Abstract
Chronic menstrual dysfunction and low female sex hormones adversely affect muscular performance in women but studies in college athletes are scarce. A cohort of 18 Japanese, female college athletes at the University of Tsukuba, Japan, were recruited and studied over 3 weeks under 2 conditions. One group had normal menstrual cycling (CYC, 9 athletes) while the other had irregular cycles (DYS, 9 athletes). Hormones and creatine kinase (CK) were measured from blood under both rest (RE) and exercise (EX) conditions. Biceps femoris tendon stiffness was measured by myometry. No differences in age, height, weight, menarche age, or one-repetition maximum weight existed between the groups. The DYS group had persistently low levels of estrogen and progesterone. In the CYC group, the CK level significantly increased at each point immediately post-exercise and 24 h post-exercise compared to pre-exercise in Weeks 1 and 2, and significantly increased at 24 h post-exercise compared to pre-exercise status in Week 3. The DYS group was significantly different only between pre-exercise and 24 h post-exercise over all 3 weeks. The DYS group also suffered from higher biceps femoris tendon stiffness at 24 h post-exercise. Chronic menstrual irregularities in Japanese college athletes increase muscle damage markers in the bloodstream and muscle stiffness after acute strength training.
1. Introduction
Resistance training enhances muscle function, controls muscular glucose metabolism, and contributes to stabilization that reduces musculoskeletal injury [1]. It also requires efficient repair of muscles after Type I injury (muscle soreness that occurs 24 to 48 h after unaccustomed exercise, also known as delayed onset muscle soreness, “DOMS”) for full recovery and subsequent increases in strength [2].
However, chronically late menstruation (cycles of longer than 39 days; oligomenorrhea) negatively affects muscle performance and recovery by stiffening muscles and increasing inflammation [3,4,5,6,7]. Around 33% of Japanese college athletes suffer from menstrual dysfunction and this issue globally affects female athletes [8,9]. Persistently low female sex hormones result in poor exercise recovery and, in women with oligomenorrhea, may decrease the advantages of acute strength training due to inefficient repair [4].
Women are prone to hamstring injury, and resistance training the biceps femoris to prevent injury is a key component of female sports conditioning [10,11], but this muscle accounts for high creatine phosphokinase activity after exercise [12]. Creatine kinase (CK), released into the blood from damaged muscle tissue, is a useful biomarker for the intensity of a strength exercise while musculoskeletal stiffness during recovery, especially in the post-exercise recovery phase, may be associated with injury risk and is a valuable measure of muscular performance [13,14]. Although studies on biomarkers of tendon damage have reported that inflammatory factors such as tumor necrosis factor (TNF)-α, IL-1β, IL-1α, and IFN-γ are elevated in tendinopathies and ligament injuries, these serum cytokines may be indicative of numerous other conditions (influenza, lupus, infection, etc.) and low estrogen has been shown to elevate serum inflammatory markers [15,16]. Therefore, muscle damage markers such as CK could be a less complex indicator of tendon condition, but studies on chronically low estrogen athletes with regard to tendon stiffness and muscle damage biomarkers have not been reported [17,18,19]. As stiffness is related to the risk of tendon injury, generating a relationship between CK and tendon condition may be valuable in accurate risk assessments for athletes with oligomenorrhea [14]. Diverse ways to measure stiffness exist, including isolated movement performance and muscular oscillation, but non-invasive myometry offers benefits for stationary, repetitive muscle measurements [20,21,22,23,24].
In this study, we hypothesized that oligomenorrheic athletes with chronically low estrogen and progesterone would have higher CK and biceps femoris tendon stiffness after acute strength training.
2. Results
2.1. Anthropometric Characteristics of the Participants
Participant characteristics are summarized in Supplemental Table S1. No significant differences were observed between the groups, but the average menstrual cycle length (in days) was significantly longer in the irregular menstrual cycle (DYS) group than the cycling (CYC) group (79.6 ± 13.3 vs. 31.0 ± 1.9 days, p < 0.01).
2.2. Hormone Levels of the Participants
Hormone profiles and amounts over the study period in each group are summarized in Supplemental Table S2. Both estradiol and progesterone levels are shown at the pre-condition level. The CYC group in Weeks 2 and 3 showed significantly higher estradiol levels than the DYS group in both exercise and rest conditions (Figure 1a,b). Progesterone levels in Week 3 were significantly higher in the CYC group but in the DYS group remained chronically low (Figure 1c,d). Exercise did not materially change estradiol or progesterone levels for either group over the three-week study period.
