The Diagnostic Value of Chromogranin A in Neuroendocrine Neoplasms is Potentiated by Clinical Factors and Inflammatory Markers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Statistical Analysis
3. Results
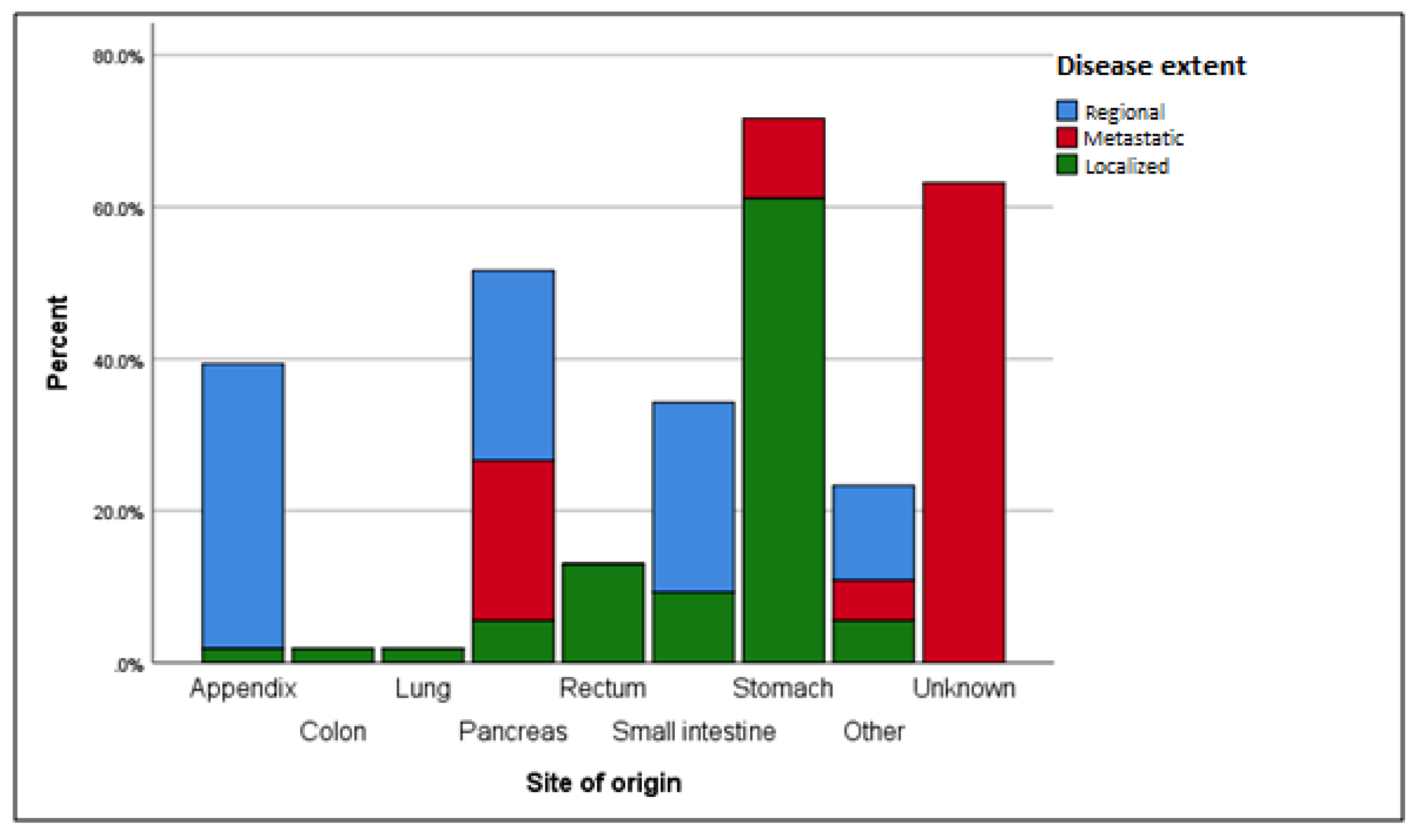
3.1. Disease Extent
3.2. Grading
3.3. Gender and Age
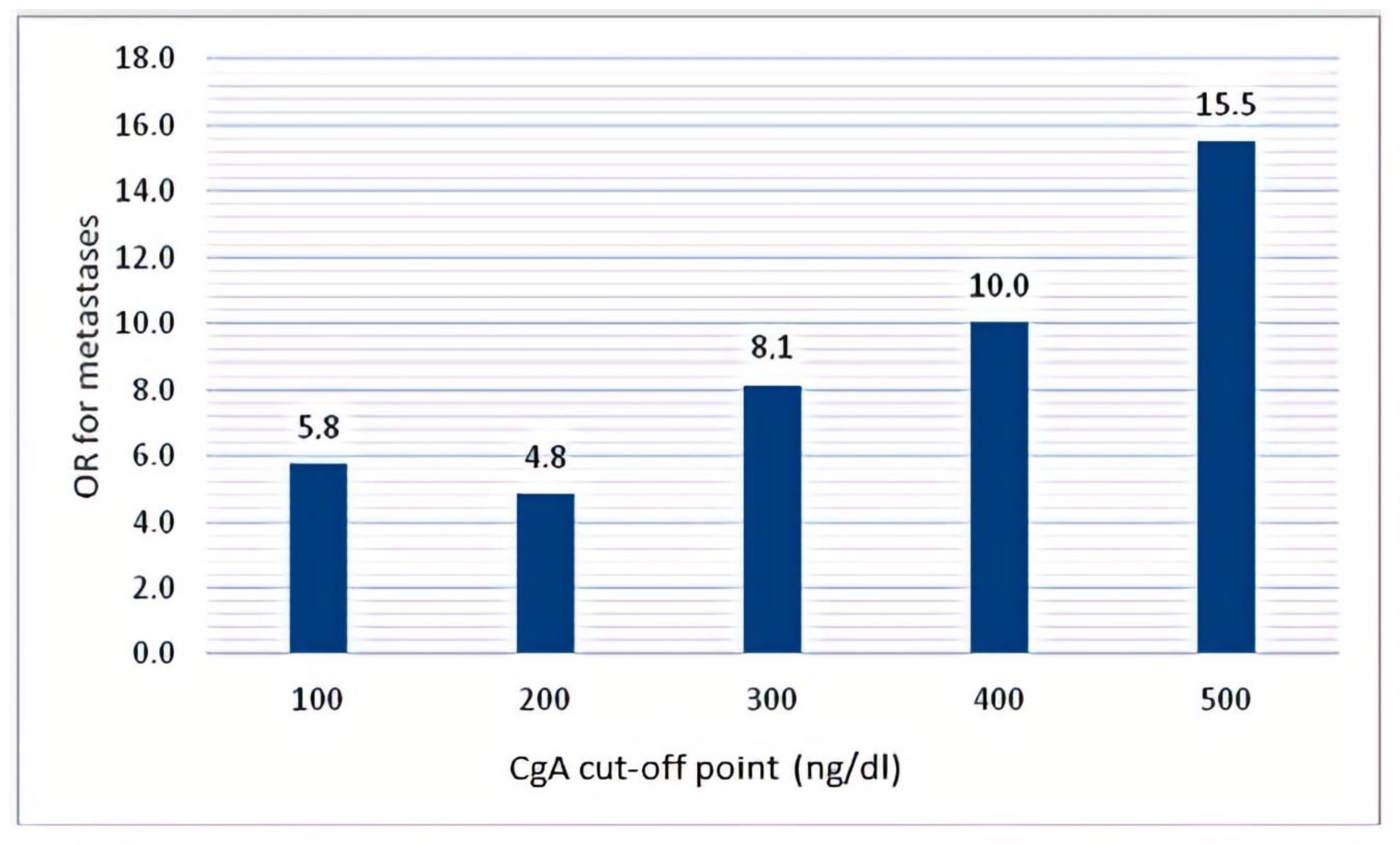
3.4. CgA and NSE
3.5. CRP and Other Inflammatory Markers
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Taal, B.G.; Visser, O. Epidemiologys of neuroendocrine tumours. Neuroendocrinology 2004, 80 (Suppl. 1), 3–7. [Google Scholar] [CrossRef] [PubMed]
- Dasari, A.; Shen, C.; Halperin, D.; Zhao, B.; Zhou, S.; Xu, Y.; Shih, T.; Yao, J.C. Trends in the Incidence, Prevalence, and Survival Outcomes in Patients with Neuroendocrine Tumors in the United States. JAMA Oncol. 2017, 3, 1335–1342. [Google Scholar] [CrossRef] [PubMed]
- Modlin, I.M.; Moss, S.F.; Chung, D.C.; Jensen, R.T.; Snyderwine, E. Priorities for improving the management of gastroenteropancreatic neuroendocrine tumors. J. Natl. Cancer Inst. 2008, 100, 1282–1289. [Google Scholar] [CrossRef] [PubMed]
- Hallet, J.; Law, C.H.; Cukier, M.; Saskin, R.; Liu, N.; Singh, S. Exploring the rising incidence of neuroendocrine tumors: A population-based analysis of epidemiology, metastatic presentation, and outcomes. Cancer 2015, 121, 589–597. [Google Scholar] [CrossRef] [PubMed]
- Caldarella, A.; Crocetti, E.; Paci, E. Distribution, incidence, and prognosis in neuroendocrine tumors: A population based study from a cancer registry. Pathol. Oncol. Res. 2011, 17, 759–763. [Google Scholar] [CrossRef]
