Low Temperature Synthesis of 3d Metal (Fe, Co, Ni, Cu)-Doped TiO2 Photocatalyst via Liquid Phase Deposition Technique
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Characterization
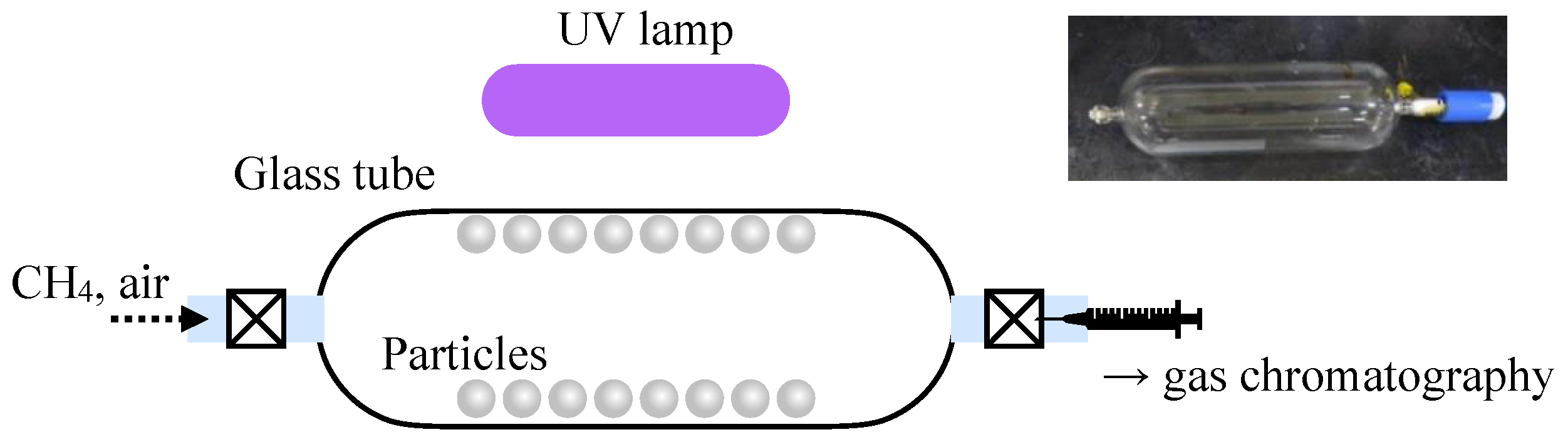
2.3. Photocatalysis Test for Reducing Methane
3. Results and Discussion
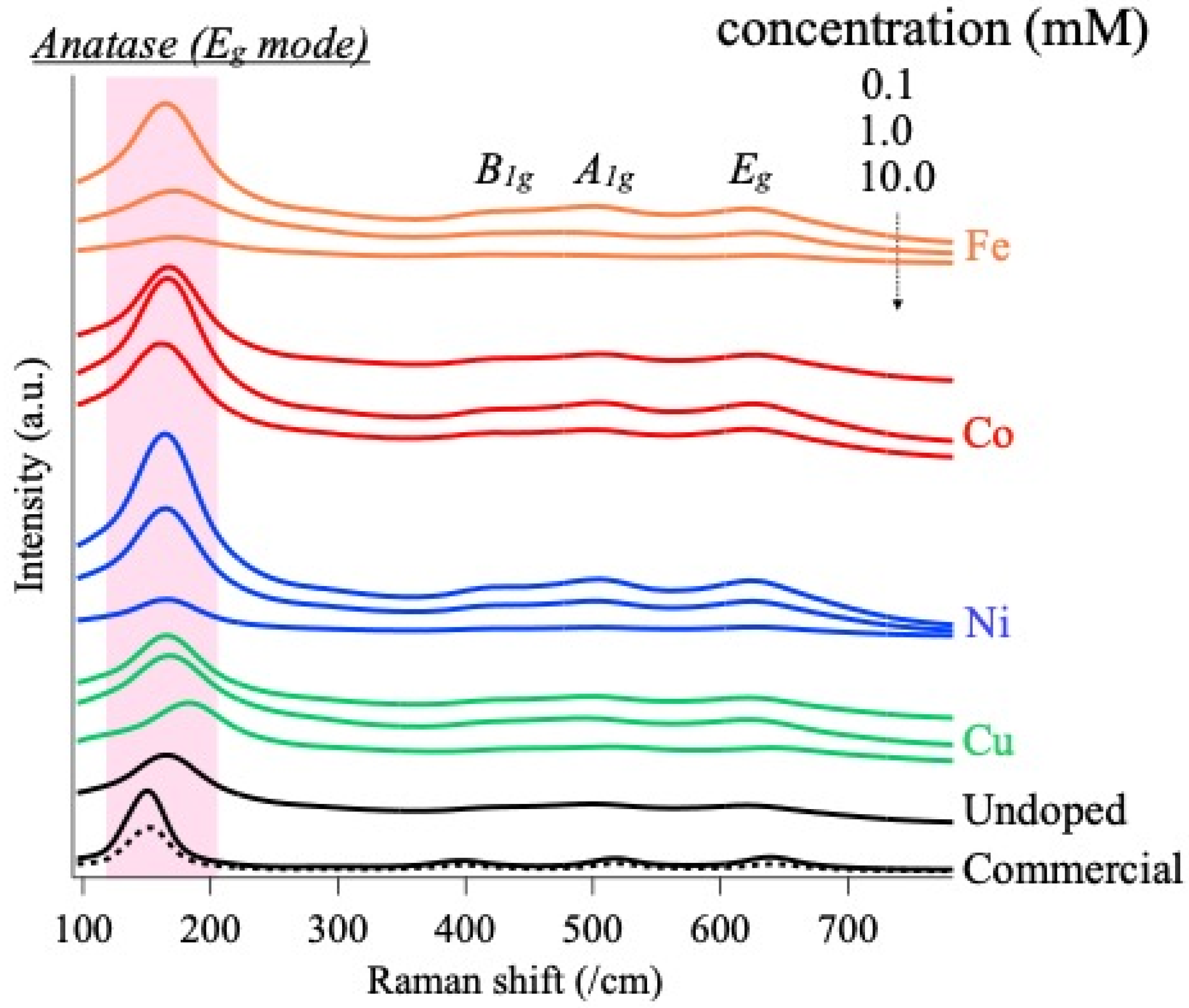
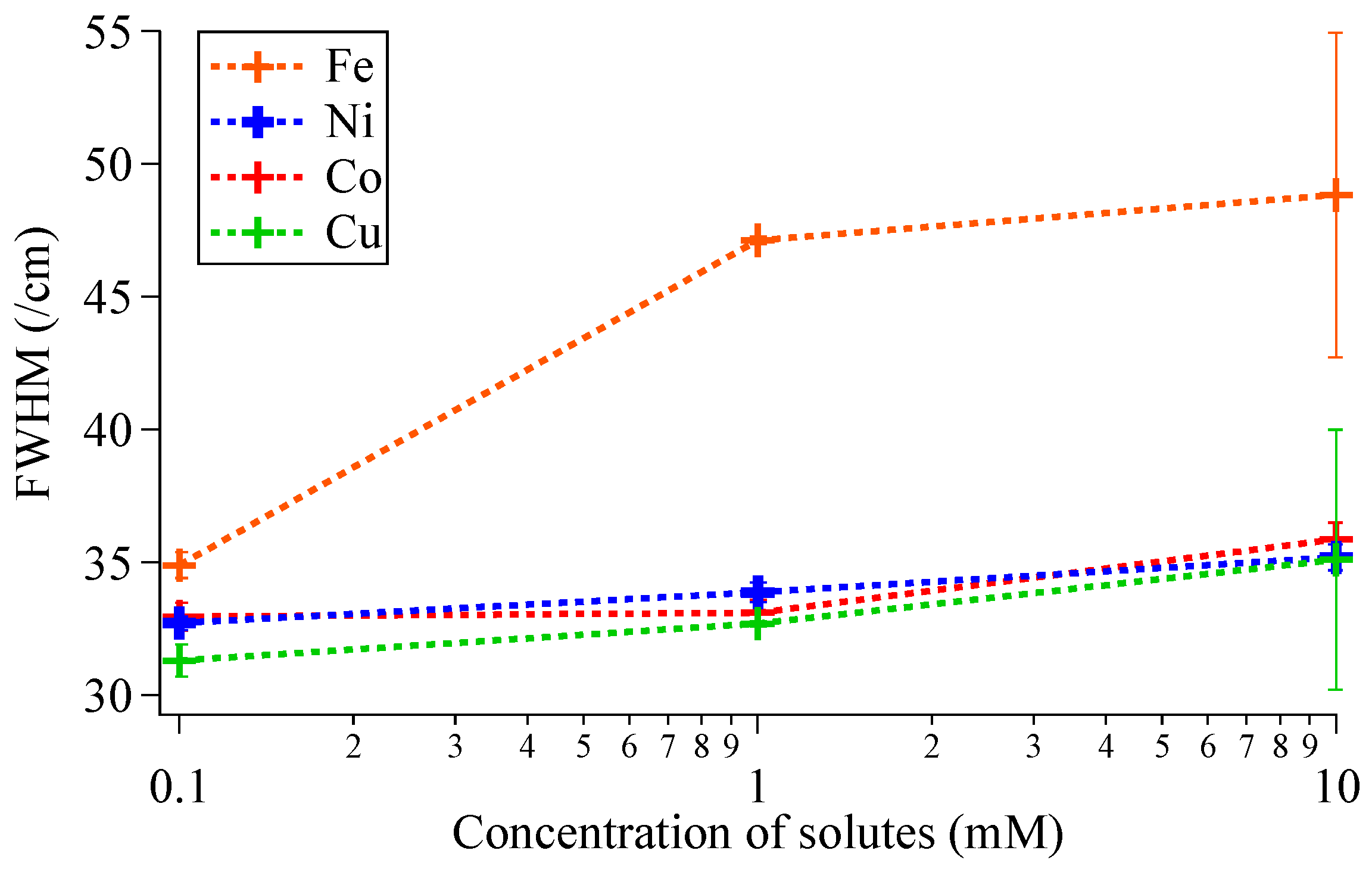
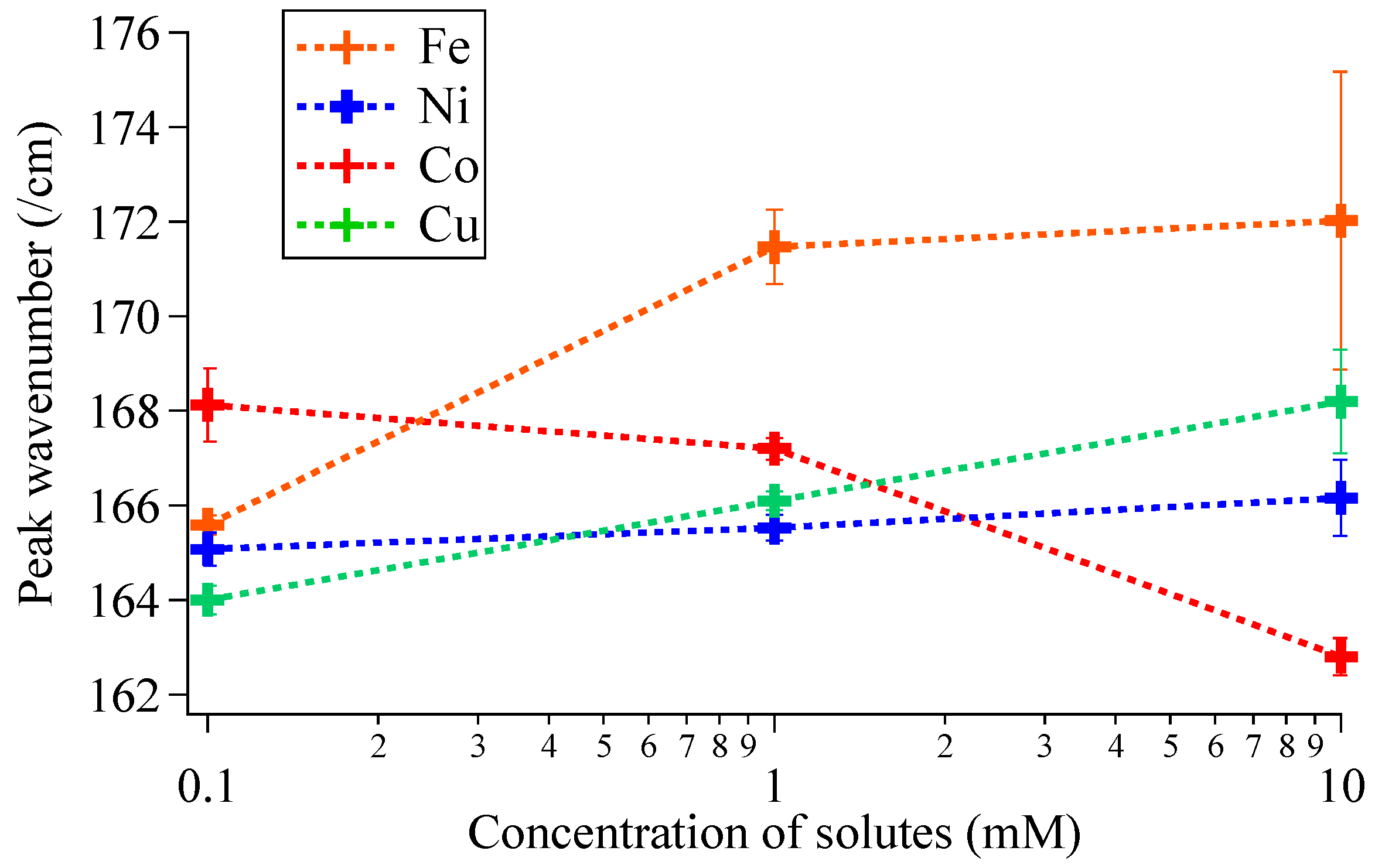
3.1. Influence of Doping on Morphology and Crystal Structures
3.2. Doping Levels and Optical Properties
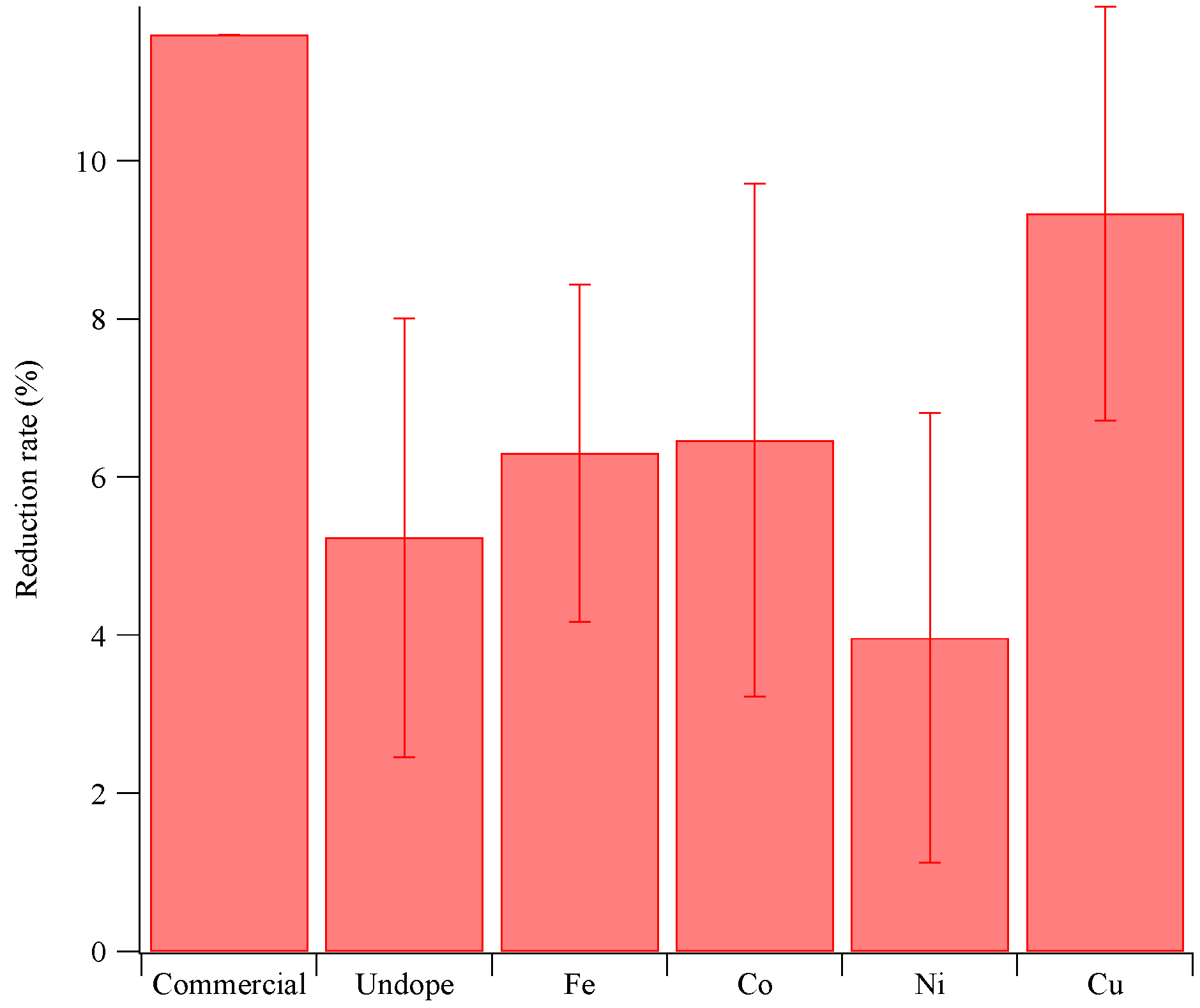
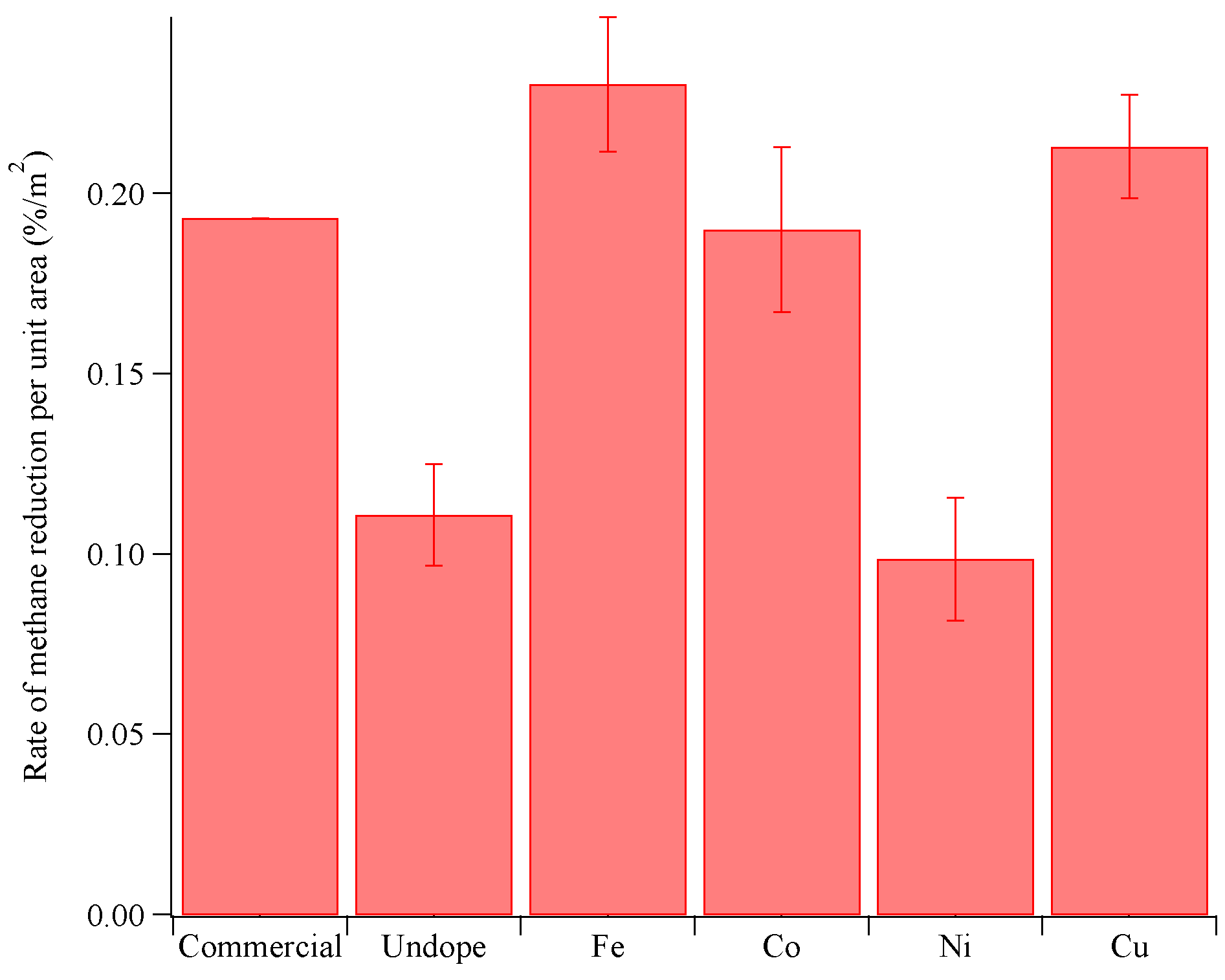
3.3. Photocatalytic Reduction of Methane
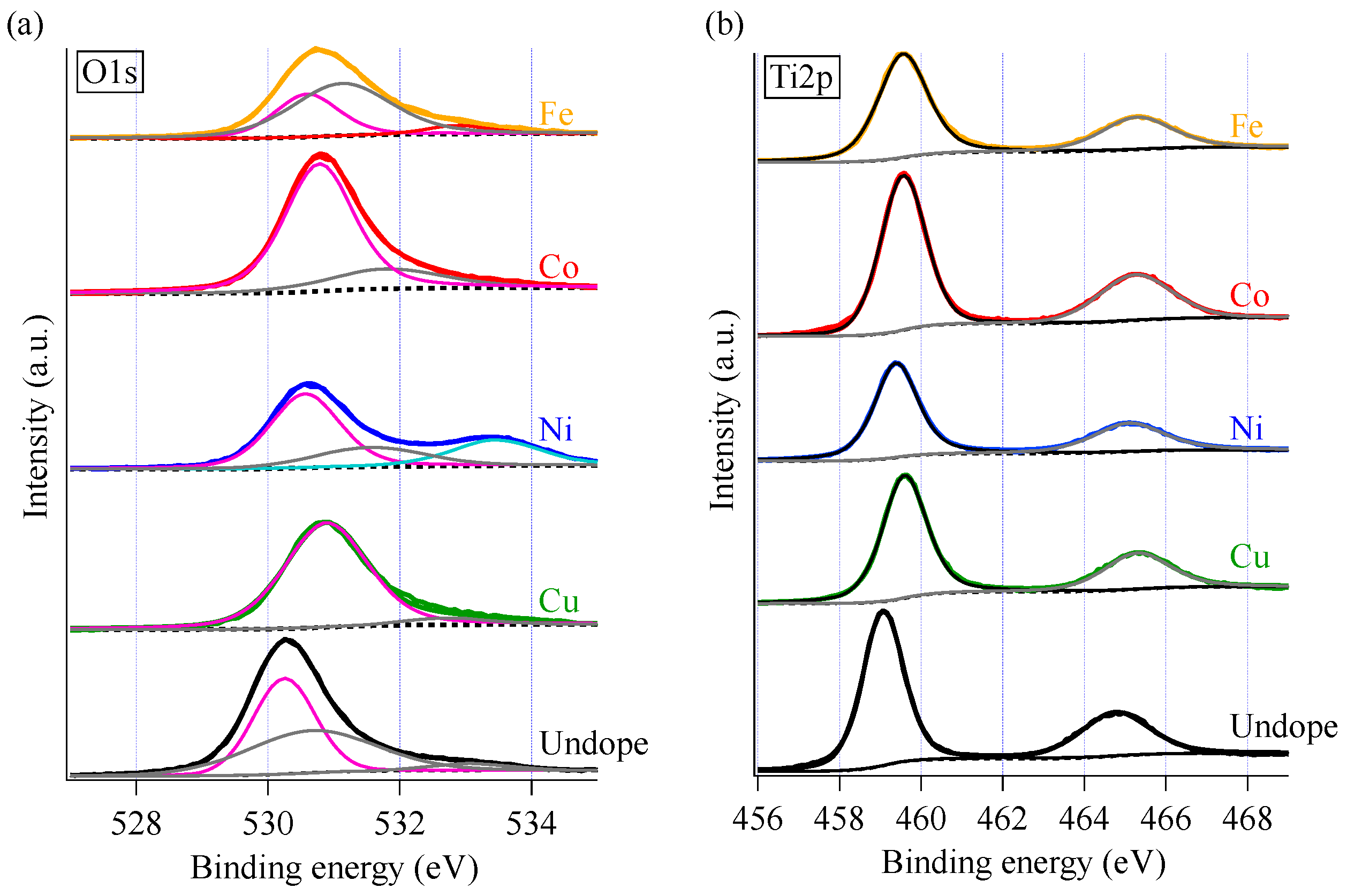
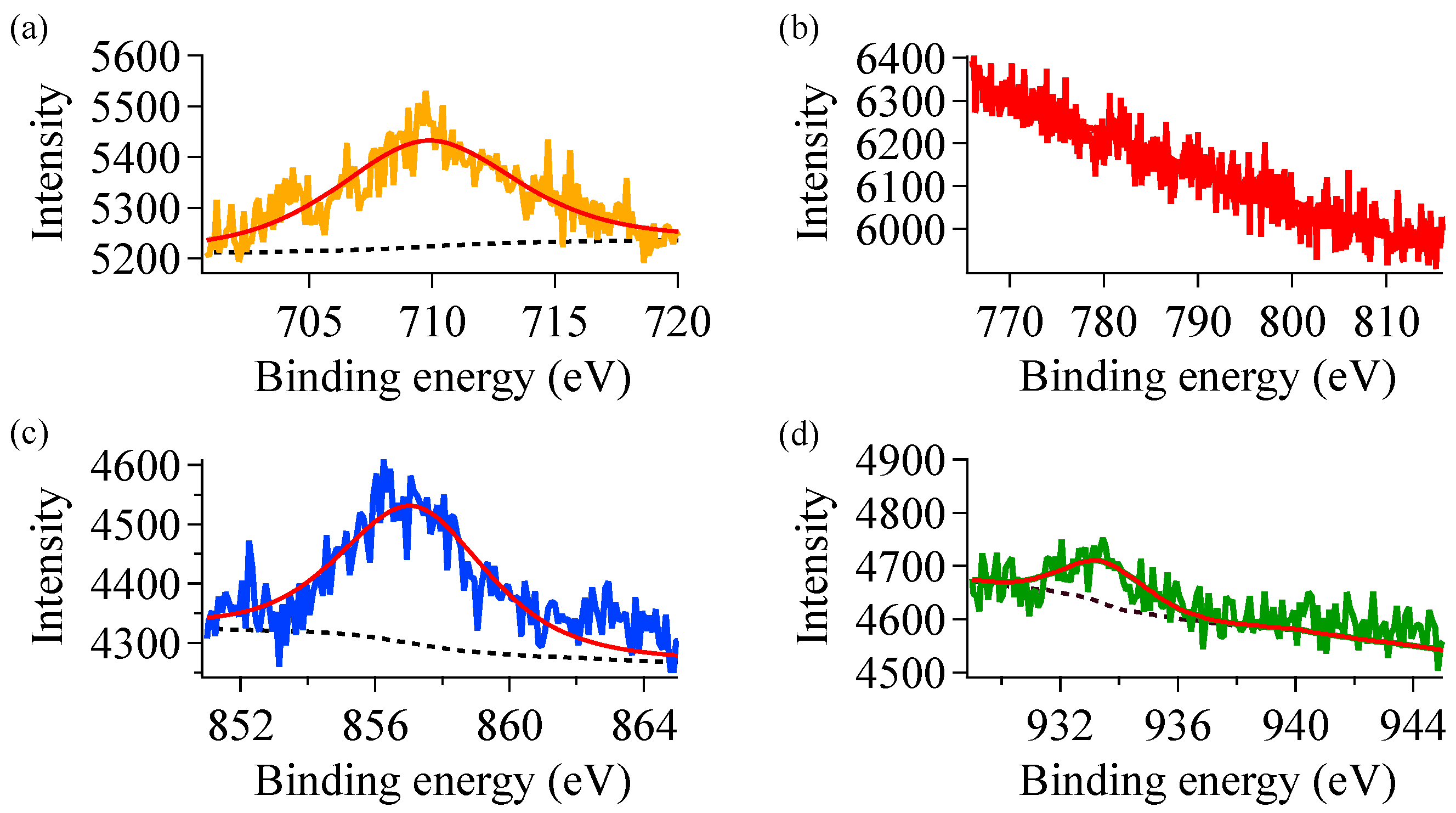
3.4. Influence of Surface Chemistry on Photocatalysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fujishima, A.; Honda, K. Electrochemical Photolysis of Water at a Semiconductor Electrode. Nature 1972, 238, 37–38. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, A.; Zhang, X.; Tryk, D.A. TiO2 Photocatalysis and Related Surface Phenomena. Surf. Sci. Rep. 2008, 63, 515–582. [Google Scholar] [CrossRef]
- Takanabe, K. Photocatalytic Water Splitting: Quantitative Approaches toward Photocatalyst by Design. ACS Catal. 2017, 7, 8006–8022. [Google Scholar] [CrossRef]
- Carcel, R.A.; Andronic, L.; Duta, A. Photocatalytic Activity and Stability of TiO2 and WO3 Thin Films. Mater. Charact. 2012, 70, 68–73. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Navalon, S.; Corma, A.; Garcia, H. Photocatalytic CO2 Reduction by TiO2 and Related Titanium Containing Solids. Energy Environ. Sci. 2012, 5, 9217. [Google Scholar] [CrossRef]
- Mino, L.; Negri, C.; Santalucia, R.; Cerrato, G.; Spoto, G.; Martra, G. Morphology, Surface Structure and Water Adsorption Properties of TiO2 Nanoparticles: A Comparison of Different Commercial Samples. Molecules 2020, 25, 4605. [Google Scholar] [CrossRef]
- Lonnen, J.; Kilvington, S.; Kehoe, S.C.; Al-Touati, F.; McGuigan, K.G. Solar and Photocatalytic Disinfection of Protozoan, Fungal and Bacterial Microbes in Drinking Water. Water Res. 2005, 39, 877–883. [Google Scholar] [CrossRef]