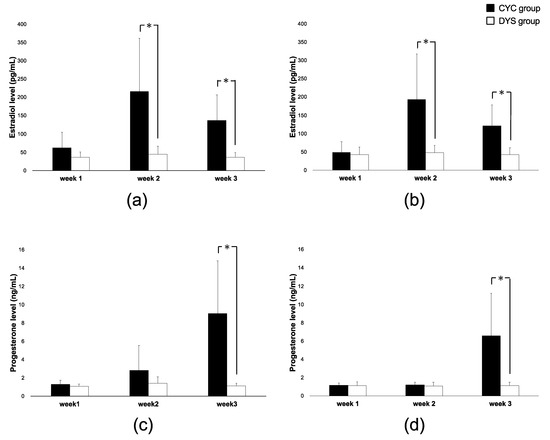
Figure 1.
Ovarian hormone levels over all weeks. (a) Estradiol concentration by group under exercise conditions, (b) estradiol concentration by group under rest conditions, (c) progesterone concentration by group under exercise conditions, and (d) progesterone concentration by group under rest conditions. The cycling (CYC) group is represented by black bars. The irregular menstrual cycle (DYS) group is represented by white bars. Error bars represent SD. * indicates p < 0.05 between CYC and DYS group in each condition.
2.3. CK Levels of the Participants
While no significant interactions in these groups with respect to time (groups x time) were observed, athletes in the DYS group experienced higher levels of serum creatine kinase over all timepoints (pre-workout, immediately after workout, and 24 h after workout) over the 3-week study period compared to the CYC group (Figure 2a–c). In the CYC group, the CK level significantly increased at each Post and Post 24 h point compared to Pre in Weeks 1 and 2 and significantly increased at Post 24 h compared to Pre in Week 3, while the DYS group was significantly different only between Pre and Post 24 h over all 3 weeks.
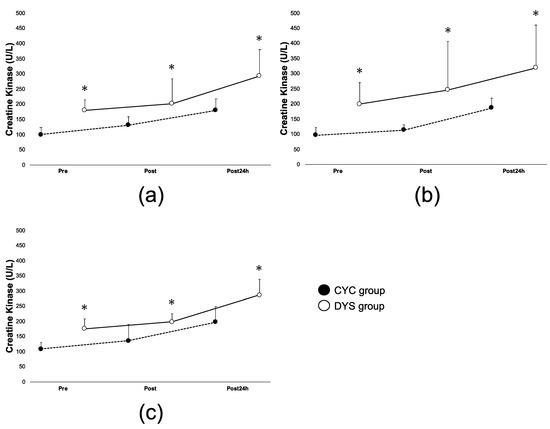
Figure 2.
Variance of creatine kinase (CK) levels in exercise (EX) condition. (a) CK level from pre-exercise to Post 24 h in Week 1, (b) CK level from pre-exercise to Post 24 h in Week 2, (c) CK level from pre-exercise to Post 24 h in Week 3. The CYC group is represented by black circle markers and the DYS group by white circle markers. Error bars represent SD. * indicates p < 0.05 between CYC and DYS group in time point.
2.4. Post-Exercise Stiffness in the Participants
Athletes in the DYS group had significantly higher stiffness in their biceps femoris tendon each week at the 24 h post-workout timepoint than the CYC group athletes (Figure 3a–c). Furthermore, stiffness in the DYS group was significantly elevated from Pre to Post 24 h over all 3 weeks.
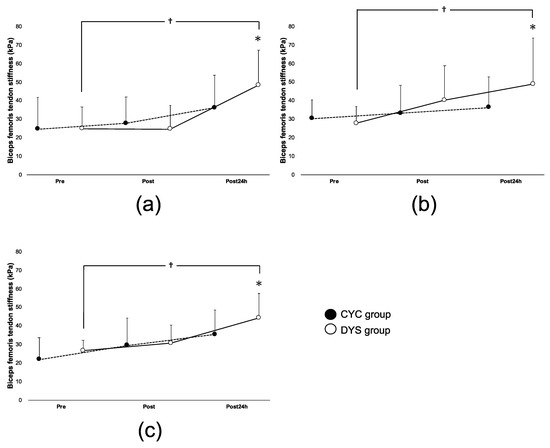
Figure 3.
