Odonata (Insecta) Communities in a Lowland Mixed Mosaic Forest in Central Kalimantan, Indonesia
Abstract
1. Introduction
2. Materials and Methods
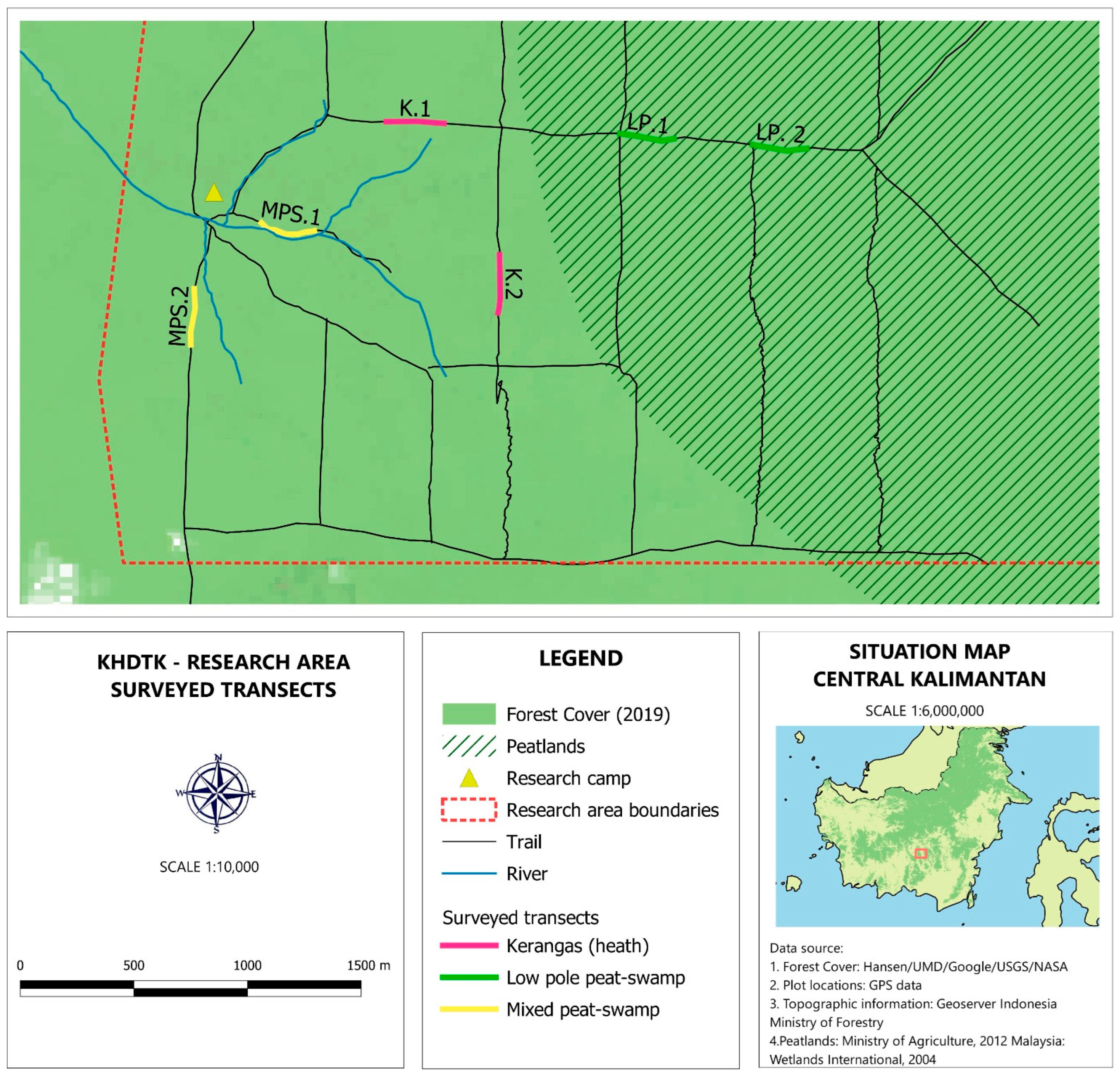
2.1. Site Description
2.2. Odonata Sampling and Sampling Design
2.3. Environmental Variables
2.4. Data Analysis
3. Results
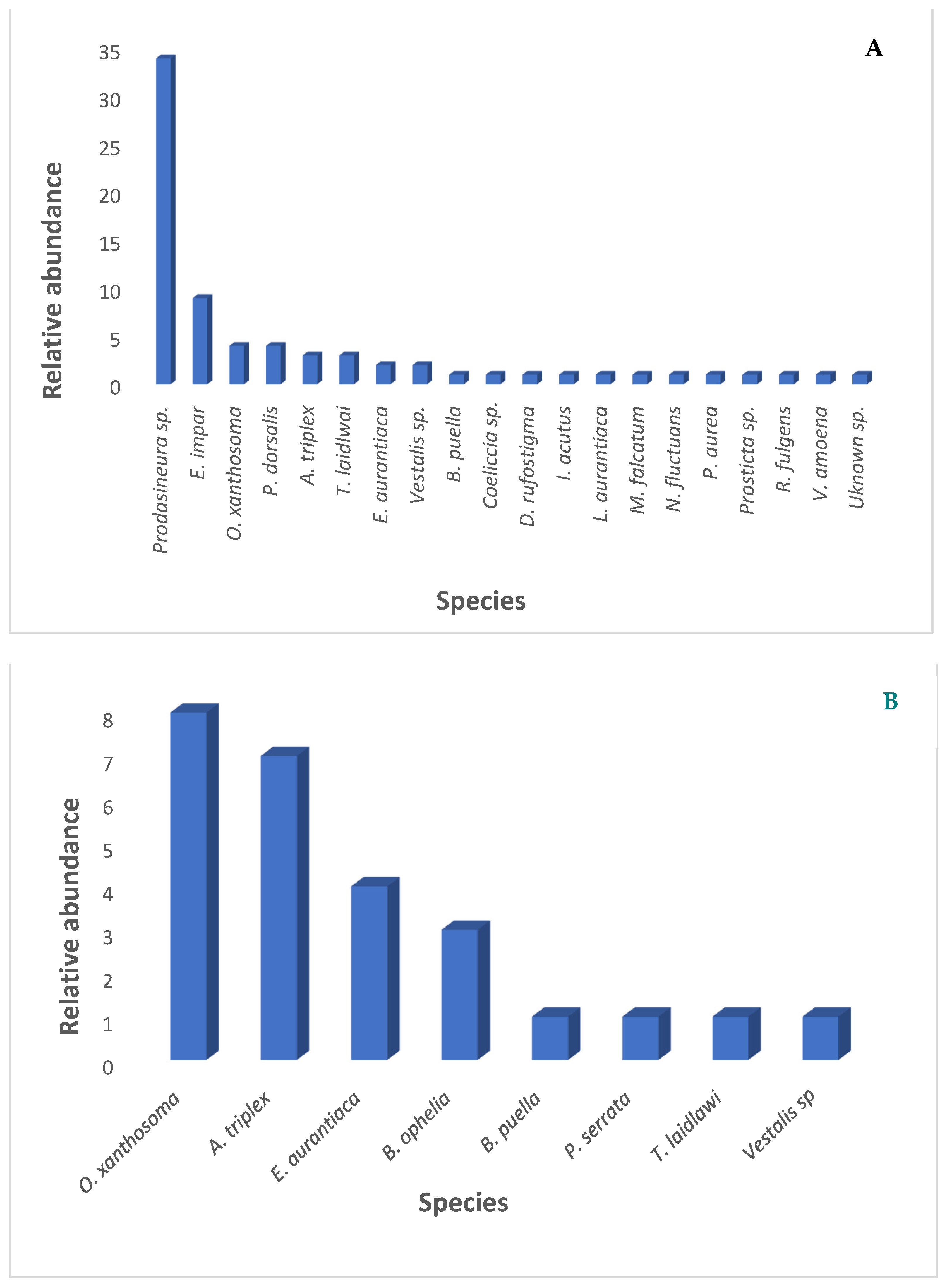
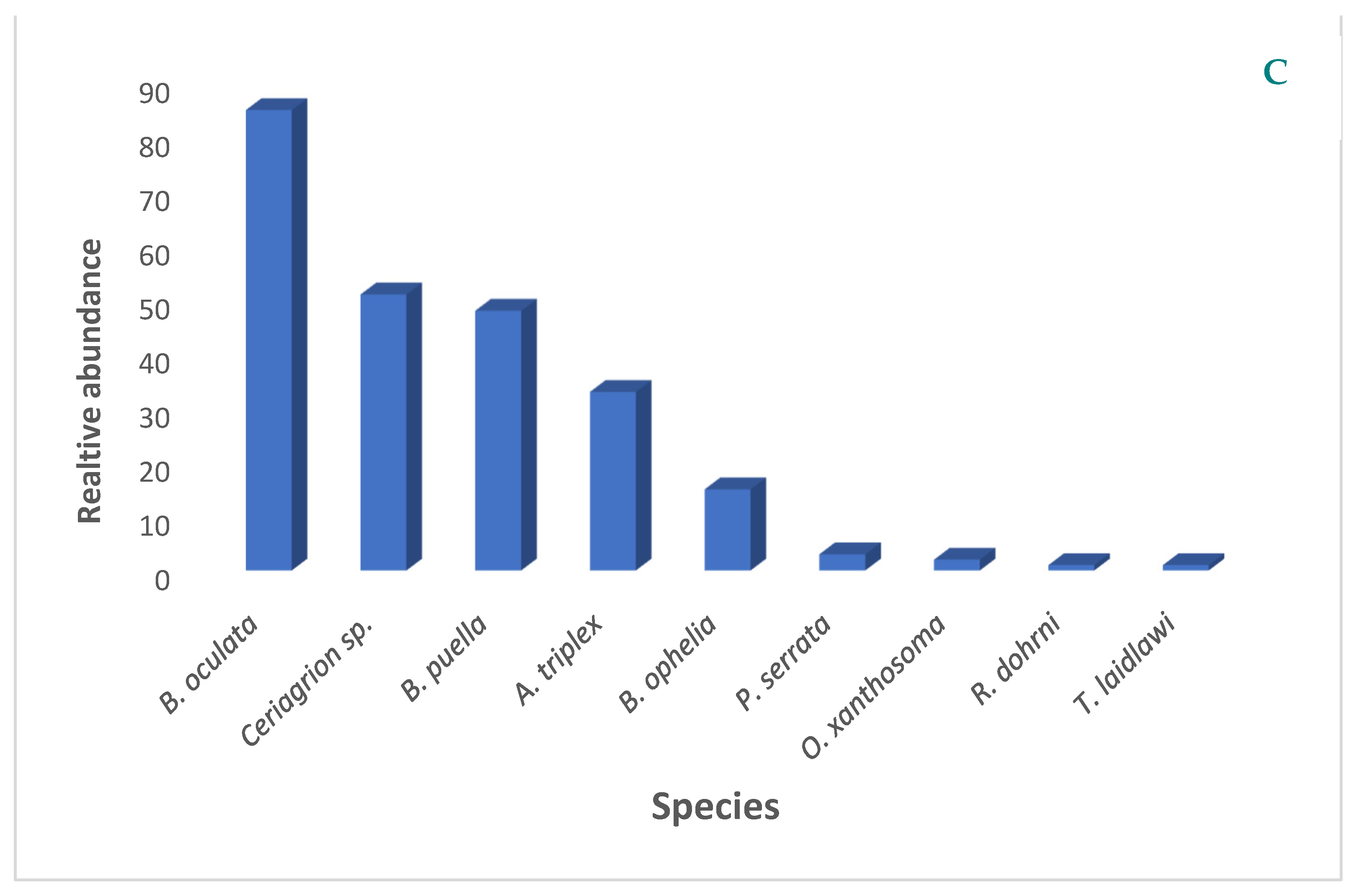
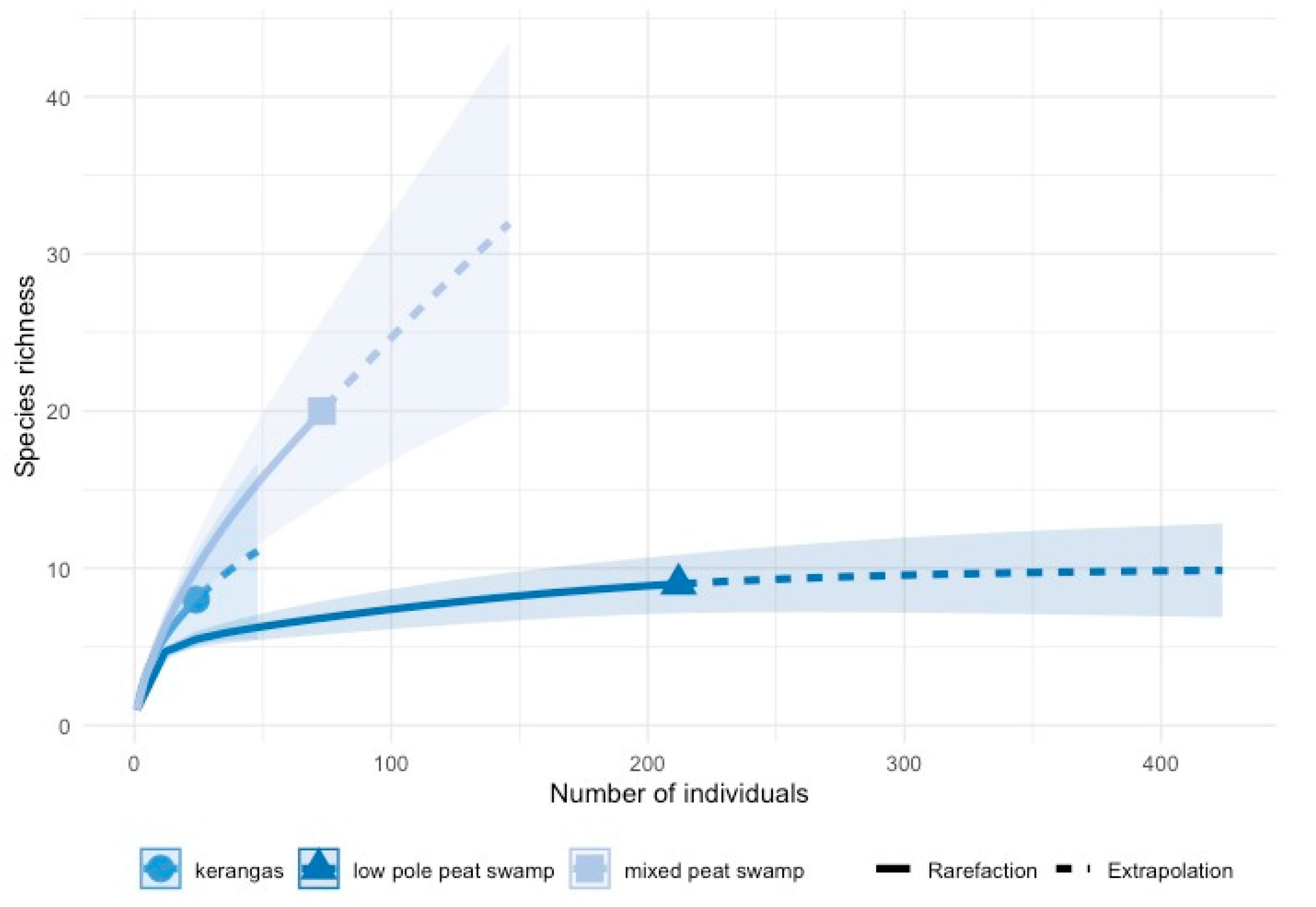
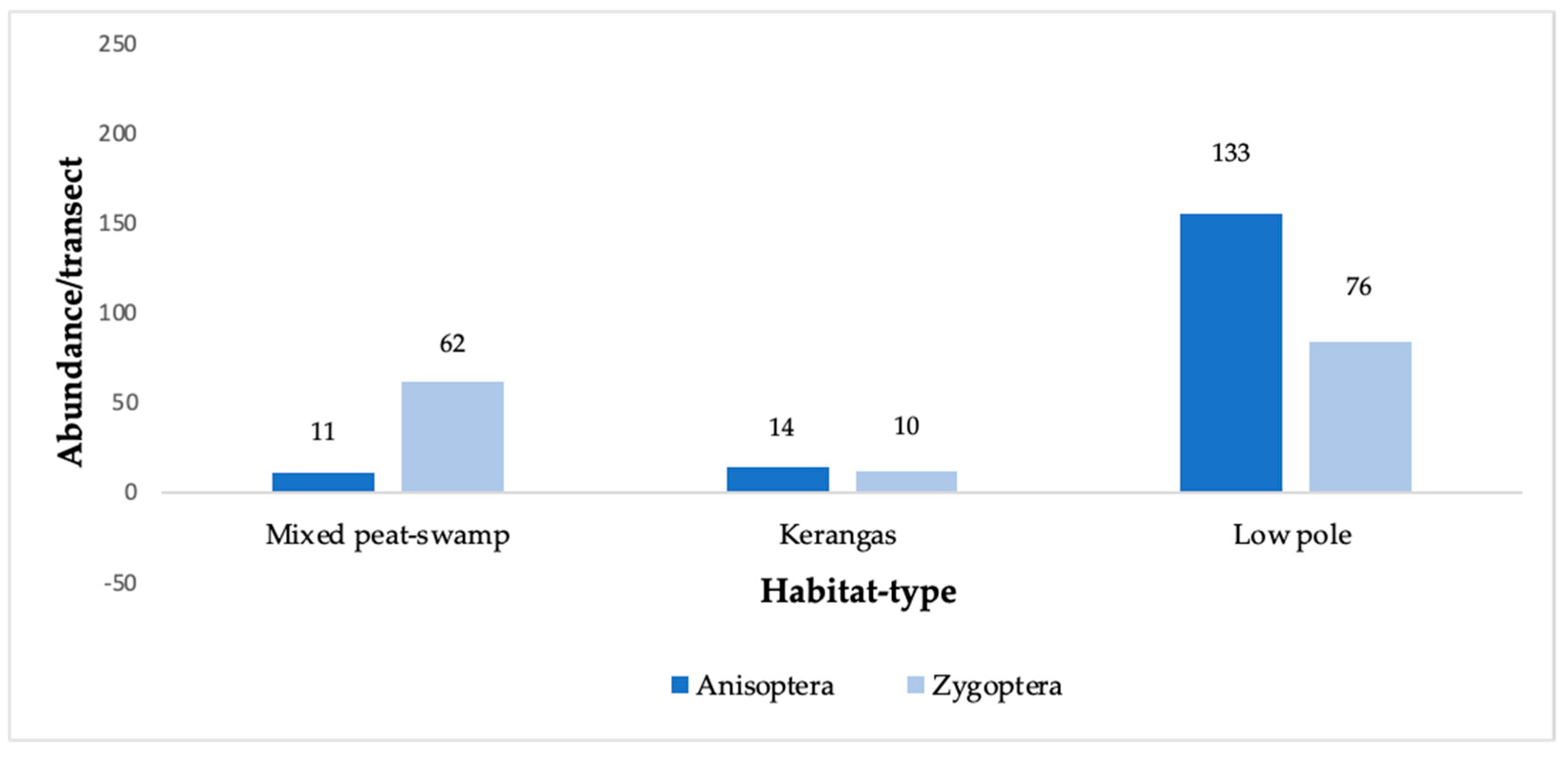
3.1. Diversity and Abundance
3.2. Environmental Variables
3.3. Species Characteristic Data
4. Discussion
4.1. Diversity, Abundance, and Communities
The Role of Mixed-Mosaic Habitat Structure on the Odonata Community in Kerangas
4.2. Morphology, Dispersal, and Habitat Selection
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Habitat Type | Suborder | Number of Species with at Least 5 Measured Records |
|---|---|---|
| Kerangas | Z | 1 |
| A | 1 | |
| Low pole peat swamp | Z | 2 |
| A | 3 | |
| Mixed peat swamp | Z | 2 |
| A | 0 |
| No | Suborder | Family | Genus | Species | Mixed Peat Swamp | Kerangas | Low Pole Peat Swamp |
|---|---|---|---|---|---|---|---|
| 1 | Anisoptera | Gomphidae | Ictinogomphus | acutus * | x | ||
| 2 | Anisoptera | Gomphidae | Leptogomphus | coomansi | |||
| 3 | Anisoptera | Libellulidae | Agrionoptera | sexlineata | x | ||
| 4 | Anisoptera | Libellulidae | Brachygonia | oculata * | x | ||
| 5 | Anisoptera | Libellulidae | Brachygonia | ophelia * | x | x | |
| 6 | Anisoptera | Libellulidae | Brachygonia | puella * | x | x | x |
| 7 | Anisoptera | Libellulidae | Lyriothemis | cleis | x | ||
| 8 | Anisoptera | Libellulidae | Nannophya | pygmaea | x | ||
| 9 | Anisoptera | Libellulidae | Neurothemis | fluctuans * | x | ||
| 10 | Anisoptera | Libellulidae | Orchithemis | xanthosoma * | x | x | x |
| 11 | Anisoptera | Libellulidae | Orthetrum | chrysis * | |||
| 12 | Anisoptera | Libellulidae | Orthetrum | sabina | |||
| 13 | Anisoptera | Libellulidae | Pornothemis | serrata * | x | x | |
| 14 | Anisoptera | Libellulidae | Raphismia | inermis | |||
| 15 | Anisoptera | Libellulidae | Rhyothemis | obsolescens | x | ||
| 16 | Anisoptera | Libellulidae | Rhyothemis | fulgens * | x | ||
