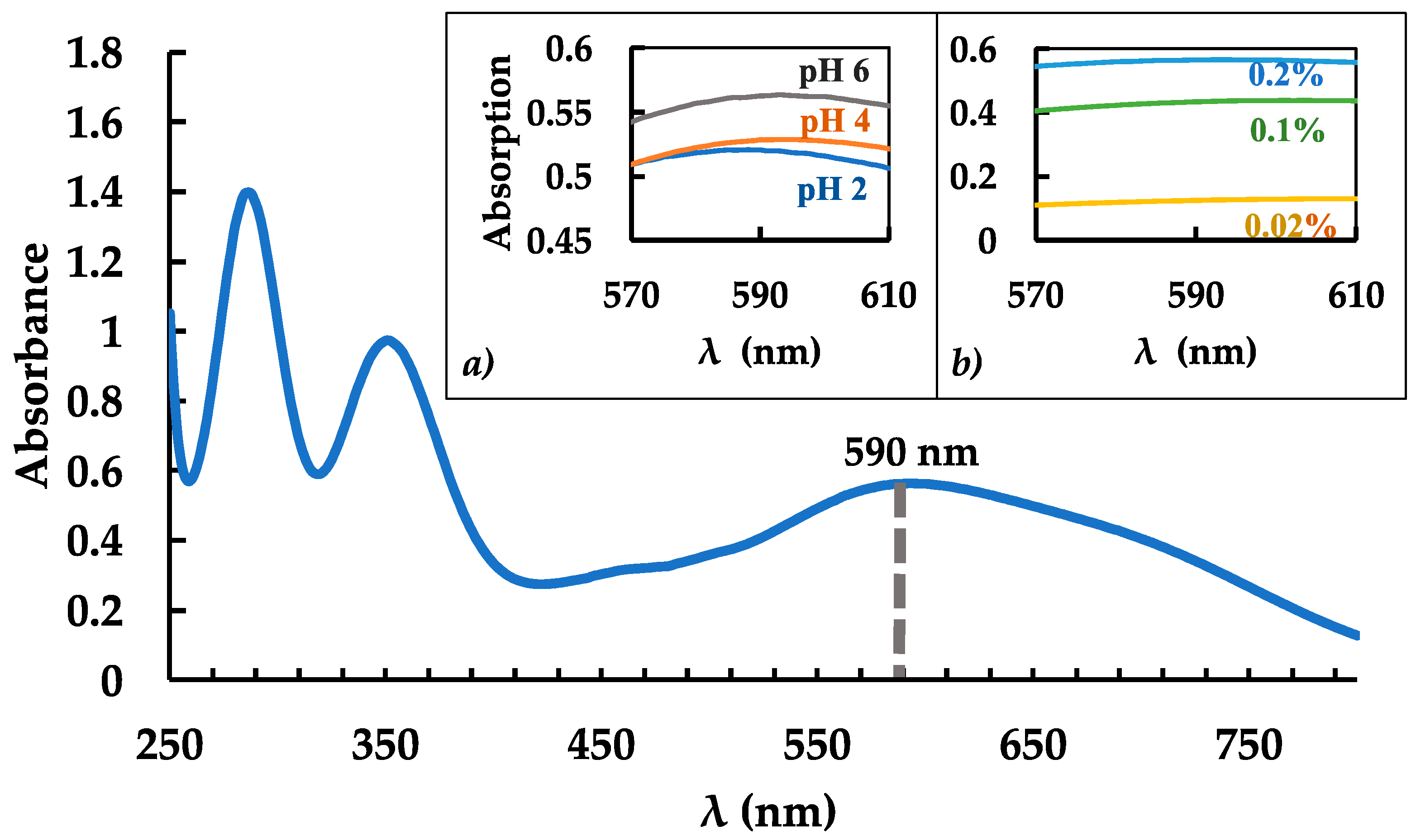
3.1. Dependance of pH and Iodine Reagent Concentration
3.2. Calibration and MDL
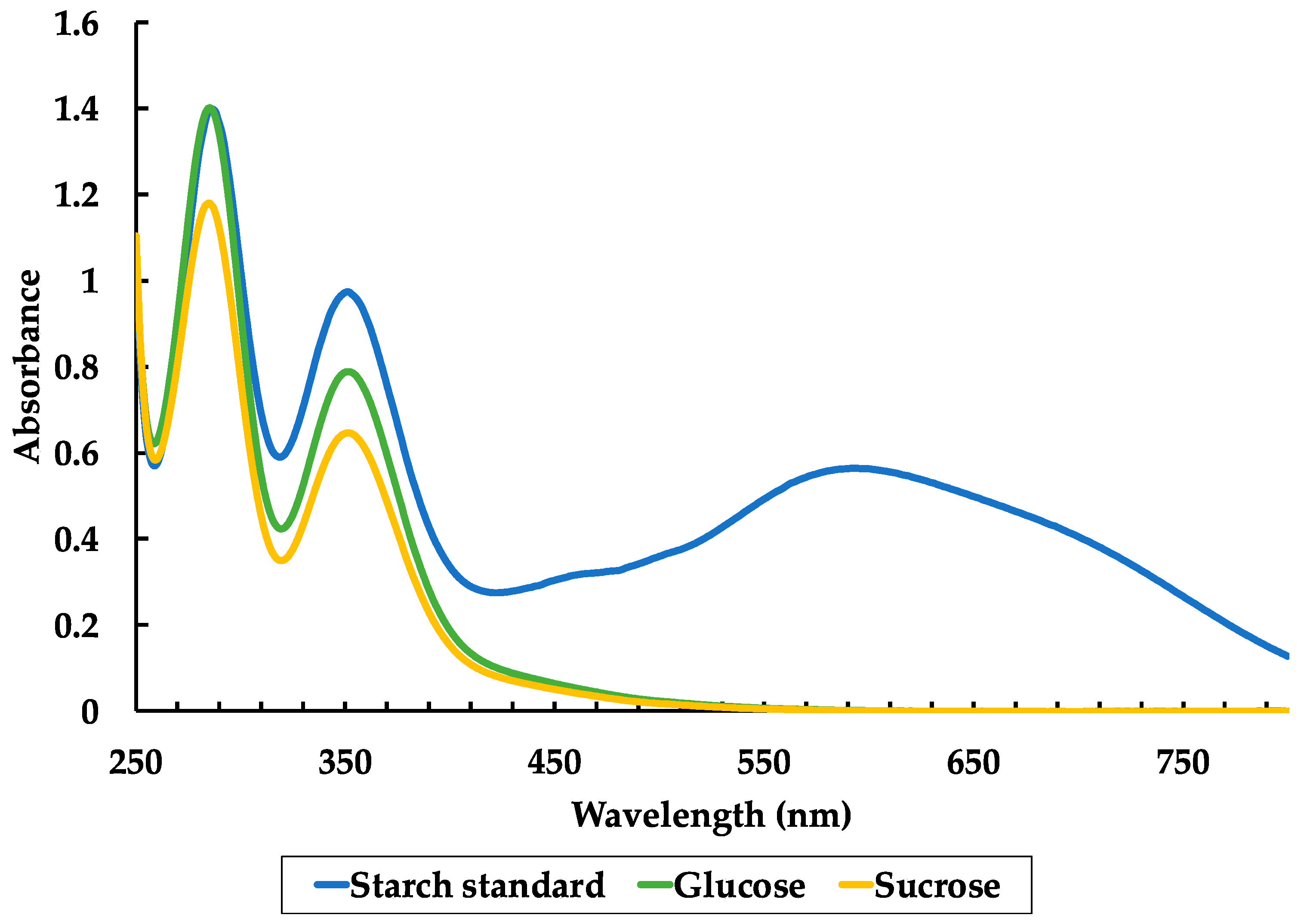
3.3. Bioaerosol and Saccharide Analysis
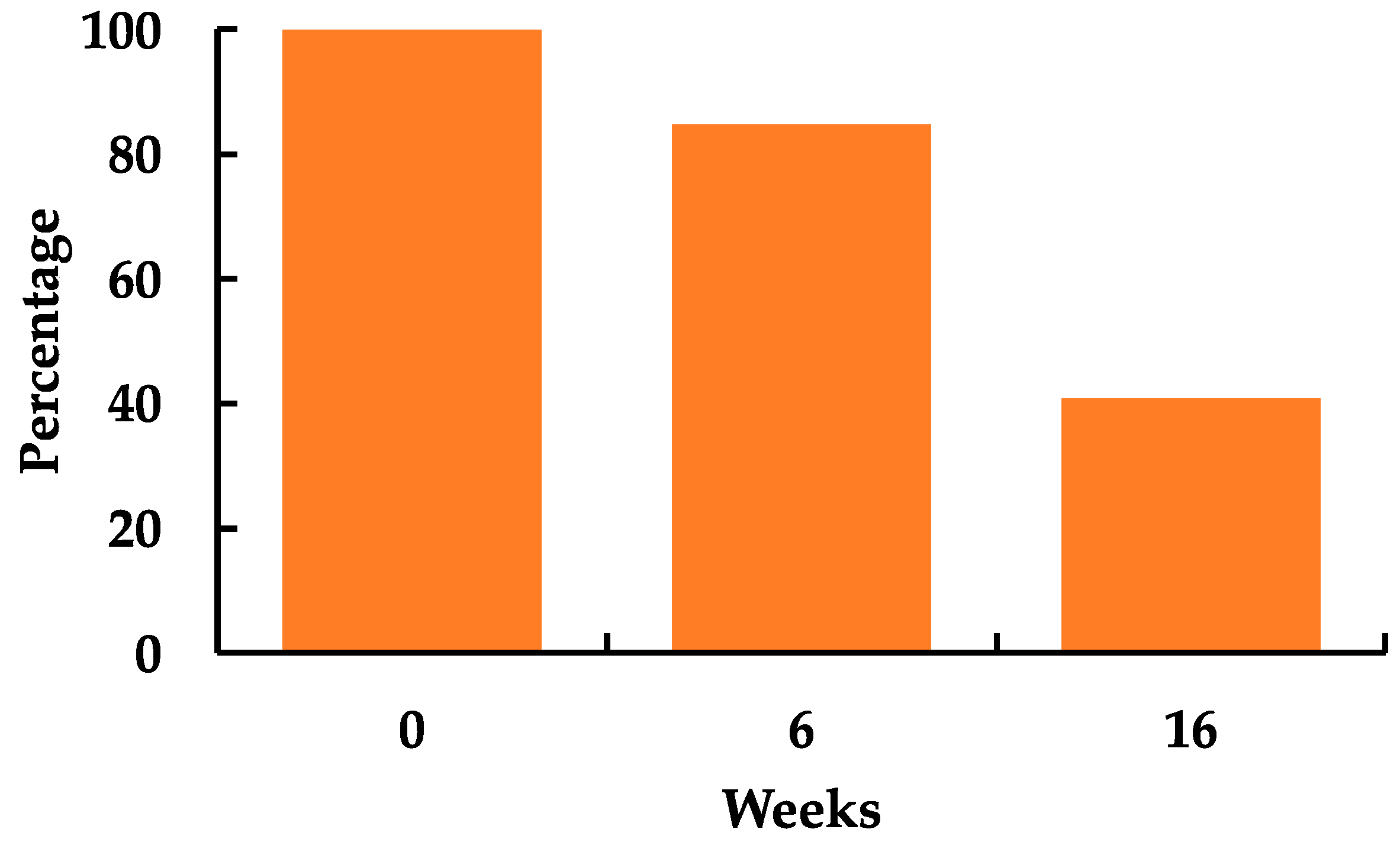
3.4. Starch Stability
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Buléon, A.; Colonna, P.; Planchot, V.; Ball, S. Mini Review Starch Granules: Structure and Biosynthesis. Int. J. Biol. Macromol. 1998, 23, 85–112. [Google Scholar] [CrossRef]
- Lafont-Mendoza, J.J.; Severiche-Sierra, C.A.; Jaimes-Morales, J. Evaluation of the Starch Quantification Methods of Musa Paradisiaca, Manihot Esculenta, and Dioscorea Trífida Using Factorial Experiments. Int. J. Food Sci. 2018, 2018, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yong, T.C.; Chiu, C.S.; Chen, C.N.N. Optimization of a Simple, Accurate and Low Cost Method for Starch Quantification in Green Microalgae. Bot. Stud. 2019, 60, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Bashir, K.; Aggarwal, M. Physicochemical, Structural and Functional Properties of Native and Irradiated Starch: A Review. J. Food Sci. Technol. 2019, 56, 513–523. [Google Scholar] [CrossRef]
- McCready, R.M.; Hassid, W.Z. The Separation and Quantitative Estimation of Amylose and Amylopectin in Potato Starch. J. Am. Chem. Soc. 1943, 65, 1154–1157. [Google Scholar] [CrossRef]
- Subroto, E.; Jeanette, G.; Meiyanasari, Y.; Luwinsky, I.; Baraddiaz, S. Review on the Analysis Methods of Starch, Amylose, Amylopectinin Food and Agricultural Products. Int. J. Emerg. Trends Eng. Res. 2020, 8, 3519–3524. [Google Scholar] [CrossRef]