Variance of biceps femoris tendon stiffness readings in EX condition. (a) Biceps tendon stiffness from pre-exercise to Post 24 h in Week 1, (b) biceps tendon stiffness from pre-exercise to Post 24 h in Week 2, (c) biceps tendon stiffness from pre-exercise to Post 24 h in Week 3. The CYC group is represented by black circle markers and the DYS group by white circle markers. Error bars represent SD. * indicates p < 0.05 between CYC and DYS group in time point. † indicates p < 0.05 compared to Pre and Post 24 h in DYS group.
3. Discussion
Our hypothesis was supported as athletes who experienced chronic menstrual delay also had higher levels of post-exercise CK and higher stiffness. As expected, we saw a higher basal level of CK at rest in the DYS group compared to the CYC group, as well as higher stiffness readings after the exercise challenge in the DYS group compared to the CYC group. This is similar to studies linking abnormal estrogen, tendon stiffness, and release of damage markers, such as CK, in the serum/local muscle through multiple modes of action [4,25,26,27].
We found that the acute, weekly exercise challenge did not affect levels of estrogen or progesterone in either group, an effect not seen in other studies [28,29]. As one mechanism for chronically low estrogen levels was reported to be a lack of body fat, thereby shutting down synthesis, the insignificant differences between CYC and DYS groups with regard to body composition cannot support this idea in our study [30]. We posit that the acute exercise, which was 90% of the 1-RM, was enough to generate CK but was not done often enough for a period of time long enough to cause the hormonal changes seen in other studies.
Stiffness readings were as predicted, with the DYS group showing higher levels at the Post 24 h timepoint over all 3 weeks. It is possible that we observed a delayed or incomplete exercise recovery and remodeling (as evidenced by stiffness) in the low-hormone group as seen in other studies with low-estrogen women [5,31,32]. Thus, evaluation of stiffness after Type I injury in oligomenorrheic women requires extended post-workout (24–48 h) measurements [33]. To the best of our knowledge, the present study is the first to track biceps femoris tendon stiffness after acute resistance training for up to 24 h after exercise in oligomenorrheic Japanese college athletes over a multi-week period.
This study had several strengths. First, this is the first study in Japanese college-level athletes to explore the effect of oligomenorrhea on muscular parameters. Second, this study was conducted in an ethnically homogenous population and is expected to be reflective of similar populations. However, limitations must be acknowledged. CK was chosen as a damage marker instead of cytokines since large muscles are reported to release CK upon injury [12]. Energy intake and expenditure were not measured during the study period, which may have affected our serum CK results. Future studies will need more participants as well as a minimum of 12 weeks of intervention while a study design with 3-factor ANOVA (groups × timepoints × 12 weeks as independent variables) will be essential for observing fluctuations in hormones from week to week. Next, this study had a small sample size, possibly imparting bias into the results. Additionally, assessing specific markers such as LD2, LD3, myoglobin, interleukin (IL)-6, IL-8, IL-10, and TNF -α may elucidate temporal mechanisms of acute strength exercise on inflammatory biomarkers that could impact athletic performance. However, this study adds to the scarce body of literature on the real effects of chronic menstrual dysfunction in Japanese college athletes and the utility of measuring muscle tendon stiffness using myometry instead of functional testing.
4. Materials and Methods
We measured blood estrogen/progesterone levels, CK, and tendon stiffness in 2 groups (eumenorrheic or oligomenorrheic college athletes) under rest or post-exercise conditions. Three time-points (before exercise, immediately after exercise, 24 h after exercise) were evaluated each week over a 3-week testing period.
A total of 22 female college athletes (ages 18–22) at the University of Tsukuba, Tsukuba, Japan, were recruited from July 2017 to March 2018. Eligible participants could have either normal menstrual cycles or irregular menstrual cycles (less than 5 times over the prior year). Of these, 1 was excluded from the study for inability to meet the measurement schedule and 3 were excluded for uncycled menstruation during the study period. All athletes were ethnically Japanese and anthropometric data can be found in Supplemental Table S1. Of the 18 remaining participants, 9 had normal menstrual cycles and 9 had delayed menstrual cycles. Inclusion criteria were as follows: physically healthy, non-smokers, familiar to resistance training, and not on medications (including oral contraceptives). All participants received an explanation of the purpose and flow of the study and signed an informed consent form prior to their inclusion. All aspects of the study were approved by the Ethics Committee of the University of Tsukuba in accordance with or equivalent to the principles of the Declaration of Helsinki (Approval #29–19).