| 17 | Anisoptera | Libellulidae | Risiophlebia | dohrni * | x | ||
| 18 | Anisoptera | Libellulidae | Tramea | phaeoneura | |||
| 19 | Anisoptera | Libellulidae | Tramea | transmarina | |||
| 20 | Anisoptera | Libellulidae | Tyriobapta | laidlawi * | x | x | x |
| 1 | Zygoptera | Calopterygidae | Vestalis | amoena * | x | ||
| 2 | Zygoptera | Calopterygidae | Vestalis | sp. * | x | x | |
| 3 | Zygoptera | Chlorocyphidae | Libellago | aurantiaca * | x | ||
| 4 | Zygoptera | Chlorocyphidae | Pachycypha | aurea * | x | ||
| 5 | Zygoptera | Coenagrionidae | Aciagrion | borneense | |||
| 6 | Zygoptera | Coenagrionidae | Agriocnemis | minima | |||
| 7 | Zygoptera | Coenagrionidae | Amphicnemis | triplex * | x | x | x |
| 8 | Zygoptera | Coenagrionidae | Archibasis | melanocyana | |||
| 9 | Zygoptera | Coenagrionidae | Ceriagrion | cerinorubellum | |||
| 10 | Zygoptera | Coenagrionidae | Mortonagrion | falcatum * | x | ||
| 11 | Zygoptera | Coenagrionidae | Pseudagrion | coomansi | |||
| 12 | Zygoptera | Coenagrionidae | Ceriagrion | sp. * | x | ||
| 13 | Zygoptera | Euphaeidae | Dysphaea | dimidiata | |||
| 14 | Zygoptera | Euphaeidae | Euphaea | impar * | x | ||
| 15 | Zygoptera | Megapodagrionidae | Podolestes | atomarius | x | ||
| 16 | Zygoptera | Platycnemididae | Coeliccia | sp. * | x | ||
| 17 | Zygoptera | Platystictidae | Drepanosticta | rufostigma * | x | ||
| 18 | Zygoptera | Platystictidae | Protosticta | sp. * | x | ||
| 19 | Zygoptera | Protoneuridae | Elattoneura | aurantiaca * | x | x | |
| 20 | Zygoptera | Protoneuridae | Prodasineura | sp. * | x | ||
| 21 | Zygoptera | Protoneuridae | Prodasineura | dorsalis * | x | ||
| 22 | Zygoptera | Not known | Unknown | sp. * | x |

References
- Wiens, J.J. The causes of species richness patterns across space, time, and clades and the role of “ecological limits”. Q. Rev. Biol. 2011, 86, 75–97. [Google Scholar] [CrossRef] [PubMed]
- Alves-Martins, F.; Calatayud, J.; Medina, N.G.; De Marco, P.; Juen, L.; Hortal, J. Drivers of regional and local diversity of Amazonian stream Odonata. Insect Conserv. Divers. 2019, 12, 251–261. [Google Scholar] [CrossRef]
