What Is New in Prophylaxis and Treatment of COVID-19 in Renal Transplant Patients? A Report from an ESOT Meeting on the Topic
Abstract
:1. Introduction
- (a)
- Vaccination;
- (b)
- Monoclonal antibodies, examining pre-exposure prophylaxis with tixagevimab and cilgavimab (Eurisheld) and other monoclonal and polyclonal antibody products;
- (c)
- Small antiviral proteins, such as Nirmatrelvir/ritonavir, Molnupivar and Remdesivir (RMD) with particular concern toward their interaction with immunosuppressive agents.
2. Vaccination
- (a)
- The immune responses to COVID-19 mRNA vaccines include both B and T cells;
- (b)
- The immune responses in SOT recipients are inferior to those obtained in both healthy controls and SOT candidates;
- (c)
- Risk factors for non-responses include short duration from TX, treatment for acute rejection and use of Mycophenolate;
- (d)
- Natural infection, and third and fourth doses improve the immune response;
- (e)
- A third dose reduces the risk of severe COVID-19;
- (f)
- A fourth dose may provide protection against the omicron variant;
- (g)
- Vaccination of all SOT candidates and SOT recipients is a priority.
3. Monoclonal Antibodies
4. Direct-Acting Small-Molecule SARS-CoV-2 Antivirals
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fishman, J.A. The Immunocompromised Transplant Recipient and SARS-CoV-2 Infection. J. Am. Soc. Nephrol. 2020, 31, 1147–1149. [Google Scholar] [CrossRef]
- Fishman, J.A.; Roberts, M.B.; Zhang, E.W.; Kumar, D.; Hirsch, H.H.; Maggiore, U. Case 29-2020: A 66-Year-Old Man with Fever and Shortness of Breath after Liver Transplantation. N. Engl. J. Med. 2020, 383, 1168–1180. [Google Scholar] [CrossRef]
- Charmetant, X.; Thanaut, O.; Grossi, P.A.; Cillo, U.; Aguado, J.M.; Cervera, C.; Potena, L. Position Statement on Measures to Reduce the Risk of COVID-19 in Solid Organ Transplant Recipients by Use of Vaccination, Immune Testing, and Public Health Policies; European Society for Organ Transplantation: Padova, Italy, 2022. [Google Scholar]
- National Institutes of Health (NIH): COVID-19 Treatment Guidelines. Special Considerations in Solid Organ Transplant, Hematopoietic Stem Cell Transplant, and Cellular Therapy Candidates, Donors and Recipients. 2022. Available online: https://www.covid19treatmentguidelines.nih.gov/special-populations/transplant/ (accessed on 1 September 2022).
- National Health Service (NHS) Blood and Transplant Organ and Tissue Donation and Transplantation (OTDT) and British Toracic Society (BTS): Joint SARSCoV-2 Vaccination in Adult Solid Organ and Islet Transplant Wait-Listed Patients and Adult Living Donor Transplant Recipients. 2021. Available online: https://www.google.com.hk/url?sa=t&rct=j&q=&esrc=s&source=web&cd=&cad=rja&uact=8&ved=2ahUKEwiK48n0t477AhUn-3MBHQLZCiMQFnoECBQQAQ&url=https%3A%2F%2Fbts.org.uk%2Fwp-content%2Fuploads%2F2021%2F01%2FDAT3911.pdf&usg=AOvVaw1pMQd7fSR1RCsJsECd9nhr (accessed on 1 September 2022).
- The Transplantation Society (TTS): Guidance on Coronavirus Disease 2019 (COVID-19) for Transplant Clinicians (2020). Available online: https://tts.org/tid-about/tid-officers-and-council?id=749 (accessed on 1 September 2022).
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. C4591001 Clinical Trial Group. Safety and Efficacy of the BNT162b2 mRNA COVID-19 Vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Baden, L.R.; El Sahly, H.M.; Essink, B.; Kotloff, K.; Frey, S.; Novak, R.; Diemert, D.; Spector, S.A.; Rouphael, N.; Creech, C.B.; et al. Efficacy and Safety of the mRNA-1273 SARS-CoV-2 Vaccine. N. Engl. J. Med. 2021, 384, 403–416. [Google Scholar] [CrossRef]
- Hamm, S.R.; Møller, D.L.; Pérez-Alós, L.; Hansen, C.B.; Pries-Heje, M.M.; Heftdal, L.D.; Hasselbalch, R.B.; Fogh, K.; Madsen, J.R.; Almagro Armenteros, J.J.; et al. Decline in Antibody Concentration 6 Months After Two Doses of SARS-CoV-2 BNT162b2 Vaccine in Solid Organ Transplant Recipients and Healthy Controls. Front. Immunol. 2022, 13, 832501. [Google Scholar] [CrossRef]
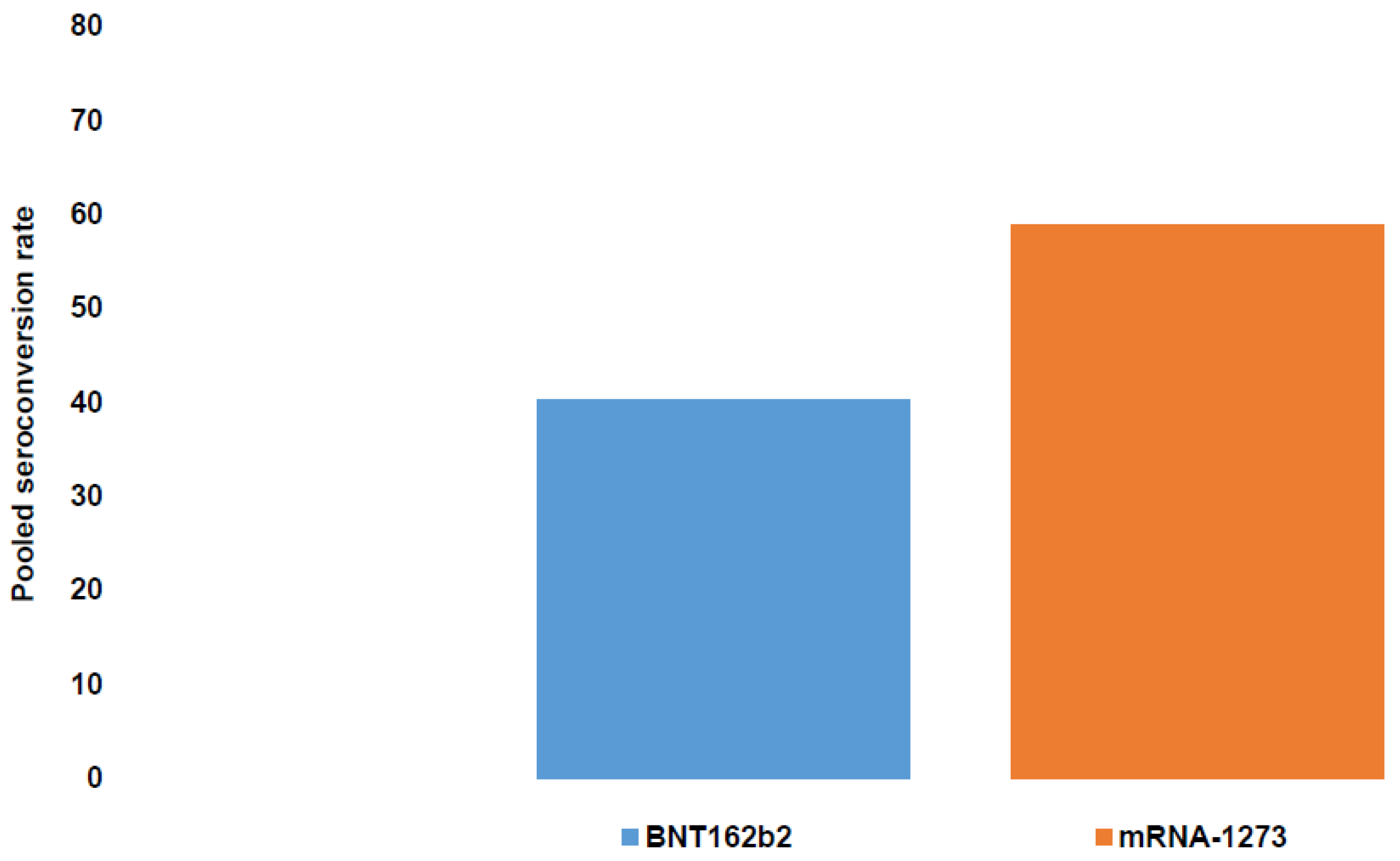
- Verleye, A.; Wijtvliet, V.; Abrams, S.; Hellemans, R.; Bougrea, R.; Massart, A.; Pipeleers, L.; Wissing, K.M.; Ariën, K.K.; De Winter, B.Y.; et al. Seroconversion rate after primary vaccination with two doses of BNT162b2 versus mRNA-1273 in solid organ transplant recipients: A systematic review and meta-analysis. Nephrol. Dial. Transplant. 2022, 37, 1566–1575. [Google Scholar] [CrossRef]
- Liefeldt, L.; Glander, P.; Klotsche, J.; Straub-Hohenbleicher, H.; Budde, K.; Eberspächer, B.; Friedersdorff, F.; Halleck, F.; Hambach, P.; Hofmann, J.; et al. Predictors of Serological Response to SARS-CoV-2 Vaccination in Kidney Transplant Patients: Baseline Characteristics, Immunosuppression, and the Role of IMPDH Monitoring. J. Clin. Med. 2022, 11, 1697. [Google Scholar] [CrossRef]