- Bates, L.F.; French, D.; Rundle, R.E. Amylose and Amylopectin Content of Starches Determined by Their Iodine Complex Formation. J. Am. Chem. Soc. 1943, 65, 142–148. [Google Scholar] [CrossRef]
- Egharevba, H.O. Chemical Properties of Starch and Its Application in the Food Industry; IntechOpen: London, UK, 2019. [Google Scholar]
- Han, J.A.; BeMiller, J.N. Preparation and Physical Characteristics of Slowly Digesting Modified Food Starches. Carbohydr. Polym. 2007, 67, 366–374. [Google Scholar] [CrossRef]
- Takeda, C.; Takeda, Y.; Hizukuri, S. Structure of Amylomaize Amylose. Cereal Chem. 1989, 66, 22–25. [Google Scholar]
- Nakayoshi, Y.; Nakamura, S.; Kameo, Y.; Shiiba, D.; Katsuragi, Y.; Ohtsubo, K. Measurement of Resistant Starch Content in Cooked Rice and Analysis of Gelatinization and Retrogradation Characteristics. Biosci. Biotechnol. Biochem. 2015, 79, 1860–1866. [Google Scholar] [CrossRef]
- Sulistyarti, H.; Atikah, A.; Fardiyah, Q.; Febriyanti, S.; Asdauna, A. A Simple and Safe Spectrophotometric Method for Iodide Determination. Makara J. Sci. 2015, 19, 43–48. [Google Scholar] [CrossRef]
- Pokhrel, S. A Review on Introduction and Applications of Starch and Its Biodegradable Polymers. Int. J. Environ. 2015, 4, 114–125. [Google Scholar] [CrossRef]
- Desai, B.S.; Modi, Z.S.; Amit, K.J.; Parmar, S.C.; Shaikh, A.I.; Aparnathi, K.D. Development of Spectroscopic Method for Quantification of Starch in Milk. Int. J. Chem. Stud. 2018, 6, 53–57. [Google Scholar]
