Simulation of Colloidal Stability and Aggregation Tendency of Magnetic Nanoflowers in Biofluids
Abstract
:1. Introduction
2. Materials and Methods
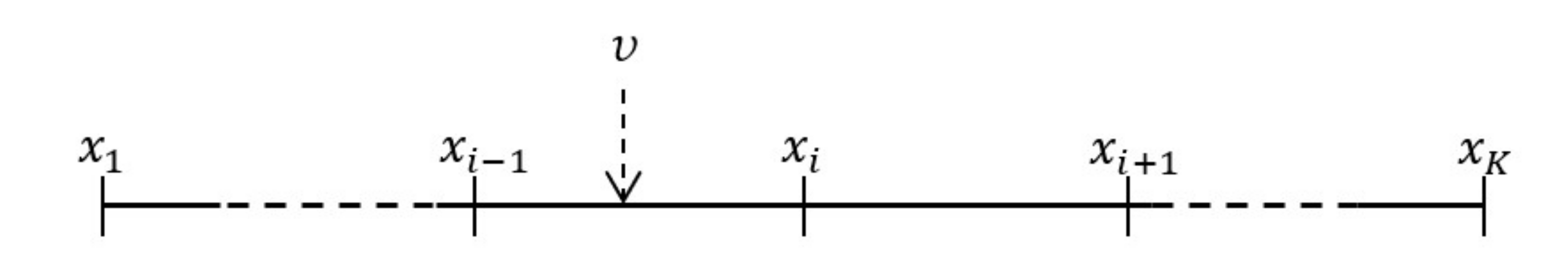
2.1. Numerical Model
2.2. Experimental Procedure
3. Results and Discussion
3.1. Model Validation
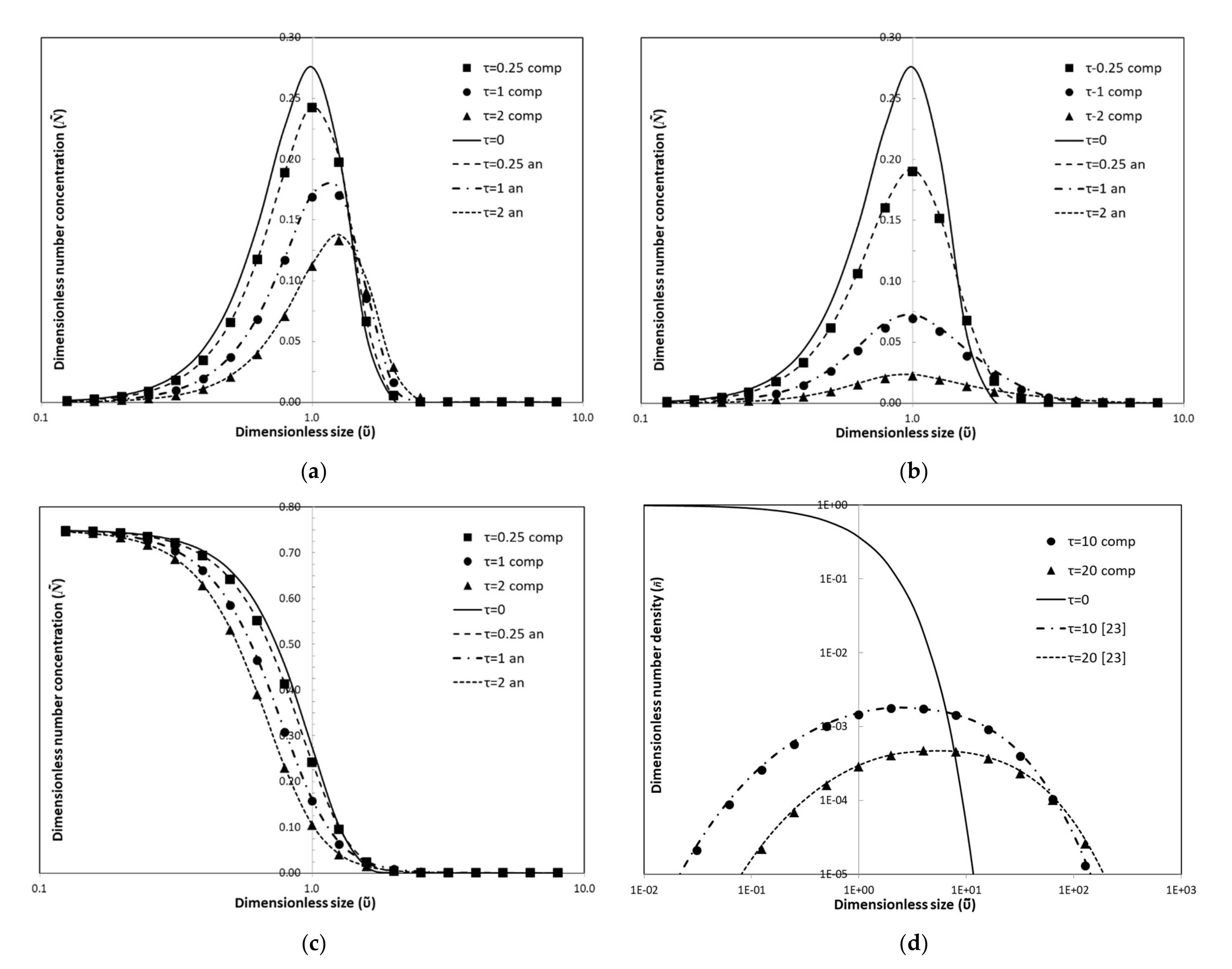
3.1.1. Constant Kernel
3.1.2. Linear Kernel
3.1.3. Quadratic Kernel
3.1.4. Brownian Kernel
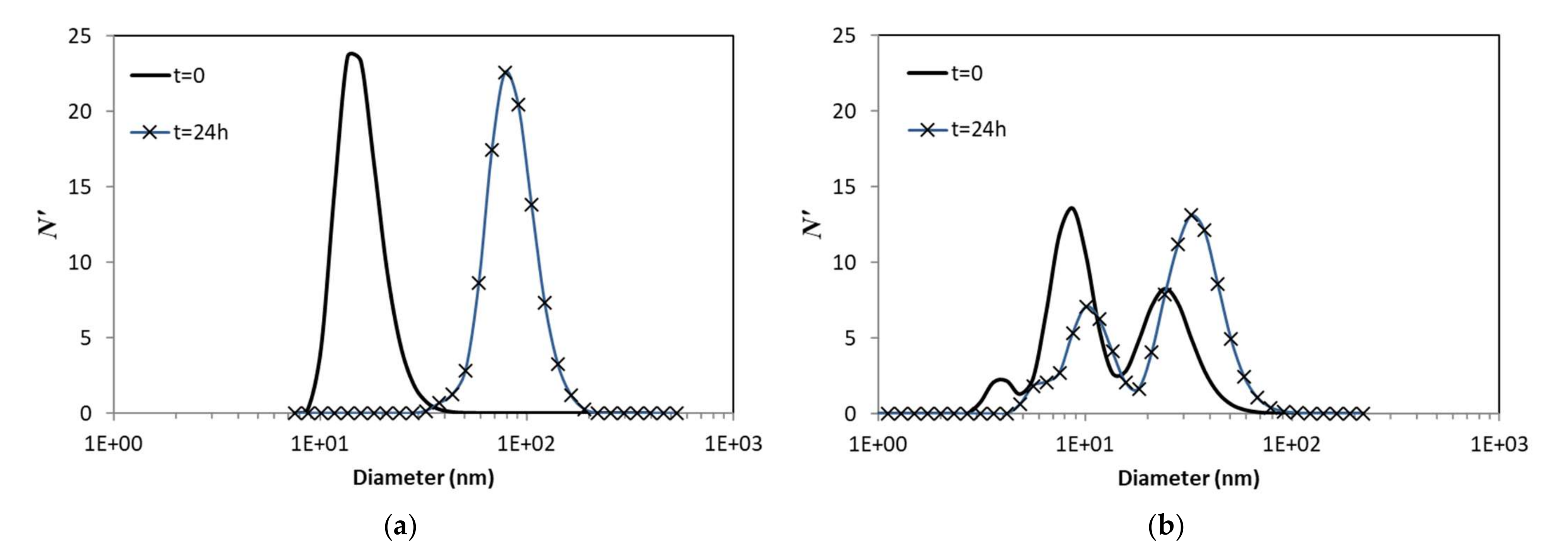
3.2. Aggregation Experiments
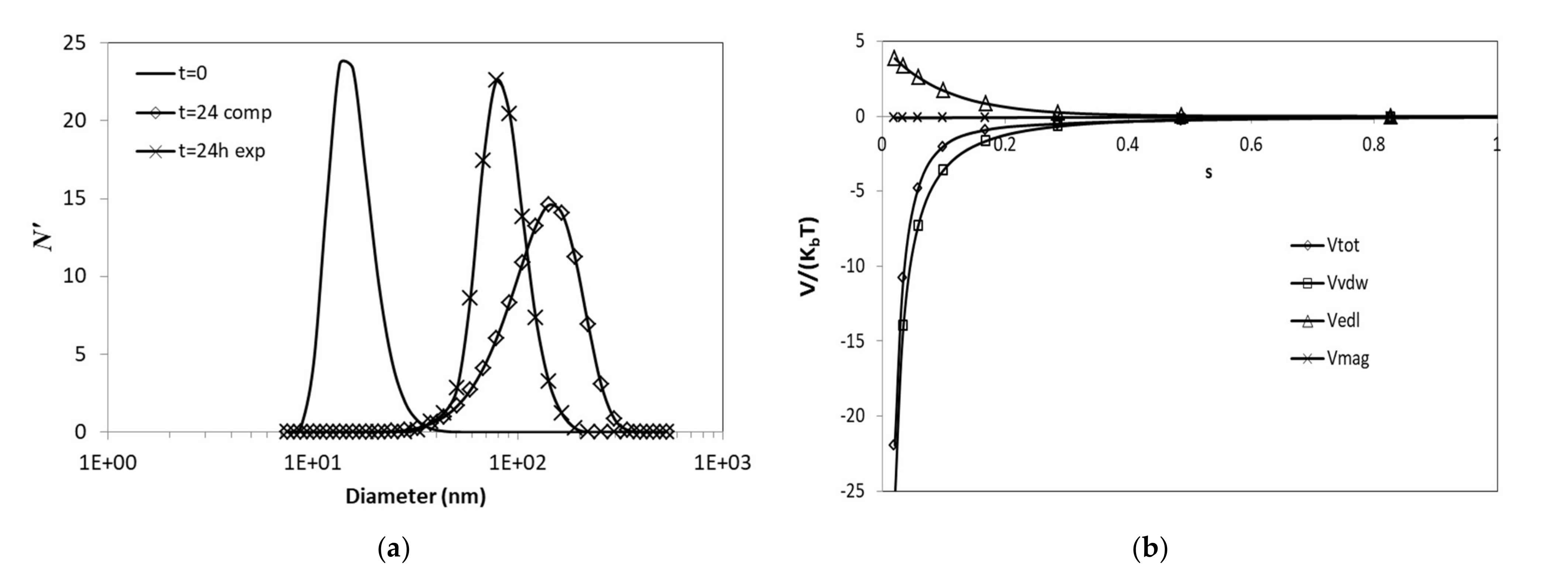
3.3. Simulations
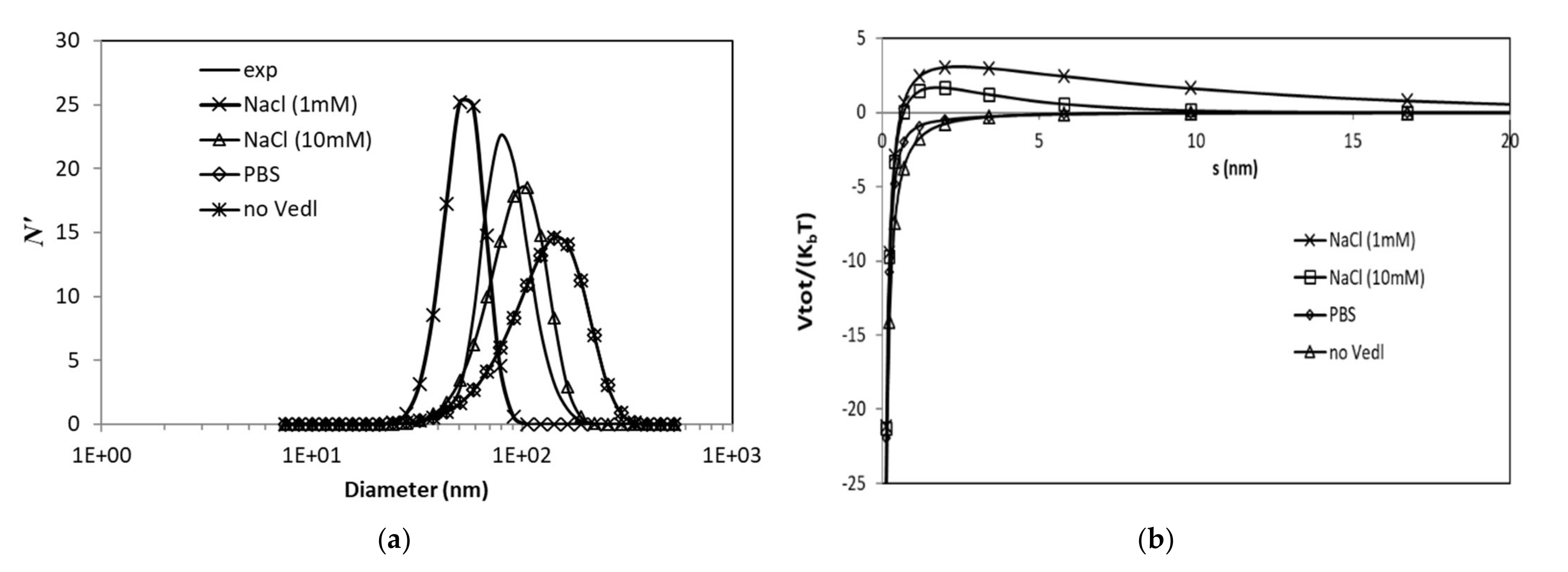
3.3.1. Effects of Ionic Strength
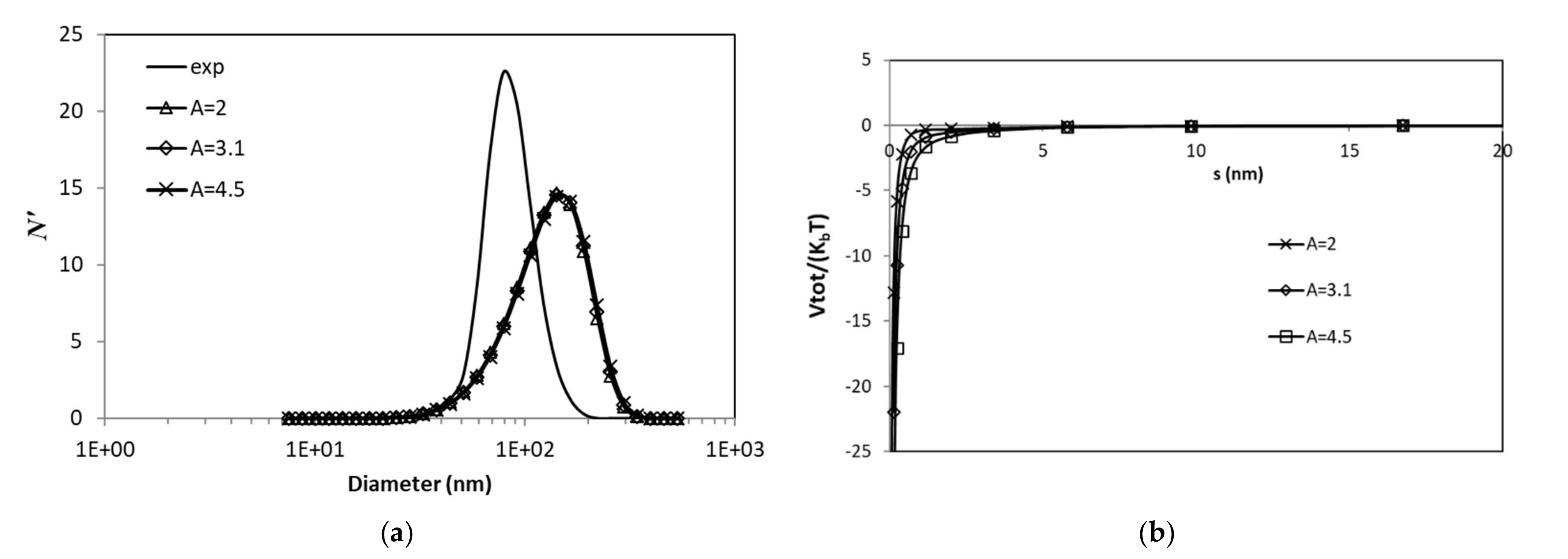
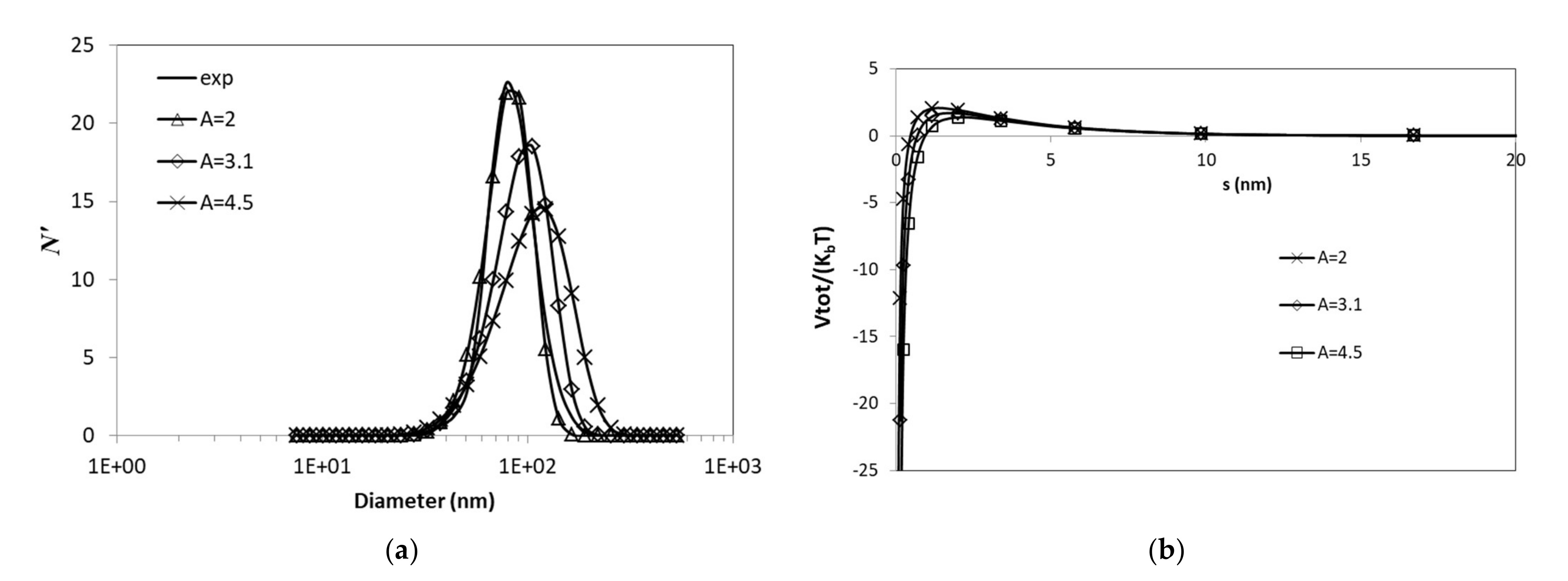
3.3.2. Effects of van der Waals Forces