- Herrmann, J.-M. Heterogeneous Photocatalysis: Fundamentals and Applications to the Removal of Various Types of Aqueous Pollutants. Catal. Today 1999, 53, 115–129. [Google Scholar] [CrossRef]
- Hager, S.; Bauer, R. Heterogeneous Photocatalytic Oxidation of Organics for Air Purification by near UV Irradiated Titanium Dioxide. Chemosphere 1999, 38, 1549–1559. [Google Scholar] [CrossRef]
- Ochiai, T.; Aoki, D.; Saito, H.; Akutsu, Y.; Nagata, M. Analysis of Adsorption and Decomposition of Odour and Tar Components in Tobacco Smoke on Non-Woven Fabric-Supported Photocatalysts. Catalysts 2020, 10, 304. [Google Scholar] [CrossRef]
- Takanabe, K.; Domen, K. Toward Visible Light Response: Overall Water Splitting Using Heterogeneous Photocatalysts. Green 2011, 1, 313. [Google Scholar] [CrossRef]
- Murakami, S.-Y.; Kominami, H.; Kera, Y.; Ikeda, S.; Noguchi, H.; Uosaki, K.; Ohtani, B. Evaluation of Electron-Hole Recombination Properties of Titanium (IV) Oxide Particles with High Photocatalytic Activity. Res. Chem. Intermed. 2007, 33, 285–296. [Google Scholar] [CrossRef]
- Khlyustova, A.; Sirotkin, N.; Kusova, T.; Kraev, A.; Titov, V.; Agafonov, A. Doped TiO2: The Effect of Doping Elements on Photocatalytic Activity. Mater. Adv. 2020, 1, 1193–1201. [Google Scholar] [CrossRef]
- Nur, A.S.M.; Sultana, M.; Mondal, A.; Islam, S.; Robel, F.N.; Islam, A.; Sumi, M.S.A. A Review on the Development of Elemental and Codoped TiO2 Photocatalysts for Enhanced Dye Degradation under UV–Vis Irradiation. J. Water Process Eng. 2022, 47, 102728. [Google Scholar] [CrossRef]
- Lu, C.-M.; Sharma, R.K.; Lin, P.-Y.; Huang, Y.-H.; Chen, J.-S.; Lee, W.-C.; Chen, C.-Y. Characteristics of Doped TiO2 Nanoparticle Photocatalysts Prepared by the Rotten Egg White. Materials 2022, 15, 4231. [Google Scholar] [CrossRef]
- Asahi, R.; Morikawa, T.; Ohwaki, T.; Aoki, K.; Taga, Y. Visible-Light Photocatalysis in Nitrogen-Doped Titanium Oxides. Science 2001, 293, 269–271. [Google Scholar] [CrossRef]