- Krajang, M.; Malairuang, K.; Sukna, J.; Rattanapradit, K.; Chamsart, S. Single-Step Ethanol Production from Raw Cassava Starch Using a Combination of Raw Starch Hydrolysis and Fermentation, Scale-up from 5-L Laboratory and 200-L Pilot Plant to 3000-L Industrial Fermenters. Biotechnol. Biofuels 2021, 14, 1–15. [Google Scholar] [CrossRef]
- Boonpo, S.; Kungwankunakorn, S. Study on Amylose Iodine Complex from Cassava Starch by Colorimetric Method. J. Adv. Agric. Technol. 2017, 4, 345–349. [Google Scholar] [CrossRef]
- Fröhlich-Nowoisky, J.; Kampf, C.J.; Weber, B.; Huffman, J.A.; Pöhlker, C.; Andreae, M.O.; Lang-Yona, N.; Burrows, S.M.; Gunthe, S.S.; Elbert, W.; et al. Bioaerosols in the Earth System: Climate, Health, and Ecosystem Interactions. Atmos. Res. 2016, 182, 346–376. [Google Scholar] [CrossRef]
- Zhang, Y.; Steiner, A.L. Projected Climate-Driven Changes in Pollen Emission Season Length and Magnitude over the Continental United States. Nat. Commun. 2022, 13, 1–10. [Google Scholar] [CrossRef]
- Després, V.R.; Huffman, J.A.; Burrows, S.M.; Hoose, C.; Safatov, A.S.; Buryak, G.; Fröhlich-Nowoisky, J.; Elbert, W.; Andreae, M.O.; Pöschl, U.; et al. Primary Biological Aerosol Particles in the Atmosphere: A Review. Tellus B Chem. Phys. Meteorol. 2012, 64, 15598. [Google Scholar] [CrossRef]
- Anderegg, W.R.L.; Abatzoglou, J.T.; Anderegg, L.D.L.; Bielory, L.; Kinney, P.L.; Ziska, L. Anthropogenic Climate Change Is Worsening North American Pollen Seasons. Proc. Natl. Acad. Sci. USA 2021, 118, e2013284118. [Google Scholar] [CrossRef]