- Cornell, H.V.; Lawton, J.H. Species interactions, local and regional processes, and limits to the richness of ecological communities: A theoretical perspective. J. Anim. Ecol. 1992, 61, 1–12. [Google Scholar] [CrossRef]
- Witman, J.D.; Etter, R.J.; Smith, F. The relationship between regional and local species diversity in marine benthic communities: A global perspective. Proc. Natl. Acad. Sci. USA 2004, 101, 15664–15669. [Google Scholar] [CrossRef]
- Brown, R.L.; Reilly, L.A.J.; Peet, R.K. Species richness: Small scale. In eLS; John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2007; pp. 1–9. [Google Scholar] [CrossRef]
- Weiher, E.; Freund, D.; Bunton, T.; Stefanski, A.; Lee, T.; Bentivenga, S. Advances, challenges, and a developing synthesis of ecological community assembly theory. Philos. Trans. R. Soc. B Biol. Sci. 2011, 366, 2403–2413. [Google Scholar] [CrossRef] [PubMed]
- Vellend, M. Conceptual synthesis in community ecology. Q. Rev. Biol. 2010, 85, 183–206. [Google Scholar] [CrossRef] [PubMed]
- Pinkert, S.; Dijkstra, K.D.B.; Zeuss, D.; Reudenbach, C.; Brandl, R.; Hof, C. Evolutionary processes, dispersal limitation and climatic history shape current diversity patterns of European dragonflies. Ecography 2018, 41, 795–804. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; Da Fonseca, G.A.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef]
- May, R.M. How many species are there on earth? Science 1988, 241, 1441–1449. [Google Scholar] [CrossRef]
- Schulze, E.D.; Mooney, H.A. Biodiversity and Ecosystem Function; Springer Study Edition: New York, NY, USA, 2012; pp. 4–525. [Google Scholar]
- MacArthur, R.H.; MacArthur, J.W. On bird species diversity. Ecology 1961, 42, 594–598. [Google Scholar] [CrossRef]
- Bastos, R.C.; Brasil, L.S.; Oliveira-Junior, J.M.B.; Carvalho, F.G.; Lennox, G.D.; Barlow, J.; Juen, L. Morphological and phylogenetic factors structure the distribution of damselfly and dragonfly species (Odonata) along an environmental gradient in Amazonian streams. Ecol. Indic. 2021, 122, 107257. [Google Scholar] [CrossRef]
- Juen, L.; De Marco, P., Jr. Odonate biodiversity in terra-firme forest streamlets in Central Amazonia: On the relative effects of neutral and niche drivers at small geographical extents. Insect Conserv. Divers. 2011, 4, 265–274. [Google Scholar] [CrossRef]