- Sirivallop, A.; Areerob, T.; Chiarakorn, S. Enhanced Visible Light Photocatalytic Activity of N and Ag Doped and Co-Doped TiO2 Synthesized by Using an In-Situ Solvothermal Method for Gas Phase Ammonia Removal. Catalysts 2020, 10, 251. [Google Scholar] [CrossRef]
- Yu, H.; Irie, H.; Hashimoto, K. Conduction Band Energy Level Control of Titanium Dioxide: Toward an Efficient Visible-Light-Sensitive Photocatalyst. J. Am. Chem. Soc. 2010, 132, 6898–6899. [Google Scholar] [CrossRef]
- Huang, F.; Yan, A.; Zhao, H. Influences of Doping on Photocatalytic Properties of TiO2 Photocatalyst. In Semiconductor Photocatalysis—Materials, Mechanisms and Applications; InTech: London, UK, 2016. [Google Scholar]
- Slamet; Nasution, H.W.; Purnama, E.; Kosela, S.; Gunlazuardi, J. Photocatalytic Reduction of CO2 on Copper-Doped Titania Catalysts Prepared by Improved-Impregnation Method. Catal. Commun. 2005, 6, 313–319. [Google Scholar] [CrossRef]
- Haque, M.M.; Khan, A.; Umar, K.; Mir, N.A.; Muneer, M.; Harada, T.; Matsumura, M. Synthesis, Characterization and Photocatalytic Activity of Visible Light Induced Ni-Doped TiO2. Energy Environ. Focus 2013, 2, 73–78. [Google Scholar] [CrossRef]
- Iwasaki, M.; Hara, M.; Kawada, H.; Tada, H.; Ito, S. Cobalt Ion-Doped TiO2 Photocatalyst Response to Visible Light. J. Colloid Interface Sci. 2000, 224, 202–204. [Google Scholar] [CrossRef] [PubMed]
- Yalçın, Y.; Kılıç, M.; Çınar, Z. Fe+3-Doped TiO2: A Combined Experimental and Computational Approach to the Evaluation of Visible Light Activity. Appl. Catal. B 2010, 99, 469–477. [Google Scholar] [CrossRef]
- Karunakaran, C.; Abiramasundari, G.; Gomathisankar, P.; Manikandan, G.; Anandi, V. Cu-Doped TiO2 Nanoparticles for Photocatalytic Disinfection of Bacteria under Visible Light. J. Colloid Interface Sci. 2010, 352, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Alotaibi, A.M.; Williamson, B.A.D.; Sathasivam, S.; Kafizas, A.; Alqahtani, M.; Sotelo-Vazquez, C.; Buckeridge, J.; Wu, J.; Nair, S.P.; Scanlon, D.O.; et al. Enhanced Photocatalytic and Antibacterial Ability of Cu-Doped Anatase TiO2 Thin Films: Theory and Experiment. ACS Appl. Mater. Interfaces 2020, 12, 15348–15361. [Google Scholar] [CrossRef]