- Ariano, R.; Canonica, G.W.; Passalacqua, G. Possible Role of Climate Changes in Variations in Pollen Seasons and Allergic Sensitizations during 27 Years. Ann. Allergy Asthma Immunol. 2010, 104, 215–222. [Google Scholar] [CrossRef]
- D’Amato, G.; Chong-Neto, H.J.; Monge Ortega, O.P.; Vitale, C.; Ansotegui, I.; Rosario, N.; Haahtela, T.; Cecchi, L.; Bergmann, C.; Ridolo, E.; et al. The Effects of Climate Change on Respiratory Allergy and Asthma Induced by Pollen and Mold Allergens. Eur. J. Allergy Clin. Immunol. 2020, 75, 2219–2228. [Google Scholar] [CrossRef] [PubMed]
- May, N.W.; Olson, N.E.; Panas, M.; Axson, J.L.; Tirella, P.S.; Kirpes, R.M.; Craig, R.L.; Gunsch, M.J.; China, S.; Laskin, A.; et al. Aerosol Emissions from Great Lakes Harmful Algal Blooms. Environ. Sci. Technol. 2018, 52, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Estillore, A.D.; Trueblood, J.v; Grassian, V.H. Atmospheric Chemistry of Bioaerosols: Heterogeneous and Multiphase Reactions with Atmospheric Oxidants and Other Trace Gases. Chem. Sci. 2016, 7, 6604–6616. [Google Scholar] [CrossRef] [PubMed]
- Axelrod, K.; Samburova, V.; Khlystov, A.Y. Relative Abundance of Saccharides, Free Amino Acids, and Other Compounds in Specific Pollen Species for Source Profiling of Atmospheric Aerosol. Sci. Total Environ. 2021, 799, 149254. [Google Scholar] [CrossRef]
- Kumar, R.; Bansal, V.; Patel, M.B.; Sarpal, A.S. Compositional Analysis of Algal Biomass in a Nuclear Magnetic Resonance (NMR) Tube. J. Algal Biomass Util. 2014, 5, 36–45. [Google Scholar]
- Chalbot, M.-C.G.; Gamboa da Costa, G.; Kavouras, I.G. NMR Analysis of the Water-Soluble Fraction of Airborne Pollen Particles. Appl. Magn. Reson. 2013, 44, 1347–1358. [Google Scholar] [CrossRef]