- Ferger, S.W.; Schleuning, M.; Hemp, A.; Howell, K.M.; Böhning-Gaese, K. Food resources and vegetation structure mediate climatic effects on species richness of birds. Glob. Ecol. Biogeogr. 2014, 23, 541–549. [Google Scholar] [CrossRef]
- Cramer, M.J.; Willig, M.R. Habitat heterogeneity, species diversity and null models. Oikos 2005, 108, 209–218. [Google Scholar] [CrossRef]
- Fischer, J.; Lindenmayer, D.B.; Manning, A.D. Biodiversity, ecosystem function, and resilience: Ten guiding principles for commodity production landscapes. Front. Ecol. Environ. 2006, 4, 80–86. [Google Scholar] [CrossRef]
- Stein, A.; Gerstner, K.; Kreft, H. Environmental heterogeneity as a universal driver of species richness across taxa, biomes and spatial scales. Ecol. Lett. 2014, 17, 866–880. [Google Scholar] [CrossRef]
- Kietzka, G.J.; Pryke, J.S.; Samways, M.J. Landscape ecological networks are successful in supporting a diverse dragonfly assemblage. Insect Conserv. Divers. 2015, 8, 229–237. [Google Scholar] [CrossRef]
- Humphrey, J.W.; Hawes, C.; Peace, A.J.; Ferris-Kaan, R.; Jukes, M.R. Relationships between insect diversity and habitat characteristics in plantation forests. For. Ecol. Manag. 1999, 113, 11–21. [Google Scholar] [CrossRef]
- Lassau, S.A.; Hochuli, D.F. Effects of habitat complexity on ant assemblages. Ecography 2004, 27, 157–164. [Google Scholar] [CrossRef]
- McGeoch, M.A.; Van Rensburg, B.J.; Botes, A. The verification and application of bioindicators: A case study of dung beetles in a savanna ecosystem. J. Appl. Ecol. 2002, 39, 661–672. [Google Scholar] [CrossRef]
- Dutra, S.; De Marco, P. Bionomic differences in odonates and their influence on the efficiency of indicator species of environmental quality. Ecol. Indic. 2015, 49, 132–142. [Google Scholar] [CrossRef]
- Chovanec, A.; Waringer, J. Ecological integrity of river–floodplain systems—Assessment by dragonfly surveys (Insecta: Odonata). Regul. Rivers Res. Manag. Int. J. Devoted River Res. Manag. 2001, 17, 493–507. [Google Scholar] [CrossRef]
- Stoks, R.; Cordoba-Aguilar, A. Evolutionary ecology of Odonata: A complex life cycle perspective. Annu. Rev. Entomol. 2012, 57, 249–265. [Google Scholar] [CrossRef]