- Mathew, S.; Ganguly, P.; Rhatigan, S.; Kumaravel, V.; Byrne, C.; Hinder, S.J.; Bartlett, J.; Nolan, M.; Pillai, S.C. Cu-Doped TiO2: Visible Light Assisted Photocatalytic Antimicrobial Activity. Appl. Sci. 2018, 8, 2067. [Google Scholar] [CrossRef]
- Li, G.; Dimitrijevic, N.M.; Chen, L.; Rajh, T.; Gray, K.A. Role of Surface/Interfacial Cu2+ Sites in the Photocatalytic Activity of Coupled CuO−TiO2 Nanocomposites. J. Phys. Chem. C 2008, 112, 19040–19044. [Google Scholar] [CrossRef]
- Wu, M.-C.; Wu, P.-Y.; Lin, T.-H.; Lin, T.-F. Photocatalytic Performance of Cu-Doped TiO2 Nanofibers Treated by the Hydrothermal Synthesis and Air-Thermal Treatment. Appl. Surf. Sci. 2018, 430, 390–398. [Google Scholar] [CrossRef]
- Colón, G.; Maicu, M.; Hidalgo, M.C.; Navío, J.A. Cu-Doped TiO2 Systems with Improved Photocatalytic Activity. Appl. Catal. B 2006, 67, 41–51. [Google Scholar] [CrossRef]
- Feng, S.-H.; Li, G.-H. Hydrothermal and Solvothermal Syntheses. In Modern Inorganic Synthetic Chemistry; Elsevier: Amsterdam, The Netherlands, 2017; pp. 73–104. [Google Scholar]
- Razali, M.H.; Ahmad-Fauzi, M.N.; Mohamed, A.R.; Sreekantan, S. Hydrothermal Synthesis and Characterisation of Cu Doped TiO2 Nanotubes for Photocatalytic Degradation of Methyl Orange. Adv. Mater. Res. 2014, 911, 126–130. [Google Scholar] [CrossRef]
- Deki, S.; Aoi, Y.; Yanagimoto, H.; Ishii, K.; Akamatsu, K.; Mizuhata, M.; Kajinami, A. Preparation and Characterization of Au-Dispersed TiO2 Thin Films by a Liquid-Phase Deposition Method. J. Mater. Chem. 1996, 6, 1879. [Google Scholar] [CrossRef]
- Maki, H.; Okumura, Y.; Ikuta, H.; Mizuhata, M. Ionic Equilibria for Synthesis of TiO2 Thin Films by the Liquid-Phase Deposition. J. Phys. Chem. C 2014, 118, 11964–11974. [Google Scholar] [CrossRef]
- Herbig, B.; Löbmann, P. TiO2 Photocatalysts Deposited on Fiber Substrates by Liquid Phase Deposition. J. Photochem. Photobiol. A Chem. 2004, 163, 359–365. [Google Scholar] [CrossRef]
