Surface Characterization of Powdered Cellulose Activated by Potassium Hydroxide in Dry Condition Through Ball Milling
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Powdered Cellulose through Ball Milling
2.3. Co-Milling Powdered Cellulose with Potassium Hydroxide through Ball Milling
2.4. Characterization of Cellulose Powder Samples
2.4.1. X-Ray Photoelectron Spectroscopy (XPS)
2.4.2. Attenuated Total Reflectance-Fourier Transform Infrared Spectroscopy (ATR-FTIR)
3. Results and Discussion
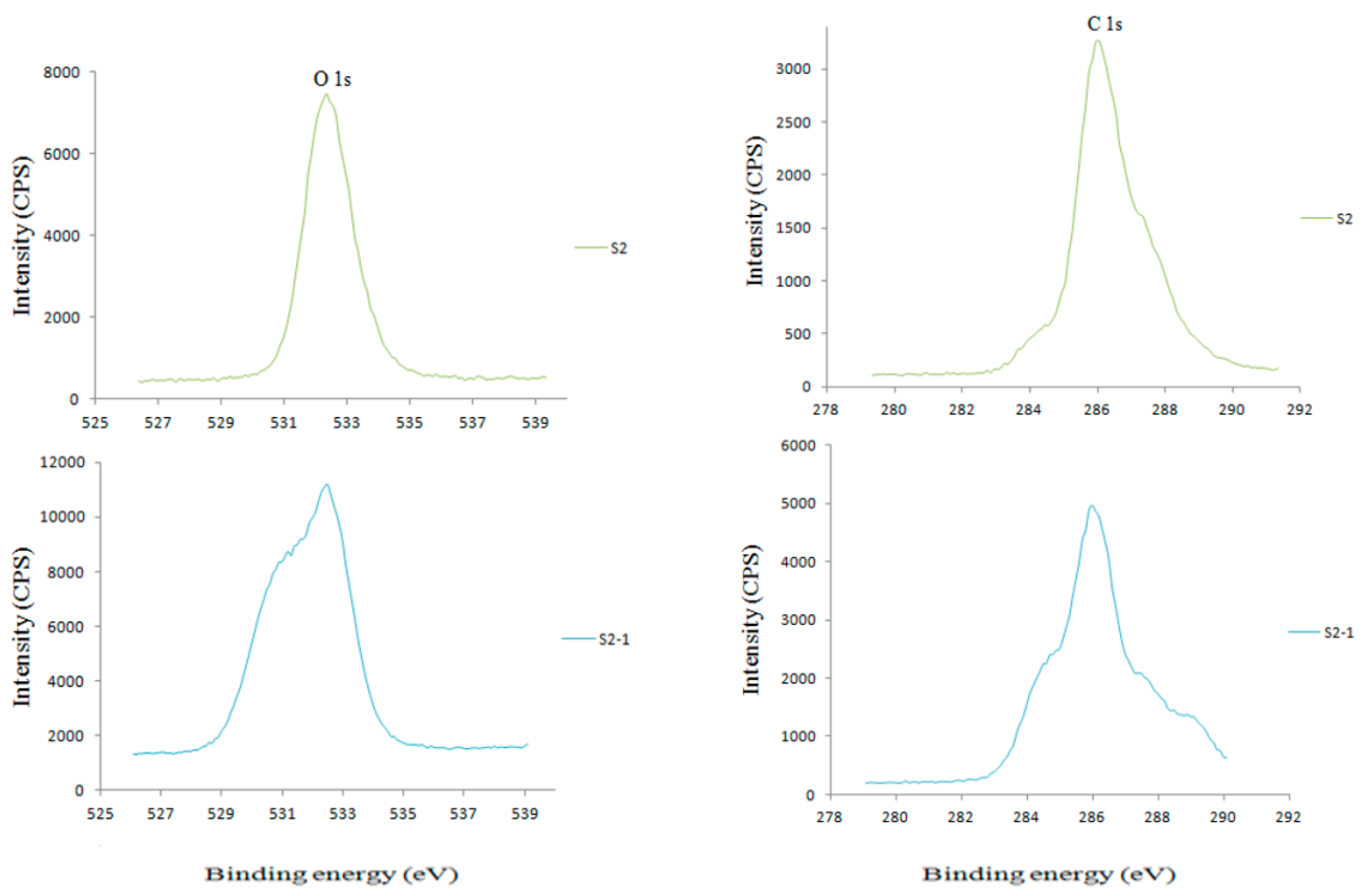
3.1. XPS Analysis
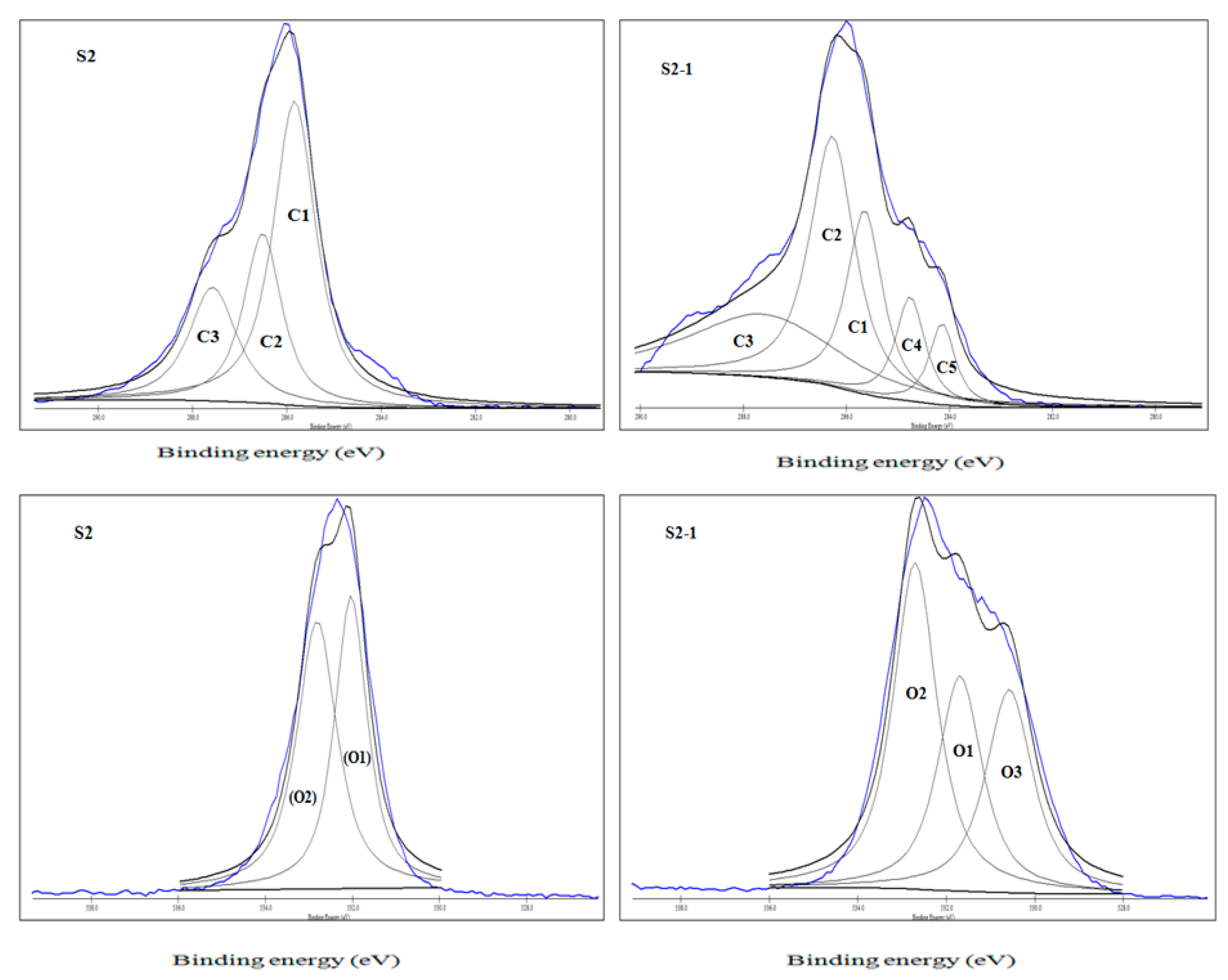
3.1.1. C1s Spectra: Deconvoluted Peaks Analysis
3.1.2. O1s Spectra: Deconvoluted Peaks
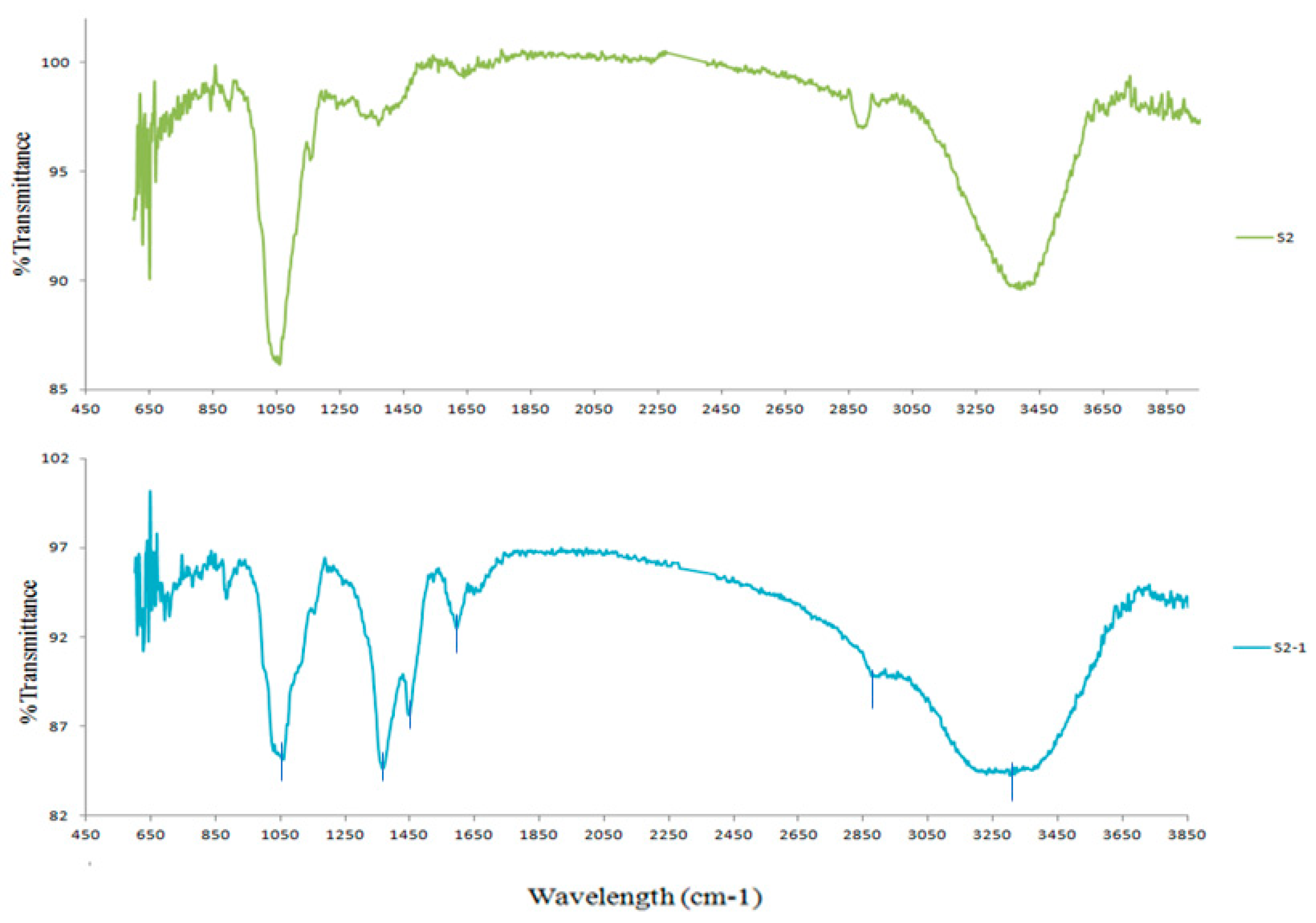
3.2. ATR-FTIR Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stütz, A.E. Glycoscience: Epimerisation, Isomerisation and Rearrangement Reactions of Carbohydrates; Springer: Berlin/Heidelberg, Germany, 2001. [Google Scholar]
- Christiansen, J.A. Hypotheses concerning the transition states in mutarotation of pyranoses: To VK La Mer on his seventieth birthday by his friend. J. Colloid Interface Sci. 1966, 1, 1–2. [Google Scholar] [CrossRef]
- Lemieux, R.U.; Stevens, J.D. The proton magnetic resonance spectra and tautomeric equilibria of aldoses in deuterium oxide. Can. J. Chem. 1966, 3, 249–262. [Google Scholar] [CrossRef]
- Ward, P.; Isbell, H. Mutarotation of sugars in solution: Part I: History, basic kinetics, and composition of sugar solutions. Adv. Carbohydr. Chem. 1968, 23, 11–57. [Google Scholar]
- Le Barch, N.; Grossel, J.M.; Looten, P.; Mathlouthi, M. Kinetic study of the mutarotation of D-glucose in concentrated aqueous solution by gas-liquid chromatography. Food Chem. 2001, 1, 119–124. [Google Scholar] [CrossRef]