- Šigutová, H.; Šipoš, J.; Dolný, A. A novel approach involving the use of Odonata as indicators of tropical forest degradation: When family matters. Ecol. Indic. 2019, 104, 229–236. [Google Scholar] [CrossRef]
- Remsburg, A.J.; Olson, A.C.; Samways, M.J. Shade alone reduces adult dragonfly (Odonata: Libellulidae) abundance. J. Insect Behav. 2008, 21, 460–468. [Google Scholar] [CrossRef]
- Thornhill, I.; Batty, L.; Death, R.G.; Friberg, N.R.; Ledger, M.E. Local and landscape scale determinants of macroinvertebrate assemblages and their conservation value in ponds across an urban land-use gradient. Biodivers. Conserv. 2017, 26, 1065. [Google Scholar] [CrossRef]
- Cunningham-Minnick, M.J.; Meyer, T.B.; Crist, T.O. Shifts in dragonfly community structure across aquatic ecotones. Int. J. Odonatol. 2019, 22, 121–133. [Google Scholar] [CrossRef]
- Buchwald, R. Vegetation and dragonfly fauna—Characteristics and examples of biocenological field studies. Vegetatio 1992, 101, 99–107. [Google Scholar] [CrossRef]
- Tews, J.; Brose, U.; Grimm, V.; Tielbörger, K.; Wichmann, M.C.; Schwager, M.; Jeltsch, F. Animal species diversity driven by habitat heterogeneity/diversity: The importance of keystone structures. J. Biogeogr. 2004, 31, 79–92. [Google Scholar] [CrossRef]
- Hykel, M.; Růžičková, J.; Dolný, A. Perch selection in Sympetrum species (Odonata: Libellulidae): Importance of vegetation structure and composition. Ecol. Entomol. 2020, 45, 90–96. [Google Scholar] [CrossRef]
- May, M.L. Thermoregulation and adaptation to temperature in dragonflies (Odonata: Anisoptera). Ecol. Monogr. 1976, 46, 1–32. [Google Scholar] [CrossRef]
- May, M.L. Thermal adaptations of dragonflies, revisited. Adv. Odonatol. 1991, 5, 71–88. [Google Scholar]
- Samejima, Y.; Tsubaki, Y. Body temperature and body size affect flight performance in a damselfly. Behav. Ecol. Sociobiol. 2010, 64, 685–692. [Google Scholar] [CrossRef]
- Fahrig, L. Effect of habitat fragmentation on the extinction threshold: A synthesis. Ecol. Appl. 2002, 12, 346–353. [Google Scholar]
- McCauley, S.J. Relationship between morphology, dispersal and habitat distribution in three species of Libellula (Odonata: Anisoptera). Aquat. Insects 2012, 34, 195–204. [Google Scholar] [CrossRef]