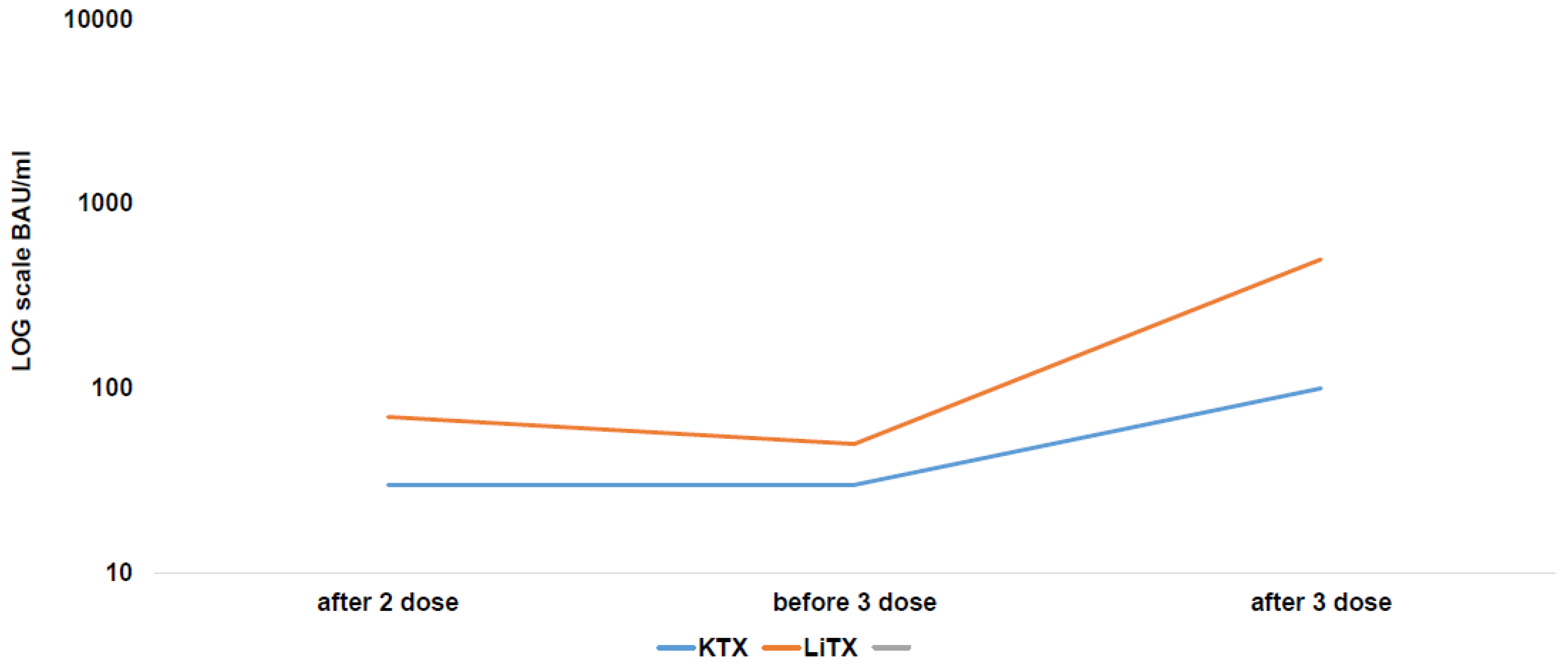
- Balsby, D.; Nilsson, A.C.; Möller, S.; Lindvig, S.O.; Davidsen, J.R.; Abazi, R.; Poulsen, M.K.; Holden, I.K.; Justesen, U.S.; Bistrup, C.; et al. Determinants of Antibody Response to a Third SARS-CoV-2 mRNA Vaccine Dose in Solid Organ Transplant Recipients: Results from the Prospective Cohort Study COVAC-Tx. Vaccines 2022, 10, 565. [Google Scholar] [CrossRef]
- Hod, T.; Ben-David, A.; Olmer, L.; Scott, N.; Ghinea, R.; Mor, E.; Levy, I.; Indenbaum, V.; Lustig, Y.; Grossman, E.; et al. BNT162b2 Third Booster Dose Significantly Increases the Humoral Response Assessed by Both RBD IgG and Neutralizing Antibodies in Renal Transplant Recipients. Transpl. Int. 2022, 35, 10239. [Google Scholar] [CrossRef]
- Dimeglio, C.; Herin, F.; Martin-Blondel, G.; Miedougé, M.; Izopet, J. Antibody titers and protection against a SARS-CoV-2 infection. J. Infect. 2022, 84, 257–259. [Google Scholar] [CrossRef]
- Dimeglio, C.; Migueres, M.; Bouzid, N.; Chapuy-Regaud, S.; Gernigon, C.; Da-Silva, I.; Porcheron, M.; Martin-Blondel, G.; Herin, F.; Izopet, J. Antibody Titers and Protection against Omicron (BA.1 and BA.2) SARS-CoV-2 Infection. Vaccines 2022, 10, 1548. [Google Scholar] [CrossRef]
- Suntronwong, N.; Assawakosri, S.; Kanokudom, S.; Yorsaeng, R.; Auphimai, C.; Thongmee, T.; Vichaiwattana, P.; Duangchinda, T.; Chantima, W.; Pakchotanon, P.; et al. Strong Correlations between the Binding Antibodies against Wild-Type and Neutralizing Antibodies against Omicron BA.1 and BA.2 Variants of SARS-CoV-2 in Individuals Following Booster (Third-Dose) Vaccination. Diagnostics 2022, 12, 1781. [Google Scholar] [CrossRef]
- Manothummetha, K.; Chuleerarux, N.; Sanguankeo, A.; Kates, O.S.; Hirankarn, N.; Thongkam, A.; Dioverti-Prono, M.V.; Torvorapanit, P.; Langsiri, N.; Worasilchai, N.; et al. Immunogenicity and Risk Factors Associated with Poor Humoral Immune Response of SARS-CoV-2 Vaccines in Recipients of Solid Organ Transplant: A Systematic Review and Meta-Analysis. JAMA Netw. Open. 2022, 5, e226822. [Google Scholar] [CrossRef]
- Benotmane, I.; Gautier-Vargas, G.; Cognard, N.; Olagne, J.; Heibel, F.; Braun-Parvez, L.; Martzloff, J.; Perrin, P.; Moulin, B.; Fafi-Kremer, S.; et al. Low immunization rates among kidney transplant recipients who received 2 doses of the mRNA-1273 SARS-CoV-2 vaccine. Kidney Int. 2021, 99, 1498–1500. [Google Scholar] [CrossRef]
- Meyer, M.; Constancias, F.; Worth, C.; Meyer, A.; Muller, M.; Boussuge, A.; Kaltenbach, G.; Schmitt, E.; Chayer, S.; Velay, A.; et al. Humoral immune response after COVID-19 infection or BNT162b2 vaccine among older adults: Evolution over time and protective thresholds. Geroscience 2022, 44, 1229–1240. [Google Scholar] [CrossRef]
- Yang, H.S.; Costa, V.; Racine-Brzostek, S.E.; Acker, K.P.; Yee, J.; Chen, Z.; Karbaschi, M.; Zuk, R.; Rand, S.; Sukhu, A.; et al. Association of Age With SARS-CoV-2 Antibody Response. JAMA Netw. Open. 2021, 4, e214302. [Google Scholar] [CrossRef]
- Mazzoni, A.; Vanni, A.; Spinicci, M.; Lamacchia, G.; Kiros, S.T.; Rocca, A.; Capone, M.; Di Lauria, N.; Salvati, L.; Carnasciali, A.; et al. SARS-CoV-2 infection and vaccination trigger long-lived B and CD4+ T lymphocytes with implications for booster strategies. J. Clin. Investig. 2022, 132, e157990. [Google Scholar] [CrossRef]
- Dan, J.M.; Mateus, J.; Kato, Y.; Hastie, K.M.; Yu, E.D.; Faliti, C.E.; Grifoni, A.; Ramirez, S.I.; Haupt, S.; Frazier, A.; et al. Immunological memory to SARS-CoV-2 assessed for up to 8 months after infection. Science 2021, 371, eabf4063. [Google Scholar] [CrossRef]
- Mazzoni, A.; Di Lauria, N.; Maggi, L.; Salvati, L.; Vanni, A.; Capone, M.; Lamacchia, G.; Mantengoli, E.; Spinicci, M.; Zammarchi, L.; et al. First-dose mRNA vaccination is sufficient to reactivate immunological memory to SARS-CoV-2 in subjects who have recovered from COVID-19. J. Clin. Investig. 2021, 131, e149150. [Google Scholar] [CrossRef]
- Mazzoni, A.; Maggi, L.; Capone, M.; Spinicci, M.; Salvati, L.; Colao, M.G.; Vanni, A.; Kiros, S.T.; Mencarini, J.; Zammarchi, L.; et al. Cell-mediated and humoral adaptive immune responses to SARS-CoV-2 are lower in asymptomatic than symptomatic COVID-19 patients. Eur. J. Immunol. 2020, 50, 2013–2024. [Google Scholar] [CrossRef]
- Mazzoni, A.; Maggi, L.; Capone, M.; Vanni, A.; Spinicci, M.; Salvati, L.; Tekle Kiros, S.; Semeraro, R.; Pengue, L.; Colao, M.G.; et al. Heterogeneous magnitude of immunological memory to SARS-CoV-2 in recovered individuals. Clin. Transl. Immunol. 2021, 10, e1281. [Google Scholar] [CrossRef] [PubMed]