- Nikou, G.C.; Pazaitou-Panayiotou, K.; Dimitroulopoulos, D.; Alexandrakis, G.; Papakostas, P.; Vaslamatzis, M.; Kaldrymidis, P.; Markussis, V.; Koumarianou, A.; “G-NET-Registry” investigators. Results of a prospective multicenter neuroendocrine tumor registry reporting on clinicopathologic characteristics of Greek patients. BMC Endocr. Disord. 2016, 16, 8. [Google Scholar] [CrossRef] [Green Version]
- Oberg, K.; Couvelard, A.; Delle Fave, G.; Gross, D.; Grossman, A.; Jensen, R.T.; Pape, U.F.; Perren, A.; Rindi, G.; Ruszniewski, P.; et al. ENETS Consensus Guidelines for Standard of Care in Neuroendocrine Tumours: Biochemical Markers. Neuroendocrinology 2017, 105, 201–211. [Google Scholar] [CrossRef]
- Marotta, V.; Zatelli, M.C.; Sciammarella, C.; Ambrosio, M.R.; Bondanelli, M.; Colao, A.; Faggiano, A. Chromogranin A as circulating marker for diagnosis and management of neuroendocrine neoplasms: More flaws than fame. Endocr. Relat. Cancer 2018, 25, R11–R29. [Google Scholar] [CrossRef]
- Massironi, S.; Rossi, R.E.; Casazza, G.; Conte, D.; Ciafardini, C.; Galeazzi, M.; Peracchi, M. Chromogranin A in diagnosing and monitoring patients with gastroenteropancreatic neuroendocrine neoplasms: A large series from a single institution. Neuroendocrinology 2014, 100, 240–249. [Google Scholar] [CrossRef]
- Rossi, R.E.; Ciafardini, C.; Sciola, V.; Conte, D.; Massironi, S. Chromogranin A in the Follow-up of Gastroenteropancreatic Neuroendocrine Neoplasms: Is It Really Game Over? A Systematic Review and Meta-analysis. Pancreas 2018, 47, 1249–1255. [Google Scholar] [CrossRef]