Basal body temperature was used to categorize menstrual cycles [34]. Starting at least 1 month prior to the measurement, the participants recorded their basal body temperatures every morning upon awakening. Normal menstrual cycles (defined as a 28–35-day cycle) and ovulation were confirmed by a gynecologist based on basal body temperature data analysis and were used to retrospectively divide athletes into the 2 groups according to ovarian status at baseline: either women with normally cycling menses (CYC group, 9 athletes) or women with a dysfunctional cycle (DYS group, 9 athletes) [35]. We measured each week and considered the first measurement in 2 conditions (rest and exercise) as the Week 1 timepoint, the second measurement as the Week 2 timepoint, and the third measurement as the Week 3 timepoint. Athletes in the CYC group (who had cyclic menstruation 1 year prior to evaluation) started their first measurement (Week 1) on the first day without club activity at least 3 days post-menstruation.
All measurements were done on days with no sports activities and participants abstained from athletics 12 h before evaluation. Measurements for each athlete were taken under 2 conditions, rest (RE) and exercise (EX), over the 3-week period. During RE, athletes sat quietly for 60 min. For the exercise protocol, athletes performed an acute heavy resistance exercise test consisting of 6 sets of 5 parallel squats at 90% of their previously determined one-repetition maximum weight (1-RM) with a 3 min rest between each set [36]. Weight values from the 1-RM exercise are shown in Supplemental Table S1. A total of 6 measurement points per week for each participant (3 measurement points at rest and 3 for the exercise condition) to reflect changes in a standard 28–35-day cycle were collected.
Body composition, blood biochemistry (including ovarian hormones), creatine kinase, and stiffness of the biceps femoris tendon were measured before (Pre), immediately after (Post), and 24 h (Post 24 h) after both (RE and EX) conditions [37].
4.1. Measurements
Basal body temperature was measured orally using digital thermometers (CTEB503L, Citizen Co, Ltd., Tokyo, Japan). A bioimpedance-based body composition analyzer (BC-118E, Tanita Co., Ltd., Tokyo, Japan) was used to obtain anthropometric data consisting of weight, body fat mass, percent body fat, and body water volume. As bioimpedance testing depends on hydration status, participants were instructed to maintain hydration status before testing. Estradiol and progesterone were evaluated using a chemiluminescent enzyme immunoassay system as described previously (PATHFAST, LSI Medience Co., Tokyo, Japan) and CK levels were analyzed enzymatically by an independent laboratory (LSI Medience, Ibaraki, Japan) according to the transferable method of the Japan Society of Chemistry using serum samples. PATHFAST assay ranges were from 20–2000 pg/mL for estradiol and 0.2–40 ng/mL for progesterone. Stiffness of the biceps femoris tendon in all athletes at all timepoints was assessed by the same skilled athletic trainer using SOFTGRAM (Shinko Denshi, Co., Ltd., Tokyo, Japan). SOFTGRAM was developed to non-invasively evaluate tendon stiffness through spherical indentation and damped oscillation measurement with a demonstrated variability of 5% or less according to previous reports [20,23,24]. All equipment was operated according to the manufacturers’ instructions.
4.2. Statistical Analyses
All data are presented as means ± SDs. We used Statistical Package for the Social Sciences (SPSS) version 25.0 (IBM Inc., Armonk, NY, USA) and data were analyzed using group × time repeated-measure two-way ANOVA with Bonferroni’s post hoc test (if required). Mauchly’s test of sphericity was used to assess homogeneity of variance in the design of the measurement and a Greenhouse–Geisser correction was used if Mauchly’s test was significant. One-way ANOVA was used to analyze the significance in each group (CYC, DYS) in the exercised group (EX) over the time points (Pre, Post, Post 24 h). It was also used to compare each of the 3 weeks with respect to the same conditions/groups and a Bonferroni post hoc test was conducted when a significant main effect was shown. p < 0.05 was considered statistically significant. Hormone and tendon stiffness data for each week, condition, and group are summarized in Supplemental Table S2.