- Buehrle, D.J.; Sutton, R.R.; McCann, E.L.; Lucas, A.E. A Review of Treatment and Prevention of Coronavirus Disease 2019 among Solid Organ Transplant Recipients. Viruses 2021, 13, 1706. [Google Scholar] [CrossRef] [PubMed]
- Dęborska-Materkowska, D.; Kamińska, D. The Immunology of SARS-CoV-2 Infection and Vaccines in Solid Organ Transplant Recipients. Viruses 2021, 13, 1879. [Google Scholar] [CrossRef]
- Rozen-Zvi, B.; Yahav, D.; Agur, T.; Zingerman, B.; Ben-Zvi, H.; Atamna, A.; Tau, N.; Mashraki, T.; Nesher, E.; Rahamimov, R. Antibody response to SARS-CoV-2 mRNA vaccine among kidney transplant recipients: A prospective cohort study. Clin. Microbiol. Infect. 2021, 27, 1173.e1–1173.e4. [Google Scholar] [CrossRef] [PubMed]
- Marion, O.; Del Bello, A.; Abravanel, F.; Couat, C.; Faguer, S.; Esposito, L.; Hebral, A.L.; Izopet, J.; Kamar, N. Safety and Immunogenicity of Anti-SARS-CoV-2 Messenger RNA Vaccines in Recipients of Solid Organ Transplants. Ann. Int. Med. 2021, 174, 1336–1338. [Google Scholar] [CrossRef] [PubMed]
- Boyarsky, B.J.; Werbel, W.A.; Avery, R.K.; Tobian, A.A.R.; Massie, A.B.; Segev, D.L.; Garonzik-Wang, J.M. Antibody Response to 2-Dose SARS-CoV-2 mRNA Vaccine Series in Solid Organ Transplant Recipients. JAMA 2021, 325, 2204–2206. [Google Scholar] [CrossRef]
- Hall, V.G.; Ferreira, V.H.; Ku, T.; Ierullo, M.; Majchrzak-Kita, B.; Chaparro, C.; Selzner, N.; Schiff, J.; McDonald, M.; Tomlinson, G.; et al. Randomized Trial of a Third Dose of mRNA-1273 Vaccine in Transplant Recipients. N. Engl. J. Med. 2021, 385, 1244–1246. [Google Scholar] [CrossRef]
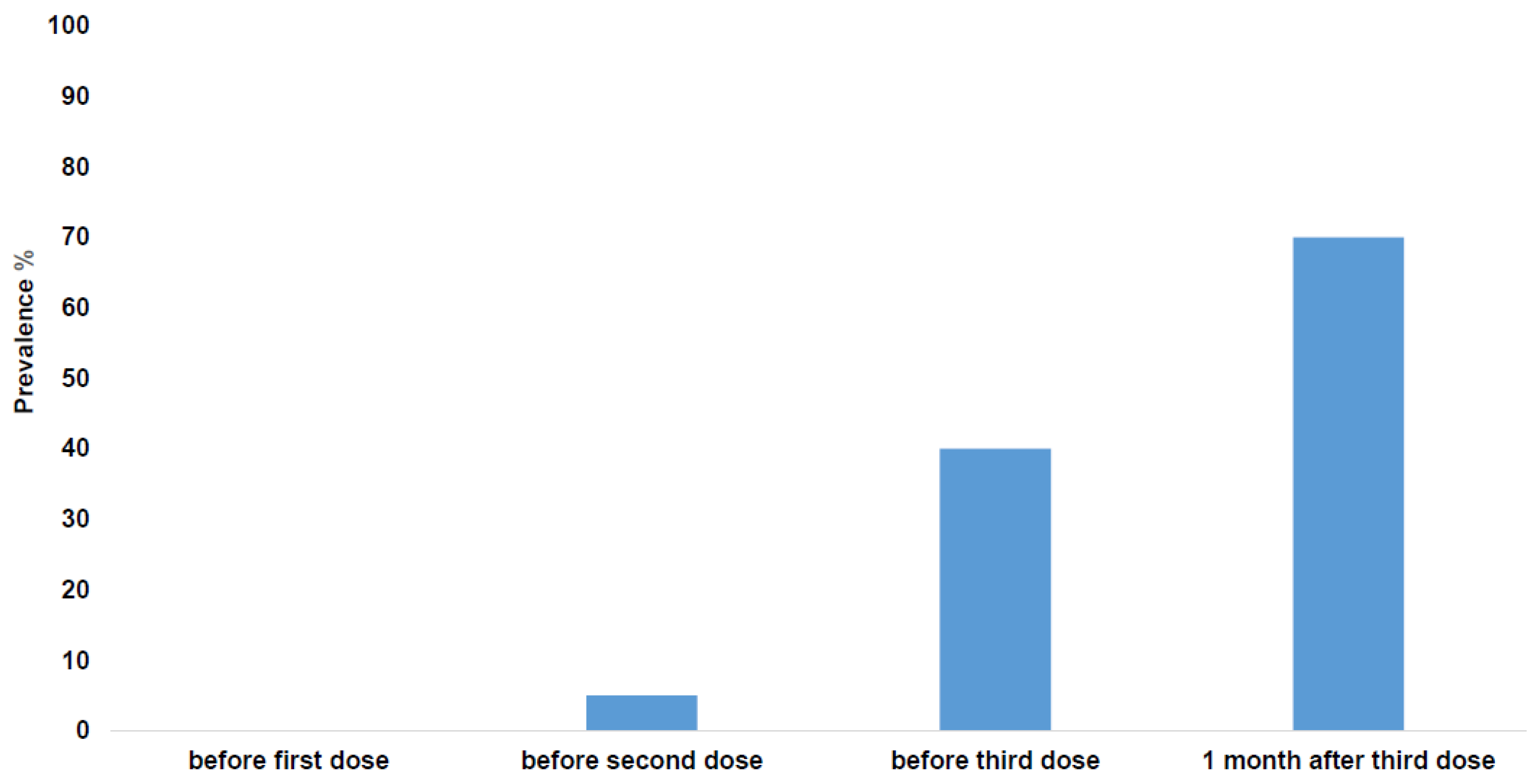
- Kamar, N.; Abravanel, F.; Marion, O.; Couat, C.; Izopet, J.; Del Bello, A. Three Doses of an mRNA COVID-19 Vaccine in Solid-Organ Transplant Recipients. N. Engl. J. Med. 2021, 385, 661–662. [Google Scholar] [CrossRef]
- Kamar, N.; Abravanel, F.; Marion, O.; Esposito, L.; Hebral, A.L.; Médrano, C.; Guitard, J.; Lavayssière, L.; Cointault, O.; Nogier, M.B.; et al. Anti-SARS-CoV-2 spike protein and neutralizing antibodies at 1 and 3 months after three doses of SARS-CoV-2 vaccine in a large cohort of solid organ transplant patients. Am. J. Transplant. 2022, 22, 1467–1474. [Google Scholar] [CrossRef]
- Kamar, N.; Abravanel, F.; Marion, O.; Romieu-Mourez, R.; Couat, C.; Del Bello, A.; Izopet, J. Assessment of 4 Doses of SARS-CoV-2 Messenger RNA-Based Vaccine in Recipients of a Solid Organ Transplant. JAMA Netw. Open. 2021, 4, e2136030. [Google Scholar] [CrossRef]
- Cristelli, M.P.; Nakamura, M.R.; Viana, L.A.; Tedesco-Silva, H.; Medina-Pestana, J. The Fourth Dose of CoronaVac Vaccine Results in a Small Increase of Seroconversion and Antibody Values Among Kidney Transplant Recipients. Transplantation, 2022; online ahead of print. [Google Scholar] [CrossRef]
- Mitchell, J.; Kim, J.; Alejo, J.L.; Chiang, T.P.; Karaba, A.H.; Blankson, J.N.; Aytenfisu, T.Y.; Chang, A.; Abedon, A.T.; Avery, R.K.; et al. Antibody Response to a Fourth Dose of SARS-CoV-2 Vaccine in Solid Organ Transplant Recipients: An Update. Transplantation 2022, 106, e338–e340. [Google Scholar] [CrossRef]
- Caillard, S.; Thaunat, O.; Benotmane, I.; Masset, C.; Blancho, G. Antibody Response to a Fourth Messenger RNA COVID-19 Vaccine Dose in Kidney Transplant Recipients: A Case Series. Ann. Int. Med. 2022, 175, 455–456. [Google Scholar] [CrossRef] [PubMed]
- Benotmane, I.; Bruel, T.; Planas, D.; Fafi-Kremer, S.; Schwartz, O.; Caillard, S. A fourth dose of the mRNA-1273 SARS-CoV-2 vaccine improves serum neutralization against the Delta variant in kidney transplant recipients. Kidney Int. 2022, 101, 1073–1076. [Google Scholar] [CrossRef] [PubMed]
- Masset, C.; Benotmane, I.; Dantal, J.; Garandeau, C.; Gauthier-Vargas, G.; Cantarovich, D.; Meurette, A.; Giral, M.; Caillard, S.; Blancho, G. A fourth SARS-CoV-2 mRNA vaccine in strictly seronegative kidney transplant recipients. Kidney Int. 2022, 10, 825–826. [Google Scholar] [CrossRef] [PubMed]
- Iketani, S.; Liu, L.; Guo, Y.; Liu, L.; Chan, J.F.; Huang, Y.; Wang, M.; Luo, Y.; Yu, J.; Chu, H.; et al. Antibody evasion properties of SARS-CoV-2 Omicron sublineages. Nature 2022, 604, 553–556. [Google Scholar] [CrossRef] [PubMed]
- Shen, X. Boosting immunity to Omicron. Nat. Med. 2022, 28, 445–446. [Google Scholar] [CrossRef]
- Pérez-Then, E.; Lucas, C.; Monteiro, V.S.; Miric, M.; Brache, V.; Cochon, L.; Vogels, C.B.F.; Malik, A.A.; De la Cruz, E.; Jorge, A.; et al. Neutralizing antibodies against the SARS-CoV-2 Delta and Omicron variants following heterologous CoronaVac plus BNT162b2 booster vaccination. Nat. Med. 2022, 28, 481–485. [Google Scholar] [CrossRef]