- Sciola, V.; Massironi, S.; Conte, D.; Caprioli, F.; Ferrero, S.; Ciafardini, C.; Peracchi, M.; Bardella, M.T.; Piodi, L. Plasma chromogranin a in patients with inflammatory bowel disease. Inflamm. Bowel Dis. 2009, 15, 867–871. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Granberg, D. Biochemical Testing in Patients with Neuroendocrine Tumors. Front. Horm. Res. 2015, 44, 24–39. [Google Scholar] [CrossRef] [PubMed]
- Hofland, J.; Kaltsas, G.; de Herder, W.W. Advances in the diagnosis and management of well-differentiated neuroendocrine neoplasms. Endocr. Rev. 2019, 41, bnz004. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef]
- Lu, H.; Ouyang, W.; Huang, C. Inflammation, a key event in cancer development. Mol. Cancer Res. 2006, 4, 221–233. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, J.V.; Cobucci, R.N.; Jatoba, C.A.; Fernandes, T.A.; de Azevedo, J.W.; de Araujo, J.M. The role of the mediators of inflammation in cancer development. Pathol. Oncol. Res. 2015, 21, 527–534. [Google Scholar] [CrossRef] [PubMed]
- Shrotriya, S.; Walsh, D.; Nowacki, A.S.; Lorton, C.; Aktas, A.; Hullihen, B.; Benanni-Baiti, N.; Hauser, K.; Ayvaz, S.; Estfan, B. Serum C-reactive protein is an important and powerful prognostic biomarker in most adult solid tumors. PLoS ONE 2018, 13, e0202555. [Google Scholar] [CrossRef]
- Mahmoud, F.A.; Rivera, N.I. The role of C-reactive protein as a prognostic indicator in advanced cancer. Curr. Oncol. Rep. 2002, 4, 250–255. [Google Scholar] [CrossRef]
- Brenner, D.R.; Scherer, D.; Muir, K.; Schildkraut, J.; Boffetta, P.; Spitz, M.R.; Le Marchand, L.; Chan, A.T.; Goode, E.L.; Ulrich, C.M.; et al. A review of the application of inflammatory biomarkers in epidemiologic cancer research. Cancer Epidemiol. Biomark. Prev. 2014, 23, 1729–1751. [Google Scholar] [CrossRef] [Green Version]