- Yu, H.; Lee, S.C.; Yu, J.; Ao, C.H. Photocatalytic Activity of Dispersed TiO2 Particles Deposited on Glass Fibers. J. Mol. Catal. A Chem. 2006, 246, 206–211. [Google Scholar] [CrossRef]
- Listiani, P.; Sanusi; Honda, M.; Oya, H.; Horio, Y.; Ichikawa, Y. Optimization of Hydrolysis Temperature in Liquid Phase Deposition for TiO2 Photocatalysis. Jpn. J. Appl. Phys. 2022, 61, 075508. [Google Scholar] [CrossRef]
- Honda, M.; Ochiai, T.; Listiani, P.; Yamaguchi, Y.; Ichikawa, Y. Low-Temperature Synthesis of Cu-Doped Anatase TiO2 Nanostructures via Liquid Phase Deposition Method for Enhanced Photocatalysis. Materials 2023, 16, 639. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Liu, B.; Yin, S.; Sato, T.; Wang, Y. Visible Light-Driven Photocatalytic Activity of Oleic Acid-Coated TiO2 Nanoparticles Synthesized from Absolute Ethanol Solution. Nanoscale Res. Lett. 2015, 10, 415. [Google Scholar] [CrossRef]
- Louwerse, M.J.; Piccinini, M.; de Jong, K.P. Diffusion Mechanisms for Ions over Hydroxylated Surfaces: Cu on γ-Al2O3. J. Phys. Chem. C 2019, 123, 18502–18507. [Google Scholar] [CrossRef]
- Challagulla, S.; Tarafder, K.; Ganesan, R.; Roy, S. Structure Sensitive Photocatalytic Reduction of Nitroarenes over TiO2. Sci. Rep. 2017, 7, 8783. [Google Scholar] [CrossRef]
- Durante, O.; Di Giorgio, C.; Granata, V.; Neilson, J.; Fittipaldi, R.; Vecchione, A.; Carapella, G.; Chiadini, F.; DeSalvo, R.; Dinelli, F.; et al. Emergence and Evolution of Crystallization in TiO2 Thin Films: A Structural and Morphological Study. Nanomaterials 2021, 11, 1409. [Google Scholar] [CrossRef]
- Shannon, R.D. Revised Effective Ionic Radii and Systematic Studies of Interatomic Distances in Halides and Chalcogenides. Acta Crystallogr. Sect. A 1976, 32, 751–767. [Google Scholar] [CrossRef]
- Choi, J.; Park, H.; Hoffmann, M.R. Effects of Single Metal-Ion Doping on the Visible-Light Photoreactivity of TiO2. J. Phys. Chem. C 2010, 114, 783–792. [Google Scholar] [CrossRef]