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hegedűs, T.; Takács, D.; Vásárhelyi, L.; Szilágyi, I.; Kónya, Z. Specific Ion Effects on Aggregation and Charging Properties of Boron Nitride Nanospheres. Langmuir 2021, 37, 2466–2475. [Google Scholar] [CrossRef]
- Yu, W.; Du, N.; Gu, Y.; Yan, J.; Hou, W. Specific Ion Effects on the Colloidal Stability of Layered Double Hydroxide Single-layer Nanosheets. Langmuir 2020, 36, 6557–6568. [Google Scholar] [CrossRef] [PubMed]
- Katana, B.; Takács, D.; Csapó, E.; Szabó, T.; Jamnik, A.; Szilagyi, I. Ion Specific Effects on the Stability of Halloysite Nanotube Colloids-Inorganic Salts versus Ionic Liquids. J. Phys. Chem. B 2020, 124, 9757–9765. [Google Scholar] [CrossRef] [PubMed]
- Hounslow, M.J.; Ryall, R.L.; Marshall, V.R. A discretized population balance for nucleation, growth and aggregation. AIChE J. 1988, 34, 1821–1832. [Google Scholar] [CrossRef]
- Kumar, S.; Ramkrishna, D. On the solution of population balance equations by discretization—I. A fixed pivot technique. Chem. Eng. Sci. 1996, 51, 1311–1332. [Google Scholar] [CrossRef]
- Vanni, M. Approximate Population Balance Equations for Aggregation–Breakage Processes. J. Colloid Interface Sci. 2000, 221, 143–160. [Google Scholar] [CrossRef]
- Biggs, C.A.; Lant, P.A. Modelling activated sludge flocculation using population balances. Powder Technol. 2002, 124, 201–211. [Google Scholar] [CrossRef]
- Runkana, V.; Somasundaran, P.; Kapur, P.C. Mathematical modelling of polymer-induced flocculation by charge neutralization. J. Colloid Interface Sci. 2004, 270, 347–358. [Google Scholar] [CrossRef]
- Atmuri, A.K.; Henson, M.A.; Bhatia, S.R. A population balance equation model to predict regimes of controlled nanoparticle aggregation. Colloids Surf. A Physicochem. Eng. Asp. 2013, 436, 325–332. [Google Scholar] [CrossRef]