- Berkovic, M.C.; Jokic, M.; Marout, J.; Radosevic, S.; Zjacic-Rotkvic, V.; Kapitanovic, S. IL-2 -330 T/G SNP and serum values-potential new tumor markers in neuroendocrine tumors of the gastrointestinal tract and pancreas (GEP-NETs). J. Mol. Med. 2010, 88, 423–429. [Google Scholar] [CrossRef]
- Cigrovski Berkovic, M.; Cacev, T.; Catela Ivkovic, T.; Zjacic-Rotkvic, V.; Kapitanovic, S. New insights into the role of chronic inflammation and cytokines in the etiopathogenesis of gastroenteropancreatic neuroendocrine tumors. Neuroendocrinology 2014, 99, 75–84. [Google Scholar] [CrossRef]
- Klimstra, D.S.; Modlin, I.R.; Coppola, D.; Lloyd, R.V.; Suster, S. The pathologic classification of neuroendocrine tumors: A review of nomenclature, grading, and staging systems. Pancreas 2010, 39, 707–712. [Google Scholar] [CrossRef]
- Jensen, R.; Norton, J. Carcinoid tumors and the carcinoid syndrome (Chapter 34.6 Cancer of the Endocrine System). In Cancer Principles and Practice of Oncology; DeVita, V.T., Jr., Hellman, S., Rosenberg, S.A., Eds.; Williams and Wilkins: Philadelphia, PA, USA, 2005. [Google Scholar]
- Komac, O.; Bengi, G.; Sagol, O.; Akarsu, M. C-reactive protein may be a prognostic factor for the whole gastroenteropancreatic neuroendocrine tumor group. World J. Gastrointest. Oncol. 2019, 11, 139–152. [Google Scholar] [CrossRef] [PubMed]
- Noone, A.M.; Howlader, N.; Krapcho, M.; Miller, D.; Brest, A.; Yu, M.; Ruhl, J.; Tatalovich, Z.; Mariotto, A.; Lewis, D.R.; et al. SEER Cancer Statistics Review, 1975–2015; National Cancer Institute: Bethesda, MD, USA, 2017. Available online: https://seer.cancer.gov/csr/1975_2015/ (accessed on 1 September 2019).