5. Conclusions
In conclusion, Japanese college athletes experiencing chronic menstrual delays may suffer from increased serum creatine kinase and resultant stiffness after acute strength training. Further studies in larger populations that mechanistically link chronically low sex hormones and athletic parameters in Asian women are needed. A large database of standard values (muscle stiffness, additional muscle damage markers, etc.) in Asian women would allow for more precise comparative studies with other ethnicities and we are currently planning larger studies to this effect. Our future studies will also check the effect of low estrogen on maximum weight and strength increases in this understudied population.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/women1020007/s1. Table S1: Anthropometric characteristics; Table S2: Hormone and tendon stiffness data for each week and group in Exercise condition.
Author Contributions
Conceptualization, A.S. and B.J.M.; methodology, A.S.; software, A.S. and Y.W.; validation, A.S., R.M., A.Z., Y.W. and B.J.M.; formal analysis, A.S. and B.J.M.; investigation, A.S. and R.M.; resources, A.S.; data curation, A.S. and B.J.M.; writing—original draft preparation, A.S. and B.J.M.; writing—review and editing, A.S., R.M., A.Z., Y.W. and B.J.M.; visualization, A.S. and Y.W.; supervision, B.J.M.; project administration, A.S.; funding acquisition, Y.W. All authors have read and agreed to the published version of the manuscript.
Funding
The Shinko Denshi Company, makers of the SOFTGRAM software used in this study, provided funding (grant # CFE29048). However, Shinko Denshi played no role in the study design, data collection/analysis, conclusions, manuscript preparation, or the decision to publish.
Institutional Review Board Statement
All aspects of this study were reviewed and approved by the Ethics Committee of the University of Tsukuba in accordance with or equivalent to the principles of the Declaration of Helsinki (Approval #29-19).
Informed Consent Statement
Informed consent (including the right to opt out) was obtained from all participants involved in the study.
Data Availability Statement
Data available on request due to privacy restrictions. The data presented in this study are available on request from the corresponding author.
Acknowledgments
The authors would like to thank the study volunteers for their participation. The Shinko Denshi Company, makers of the SOFTGRAM software used in this study, also provided funding (grant # CFE29048). However, Shinko Denshi played no role in the study design, data collection/analysis, conclusions, manuscript preparation, or the decision to publish.
Conflicts of Interest
Other than the above, the authors declare no other conflict of interest.
References
- Armstrong, M.J.; Colberg, S.R.; Sigal, R.J. Moving Beyond Cardio: The Value of Resistance Training, Balance Training, and Other Forms of Exercise in the Management of Diabetes. Diabetes Spectr. 2015, 28, 14–23. [Google Scholar] [CrossRef] [PubMed]
- Safran, M.R.; Seaber, A.V.; Garrett, W.E., Jr. Warm-up and muscular injury prevention. An update. Sports Med. 1989, 8, 239–249. [Google Scholar] [CrossRef] [PubMed]
- Adachi, T. Diagnosis, Treatment and Management of Gynecologic Disease. Acta Obstet. Gynaecol. Jpn. 2009, 61, N643–N657. [Google Scholar]
- Chidi-Ogbolu, N.; Baar, K. Effect of Estrogen on Musculoskeletal Performance and Injury Risk. Front. Physiol. 2019. [CrossRef]
- Enns, D.L.; Tiidus, P.M. The influence of estrogen on skeletal muscle: Sex matters. Sports Med. 2010, 40, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Pallavi, L.C.; De Souza, U.J.; Shivaprakash, G. Assessment of Musculoskeletal Strength and Levels of Fatigue during Different Phases of Menstrual Cycle in Young Adults. J. Clin. Diagnostic Res. 2017, 11, CC11–CC13. [Google Scholar] [CrossRef]