- Hughes, D.D.; Mampage, C.B.A.; Jones, L.M.; Liu, Z.; Stone, E.A. Characterization of Atmospheric Pollen Fragments during Springtime Thunderstorms. Environ. Sci. Technol. Lett. 2020, 7, 409–414. [Google Scholar] [CrossRef]
- Mampage, C.B.A.; Hughes, D.D.; Jones, L.M.; Metwali, N.; Thorne, P.S.; Stone, E.A. Characterization of Sub-Pollen Particles in Size-Resolved Atmospheric Aerosol Using Chemical Tracers. Atmos. Environ. X 2022, 15, 100177. [Google Scholar] [CrossRef]
- Burkart, J.; Gratzl, J.; Seifried, T.M.; Bieber, P.; Grothe, H. Isolation of Subpollen Particles (SPPs) of Birch: SPPs Are Potential Carriers of Ice Nucleating Macromolecules. Biogeosciences 2021, 18, 5751–5765. [Google Scholar] [CrossRef]
- Noranizan, M.A.; Dzulkifly, M.H.; Russly, A.R. Effect of Heat Treatment on the Physico-Chemical Properties of Starch from Different Botanical Sources. Int. Food Res. J. 2010, 17, 127–135. [Google Scholar]
- Roberts, S.A.; Cameron, R.E. The Effects of Concentration and Sodium Hydroxide on the Rheological Properties of Potato Starch Gelatinisation. Carbohydr. Polym. 2022, 50, 133–143. [Google Scholar] [CrossRef]
- Sun, Q.; Gong, M.; Li, Y.; Xiong, L. Effect of Dry Heat Treatment on the Physicochemical Properties and Structure of Proso Millet Flour and Starch. Carbohydr. Polym. 2014, 110, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, R.R.; Bear, R.S.; Rundle, R.E. The Relation of Starch-Iodine Absorption Spectra to the Structure of Starch and Starch Components. J. Am. Chem. Soc. 1944, 66, 111–115. [Google Scholar] [CrossRef]