- Liang, L.; Wang, Y.; Pan, Z. Prediction of sedimentation behavior of submicron-sized particles of praseodymium-doped zirconium silicate in aqueous suspension by population balance model. Particuology 2016, 25, 83–92. [Google Scholar] [CrossRef]
- Huang, G.; Pan, Z.; Wang, Y. Dispersion of praseodymium-doped zirconium silicate pigment in aqueous suspension by modified hydroxyl copolymer. Chem Eng. Res. Des. 2020, 154, 86–100. [Google Scholar] [CrossRef]
- Arias, L.S.; Pessan, J.P.; Vieira, A.P.M.; Lima, T.M.T.; Delbem, A.C.B.; Monteiro, D.R. Iron Oxide Nanoparticles for Biomedical Applications: A Perspective on Synthesis, Drugs, Antimicrobial Activity, and Toxicity. Antibiotics 2018, 7, 46. [Google Scholar] [CrossRef] [Green Version]
- Guardia, P.; Labarta, A.; Batlle, X. Tuning the Size, the Shape, and the Magnetic Properties of Iron Oxide Nanoparticles. J. Phys. Chem. C. 2011, 115, 390–396. [Google Scholar] [CrossRef]
- Hemery, G.; Keyes, A.C., Jr.; Garaio, E.; Rodrigo, I.; Garcia, J.A.; Plazaola, F.; Garanger, E.; Sandre, O. Tuning Sizes, Morphologies, and Magnetic Properties of Monocore Versus Multicore Iron Oxide Nanoparticles through the Controlled Addition of Water in the Polyol Synthesis. Inorg. Chem. 2017, 56, 8232–8243. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fievet, F.; Lagier, J.P.; Blin, B.; Beaudoin, B.; Figlarz, M. Homogeneous and heterogeneous nucleations in the polyol process for the preparation of micron and submicron size metal particles. Solid State Ionics 1989, 32–33, 198–205. [Google Scholar] [CrossRef]