- Cheng, S.M.S.; Mok, C.K.P.; Leung, Y.W.Y.; Ng, S.S.; Chan, K.C.K.; Ko, F.W.; Chen, C.; Yiu, K.; Lam, B.H.S.; Lau, E.H.Y.; et al. Neutralizing antibodies against the SARS-CoV-2 Omicron variant BA.1 following homologous and heterologous CoronaVac or BNT162b2 vaccination. Nat. Med. 2022, 28, 486–489. [Google Scholar] [CrossRef]
- Gruell, H.; Vanshylla, K.; Tober-Lau, P.; Hillus, D.; Schommers, P.; Lehmann, C.; Kurth, F.; Sander, L.E.; Klein, F. mRNA booster immunization elicits potent neutralizing serum activity against the SARS-CoV-2 Omicron variant. Nat. Med. 2022, 28, 477–480. [Google Scholar] [CrossRef]
- VanBlargan, L.A.; Errico, J.M.; Halfmann, P.J.; Zost, S.J.; Crowe, J.E., Jr.; Purcell, L.A.; Kawaoka, Y.; Corti, D.; Fremont, D.H.; Diamond, M.S. An infectious SARS-CoV-2 B.1.1.529 Omicron virus escapes neutralization by therapeutic monoclonal antibodies. Nat. Med. 2022, 28, 490–495. [Google Scholar] [CrossRef] [PubMed]
- Bar-On, Y.M.; Goldberg, Y.; Mandel, M.; Bodenheimer, O.; Amir, O.; Freedman, L.; Alroy-Preis, S.; Ash, N.; Huppert, A.; Milo, R. Protection by a Fourth Dose of BNT162b2 against Omicron in Israel. N. Engl. J. Med. 2022, 386, 1712–1720. [Google Scholar] [CrossRef] [PubMed]
- Wohl, A.; Leibowitz, R. Protection by a Fourth Dose of BNT162b2 against Omicron in Israel. N. Engl. J. Med. 2022, 386, 2441. [Google Scholar]
- Zhang, J.; Chen, N.; Zhao, D.; Zhang, J.; Hu, Z.; Tao, Z. Clinical Characteristics of COVID-19 Patients Infected by the Omicron Variant of SARS-CoV-2. Front. Med. 2022, 9, 912367. [Google Scholar] [CrossRef] [PubMed]
- Al Jurdi, A.; Gassen, R.B.; Borges, T.J.; Lape, I.T.; Morena, L.; Efe, O.; Solhjou, Z.; El Fekih, R.; Deban, C.; Bohan, B.; et al. Suboptimal antibody response against SARS-CoV-2 Omicron variant after third dose of mRNA vaccine in kidney transplant recipients. Kidney Int. 2022, 101, 1282–1286. [Google Scholar] [CrossRef]
- Dejnirattisai, W.; Shaw, R.H.; Supasa, P.; Liu, C.; Stuart, A.S.; Pollard, A.J.; Liu, X.; Lambe, T.; Crook, D.; Stuart, D.I.; et al. Reduced neutralisation of SARS-CoV-2 omicron B.1.1.529 variant by post-immunisation serum. Lancet 2022, 399, 234–236. [Google Scholar] [CrossRef]
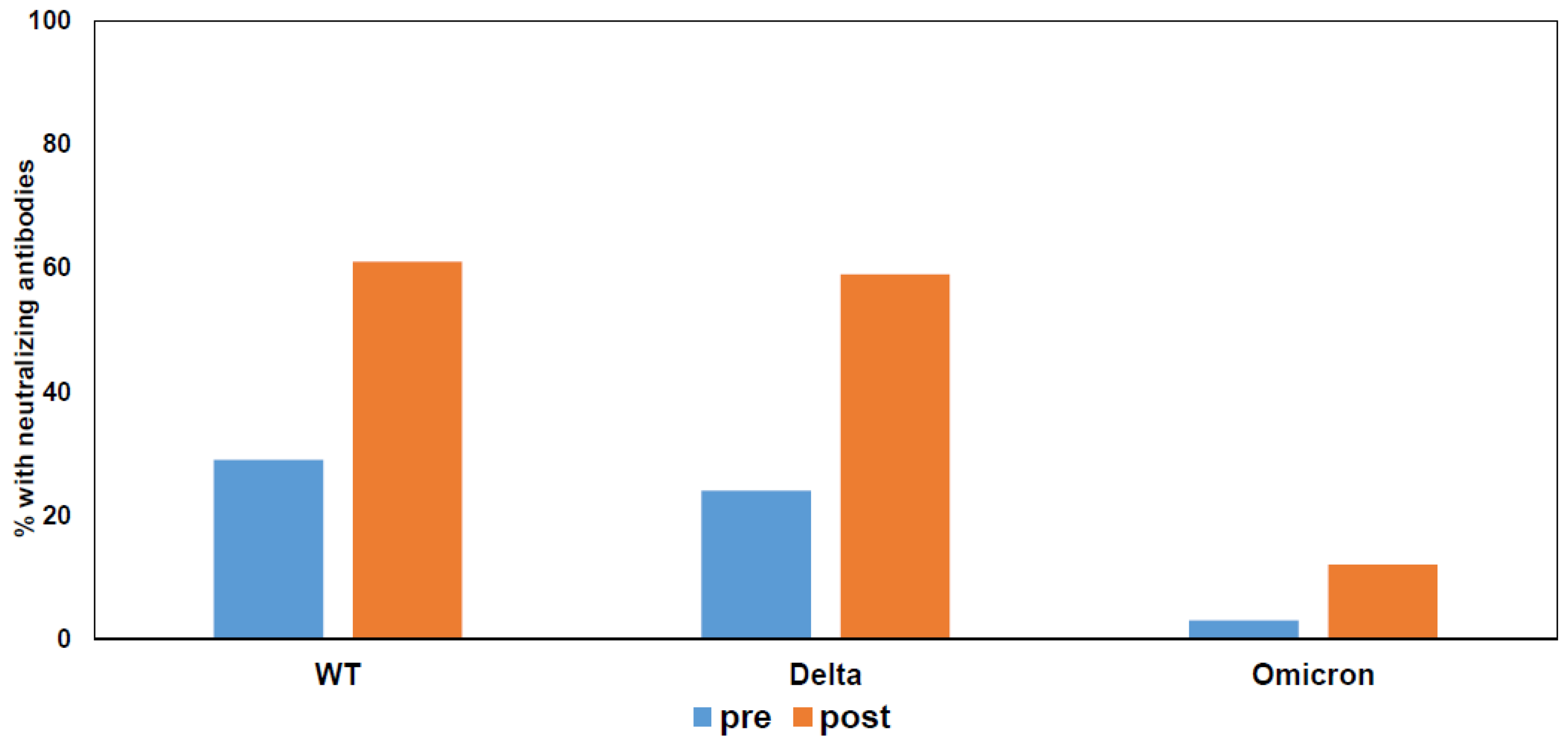
- Karaba, A.H.; Zhu, X.; Liang, T.; Wang, K.H.; Rittenhouse, A.G.; Akinde, O.; Eby, Y.; Ruff, J.E.; Blankson, J.N.; Abedon, A.T.; et al. A third dose of SARS-CoV-2 vaccine increases neutralizing antibodies against variants of concern in solid organ transplant recipients. Am. J. Transplant. 2022, 22, 1253–1260. [Google Scholar] [CrossRef]
- Karaba, A.H.; Johnston, T.S.; Aytenfisu, T.Y.; Akinde, O.; Eby, Y.; Ruff, J.E.; Abedon, A.T.; Alejo, J.L.; Blankson, J.N.; Cox, A.L.; et al. A Fourth Dose of COVID-19 Vaccine Does Not Induce Neutralization of the Omicron Variant Among Solid Organ Transplant Recipients with Suboptimal Vaccine Response. Transplantation 2022, 106, 1440–1444. [Google Scholar] [CrossRef]
- Sakuraba, A.; Luna, A.; Micic, D. A Systematic Review and Meta-Analysis of Serologic Response following Coronavirus Disease 2019 (COVID-19) Vaccination in Solid Organ Transplant Recipients. Viruses 2022, 14, 1822. [Google Scholar] [CrossRef]
- Massa, F.; Cremoni, M.; Gérard, A.; Grabsi, H.; Rogier, L.; Blois, M.; Couzin, C.; Hassen, N.B.; Rouleau, M.; Barbosa, S.; et al. Safety and cross-variant immunogenicity of a three-dose COVID-19 mRNA vaccine regimen in kidney transplant recipients. EBioMedicine 2021, 73, 103679. [Google Scholar] [CrossRef]
- Masset, C.; Kerleau, C.; Garandeau, C.; Ville, S.; Cantarovich, D.; Hourmant, M.; Kervella, D.; Houzet, A.; Guillot-Gueguen, C.; Guihard, I.; et al. A third injection of the BNT162b2 mRNA COVID-19 vaccine in kidney transplant recipients improves the humoral immune response. Kidney Int. 2021, 100, 1132–1135. [Google Scholar] [CrossRef] [PubMed]
- Baum, A.; Fulton, B.O.; Wloga, E.; Copin, R.; Pascal, K.E.; Russo, V.; Giordano, S.; Lanza, K.; Negron, N.; Ni, M.; et al. Antibody cocktail to SARS-CoV-2 spike protein prevents rapid mutational escape seen with individual antibodies. Science 2020, 369, 1014–1018. [Google Scholar] [CrossRef] [PubMed]
- Copin, R.; Baum, A.; Wloga, E.; Pascal, K.E.; Giordano, S.; Fulton, B.O.; Zhou, A.; Negron, N.; Lanza, K.; Chan, N.; et al. The monoclonal antibody combination REGEN-COV protects against SARS-CoV-2 mutational escape in preclinical and human studies. Cell 2021, 184, 3949–3961. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, M.P.; Forleo-Neto, E.; Musser, B.J.; Isa, F.; Chan, K.C.; Sarkar, N.; Bar, K.J.; Barnabas, R.V.; Barouch, D.H.; Cohen, M.S.; et al. COVID-19 Phase 3 Prevention Trial Team. Subcutaneous REGEN-COV Antibody Combination to Prevent COVID-19. N. Engl. J. Med. 2021, 385, 1184–1195. [Google Scholar] [CrossRef]