- Tiidus, P.M.; Holden, D.; Bombardier, E.; Zajchowski, S.; Enns, D.; Belcastro, A. Estrogen effect on post-exercise skeletal muscle neutrophil infiltration and calpain activity. Can. J. Physiol. Pharmacol. 2001, 79, 400–406. [Google Scholar] [CrossRef]
- Nose, S.; Dohi, M.; Namba, A.; Akimori, K.; Mesaki, N.; Komatsu, Y.; Akama, T.; Kawahara, T. Investigation of amenorrhea and stress fractures in elite female athletes. Jpn. J. Soc. Clin. Sports Med. 2014, 22, 67–74. [Google Scholar]
- Sawai, A.; Tochigi, Y.; Kavaliova, N.; Zaboronok, A.; Warashina, Y.; Mathis, B.J.; Mesaki, N.; Shiraki, H.; Watanabe, K. MRI reveals menstrually-related muscle edema that negatively affects athletic agility in young women. PLoS ONE 2018, 13, e0191022. [Google Scholar] [CrossRef]
- Bell, D.R.; Blackburn, J.T.; Norcorss, M.F.; Ondrak, K.S.; Hudson, J.D.; Hackney, A.C.; Padua, D.A. Estrogen and muscle stiffness have a negative relationship in females. Knee Surg. Sports Traumatol. Arthrosc. 2012, 20, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Blackburn, J.T.; Bell, D.R.; Norcross, M.F.; Hudson, J.D.; Kimsey, M.H. Sex comparison of hamstring structural and material properties. Clin. Biomech. 2009, 24, 65–70. [Google Scholar] [CrossRef]
- Doize, F.; Laporte, R.; Deroth, L. Effects of exercise on skeletal muscle and serum enzyme activities in pigs. Vet. Res. Commun. 1989, 13, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Baird, M.F.; Graham, S.M.; Baker, J.S.; Bickerstaff, G.F. Creatine-kinase- and exercise-related muscle damage implications for muscle performance and recovery. J. Nutr. Metab. 2012. [Google Scholar] [CrossRef] [PubMed]
- Lorimer, A.V.; Hume, P.A. Stiffness as a Risk Factor for Achilles Tendon Injury in Running Athletes. Sports Med. 2016, 46, 1921–1938. [Google Scholar] [CrossRef] [PubMed]
- Dragin, N.; Nancy, P.; Villegas, J.; Roussin, R.; Le Panse, R.; Berrih-Aknin, S. Balance between Estrogens and Proinflammatory Cytokines Regulates Chemokine Production Involved in Thymic Germinal Center Formation. Sci. Rep. 2017, 7, 7970. [Google Scholar] [CrossRef]
- Shivers, K.Y.; Amador, N.; Abrams, L.; Hunter, D.; Jenab, S.; Quiñones-Jenab, V. Estrogen alters baseline and inflammatory-induced cytokine levels independent from hypothalamic-pituitary-adrenal axis activity. Cytokine 2015, 72, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Gaida, J.E.; Alfredson, H.; Forsgren, S.; Cook, J.L. A pilot study on biomarkers for tendinopathy: Lower levels of serum TNF-α and other cytokines in females but not males with Achilles tendinopathy. BMC Sports Sci. Med. Rehabil. 2016, 8, 5. [Google Scholar] [CrossRef]
- Harkey, M.S.; Luc, B.A.; Golightly, Y.M.; Thomas, A.C.; Driban, J.B.; Hackney, A.C.; Pietrosimone, B. Osteoarthritis-related biomarkers following anterior cruciate ligament injury and reconstruction: A systematic review. Osteoarthr. Cartil. 2015, 23, 1–12. [Google Scholar] [CrossRef]
- Lattermann, C.; Conley, C.E.; Johnson, D.L.; Reinke, E.K.; Huston, L.J.; Huebner, J.L.; Chou, C.H.; Kraus, V.B.; Spindler, K.P.; Jacobs, C.A. Select Biomarkers on the Day of Anterior Cruciate Ligament Reconstruction Predict Poor Patient-Reported Outcomes at 2-Year Follow-Up: A Pilot Study. BioMed. Res. Int. 2018, 2018, 9387809. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.N.; Li, Y.P.; Liu, C.L.; Zhang, Z.J. Assessing the elastic properties of skeletal muscle and tendon using shearwave ultrasound elastography and MyotonPRO. Sci. Rep. 2018, 8, 1–9. [Google Scholar] [CrossRef]
- Zinder, S.M.; Padua, D.A. Reliability, Validity, and Precision of a Handheld Myometer for Assessing in Vivo Muscle Stiffness. J. Sport Rehabil. 2011, 20, jsr.2010-0051. [Google Scholar] [CrossRef]