- Scanlon, D.O.; Dunnill, C.W.; Buckeridge, J.; Shevlin, S.A.; Logsdail, A.J.; Woodley, S.M.; Catlow, C.R.A.; Powell, M.J.; Palgrave, R.G.; Parkin, I.P.; et al. Band Alignment of Rutile and Anatase TiO2. Nat. Mater. 2013, 12, 798–801. [Google Scholar] [CrossRef] [PubMed]
- Brus, L. Electronic Wave Functions in Semiconductor Clusters: Experiment and Theory. J. Phys. Chem. 1986, 90, 2555–2560. [Google Scholar] [CrossRef]
- Thompson, T.L.; Yates, J.T. TiO2-Based Photocatalysis: Surface Defects, Oxygen and Charge Transfer. Top. Catal. 2005, 35, 197–210. [Google Scholar] [CrossRef]
- Honda, M.; Saito, Y.; Kawata, S. Individual TiO2 Nanocrystals Probed by Resonant Rayleigh Scattering Spectroscopy. Appl. Phys. Express 2014, 7, 112402. [Google Scholar] [CrossRef]
- Kusumawardani, L.J.; Syahputri, Y. Study of Structural and Optical Properties of Fe(III)-Doped TiO2 Prepared by Sol-Gel Method. IOP Conf. Ser. Earth Environ. Sci. 2019, 299, 012066. [Google Scholar] [CrossRef]
- Vega, M.P.B.; Hinojosa-Reyes, M.; Hernández-Ramírez, A.; Mar, J.L.G.; Rodríguez-González, V.; Hinojosa-Reyes, L. Visible Light Photocatalytic Activity of Sol–Gel Ni-Doped TiO2 on p-Arsanilic Acid Degradation. J. Solgel Sci. Technol. 2018, 85, 723–731. [Google Scholar] [CrossRef]
- Boutlala, A.; Bourfaa, F.; Mahtili, M.; Bouaballou, A. Deposition of Co-Doped TiO2 Thin Films by Sol-Gel Method. IOP Conf. Ser. Mater. Sci. Eng. 2016, 108, 012048. [Google Scholar] [CrossRef]
- Wtulich, M.; Szkoda, M.; Gajowiec, G.; Gazda, M.; Jurak, K.; Sawczak, M.; Lisowska-Oleksiak, A. Hydrothermal Cobalt Doping of Titanium Dioxide Nanotubes towards Photoanode Activity Enhancement. Materials 2021, 14, 1507. [Google Scholar] [CrossRef]
- Nguyen, V.N.; Nguyen, N.K.T.; Nguyen, P.H. Hydrothermal Synthesis of Fe-Doped TiO2 Nanostructure Photocatalyst. Adv. Nat. Sci. Nanosci. Nanotechnol. 2011, 2, 035014. [Google Scholar] [CrossRef]