- Hugounenq, P.; Levy, M.; Alloyeau, D.; Lartigue, L.; Dubois, E.; Cabuil, V.; Ricolleau, C.; Roux, S.; Wilhelm, C.; Gazeau, F.; et al. Iron Oxide Monocrystalline Nanoflowers for Highly Efficient Magnetic Hyperthermia. J. Phys. Chem. C 2012, 116, 15702–15712. [Google Scholar] [CrossRef]
- Hemery, G.; Genevois, C.; Couillaud, F.; Lacomme, S.; Gontier, E.; Ibarboure, E.; Lecommandoux, S.; Garanger, E.; Sandre, O. Monocore vs multicore magnetic iron oxide nanoparticles: Uptake by glioblastoma cells and efficiency for magnetic hyperthermia. Mol. Syst. Des. Eng. 2017, 2, 629–639. [Google Scholar] [CrossRef] [Green Version]
- Von Smoluchowski, M. Versucheiner mathematischen theorie der koagulations kinetic kolloider losungen. Z. Phys. Chem. 1917, 92, 129–168. [Google Scholar]
- Jeldres, R.I.; Fawell, P.D.; Florio, B.J. Population balance modelling to describe the particle aggregation process: A review. Powder Technol. 2018, 326, 190–207. [Google Scholar] [CrossRef]
- Somasundaran, P.; Runkana, V. Modeling flocculation of colloidal mineral suspensions using population balances. Int. J. Miner. Process. 2003, 72, 33–55. [Google Scholar] [CrossRef]
- De Vicente, J.; Delgado, A.V.; Plaza, R.C.; Duran, J.D.G.; Gonzalez-Caballero, F. Stability of cobalt ferrite colloidal particles: Effect of pH and applied magnetic fields. Langmuir 2000, 16, 7954–7961. [Google Scholar] [CrossRef]
- Hindmarsh, A.C. ODEPACK: “Ordinary Differential Equation Solver Library”. Astrophysics Source Code Library 2019. Available online: https://ui.adsabs.harvard.edu/#abs/2019ascl.soft05021H/abstract (accessed on 6 November 2021).