- Pruyn, E.C.; Watsford, M.L.; Murphy, A.J. Validity and reliability of three methods of stiffness assessment. J. Sport Health Sci. 2016, 5, 476–483. [Google Scholar] [CrossRef]
- Tani, M.; Sakuma, A. Applicability Evaluation of Young’s Modulus Measurement using Equivalent Indentation Strain in Spherical Indentation Testing for Soft Materials. Trans. Jpn. Soc. Mech. Eng. A 2010, 76, 102–108. [Google Scholar] [CrossRef][Green Version]
- Tani, M.; Sakuma, A.; Shinomiya, M. Evaluation of Thickness and Young’s Modulus of Soft Materials by using Spherical Indentation Testing. Trans. Jpn. Soc. Mech. Eng. A 2009, 75, 901–908. [Google Scholar] [CrossRef]
- Kubo, K.; Miyamoto, M.; Tanaka, S.; Maki, A.; Tsunoda, N.; Kanehisa, H. Muscle and tendon properties during menstrual cycle. Int. J. Sports Med. 2009, 30, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Lowe, D.A.; Baltgalvis, K.A.; Greising, S.M. Mechanisms behind estrogen’s beneficial effect on muscle strength in females. Exerc. Sport Sci. Rev. 2010, 38, 61–67. [Google Scholar] [CrossRef]
- Zhao, Z.; Han, F.; Yang, S.; Wu, J.; Zhan, W. Oxamate-mediated inhibition of lactate dehydrogenase induces protective autophagy in gastric cancer cells: Involvement of the Akt–mTOR signaling pathway. Cancer Lett. 2015, 358, 17–26. [Google Scholar] [CrossRef]
- Copeland, J.L.; Consitt, L.A.; Tremblay, M.S. Hormonal responses to endurance and resistance exercise in females aged 19-69 years. J. Gerontol. A Biol. Sci. Med. Sci. 2002, 57, B158–B165. [Google Scholar] [CrossRef]
- McTiernan, A.; Tworoger, S.S.; Ulrich, C.M.; Yasui, Y.; Irwin, M.L.; Rajan, K.B.; Sorensen, B.; Rudolph, R.E.; Bowen, D.; Stanczyk, F.Z.; et al. Effect of exercise on serum estrogens in postmenopausal women: A 12-month randomized clinical trial. Cancer Res. 2004, 64, 2923–2928. [Google Scholar] [CrossRef]
- De Crée, C. Sex steroid metabolism and menstrual irregularities in the exercising female. A review. Sports Med. 1998, 25, 369–406. [Google Scholar] [CrossRef]
- Hansen, M.; Skovgaard, D.; Reitelseder, S.; Holm, L.; Langbjerg, H.; Kjaer, M. Effects of Estrogen Replacement and Lower Androgen Status on Skeletal Muscle Collagen and Myofibrillar Protein Synthesis in Postmenopausal Women. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 1005–1013. [Google Scholar] [CrossRef]
- Hansen, M. Female hormones: Do they influence muscle and tendon protein metabolism? Proc. Nutr. Soc. 2018, 77, 32–41. [Google Scholar] [CrossRef]
- Doma, K.; Nicholls, A.; Gahreman, D.; Damas, F.; Libardi, C.A.; Sinclair, W. The Effect of a Resistance Training Session on Physiological and Thermoregulatory Measures of Sub-maximal Running Performance in the Heat in Heat-Acclimatized Men. Sports Med. Open 2019, 5, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fukaya, K.; Kawamori, A.; Osada, Y.; Kitazawa, M.; Ishiguro, M. The forecasting of menstruation based on a state-space modeling of basal body temperature time series. Stat. Med. 2017, 36, 3361–3379. [Google Scholar] [CrossRef] [PubMed]
- Vanheest, J.L.; Rodgers, C.D.; Mahoney, C.E.; De Souza, M.J. Ovarian suppression impairs sport performance in junior elite female swimmers. Med. Sci. Sports Exerc. 2014, 46, 156–166. [Google Scholar] [CrossRef] [PubMed]
- Wolf, M.R.; Fragala, M.S.; Volek, J.S.; Denegar, C.R.; Anderson, J.M.; Comstock, B.A.; Dunn-Lewis, C.; Hooper, D.R.; Szivak, T.K.; Luk, H.Y.; et al. Sex differences in creatine kinase after acute heavy resistance exercise on circulating granulocyte estradiol receptors. Eur. J. Appl. Physiol. 2012, 112, 3335–3340. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, B.M.; Dantas, E.; de Salles, B.F.; Miranda, H.; Koch, A.J.; Willardson, J.M.; Simão, R. Creatine kinase and lactate dehydrogenase responses after upper-body resistance exercise with different rest intervals. J. Strength Cond. Res. 2010, 24, 1657–1662. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).