- Merison, T.; Goldman, A.; Bomze, D. Subcutaneous REGEN-COV Antibody Combination to Prevent COVID-19. N. Engl. J. Med. 2021, 385, e70. [Google Scholar]
- Dougan, M.; Nirula, A.; Azizad, M.; Mocherla, B.; Gottlieb, R.L.; Chen, P.; Hebert, C.; Perry, R.; Boscia, J.; Heller, B.; et al. Bamlanivimab plus Etesevimab in Mild or Moderate COVID-19. N. Engl. J. Med. 2021, 385, 1382–1392. [Google Scholar] [CrossRef]
- National French Health Authority. RONAPREVE (Casirivimab-Imdevimab) (Prophylaxie Pré-Exposition de L’infection à SARS-CoV-2). Available online: https://www.has-sante.fr/jcms/p_3281544/fr/ronapreve-casirivimab-imdevimab-prophylaxie-pre-expositionj-de-l-infectiona-sars-cov-2 (accessed on 6 August 2022).
- Dimeglio, C.; Del Bello, A.; Chapuy-Regaud, S.; Esposito, L.; Danet, C.; Couat, C.; Izopet, J.; Kamar, N. Casirivimab-imdevimab to Prevent SARS-CoV-2 Infections in Solid Organ Transplant Recipients. Transplantation 2022, 106, e275–e276. [Google Scholar] [CrossRef]
- Ducloux, D.; Courivaud, C. REGEN-Cov antibody combination to prevent COVID-19 infection in kidney transplant recipient without detectable antibody response to optimal vaccine scheme. Kidney Int. 2022, 101, 645–646. [Google Scholar] [CrossRef]
- Kamar, N.; Gouin, A.; Izopet, J. Omicron breakthrough infection in a kidney-transplant patient given pre-exposition casirivimab and imdevimab monoclonal antibodies. Transpl. Infect. Dis. 2022, 24, e13803. [Google Scholar] [CrossRef]
- Planas, D.; Saunders, N.; Maes, P.; Guivel-Benhassine, F.; Planchais, C.; Buchrieser, J.; Bolland, W.H.; Porrot, F.; Staropoli, I.; Lemoine, F.; et al. Considerable escape of SARS-CoV-2 Omicron to antibody neutralization. Nature 2022, 602, 671–675. [Google Scholar] [CrossRef]
- Taylor, P.C.; Adams, A.C.; Hufford, M.M.; de la Torre, I.; Winthrop, K.; Gottlieb, R.L. Neutralizing monoclonal antibodies for treatment of COVID-19. Nat. Rev. Immunol. 2021, 21, 382–393. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Wang, J.; Jian, F.; Xiao, T.; Song, W.; Yisimayi, A.; Huang, W.; Li, Q.; Wang, P.; An, R.; et al. Omicron escapes the majority of existing SARS-CoV-2 neutralizing antibodies. Nature 2022, 602, 657–663. [Google Scholar] [CrossRef] [PubMed]
- Cameroni, E.; Bowen, J.E.; Rosen, L.E.; Saliba, C.; Zepeda, S.K.; Culap, K.; Pinto, D.; VanBlargan, L.A.; De Marco, A.; di Iulio, J.; et al. Broadly neutralizing antibodies overcome SARS-CoV-2 Omicron antigenic shift. Nature 2022, 602, 664–670. [Google Scholar] [CrossRef]
- Loo, Y.M.; McTamney, P.M.; Arends, R.H.; Abram, M.E.; Aksyuk, A.A.; Diallo, S.; Flores, D.J.; Kelly, E.J.; Ren, K.; Roque, R.; et al. The SARS-CoV-2 monoclonal antibody combination, AZD7442, is protective in nonhuman primates and has an extended half-life in humans. Sci. Transl. Med. 2022, 14, eabl8124. [Google Scholar] [CrossRef] [PubMed]
- Zost, S.J.; Gilchuk, P.; Case, J.B.; Binshtein, E.; Chen, R.E.; Nkolola, J.P.; Schäfer, A.; Reidy, J.X.; Trivette, A.; Nargi, R.S.; et al. Potently neutralizing and protective human antibodies against SARS-CoV-2. Nature 2020, 584, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Zost, S.J.; Greaney, A.J.; Starr, T.N.; Dingens, A.S.; Chen, E.C.; Chen, R.E.; Case, J.B.; Sutton, R.E.; Gilchuk, P.; et al. Genetic and structural basis for SARS-CoV-2 variant neutralization by a two-antibody cocktail. Nat. Microbiol. 2021, 6, 1233–1244. [Google Scholar] [CrossRef] [PubMed]
- Levin, M.J.; Ustianowski, A.; De Wit, S.; Launay, O.; Avila, M.; Templeton, A.; Yuan, Y.; Seegobin, S.; Ellery, A.; Levinson, D.J.; et al. Intramuscular AZD7442 (Tixagevimab-Cilgavimab) for Prevention of COVID-19. N. Engl. J. Med. 2022, 386, 2188–2200. [Google Scholar] [CrossRef]
- Bertrand, D.; Laurent, C.; Lemée, V.; Lebourg, L.; Hanoy, M.; Le Roy, F.; Nezam, D.; Pruteanu, D.; Grange, S.; de Nattes, T.; et al. Efficacy of anti-SARS-CoV-2 monoclonal antibody prophylaxis and vaccination on the Omicron variant of COVID-19 in kidney transplant recipients. Kidney Int. 2022, 102, 440–442. [Google Scholar] [CrossRef]
- Benotmane, I.; Velay, A.; Gautier-Vargas, G.; Olagne, J.; Thaunat, O.; Fafi-Kremer, S.; Caillard, S. Pre-exposure prophylaxis with 300 mg Evusheld elicits limited neutralizing activity against the Omicron variant. Kidney Int. 2022, 102, 442–444. [Google Scholar] [CrossRef]
- US Food & Drug Administration. FDA Authorizes Revisions to Evusheld Dosing. Available online: https://www.fda.gov/drugs/drug-safety-and-availability/fda-authorizes-revisions-evusheld-dosing (accessed on 24 May 2002).