- McGrance, S.J.; Cornell, H.J.; Rix, C.J. A Simple and Rapid Colorimetric Method for the Determination of Amylose in Starch Products. Starch/Staerke 1998, 50, 158–163. [Google Scholar] [CrossRef]
- Hermansson, A.-M.; Svegmark, K. Developments in the Understanding of Starch Functionality. Trends Food Sci. Technol. 1996, 7, 345–353. [Google Scholar] [CrossRef]
- Ripp, J. Analytical Detection Limit Guidance & Laboratory Guide for Determining Method Detection Limits; Wisconsin Department of Natural Resources, Laboratory Certification Program: Madison, WI, USA, 1996. [Google Scholar]
- Brust, H.; Orzechowski, S.; Fettke, J. Starch and Glycogen Analyses: Methods and Techniques. Biomolecules 2020, 10, 1020. [Google Scholar] [CrossRef]
- Ross, A.; Willson, V.L. Independent Samples T-Test. In Basic and Advanced Statistical Tests; Sense Publishers: Rotterdam, The Netherlands, 2017; pp. 13–16. [Google Scholar]
- Pacini, E.; Guarnieri, M.; Nepi, M. Pollen Carbohydrates and Water Content during Development, Presentation, and Dispersal: A Short Review. Protoplasma 2006, 228, 73–77. [Google Scholar] [CrossRef]
- Qin, Y.; Zhang, H.; Dai, Y.; Hou, H.; Dong, H. Effect of Alkali Treatment on Structure and Properties of High Amylose Corn Starch Film. Materials 2019, 12, 1705. [Google Scholar] [CrossRef]
- Gou, M.; Wu, H.; Saleh, A.S.M.; Jing, L.; Liu, Y.; Zhao, K.; Su, C.; Zhang, B.; Jiang, H.; Li, W. Effects of Repeated and Continuous Dry Heat Treatments on Properties of Sweet Potato Starch. Int. J. Biol. Macromol. 2019, 129, 869–877. [Google Scholar] [CrossRef]