- Lawrence, B.; Gustafsson, B.I.; Kidd, M.; Pavel, M.; Svejda, B.; Modlin, I.M. The clinical relevance of chromogranin A as a biomarker for gastroenteropancreatic neuroendocrine tumors. Endocrinol. Metab. Clin. North Am. 2011, 40, 111–134. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.; Turner, G.; King, B.; Jones, L.; Culliford, D.; McCance, D.; Ardill, J.; Johnston, B.T.; Poston, G.; Rees, M.; et al. Midgut neuroendocrine tumours with liver metastases: Results of the UKINETS study. Endocr. Relat. Cancer 2009, 16, 885–894. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arnold, R.; Wilke, A.; Rinke, A.; Mayer, C.; Kann, P.H.; Klose, K.J.; Scherag, A.; Hahmann, M.; Muller, H.H.; Barth, P. Plasma chromogranin A as marker for survival in patients with metastatic endocrine gastroenteropancreatic tumors. Clin. Gastroenterol. Hepatol. 2008, 6, 820–827. [Google Scholar] [CrossRef]
- Campana, D.; Nori, F.; Piscitelli, L.; Morselli-Labate, A.M.; Pezzilli, R.; Corinaldesi, R.; Tomassetti, P. Chromogranin A: Is it a useful marker of neuroendocrine tumors? J. Clin. Oncol. 2007, 25, 1967–1973. [Google Scholar] [CrossRef] [Green Version]
- Rossi, R.E.; Garcia-Hernandez, J.; Meyer, T.; Thirlwell, C.; Watkins, J.; Martin, N.G.; Caplin, M.E.; Toumpanakis, C. Chromogranin A as a predictor of radiological disease progression in neuroendocrine tumours. Ann. Transl. Med. 2015, 3, 118. [Google Scholar] [CrossRef]
- Citterio, D.; Pusceddu, S.; Facciorusso, A.; Coppa, J.; Milione, M.; Buzzoni, R.; Bongini, M.; de Braud, F.; Mazzaferro, V. Primary tumour resection may improve survival in functional well-differentiated neuroendocrine tumours metastatic to the liver. Eur. J. Surg. Oncol. 2017, 43, 380–387. [Google Scholar] [CrossRef]
- Yao, J.C.; Pavel, M.; Phan, A.T.; Kulke, M.H.; Hoosen, S.; St Peter, J.; Cherfi, A.; Oberg, K.E. Chromogranin A and neuron-specific enolase as prognostic markers in patients with advanced pNET treated with everolimus. J. Clin. Endocrinol. Metab. 2011, 96, 3741–3749. [Google Scholar] [CrossRef] [Green Version]
- Pavel, M.E.; Baudin, E.; Oberg, K.E.; Hainsworth, J.D.; Voi, M.; Rouyrre, N.; Peeters, M.; Gross, D.J.; Yao, J.C. Efficacy of everolimus plus octreotide LAR in patients with advanced neuroendocrine tumor and carcinoid syndrome: Final overall survival from the randomized, placebo-controlled phase 3 RADIANT-2 study. Ann. Oncol. 2017, 28, 1569–1575. [Google Scholar] [CrossRef] [PubMed]
- Mirkin, K.A.; Hollenbeak, C.S.; Wong, J. Impact of chromogranin A, differentiation, and mitoses in nonfunctional pancreatic neuroendocrine tumors </= 2 cm. 2017, 211, 206–214. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, D.; Grossman, A.; Gross, D.; Delle Fave, G.; Barkmanova, J.; O’Connor, J.; Pape, U.F.; Plockinger, U.; Mallorca Consensus Conference Participants; European Neuroendocrine Tumor Society. ENETS Consensus Guidelines for the Standards of Care in Neuroendocrine Tumors: Biochemical markers. Neuroendocrinology 2009, 90, 194–202. [Google Scholar] [CrossRef] [Green Version]
- van Adrichem, R.C.; Kamp, K.; Vandamme, T.; Peeters, M.; Feelders, R.A.; de Herder, W.W. Serum neuron-specific enolase level is an independent predictor of overall survival in patients with gastroenteropancreatic neuroendocrine tumors. Ann. Oncol. 2016, 27, 746–747. [Google Scholar] [CrossRef] [PubMed]