- Rony, P.R. Polyfunctional catalysis. I. Activation parameters for the mutarotation of tetramethyl-D-glucose in benzene. J. Am. Chem. Soc. 1968, 11, 2824–2831. [Google Scholar] [CrossRef]
- Fiandanese, V.; Naso, F. Benzamidine-catalysed mutarotation of 2, 3, 4, 6-tetra-O-methyl-D-glucose. J. Chem. Soc. Perkin Trans. 1977, 8, 1047–1051. [Google Scholar] [CrossRef]
- Morpurgo, S.; Grandi, A.; Zazza, C.; Bossa, M. A theoretical study on the sugars’ mutarotation: The epimerisation of 2-tetrahydropyranol catalysed by formamidine, benzamidine and by the 2-aminopyridine/2-iminopyridine tautomeric couple. J. Mol. Struct. THEOCHEM 2005, 1, 71–82. [Google Scholar] [CrossRef]
- Rony, P.R.; McCormac, W.E.; Wunderly, S.W. Polyfunctional catalysis. II. General base catalysis of the mutarotation of tetramethyl-D-gluose in benzene and methanol-benzene. J. Am. Chem. Soc. 1969, 15, 4244–4251. [Google Scholar] [CrossRef]
- Brown, G.M.; Levy, H.A. α-d-Glucose: Further refinement based on neutron-diffraction data. Acta Crystallographica Sect. B Struct. Crystallogr. Cryst. Chem. 1979, 3, 656–659. [Google Scholar] [CrossRef]
- Fisher, J. Modern NMR Techniques for Synthetic Chemistry; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Dujardin, N.; Willart, J.F.; Dudognon, E.; Danede, F.; Descamps, M. First obtaining of glass solutions and phase diagram of glucose with fully tunable anomeric concentration. J. Pharm. Sci. 2010, 3, 1476–1483. [Google Scholar] [CrossRef]
- Dujardin, N.; Willart, J.F.; Dudognon, E.; Hédoux, A.; Guinet, Y.; Paccou, L.; Chazallon, B.; Descamps, M. Solid state vitrification of crystalline α and β-D-glucose by mechanical milling. Solid State Commun. 2008, 1, 78–82. [Google Scholar] [CrossRef]
- Willart, J.F.; Carpentier, L.; Danède, F.; Descamps, M. Solid-state vitrification of crystalline griseofulvin by mechanical milling. J. Pharm. Sci. 2012, 4, 1570–1577. [Google Scholar] [CrossRef] [PubMed]