- Mazzotta, V.; Cozzi-Lepri, A.; Colavita, F.; Lanini, S.; Rosati, S.; Lalle, E.; Mastrorosa, I.; Cimaglia, C.; Vergori, A.; Bevilacqua, N.; et al. Emulation of a Target Trial from Observational Data to Compare Effectiveness of Casirivimab/Imdevimab and Bamlanivimab/Etesevimab for Early Treatment of Non-Hospitalized Patients With COVID-19. Front. Immunol. 2022, 13, 868020. [Google Scholar] [CrossRef]
- Gottlieb, R.L.; Nirula, A.; Chen, P.; Boscia, J.; Heller, B.; Morris, J.; Huhn, G.; Cardona, J.; Mocherla, B.; Stosor, V.; et al. Effect of Bamlanivimab as Monotherapy or in Combination With Etesevimab on Viral Load in Patients With Mild to Moderate COVID-19: A Randomized Clinical Trial. JAMA 2021, 325, 632–644. [Google Scholar] [CrossRef] [PubMed]
- Dougan, M.; Azizad, M.; Mocherla, B.; Gottlieb, R.L.; Chen, P.; Hebert, C.; Perry, R.; Boscia, J.; Heller, B.; Morris, J.; et al. A Randomized, Placebo-Controlled Clinical Trial of Bamlanivimab and Etesevimab Together in High-Risk Ambulatory Patients With COVID-19 and Validation of the Prognostic Value of Persistently High Viral Load. Clin. Infect. Dis. 2022, 75, e440–e449. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Gonzalez-Rojas, Y.; Juarez, E.; Crespo Casal, M.; Moya, J.; Falci, D.R.; Sarkis, E.; Solis, J.; Zheng, H.; Scott, N.; et al. Early Treatment for COVID-19 with SARS-CoV-2 Neutralizing Antibody Sotrovimab. N. Engl. J. Med. 2021, 385, 1941–1950. [Google Scholar] [CrossRef]
- Pinto, D.; Park, Y.J.; Beltramello, M.; Walls, A.C.; Tortorici, M.A.; Bianchi, S.; Jaconi, S.; Culap, K.; Zatta, F.; De Marco, A.; et al. Cross-neutralization of SARS-CoV-2 by a human monoclonal SARS-CoV antibody. Nature 2020, 583, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Vellas, C.; Trémeaux, P.; Del Bello, A.; Latour, J.; Jeanne, N.; Ranger, N.; Danet, C.; Martin-Blondel, G.; Delobel, P.; Kamar, N.; et al. Resistance mutations in SARS-CoV-2 omicron variant in patients treated with sotrovimab. Clin. Microbiol. Infect. 2022, 28, 1297–1299. [Google Scholar] [CrossRef]
- Rockett, R.; Basile, K.; Maddocks, S.; Fong, W.; Agius, J.E.; Johnson-Mackinnon, J.; Arnott, A.; Chandra, S.; Gall, M.; Draper, J.; et al. Resistance Mutations in SARS-CoV-2 Delta Variant after Sotrovimab Use. N. Engl. J. Med. 2022, 386, 1477–1479. [Google Scholar] [CrossRef]
- Gueguen, J.; Colosio, C.; Del Bello, A.; Scemla, A.; N’Guyen, Y.; Rouzaud, C.; Carvalho-Schneider, C.; Gautier Vargas, G.; Tremolières, P.; Eddine, A.J.; et al. Early Administration of Anti-SARS-CoV-2 Monoclonal Antibodies Prevents Severe COVID-19 in Kidney Transplant Patients. Kidney Int. Rep. 2022, 7, 1241–1247. [Google Scholar] [CrossRef]
- Dhand, A.; Lobo, S.A.; Wolfe, K.; Feola, N.; Lee, L.; Nog, R.; Chen, D.; Glicklich, D.; Diflo, T.; Nabors, C. Casirivimab-imdevimab for Treatment of COVID-19 in Solid Organ Transplant Recipients: An Early Experience. Transplantation 2021, 105, e68–e69. [Google Scholar] [CrossRef]
- Dhand, A.; Lobo, S.A.; Wolfe, K.; Feola, N.; Nabors, C. Bamlanivimab for treatment of COVID-19 in solid organ transplant recipients: Early single-center experience. Clin. Transplant. 2021, 35, e14245. [Google Scholar] [CrossRef]
- Kutzler, H.L.; Kuzaro, H.A.; Serrano, O.K.; Feingold, A.; Morgan, G.; Cheema, F. Initial experience of bamlanivimab monotherapy use in solid organ transplant recipients. Transpl. Infect. Dis. 2021, 23, e13662. [Google Scholar] [CrossRef]
- Vellas, C.; Del Bello, A.; Debard, A.; Steinmeyer, Z.; Tribaudeau, L.; Ranger, N.; Jeanne, N.; Martin-Blondel, G.; Delobel, P.; Kamar, N.; et al. Influence of treatment with neutralizing monoclonal antibodies on the SARS-CoV-2 nasopharyngeal load and quasispecies. Clin. Microbiol. Infect. 2022, 28, 139.e5–139.e8. [Google Scholar] [CrossRef] [PubMed]
- Biscarini, S.; Villa, S.; Genovese, C.; Tomasello, M.; Tonizzo, A.; Fava, M.; Iannotti, N.; Bolis, M.; Mariani, B.; Valzano, A.G.; et al. Safety Profile and Outcomes of Early COVID-19 Treatments in Immunocompromised Patients: A Single-Centre Cohort Study. Biomedicines 2022, 10, 2002. [Google Scholar] [CrossRef] [PubMed]
- Birk, N.K.; Jain, S.; Massoud, L.; Ramesh, D.; Monday, L.; Muma, B.; Williams, J.; Alangaden, G.; Ramesh, M. Real-world Experience of Sotrovimab in High-risk, Immunocompromised COVID-19 Patients. Open Forum. Infect. Dis. 2022, 9, ofac282. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention. COVID Data Trecker. 2022. Available online: https://covid.cdc.gov/covid-data-tracker (accessed on 21 July 2022).