- Dow, R.; Silvius, M. Results of an Odonata survey carried out in the peatlands of Central Kalimantan, Indonesia, in 2012. Faun. Stud. South-East Asian Pac. Isl. Odonata 2014, 7, 1–37. [Google Scholar]
- Husson, S.J.; Limin, S.H.; Boyd, N.S.; Brousseau, J.J.; Collier, S.; Cheyne, S.M.; Arcy, L.J.D.; Dow, R.A.; Dowds, N.W.; Dragiewicz, M.L.; et al. Biodiversity of the Sebangau tropical peat swamp forest, Indonesian Borneo. Mires Peat 2018, 22, 1–50. [Google Scholar]
- Cleary, D.F.; Mooers, A.Ø.; Eichhorn, K.A.; Van Tol, J.; De Jong, R.; Menken, S.B. Diversity and community composition of butterflies and odonates in an ENSO-induced fire affected habitat mosaic: A case study from East Kalimantan, Indonesia. Oikos 2004, 105, 426–448. [Google Scholar] [CrossRef]
- Dolný, A.; Bárta, D.; Lhota, S.; Drozd, P. Dragonflies (Odonata) in the Bornean rain forest as indicators of changes in biodiversity resulting from forest modification and destruction. Trop. Zool. 2011, 24, 63–68. [Google Scholar]
- Dolný, A.; Harabiš, F.; Bárta, D.; Lhota, S.; Drozd, P. Aquatic insects indicate terrestrial habitat degradation: Changes in taxonomical structure and functional diversity of dragonflies in tropical rainforest of East Kalimantan. Trop. Zool. 2012, 25, 141–157. [Google Scholar] [CrossRef]
- MacKinnon, K.; Hatta, G.; Halim, H.; Mangalir, A. The Ecology of Kalimantan; Oxford University Press: Oxford, UK, 1996; Volume III, pp. 723–724. [Google Scholar]
- Buckley, J.W.B.; Capilla, B.R.; Maimunah, S.; Armadyanto, A.; Boyd, N.; Cheyne, S.M.; Iwan; Husson, S.J.; Salahudin, S.; Ferisa, A.; et al. Biodiversity, Forest Structure and Conservation Importance of the Mungku Baru Education forest, Rungan, Central Kalimantan, Indonesia. Borneo Nature Foundation Internal Report (2018). Available online: https://www.google.com.hk/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&ved=2ahUKEwjatsKq2ev8AhWHV2wGHb7MBfwQFnoECA8QAQ&url=https%3A%2F%2Fdocplayer.net%2F139261754-Biodiversity-forest-structure-conservation-importance-of-the-mungku-baru-education-forest-rungan-central-kalimantan-indonesia.html&usg=AOvVaw2kntetM0leuZumAfVmGaoZ (accessed on 10 October 2022).