- Guan, B.; Yu, J.; Guo, S.; Yu, S.; Han, S. Porous Nickel Doped Titanium Dioxide Nanoparticles with Improved Visible Light Photocatalytic Activity. Nanoscale Adv. 2020, 2, 1352–1357. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.-P.; Chen, H.; Nakaruk, A.; Koshy, P.; Sorrell, C.C. Effect of Annealing Temperature on the Photocatalytic Activity of TiO2 Thin Films. Energy Procedia 2013, 34, 627–636. [Google Scholar] [CrossRef]
- Li, D.; Song, H.; Meng, X.; Shen, T.; Sun, J.; Han, W.; Wang, X. Effects of Particle Size on the Structure and Photocatalytic Performance by Alkali-Treated TiO2. Nanomaterials 2020, 10, 546. [Google Scholar] [CrossRef]
- Erdem, B.; Hunsicker, R.A.; Simmons, G.W.; Sudol, E.D.; Dimonie, V.L.; El-Aasser, M.S. XPS and FTIR Surface Characterization of TiO2 Particles Used in Polymer Encapsulation. Langmuir 2001, 17, 2664–2669. [Google Scholar] [CrossRef]
- Zhu, L.; Lu, Q.; Lv, L.; Wang, Y.; Hu, Y.; Deng, Z.; Lou, Z.; Hou, Y.; Teng, F. Ligand-Free Rutile and Anatase TiO2 Nanocrystals as Electron Extraction Layers for High Performance Inverted Polymer Solar Cells. RSC Adv. 2017, 7, 20084–20092. [Google Scholar] [CrossRef]
- Jackman, M.J.; Thomas, A.G.; Muryn, C. Photoelectron Spectroscopy Study of Stoichiometric and Reduced Anatase TiO2 (101) Surfaces: The Effect of Subsurface Defects on Water Adsorption at Near-Ambient Pressures. J. Phys. Chem. C 2015, 119, 13682–13690. [Google Scholar] [CrossRef]
- An, H.; Wang, D.; Miao, S.; Yang, Q.; Zhao, X.; Wang, Y. Preparation of Ni-IL/SiO2 and Its Catalytic Performance for One-Pot Sequential Synthesis of 2-Propylheptanol from n-Valeraldehyde. RSC Adv. 2020, 10, 28100–28105. [Google Scholar] [CrossRef]
- Yang, S.; Song, X.; Zhang, P.; Sun, J.; Gao, L. Self-Assembled A-Fe2O3 Mesocrystals/Graphene Nanohybrid for Enhanced Electrochemical Capacitors. Small 2014, 10, 2270–2279. [Google Scholar] [CrossRef]
- You, M.; Kim, T.G.; Sung, Y.-M. Synthesis of Cu-Doped TiO2 Nanorods with Various Aspect Ratios and Dopant Concentrations. Cryst. Growth Des. 2010, 10, 983–987. [Google Scholar] [CrossRef]
- Li, L.; Cai, Y.; Li, G.; Mu, X.; Wang, K.; Chen, J. Synergistic Effect on the Photoactivation of the Methane CH Bond over Ga3+-Modified ETS-10. Angew. Chem. Int. Ed. 2012, 51, 4702–4706. [Google Scholar] [CrossRef]