- Piella, J.; Bastus, N.G.; Puntes, V. Size-Dependent Protein–Nanoparticle Interactions in Citrate-Stabilized Gold Nanoparticles: The Emergence of the Protein Corona. Bioconjug. Chem. 2017, 28, 88–97. [Google Scholar] [CrossRef]
- Gelbard, F.; Seinfeld, J.H. Numerical solution of the dynamic equation for particulate systems. J. Comput. Phys. 1978, 28, 357–375. [Google Scholar] [CrossRef]
- Ernst, M.H.; Ziff, R.M.; Hendriks, E.M. Coagulation processes with a phase transition. J. Colloid Interface Sci. 1984, 97, 266–277. [Google Scholar] [CrossRef]
- Liu, A.; Rigopoulos, S. A conservative method for numerical solution of the population balance equation, and application to soot formation. Combust. Flame 2019, 2015, 506–521. [Google Scholar] [CrossRef]
- Barbero, F.; Russo, L.; Vitali, M.; Piella, J.; Salvo, I.; Borrajo, M.L.; Busquets-Fité, M.; Grandori, R.; Bastús, N.G.; Casals, E.; et al. Formation of the Protein Corona: The Interface between Nanoparticles and the Immune System. Semin. Immunol. 2017, 34, 52–60. [Google Scholar] [CrossRef]
- Lima, T.; Bernfur, K.; Vilanova, M.; Cedervall, T. Understanding the Lipid and Protein Corona Formation on Different Sized Polymeric Nanoparticles. Sci. Rep. 2020, 10, 1129. [Google Scholar] [CrossRef]
- Zhanov, V.P. Nanoparticles without and with protein corona: Van der Waals and hydration interaction. J. Biol. Phys. 2019, 45, 307–316. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Faure, B. Particle Interactions at the Nanoscale. Ph.D. Thesis, Stockholm University, Stockholm, Sweden, 2012. [Google Scholar]
- Antonopoulou, E.; Rohmann-Shaw, C.F.; Sykes, T.C.; Cayre, O.J.; Hunter, T.N.; Jimack, P.K. Numerical and experimental analysis of the sedimentation of spherical colloidal suspensions under centrifugal force. Phys. Fluids 2018, 30, 030702. [Google Scholar] [CrossRef]
- Galli, M.; Sáringer, S.; Szilágyi, I.; Trefalt, G. A Simple Method to Determine Critical Coagulation Concentration from Electrophoretic Mobility. Colloids Interfaces 2020, 4, 20. [Google Scholar] [CrossRef]
- Li, W.; Liu, D.; Wu, J.; Kim, C.; Fortner, J.D. Aqueous Aggregation and Surface Deposition Processes of Engineered Superparamagnetic Iron Oxide Nanoparticles for Environmental Applications. Environ. Sci. Technol. 2014, 48, 11892–11900. [Google Scholar] [CrossRef] [PubMed]
- Ezzaier, H.; Marins, J.A.; Claudet, C.; Hemery, G.; Sandre, O.; Kuzhir, P. Kinetics of Aggregation and Magnetic Separation of Multicore Iron Oxide Nanoparticles: Effect of the Grafted Layer Thickness. Nanomaterials 2018, 8, 623. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Parameter | Value |
|---|---|
| Μ 1 | 0.6864 cP |
| T 2 | 310 K |
| zc 3 | 1 |
| εΓ 4 | 78.5 |
| Φ 5 | −0.027 V |
| M 6 | 0.7346 A/m |
| A 7 | 3.1 × 10−20 J |
| PBS 8 | KCl 2.68 mM KH2PO4 1.47 mM NaCl 136.89 mM K2HPO4 8.10 mM |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Neofytou, P.; Theodosiou, M.; Krokidis, M.G.; Efthimiadou, E.K. Simulation of Colloidal Stability and Aggregation Tendency of Magnetic Nanoflowers in Biofluids. Modelling 2022, 3, 14-26. https://doi.org/10.3390/modelling3010002
Neofytou P, Theodosiou M, Krokidis MG, Efthimiadou EK. Simulation of Colloidal Stability and Aggregation Tendency of Magnetic Nanoflowers in Biofluids. Modelling. 2022; 3(1):14-26. https://doi.org/10.3390/modelling3010002
Chicago/Turabian StyleNeofytou, Panagiotis, Maria Theodosiou, Marios G. Krokidis, and Eleni K. Efthimiadou. 2022. "Simulation of Colloidal Stability and Aggregation Tendency of Magnetic Nanoflowers in Biofluids" Modelling 3, no. 1: 14-26. https://doi.org/10.3390/modelling3010002
APA StyleNeofytou, P., Theodosiou, M., Krokidis, M. G., & Efthimiadou, E. K. (2022). Simulation of Colloidal Stability and Aggregation Tendency of Magnetic Nanoflowers in Biofluids. Modelling, 3(1), 14-26. https://doi.org/10.3390/modelling3010002