- Orr, A.G. An annotated checklist of the Odonata of Brunei with ecological notes and descriptions of hitherto unknown males and larvae. Int. J. Odonatol. 2001, 4, 167–220. [Google Scholar] [CrossRef]
- Orr, A.G. Odonata in Bornean tropical rain forest formations: Diversity, endemicity and implications for conservation management. In Forest and Dragonflies; Cordero Rivera, A., Ed.; Pensoft Publishers: Sofia, Bulgaria, 2006; pp. 51–78. [Google Scholar]
- Purwanto, P.B.; Zaman, M.N.; Akbar, M.; Arief, M. Study of Odonata Diversity in Kerangas Forest Sukadamai Village and Punai Beach Simpang Pesak, Belitung Timur. Proc. Int. Conf. Sci. Eng. 2019, 2, 133–136. [Google Scholar] [CrossRef]
- Katagiri, S.; Yamakura, T.; Lee, S.H. Properties of soils in kerangas forest on sandstone at Bako National Park, Sarawak, East Malaysia. Jpn. J. Southeast Asian Stud. 1991, 29, 35–48. [Google Scholar]
- Pollard, E. A method for assessing changes in the abundance of butterflies. Biol. Conserv. 1977, 12, 115–134. [Google Scholar] [CrossRef]
- Walpole, M.J.; Sheldon, I.R. Sampling butterflies in tropical rainforest: An evaluation of a transect walk method. Biol. Conserv. 1999, 87, 85–91. [Google Scholar] [CrossRef]
- Oppel, S. Using distance sampling to quantify Odonata density in tropical rainforests. Int. J. Odonatol. 2006, 9, 81–88. [Google Scholar] [CrossRef]
- Van Swaay, C.A.M. Butterfly densities on line transects in The Netherlands from 1990–2001. Entomol. Ber.-Ned. Entomol. Vereenigung 2003, 63, 82–87. [Google Scholar]
- Cordero-Rivera, A.; Stoks, R. Mark-recapture studies and demography. In Dragonflies and Damselflies: Model Organisms for Ecological and Evolutionary Research; Córdoba-Aguilar, A., Ed.; Oxford University Press: Oxford, UK, 2008; pp. 7–20. [Google Scholar]
- Anderson, C.N.; Cordoba-Aguilar, A.; Druru, J.P.; Grether, G.F. An assessment of marking techniques for odonates in the family Calopterygidae. Entomol. Exp. Et Appl. 2011, 141, 258–261. [Google Scholar] [CrossRef]
- Alvarez-Covelli, C.; Álvarez-Covelli, M.A.; Palacino-Rodríguez, F. Abdomen or wings? Comparing two body places for marking in Mesamphiagrion laterale (Odonata: Coenagrionidae). Odonatologica 2015, 44, 343–348. [Google Scholar]
- Quisil, S.J.C.; Arreza, J.D.Z.E.; Nuñeza, O.M.; Villanueva, R.T.J. Species richness of Odonata in Lanuza and San Agustin, Surigao del Sur, Philippines. Adv. Environ. Sci. 2013, 5, 245–260. [Google Scholar]
- Jost, L. Entropy and diversity. Oikos 2006, 113, 363–375. [Google Scholar] [CrossRef]
- Chao, A.; Chiu, C.H.; Jost, L. Unifying species diversity, phylogenetic diversity, functional diversity, and related similarity and differentiation measures through Hill numbers. Annu. Rev. Ecol. Evol. Syst. 2014, 45, 297–324. [Google Scholar] [CrossRef]
- Hsieh, T.C.; Ma, K.H.; Chao, A. iNEXT: Interpolation and Extrapolation for Species Diversity. Available online: https://cran.r-project.org/web/packages/iNEXT/iNEXT.pdf (accessed on 10 October 2022).
- Wickham, H. ggplot2: Elegant Graphics for Data Analysis; Springer: New York, NY, USA, 2016. [Google Scholar]
- Length, R.V. emmeans: Estimated Marginal Means, aka Least-Squares Means. R Package Version 1.8.3. 2022. Available online: https://CRAN.R-project.org/package=emmeans (accessed on 10 October 2022).
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 10 October 2022).
- Florian Hartig. DHARMa: Residual Diagnostics for Hierarchical (Multi-Level/Mixed) Regression Models. R Package Version 0.4.6. 2022. Available online: http://florianhartig.github.io/DHARMa/ (accessed on 10 October 2022).
- De Marco, P., Jr.; Resende, D.C. Activity patterns and thermoregulation in a tropical dragonfly assemblage. Odonatologica 2002, 31, 129–138. [Google Scholar]
- Loreau, M.; Naeem, S.; Inchausti, P.; Bengtsson, J.; Grime, J.P.; Hector, A.; Hooper, D.U.; Huston, M.A.; Rafaelli, D.; Schmid, B.; et al. Biodiversity and ecosystem functioning: Current knowledge and future challenges. Science 2001, 294, 804–808. [Google Scholar] [CrossRef]
- Miyamoto, K.; Kohyama, T.S.; Rahajoe, J.S.; Mirmanto, E.; Simbolon, H. Forest structure and productivity of tropical heath and peatland forests. In Tropical Peatland Ecosystems; Osaki, M., Tsuji, N., Eds.; Springer: Tokyo, Japan, 2016; pp. 151–166. [Google Scholar]
- Corbet, P.S.; May, M.L. Fliers and perchers among Odonata: Dichotomy or multidimensional continuum? A provisional reappraisal. Int. J. Odonatol. 2008, 11, 155–171. [Google Scholar] [CrossRef]
- De Oliveira-Junior, J.M.B.; Shimano, Y.; Gardner, T.A.; Hughes, R.M.; de Marco Júnior, P.; Juen, L. Neotropical dragonflies (I nsecta: O donata) as indicators of ecological condition of small streams in the eastern A mazon. Austral Ecol. 2015, 40, 733–744. [Google Scholar] [CrossRef]
- Juen, L.; De Marco, P. Dragonfly endemism in the Brazilian Amazon: Competing hypotheses for biogeographical patterns. Biodivers. Conserv. 2012, 21, 3507–3521. [Google Scholar] [CrossRef]
- Brasil, L.S.; Vieira, T.B.; de Oliveira-Junior, J.M.B.; Dias-Silva, K.; Juen, L. Elements of metacommunity structure in Amazonian Zygoptera among streams under different spatial scales and environmental conditions. Ecol. Evol. 2017, 7, 3190–3200. [Google Scholar] [CrossRef]