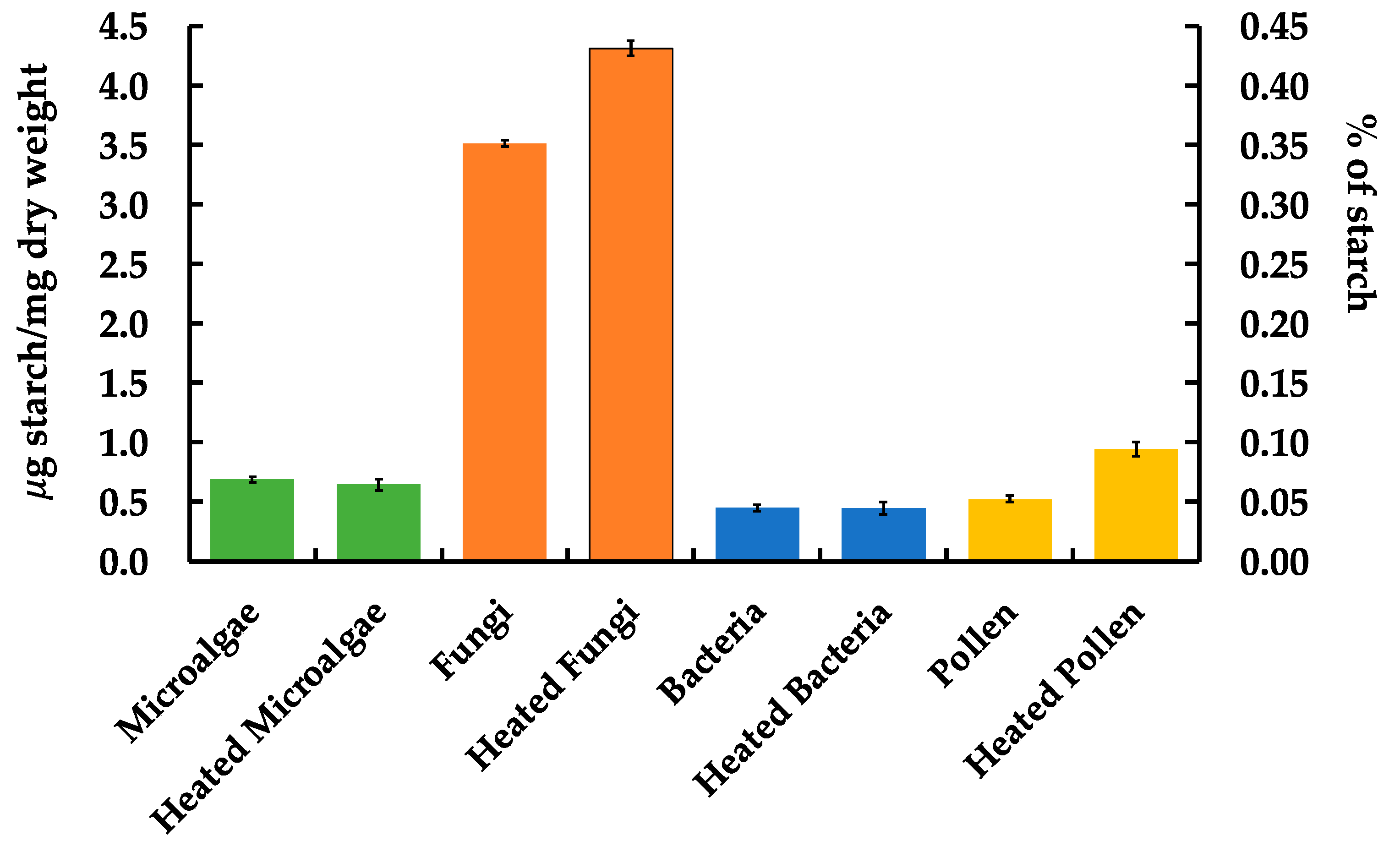
| Bioaerosol Type | Common Name | Botanical Name | Origin |
|---|---|---|---|
| Pollen | Lodgepole Pine | Pinus contorta | Collected in North Lake Tahoe, NV, USA (39°18′03″ N 119°55′22″ W) on 7 July 2020 |
| Fungi | Western Gall Rust | Endocronartium harknessii | Collected in Mt. Shasta, CA, USA on 31 May 2021 |
| Bacteria | Hay Bacillus | Bacillus subtilis | Cultured in the Molecular Microbial Ecology and Genomics Lab at the Desert Research Institute, NV, USA |
| Microalgae | Spirulina | Arthrospira platensis | Purchased commercially from Amazon, Inc. (Seattle, WA, USA) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bahdanovich, P.; Axelrod, K.; Khlystov, A.Y.; Samburova, V. Optimized Spectrophotometry Method for Starch Quantification. Analytica 2022, 3, 394-405. https://doi.org/10.3390/analytica3040027
Bahdanovich P, Axelrod K, Khlystov AY, Samburova V. Optimized Spectrophotometry Method for Starch Quantification. Analytica. 2022; 3(4):394-405. https://doi.org/10.3390/analytica3040027
Chicago/Turabian StyleBahdanovich, Palina, Kevin Axelrod, Andrey Y. Khlystov, and Vera Samburova. 2022. "Optimized Spectrophotometry Method for Starch Quantification" Analytica 3, no. 4: 394-405. https://doi.org/10.3390/analytica3040027
APA StyleBahdanovich, P., Axelrod, K., Khlystov, A. Y., & Samburova, V. (2022). Optimized Spectrophotometry Method for Starch Quantification. Analytica, 3(4), 394-405. https://doi.org/10.3390/analytica3040027