- Broido, A.; Houminer, Y.; Patai, S. Pyrolytic reactions of carbohydrates. Part I. Mutarotation of molten D-glucose. J. Chem. Soc. B Phys. Org. 1966, 411–414. [Google Scholar] [CrossRef]
- Ribeiro, L.S.; Órfão, J.J.; Pereira, M.F.R. Enhanced direct production of sorbitol by cellulose ball-milling. Green Chem. 2015, 5, 2973–2980. [Google Scholar] [CrossRef]
- Paakkari, T.; Serimaa, R.; Fink, H.P. Structure of amorphous cellulose. Acta Polymerica 1989, 12, 731–734. [Google Scholar] [CrossRef]
- Jiang, J.; Wang, J.; Zhang, X.; Wolcott, M. Evaluation of physical structural features on influencing enzymatic hydrolysis efficiency of micronized wood. RSC Adv. 2016, 105, 103026–103034. [Google Scholar] [CrossRef]
- Mikushina, I.V.; Troitskaya, I.B.; Dushkin, A.V.; Bazarnova, N.G. Changes in the chemical composition of wood during mechanochemical treatment. Chem. Sust. Dev. 2002, 4, 441–445. [Google Scholar]
- Azadfar, M.; Graham, M.R.; Wolcott, M.P. Effect of Cellulose Reducing Ends on the Reinforcing Capacity of Powdered Cellulose in Polypropylene Composites. J. Compos. Sci. 2019, 3, 98. [Google Scholar] [CrossRef]
- Yaylayan, V.A.; Ismail, A.A. Investigation of the enolization and carbonyl group migration in reducing sugars by FTIR spectroscopy. Carbohydr. Res. 1995, 2, 253–265. [Google Scholar] [CrossRef]
- Shaffer, P.A.; Friedemann, T.E. Sugar activation by alkali I. Formation of lactic and saccharinic acids. J. Biol. Chem. 1930, 1, 345–374. [Google Scholar]
- Braun, M. Modern Enolate Chemistry: From Preparation to Applications in Asymmetric Synthesis; John Wiley & Sons: Hoboken, NJ, USA, 2015. [Google Scholar]
- Sinnott, M. Carbohydrate Chemistry and Biochemistry: Structure and Mechanism; Royal Society of Chemistry: London, UK, 2007. [Google Scholar]