- Kalkman, V.J.; Clausnitzer, V.; Dijkstra, K.D.B.; Orr, A.G.; Paulson, D.R.; van Tol, J. Global diversity of dragonflies (Odonata) in freshwater. Hydrobiologia 2007, 595, 351–363. [Google Scholar] [CrossRef]
- Calvão, L.B.; Vital, M.V.C.; Juen, L.; Lima Filho, G.F.D.; Oliveira Júnior, J.M.B.D.; Pinto, N.S.; Marco Júnior, P.D. Thermoregulation and microhabitat choice in Erythrodiplax latimaculata Ris males (Anisoptera: Libellulidae). Odonatologica 2013, 42, 97–108. [Google Scholar] [CrossRef]
- Oliveira-Junior, J.M.B.; Juen, L. The Zygoptera/Anisoptera ratio (Insecta: Odonata): A new tool for habitat alterations assessment in Amazonian streams. Neotrop. Entomol. 2019, 48, 552–560. [Google Scholar] [CrossRef] [PubMed]
- Page, S.E.; Rieley, J.O.; Shotyk, Ø.W.; Weiss, D. Interdependence of peat and vegetation in a tropical peat swamp forest. In Changes and Disturbance in Tropical Rainforest in South-East Asia; Newbury, D.M., Clutton-Brock, T.H., Prance, G.T., Eds.; The Royal Society: London, UK, 1999; pp. 161–173. [Google Scholar]
- Kietzka, G.J.; Pryke, J.S.; Samways, M.J. Aerial adult dragonflies are highly sensitive to in-water conditions across an ancient landscape. Divers. Distrib. 2017, 23, 14–26. [Google Scholar] [CrossRef]
- Rosenzweig, M.L. A theory of habitat selection. Ecology 1981, 62, 327–335. [Google Scholar] [CrossRef]
- Orr, A.G. Territorial behaviour associated with feeding in both sexes of the tropical zygopteran, Libellago hyalina (Odonata: Chlorocyphidae). Int. J. Odonatol. 2004, 7, 493–504. [Google Scholar] [CrossRef]
- Dow, R.A. An annotated checklist of the Odonata (Insecta) known from Sarawak with records to district level. Sarawak Mus. J. 2021, 81, 313–422. [Google Scholar]
- Dobson, M.; Frid, C. Ecology of Aquatic Systems, 2nd ed.; Oxford University Press: Oxford, UK, 1998. [Google Scholar]
- Hof, C.; Brändle, M.; Brandl, R. Lentic odonates have larger and more northern ranges than lotic species. J. Biogeogr. 2006, 33, 63–70. [Google Scholar] [CrossRef]
- Schilder, R.J.; Marden, J.H. A hierarchical analysis of the scaling of force and power production by dragonfly flight motors. J. Exp. Biol. 2004, 207, 767–776. [Google Scholar] [CrossRef]
- Rundle, S.D.; Bilton, D.T.; Abbott, J.C.; Foggo, A. Range size in North American Enallagma damselflies correlates with wing size. Freshw. Biol. 2007, 52, 471–477. [Google Scholar] [CrossRef]
- May, M.L. A critical overview of progress in studies of migration of dragonflies (Odonata: Anisoptera), with emphasis on North America. J. Insect Conserv. 2013, 17, 1–15. [Google Scholar] [CrossRef]


| Morphological Trait | Habitat Comparison | Suborder | Estimate | Standard Error | df | t Ratio | p Value |
|---|---|---|---|---|---|---|---|
| Body | LP–MPS | Z | 0.025 | 0.022 | 217 | 1.16 | 0.478 |
| LP–K | Z | −0.053 | 0.041 | 217 | −1.293 | 0.4 | |
| MPS–K | Z | −0.079 | 0.042 | 217 | −1.883 | 0.146 | |
| LP–MPS | A | −0.217 | 0.046 | 217 | −4.753 | 0.001 | |
| LP–K | A | −0.255 | 0.035 | 217 | −7.37 | 0.001 | |
| MPS–K | A | −0.038 | 0.054 | 217 | −0.703 | 0.762 | |
| Thorax | LP–MPS | Z | −0.005 | 0.028 | 221 | −0.186 | 0.981 |
| LP–K | Z | 0.088 | 0.054 | 221 | 1.632 | 0.235 | |
| MPS–K | Z | 0.093 | 0.054 | 221 | 1.711 | 0.203 | |
| LP–MPS | A | −0.141 | 0.053 | 221 | −2.668 | 0.022 | |
| LP–K | A | −0.198 | 0.045 | 221 | −4.4 | 0.001 | |
| MPS–K | A | −0.057 | 0.065 | 221 | −0.869 | 0.66 | |
| Hindwing | LP–MPS | Z | −0.012 | 0.028 | 216 | −0.421 | 0.907 |
| LP–K | Z | −0.086 | 0.053 | 216 | −1.622 | 0.238 | |
| MPS–K | Z | −0.074 | 0.053 | 216 | −1.39 | 0.348 | |
| LP–MPS | A | −0.248 | 0.055 | 216 | −4.531 | 0.001 | |
| LP–K | A | −0.256 | 0.044 | 216 | −5.807 | 0.001 | |
| MPS–K | A | −0.008 | 0.066 | 216 | −0.126 | 0.991 | |
| Hindwing:Body | LP–MPS | Z | −0.068 | 0.019 | 218 | −3.55 | 0.001 |
| LP–K | Z | −0.051 | 0.035 | 218 | −1.481 | 0.302 | |
| MPS–K | Z | 0.017 | 0.035 | 218 | 0.479 | 0.881 | |
| LP–MPS | A | 0.039 | 0.036 | 218 | 1.084 | 0.525 | |
| LP–K | A | 0 | 0.03 | 218 | −0.004 | 1 | |
| MPS–K | A | −0.039 | 0.044 | 218 | −0.882 | 0.652 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hendriks, J.A.; Mariaty; Maimunah, S.; Anirudh, N.B.; Holly, B.A.; Erkens, R.H.J.; Harrison, M.E. Odonata (Insecta) Communities in a Lowland Mixed Mosaic Forest in Central Kalimantan, Indonesia. Ecologies 2023, 4, 55-73. https://doi.org/10.3390/ecologies4010006
Hendriks JA, Mariaty, Maimunah S, Anirudh NB, Holly BA, Erkens RHJ, Harrison ME. Odonata (Insecta) Communities in a Lowland Mixed Mosaic Forest in Central Kalimantan, Indonesia. Ecologies. 2023; 4(1):55-73. https://doi.org/10.3390/ecologies4010006
Chicago/Turabian StyleHendriks, Jorian A., Mariaty, Siti Maimunah, Namrata B. Anirudh, Brendan A. Holly, Roy H. J. Erkens, and Mark E. Harrison. 2023. "Odonata (Insecta) Communities in a Lowland Mixed Mosaic Forest in Central Kalimantan, Indonesia" Ecologies 4, no. 1: 55-73. https://doi.org/10.3390/ecologies4010006
APA StyleHendriks, J. A., Mariaty, Maimunah, S., Anirudh, N. B., Holly, B. A., Erkens, R. H. J., & Harrison, M. E. (2023). Odonata (Insecta) Communities in a Lowland Mixed Mosaic Forest in Central Kalimantan, Indonesia. Ecologies, 4(1), 55-73. https://doi.org/10.3390/ecologies4010006