- Vathsala, A.; Somani, J.; Ross D’Costa, M.; Lum, L.; Wong, E.T.; Sran, H.K. Comparative outcomes after early Sotrovimab administration in vaccinated and unvaccinated kidney transplant recipients with SARS-CoV-2 infection during the Delta and Omicron BA.1 surges. Transpl. Infect. Dis. 2022, e13930. [Google Scholar] [CrossRef]
- Lauring, A.S.; Tenforde, M.W.; Chappell, J.D.; Gaglani, M.; Ginde, A.A.; McNeal, T.; Ghamande, S.; Douin, D.J.; Talbot, H.K.; Casey, J.D.; et al. Influenza and Other Viruses in the Acutely Ill (IVY) Network. Clinical severity of, and effectiveness of mRNA vaccines against, COVID-19 from omicron, delta, and alpha SARS-CoV-2 variants in the United States: Prospective observational study. BMJ 2022, 376, e069761. [Google Scholar] [CrossRef]
- Villanego, F.; Mazuecos, A.; Cubillo, B.; Merino, M.J.; Poveda, I.; Saura, I.M.; Segurado, Ó.; Cruzado, L.; Eady, M.; Zárraga, S.; et al. Treatment with sotrovimab for SARS-CoV-2 infection in a cohort of high-risk kidney transplant recipients. Clin. Kidney J. 2022, 15, 1847–1855. [Google Scholar] [CrossRef]
- Vangeel, L.; Chiu, W.; De Jonghe, S.; Maes, P.; Slechten, B.; Raymenants, J.; André, E.; Leyssen, P.; Neyts, J.; Jochmans, D. Remdesivir, Molnupiravir and Nirmatrelvir remain active against SARS-CoV-2 Omicron and other variants of concern. Antivir. Res. 2022, 198, 105252. [Google Scholar] [CrossRef]
- Painter, W.P.; Holman, W.; Bush, J.A.; Almazedi, F.; Malik, H.; Eraut, N.C.J.E.; Morin, M.J.; Szewczyk, L.J.; Painter, G.R. Human Safety, Tolerability, and Pharmacokinetics of Molnupiravir, a Novel Broad-Spectrum Oral Antiviral Agent with Activity Against SARS-CoV-2. Antimicrob. Agents Chemother. 2021, 65, e02428-20. [Google Scholar] [CrossRef]
- Owen, D.R.; Allerton, C.M.N.; Anderson, A.S.; Aschenbrenner, L.; Avery, M.; Berritt, S.; Boras, B.; Cardin, R.D.; Carlo, A.; Coffman, K.J.; et al. An oral SARS-CoV-2 Mpro inhibitor clinical candidate for the treatment of COVID-19. Science 2021, 374, 1586–1593. [Google Scholar] [CrossRef]
- Schöning, V.; Kern, C.; Chaccour, C.; Hammann, F. Effectiveness of Antiviral Therapy in Highly-Transmissible Variants of SARS-CoV-2: A Modeling and Simulation Study. Front. Pharmacol. 2022, 13, 816429. [Google Scholar] [CrossRef]
- WHO. Update to living WHO guideline on drugs for COVID-19. BMJ 2022, 378, o1713. [Google Scholar]
- Wong, C.K.H.; Au, I.C.H.; Lau, K.T.K.; Lau, E.H.Y.; Cowling, B.J.; Leung, G.M. Real-world effectiveness of early molnupiravir or nirmatrelvir-ritonavir in hospitalised patients with COVID-19 without supplemental oxygen requirement on admission during Hong Kong’s omicron BA.2 wave: A retrospective cohort study. Lancet Infect. Dis. 2022; in press. [Google Scholar] [CrossRef]
- Elec, A.D.; Oltean, M.; Goldis, P.; Cismaru, C.; Lupse, M.; Muntean, A.; Elec, F.I. COVID-19 after kidney transplantation: Early outcomes and renal function following antiviral treatment. Int. J. Infect. Dis. 2021, 104, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Colaneri, M.; Amarasinghe, N.; Rezzonico, L.; Pieri, T.C.; Segalini, E.; Sambo, M.; Roda, S.; Meloni, F.; Gregorini, M.; Rampino, T.; et al. Early remdesivir to prevent severe COVID-19 in recipients of solid organ transplant: A real-life study from Northern Italy. Int. J. Infect. Dis. 2022, 121, 157–160. [Google Scholar] [CrossRef] [PubMed]
- Elec, F.; Magnusson, J.; Elec, A.; Muntean, A.; Antal, O.; Moisoiu, T.; Cismaru, C.; Lupse, M.; Oltean, M. COVID-19 and kidney transplantation: The impact of remdesivir on renal function and outcome—A retrospective cohort study. Int. J. Infect. Dis. 2022, 118, 247–253. [Google Scholar] [CrossRef]
- Radcliffe, C.; Palacios, C.F.; Azar, M.M.; Cohen, E.; Malinis, M. Real-world experience with available, outpatient COVID-19 therapies in solid organ transplant recipients during the omicron surge. Am. J. Transplant. 2022; online ahead of print. [Google Scholar] [CrossRef]
- Hedvat, J.; Lange, N.W.; Salerno, D.M.; DeFilippis, E.M.; Kovac, D.; Corbo, H.; Chen, J.K.; Choe, J.Y.; Lee, J.H.; Anamisis, A.; et al. COVID-19 therapeutics and outcomes among solid organ transplant recipients during the Omicron BA.1 era. Am. J. Transplant. 2022; online ahead of print. [Google Scholar] [CrossRef]
- Gandolfini, I.; Crespo, M.; Hellemans, R.; Maggiore, U.; Mariat, C.; Mjoen, G.; Oniscu, G.C.; Peruzzi, L.; Sever, M.S.; Watschinger, B.; et al. Issues regarding COVID-19 in kidney transplantation in the era of the omicron variant: A commentary by the era descartes working group. Nephrol. Dial. Transplant. 2022, 37, 1824–1829. [Google Scholar] [CrossRef]
- Salerno, D.M.; Jennings, D.L.; Lange, N.W.; Kovac, D.B.; Shertel, T.; Chen, J.K.; Hedvat, J.; Scheffert, J.; Brown, R.S., Jr.; Pereira, M.R. Early clinical experience with nirmatrelvir/ritonavir for the treatment of COVID-19 in solid organ transplant recipients. Am. J. Transplant. 2022, 22, 2083–2088. [Google Scholar] [CrossRef]
- Prikis, M.; Cameron, A. Paxlovid (Nirmatelvir/Ritonavir) and Tacrolimus Drug-Drug Interaction in a Kidney Transplant Patient with SARS-2-CoV infection: A Case Report. Transplant. Proc. 2022, 54, 1557–1560. [Google Scholar] [CrossRef]
- Wang, A.X.; Koff, A.; Hao, D.; Tuznik, N.M.; Huang, Y. Effect of nirmatrelvir/ritonavir on calcineurin inhibitor levels: Early experience in four SARS-CoV-2 infected kidney transplant recipients. Am. J. Transplant. 2022, 22, 2117–2119. [Google Scholar] [CrossRef] [PubMed]
- Lange, N.W.; Salerno, D.M.; Jennings, D.L.; Choe, J.; Hedvat, J.; Kovac, D.B.; Scheffert, J.; Shertel, T.; Ratner, L.E.; Brown, R.S., Jr.; et al. Nirmatrelvir/ritonavir use: Managing clinically significant drug-drug interactions with transplant immunosuppressants. Am. J. Transplant. 2022, 22, 1925–1926. [Google Scholar] [CrossRef] [PubMed]
- Lemaitre, F.; Grégoire, M.; Monchaud, C.; Bouchet, S.; Saint-Salvi, B.; Polard, E.; SFPT Therapeutic Drug Monitoring and Treatment Personalization Group (STP-PT) of the French Society of Pharmacology and Therapeutics (SFPT); French Pharmacovigilance Network (CRPV); ANRS-MIE AC-43 Clinical Pharmacology Committee, Joint Working Group; SFPT Therapeutic Drug Monitoring and Treatment Personalization Group (STP-PT) of the French Society of Pharmacology and Therapeutics (SFPT); et al. Management of drug-drug interactions with nirmatrelvir/ritonavir in patients treated for COVID-19: Guidelines from the French Society of Pharmacology and Therapeutics (SFPT). Therapie 2022, 77, 509–521. [Google Scholar] [CrossRef] [PubMed]
| Multivariate Analysis | |||
|---|---|---|---|
| Factors | OR | 95% CI | p Value |
| (a) | |||
| Age at 2nd vaccination | 0.98 | 0.96; 1.00 | 0.039 |
| Time after kidney TX | 1.06 | 1.02; 1.10 | 0.001 |
| TAC + MPA + Steroid | 0.15 | 0.08; 0.28 | <0.001 |
| CyA + MPA + Steroid | 0.51 | 0.27; 0.96 | 0.038 |
| TAC/CyA + Steroid | 4.11 | 1.71; 9.90 | 0.002 |
| eGFR at vaccination | 1.03 | 1.02; 1.04 | <0.001 |
| Lymphocyte count at vaccination | 1.12 | 1.06; 1.18 | <0.001 |