- Menuel, S.; Doumert, B.; Saitzek, S.; Ponchel, A.; Delevoye, L.; Monflier, E.; Hapiot, F. Selective secondary face modification of cyclodextrins by mechanosynthesis. J. Org. Chem. 2015, 12, 6259–6266. [Google Scholar] [CrossRef] [PubMed]
- Matuana, L.M.; Balatinecz, J.J.; Park, C.B.; Sodhi, R.N.S. X-ray photoelectron spectroscopy study of silane-treated newsprint-fibers. Wood Sci. Technol. 1999, 4, 259–270. [Google Scholar] [CrossRef]
- Kovac, J. Surface characterization of polymers by XPS and SIMS techniques. Mater. Tehnol. 2011, 45, 191–197. [Google Scholar]
- Azadfar, M.; Gao, A.H.; Bule, M.V.; Chen, S. Structural characterization of lignin: A potential source of antioxidants guaiacol and 4-vinylguaiacol. Int. J. Biol. Macromol. 2015, 75, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Kamdem, D.P.; Riedl, B.; Adnot, A.; Kaliaguine, S. ESCA spectroscopy of poly (methyl methacrylate) grafted onto wood fibers. J. Appl. Polym. Sci. 1991, 10, 1901–1912. [Google Scholar] [CrossRef]
- Johansson, L.S.; Campbell, J.M. Reproducible XPS on biopolymers: Cellulose studies. Surf. Interface Anal. 2004, 8, 1018–1022. [Google Scholar] [CrossRef]
- Johansson, L.S.; Campbell, J.M.; Hänninen, T.; Ganne-Chèdeville, C.; Vuorinen, T.; Hughes, M.; Laine, J. XPS and the medium-dependent surface adaptation of cellulose in wood. Surf. Interface Anal. 2012, 8, 899–903. [Google Scholar] [CrossRef]
- Stark, N.M.; Matuana, L.M. Surface chemistry changes of weathered HDPE/wood-flour composites studied by XPS and FTIR spectroscopy. Polym. Degrad. Stab. 2004, 1, 1–9. [Google Scholar] [CrossRef]
- Liu, F.P.; Rials, T.G.; Simonsen, J. Relationship of wood surface energy to surface composition. Langmuir 1998, 2, 536–541. [Google Scholar] [CrossRef]