- Li, Q.; Ouyang, Y.; Li, H.; Wang, L.; Zeng, J. Photocatalytic Conversion of Methane: Recent Advancements and Prospects. Angew. Chem. Int. Ed. 2022, 61, e202108069. [Google Scholar] [CrossRef]



| Conc. (mM) | Fe | Co | Ni | Cu |
|---|---|---|---|---|
| 0.1 | 570 ± 150 | 550 ± 70 | 450 ± 60 | 1460 ± 190 |
| 1.0 | 500 ± 150 | 450 ± 110 | 580 ± 80 | 1420 ± 170 |
| 10 | 270 ± 40 | 620 ± 130 | 650 ± 100 | 1290 ± 130 |
| Temperature | Pressure | Reaction Time | Bandgaps | Refs. | |
|---|---|---|---|---|---|
| This study | RT~80 °C | atmospheric | 3 h | 2.4~3.2 eV | - |
| Sol-gel | 500~600 °C | atmospheric | 4–7 h | 2.0~3.0 eV | [20,29,48,49,50] |
| Hydrothermal | 120~180 °C | several GPa | 1–12 h | 2.85~3.0 eV | [28,30,31,51,52,53] |
| Aerosol assisted CVD | ~450 °C | atmospheric | N/A | 3.2 eV | [25] |
| Sample | Surface Area (m2/g) |
|---|---|
| Commercial | 250.26 |
| Undoped | 196.84 |
| Fe | 114.00 |
| Co | 141.86 |
| Ni | 141.86 |
| Cu | 182.61 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Honda, M.; Yoshii, Y.; Okayama, N.; Ichikawa, Y. Low Temperature Synthesis of 3d Metal (Fe, Co, Ni, Cu)-Doped TiO2 Photocatalyst via Liquid Phase Deposition Technique. Sustain. Chem. 2025, 6, 1. https://doi.org/10.3390/suschem6010001
Honda M, Yoshii Y, Okayama N, Ichikawa Y. Low Temperature Synthesis of 3d Metal (Fe, Co, Ni, Cu)-Doped TiO2 Photocatalyst via Liquid Phase Deposition Technique. Sustainable Chemistry. 2025; 6(1):1. https://doi.org/10.3390/suschem6010001
Chicago/Turabian StyleHonda, Mitsuhiro, Yusaku Yoshii, Nobuchika Okayama, and Yo Ichikawa. 2025. "Low Temperature Synthesis of 3d Metal (Fe, Co, Ni, Cu)-Doped TiO2 Photocatalyst via Liquid Phase Deposition Technique" Sustainable Chemistry 6, no. 1: 1. https://doi.org/10.3390/suschem6010001
APA StyleHonda, M., Yoshii, Y., Okayama, N., & Ichikawa, Y. (2025). Low Temperature Synthesis of 3d Metal (Fe, Co, Ni, Cu)-Doped TiO2 Photocatalyst via Liquid Phase Deposition Technique. Sustainable Chemistry, 6(1), 1. https://doi.org/10.3390/suschem6010001