| CNI trough levels at vaccination | 0.94 | 0.90; 1.00 | 0.036 |
| (b) | |||
| Female | 0.41 | 0.20; 0.83 | 0.013 |
| Age at 2nd vaccination | 0.96 | 0.94; 0.99 | 0.002 |
| Time after kidney TX | 1.07 | 1.01; 1.13 | 0.031 |
| TAC MPA Steroids | 0.43 | 0.20; 0.95 | 0.036 |
| CyA MPA Steroids | 1.30 | 0.57–2.98 | 0.534 |
| eGFR at vaccination | 1.03 | 1.01; 1.05 | 0.014 |
| Lymphocyte count at vaccination | 1.06 | 0.99; 1.14 | 0.077 |
| CNI trough levels at vaccination | 0.92 | 0.84; 1.00 | 0.040 |
| MPA dose at vaccination | 0.72 | 0.59; 0.87 | 0.001 |
| IMPDH activity | 0.34 | 0.25; 0.46 | <0.001 |
| Before 3rd Vaccine | Post-3rd-Vaccine | p Value | |
|---|---|---|---|
| All cohort | |||
| IgG-RBD GMT (95% CI) | 0.79 (0.65–0.96) | 3.08 (2.76–3.45) | <0.0001 |
| NA GMT (95% CI) | 17.46 (12.38–24.62) | 362.2 (220.7–594.6) | <0.0001 |
| Positive responders | |||
| N (%) | 32 (32.3) | 85 (85.9) | <0.0001 |
| =35IgG-RBD GMT (95% CI) | 2.53 (2.07–3.11) | 3.57 (3.28–3.88) | <0.0001 |
| NA GMT (95% CI) | 89.12 (53.03–149.8) | 689.9 (456.3–1043) | <0.0001 |
| Negative responders | |||
| N (%) | 67 (67.7) | 14 (14.14) | <0.0001 |
| IgG-RBD GMT (95% CI) | 0.45 (0.39–052) | 1.28 (0.87–1.86) | <0.0001 |
| NA GMT (95% CI) | 8.01 (5.92–10.84) | 7.25 (2.42–21.71) | 0.85 |
| Kidney | Statistics | Transplant | Control | |||
|---|---|---|---|---|---|---|
| A Serological response compared with controls after one dose of vaccine | ||||||
| Kidney | Odds ratio | Lower limit | Upper limit | p Value | ||
| 0.0063 | 0.0025 | 0.0160 | <0.001 | 20/299 | 89/96 | |
| B Serological response compared with controls after two doses of vaccine | ||||||
| 0.0063 | 0.0025 | 0.0159 | <0.001 | 208/655 | 271/273 | |
| C Serological response compared with controls after three doses of vaccine | ||||||
| 0.669 | 0.601 | 0.732 | <0.001 | 132/197 | ||
| Outcomes | MoAb Group | Control Group | p Value |
|---|---|---|---|
| Severe COVID-19, n (%) | 3 (3.8) | 30 (19.4) | 0.001 |
| Admission to ICU, n (%) | 2 (2.5) | 24 (15.5) | 0.002 |
| Need for mechanical ventilation, n (%) | 0 (0.0) | 18 (11.6) | <0.001 |
| Death, n (%) | 1 (1.25) | 18 (11.6) | 0.005 |
| Variable | <5 Days (n = 46) | 5 Days (n = 36) | p-Value |
|---|---|---|---|
| Ventilator support, n (%) | 1 (2.2) | 13 (36.1) | <0.001 |
| ICU admission, n (%) | 1 (2.2) | 9 (25) | 0.002 |
| Death, n (%) | 1 (2.2) | 6 (16.7) | 0.02 |
| Class and Agent | Dosing | Place in Therapy | Drug-Drug Interactions | Adverse Effects | Special Consideration for TX Recipients |
|---|---|---|---|---|---|
| Monoclonal antibodies | Bemlanivimab 700 mg plus etesevimab 1400 mg IV as a single dose | NIH Guidelines # Casirivimab plus imdevimab or sotrovimab recommended for outpatients with mild to moderate COVID-19 # Recommend against use of bamlanivimab plus etesevimab # Recommend against use in hospitalized patients outside a clinical trial | May decrease the effects of COVID-19 vaccination; postpone administration of COVID-19 vaccine until at least 90 days after treatment | Hypersensitivity Pruritis Injection site reactions Fever | # Authorized under FDA EUA # Administer in healthcare settings # Monitor patient for at least 1 h post-administration # Use of bamlanivimab alone and bamlanivimab plus etesevimab is not recommended due to decreased susceptibility of SARS-CoV-2 variants |
| Casirivimab 600 mg plus imdevimab 600 mg IV/SQ as a single dose | WHO Guidelines # Class not addressed | ||||
| Sotrovimab 500 mg IV as a single dose | IDSA Guidelines # Recommended for ambulatory patients with mild to moderate COVID-19 at high risk |
| Nirmatrelvir/Ritonavir (Paxlovid) | Molnupiravir (Lagevrio) | Remdesivir (Veklury) | |
|---|---|---|---|
| Population | Age > 12 years and >40 kg. Mild to moderate COVID-19 and high risk of progression to hospitalization or death. EMA and FDA approved | Age > 18 years. Mild to moderate COVID-19 and high risk of progression or death. EMA and FDA approved | Age > 12 years and >40 kg requiring supplemental oxygen. EMA and FDA approved (in and out of hospital) |
| Efficacy (high-risk population) | NNT (number needed to treat) = 18 (all cause hospitalization or death) | NNT = 35 (all cause hospitalization or death) when administered within the first 5 days of symptom onset | NNT = 22 (all cause hospitalization or death) |
| Drug interactions | Serious concern (ritonavir strongly inhibits CYP3A4) | Negligible | Monitor when co-administered with strong CYP3A4 inducers/inhibitors |
| Common side-effects | Dysgeusia and diarrhea | Diarrhea, nausea, anemia and potentially mutagenicity | Bradycardia, drug-induced liver injury and acute kidney injury |
| Renal/hepatic impairment | Dose adjustment with moderate renal impairment | No dose adjustment required | Not recommended if eGFR < 30 mL/min |
| Pregnancy | Contraindicated | Contraindicated | Reassuring data |
| Activity vs. variants | All known variants | All known variants | All known variants |
| Immunosuppressive Drug | Nature and Magnitude of the Effect | Therapeutic Strategy |
|---|---|---|
| Tacrolimus | Increase in tacrolimus exposure by 40-fold | Administer 1/8 of the usual daily dose (DD) on day 1, then stop. Administer 1/2 of the DD on day 6, then 3/4 on day 7 and restart usual DD on day 8. Alternative for low immunological risk: start nirmatrelvir/ritonavir 12 h after the last intake of tacrolimus and restart tacrolimus at usual DD 24 h after the last antiviral dose. TDM if possible |
| Cyclosporine | Increase in cyclosporine exposure by 8-fold | Administer 1/5 of the usual DD every day of nirmatrelvir/ritonavir treatment. Administer 1/2 of the DD on day 6, then 3/4 on day 7 and restart usual DD on day 8. TDM if possible |
| Everolimus/Sirolimus | Increase in everolimus and sirolimus exposure by 15- and 11-fold, respectively | Administer 1/8 of the usual DD on day 1, day 3 and day 5. Usual DD can be restarted on day-7. TDM if possible |
| Mycophenolate mofetil | Weak interaction expected. Possible decrease in mycophenolic acid exposure | The dosage can be maintained |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salvadori, M. What Is New in Prophylaxis and Treatment of COVID-19 in Renal Transplant Patients? A Report from an ESOT Meeting on the Topic. Transplantology 2022, 3, 288-306. https://doi.org/10.3390/transplantology3040030
Salvadori M. What Is New in Prophylaxis and Treatment of COVID-19 in Renal Transplant Patients? A Report from an ESOT Meeting on the Topic. Transplantology. 2022; 3(4):288-306. https://doi.org/10.3390/transplantology3040030
Chicago/Turabian StyleSalvadori, Maurizio. 2022. "What Is New in Prophylaxis and Treatment of COVID-19 in Renal Transplant Patients? A Report from an ESOT Meeting on the Topic" Transplantology 3, no. 4: 288-306. https://doi.org/10.3390/transplantology3040030
APA StyleSalvadori, M. (2022). What Is New in Prophylaxis and Treatment of COVID-19 in Renal Transplant Patients? A Report from an ESOT Meeting on the Topic. Transplantology, 3(4), 288-306. https://doi.org/10.3390/transplantology3040030