- Nzokou, P.; Kamdem, D.P. X-ray photoelectron spectroscopy study of red oak-(Quercus rubra), black cherry-(Prunus serotina) and red pine-(Pinus resinosa) extracted wood surfaces. Surf. Interface Anal. 2005, 8, 689–694. [Google Scholar] [CrossRef]
- Pizzi, A.; Mittal, K.L. Handbook of Adhesive Technology, Revised and Expanded; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Sun, X.F.; Sun, R.; Sun, J.X. Acetylation of rice straw with or without catalysts and its characterization as a natural sorbent in oil spill cleanup. J. Agric. Food Chem. 2002, 22, 6428–6433. [Google Scholar] [CrossRef] [PubMed]
- Khalil, H.A.; Ismail, H.; Rozman, H.D.; Ahmad, M.N. The effect of acetylation on interfacial shear strength between plant fibers and various matrices. Eur. Polym. J. 2001, 5, 1037–1045. [Google Scholar] [CrossRef]
- Huang, L.; Ye, Z.; Berry, R. Modification of cellulose nanocrystals with quaternary ammonium-containing hyperbranched polyethylene ionomers by ionic assembly. ACS Sustain. Chem. Eng. 2016, 9, 4937–4950. [Google Scholar] [CrossRef]

| Sample | % C | % O | % K | O/C |
|---|---|---|---|---|
| S2 | 57.36 | 42.64 | 0.74 | |
| S2-1 | 40.76 | 41.4 | 9.79 | 1.01 |
| Group | Symbol | Carbon Atom or Oxygen Atom Bonded to |
|---|---|---|
| Carbon | ||
| I | C1 | C-C/C-H |
| II | C2 | C-OH |
| III | C3 | C=O, O-C-O |
| IV | C4 | -C*=C(OH)-CHO -C=C*(OH)-CHO -C-C(O)-C*HO |
| V | C5 | |
| Oxygen | ||
| I | O1 | C=O*: (-(C6H10O5)-)n C-O*-C: (-(C6H10O5)-)n |
| II | O2 | |
| III | O3 | -C=C(OH)-CHO* -C-C(O*)-CHO |
| Carbon Type | Binding Energy (eV) | Intensity (CPS) |
|---|---|---|
| S2 | ||
| C1 | 285.834 | 3082 |
| C2 | 286.511 | 2573.33 |
| C3 | 287.568 | 1423.33 |
| S2-1 | ||
| C1 | 285.645 | 3998 |
| C2 | 286.273 | 4782.66 |
| C3 | 287.632 | 2012.66 |
| C4 | 284.761 | 2399.33 |