- Wiese, D.; Kampe, K.; Waldmann, J.; Heverhagen, A.E.; Bartsch, D.K.; Fendrich, V. C-Reactive Protein as a New Prognostic Factor for Survival in Patients With Pancreatic Neuroendocrine Neoplasia. J. Clin. Endocrinol. Metab. 2016, 101, 937–944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaitanidis, A.; Patel, D.; Kebebew, E. ASO Author Reflections: Systemic Inflammatory Markers in Pancreatic Neuroendocrine Tumors. Ann. Surg. Oncol. 2018, 25, 874–875. [Google Scholar] [CrossRef] [PubMed]
- Qi, Q.; Geng, Y.; Sun, M.; Wang, P.; Chen, Z. Clinical implications of systemic inflammatory response markers as independent prognostic factors for advanced pancreatic cancer. Pancreatology 2015, 15, 145–150. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, D.; Lin, Y.; Yu, M.; Lu, X.; Jian, Z.; Na, N.; Hou, B. Pretreatment hematologic markers as prognostic predictors of gastroenteropancreatic neuroendocrine tumors: A systematic review and meta-analysis. Oncotargets Ther. 2018, 11, 2489–2496. [Google Scholar] [CrossRef] [Green Version]

| Anthropometric and Tumor Characteristics | Patients (N = 93) |
|---|---|
| Age (years), mean (SD) | 57.57 (±14.76) |
| Gender, n (%) | |
| Female, n (%) | 57 (61.3%) |
| Male, n (%) | 36 (38.7%) |
| Primary tumor localization, n (%) | |
| Appendix | 4 (4.3%) |
| Colon | 2 (2.2%) |
| Lung | 1 (1.1%) |
| Pancreas | 14 (15.1%) |
| Rectum | 8 (8.6%) |
| Small intestine | 10 (10.8%) |
| Stomach | 36 (38.7%) |
| Unknown | 12 (12.9%) |
| Other a | 6 (6.5%) |
| MEN associated, n (%) | 1 (1.1%) |
| Disease extent, n (%) | |
| Localized | 62 (66.7%) |
| Regional | 12 (12.9%) |
| Metastatic | 19 (20.4%) |
| Presence of metastases, n (%) | |
| Non-Metastatic | 74 (79.5%) |
| Metastatic | 19 (20.4%) |
| Grading (WHO 2010), n (%) | |
| G1 | 68 (73.1%) |
| G2 | 22 (23.6%) |
| G3 | 3 (3.2%) |
| Inflammatory Marker | Mean ± SD | Relative Risk Ratio (RR) | p-Value |
|---|---|---|---|
| C-reactive protein (CRP) (mg/L) | 6.95 ± 7.57 | 1.493 | 0.008 |
| White blood count (WBC) | 9743.24 ± 7172.8 | 1.00 | 0.546 |
| Neutrophil-to-lymphocyte ratio (NLR) | 2.67 ± 1.92 | 2.222 | 0.279 |
| Platelet-to-lymphocyte ratio (PLR) | 133.39 ± 113.5 | 1.020 | 0.336 |
| Lymphocyte-to-monocyte ratio (LMR) | 5.66 ± 4.81 | 0.501 | 0.382 |
| Mean platelet volume (MPV) | 10.52 ± 10.6 | 7.1 | 0.506 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papalou, O.; Peppa, M.; Kandaraki, E.A.; Diamanti-Kandarakis, E.; Nikou, G.C. The Diagnostic Value of Chromogranin A in Neuroendocrine Neoplasms is Potentiated by Clinical Factors and Inflammatory Markers. Endocrines 2020, 1, 1-12. https://doi.org/10.3390/endocrines1010001
Papalou O, Peppa M, Kandaraki EA, Diamanti-Kandarakis E, Nikou GC. The Diagnostic Value of Chromogranin A in Neuroendocrine Neoplasms is Potentiated by Clinical Factors and Inflammatory Markers. Endocrines. 2020; 1(1):1-12. https://doi.org/10.3390/endocrines1010001
Chicago/Turabian StylePapalou, Olga, Melpomeni Peppa, Eleni A. Kandaraki, Evanthia Diamanti-Kandarakis, and George C. Nikou. 2020. "The Diagnostic Value of Chromogranin A in Neuroendocrine Neoplasms is Potentiated by Clinical Factors and Inflammatory Markers" Endocrines 1, no. 1: 1-12. https://doi.org/10.3390/endocrines1010001
APA StylePapalou, O., Peppa, M., Kandaraki, E. A., Diamanti-Kandarakis, E., & Nikou, G. C. (2020). The Diagnostic Value of Chromogranin A in Neuroendocrine Neoplasms is Potentiated by Clinical Factors and Inflammatory Markers. Endocrines, 1(1), 1-12. https://doi.org/10.3390/endocrines1010001