| C5 | 284.139 | 1748 |
| Oxygen Type | Binding Energy (eV) | Intensity (CPS) |
|---|---|---|
| S2 | ||
| O1 | 532.032 | 6880 |
| O2 | 532.812 | 5944.44 |
| S2-1 | ||
| O1 | 531.691 | 9206.63 |
| O2 | 532.706 | 10,463.55 |
| O3 | 530.577 | 7575.66 |
| Assignment | Cellulose Powder Samples | |
|---|---|---|
| λ (cm−1) | T (%) | |
| –OH stretching vibrations | 3380 (S2), 3290(S2-1) | 89.6(S2), 84.2(S2-1) |
| –CH stretching vibrations (-CH2-, -CH3) | 2900 (S2), 2890 (S2-1) | 97.1 (S2), 89.7 (S2-1) |
| C=C stretching vibrations | 1590 (S2-1) | 92.90 (S2-1) |
| -C-H stretching vibrations | 1440–1370 (S2-1) | 87.6–84.6 (S2-1) |
| CH2 and C-H bending in cellulose | 1370 (S2) | 97.1 (S2) |
| C-O and C-O-C stretching vibrations in cellulose | 1050 (S2), 1050 (S2-1) | 86.2 (S2), 85.2 (S2-1) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Azadfar, M.; Wolcott, M.P. Surface Characterization of Powdered Cellulose Activated by Potassium Hydroxide in Dry Condition Through Ball Milling. Polysaccharides 2020, 1, 80-89. https://doi.org/10.3390/polysaccharides1010006
Azadfar M, Wolcott MP. Surface Characterization of Powdered Cellulose Activated by Potassium Hydroxide in Dry Condition Through Ball Milling. Polysaccharides. 2020; 1(1):80-89. https://doi.org/10.3390/polysaccharides1010006
Chicago/Turabian StyleAzadfar, Mohammadali, and Michael P. Wolcott. 2020. "Surface Characterization of Powdered Cellulose Activated by Potassium Hydroxide in Dry Condition Through Ball Milling" Polysaccharides 1, no. 1: 80-89. https://doi.org/10.3390/polysaccharides1010006
APA StyleAzadfar, M., & Wolcott, M. P. (2020). Surface Characterization of Powdered Cellulose Activated by Potassium Hydroxide in Dry Condition Through Ball Milling. Polysaccharides, 1(1), 80-89. https://doi.org/10.3390/polysaccharides1010006






