A Machine Learning Algorithm Predicting Infant Psychomotor Developmental Delay Using Medical and Social Determinants
Abstract
1. Introduction
1.1. Predictors of Developmental Delay
1.2. Data Analytic Approach
2. Methods
2.1. Population and Sample
2.2. Outcome Variable
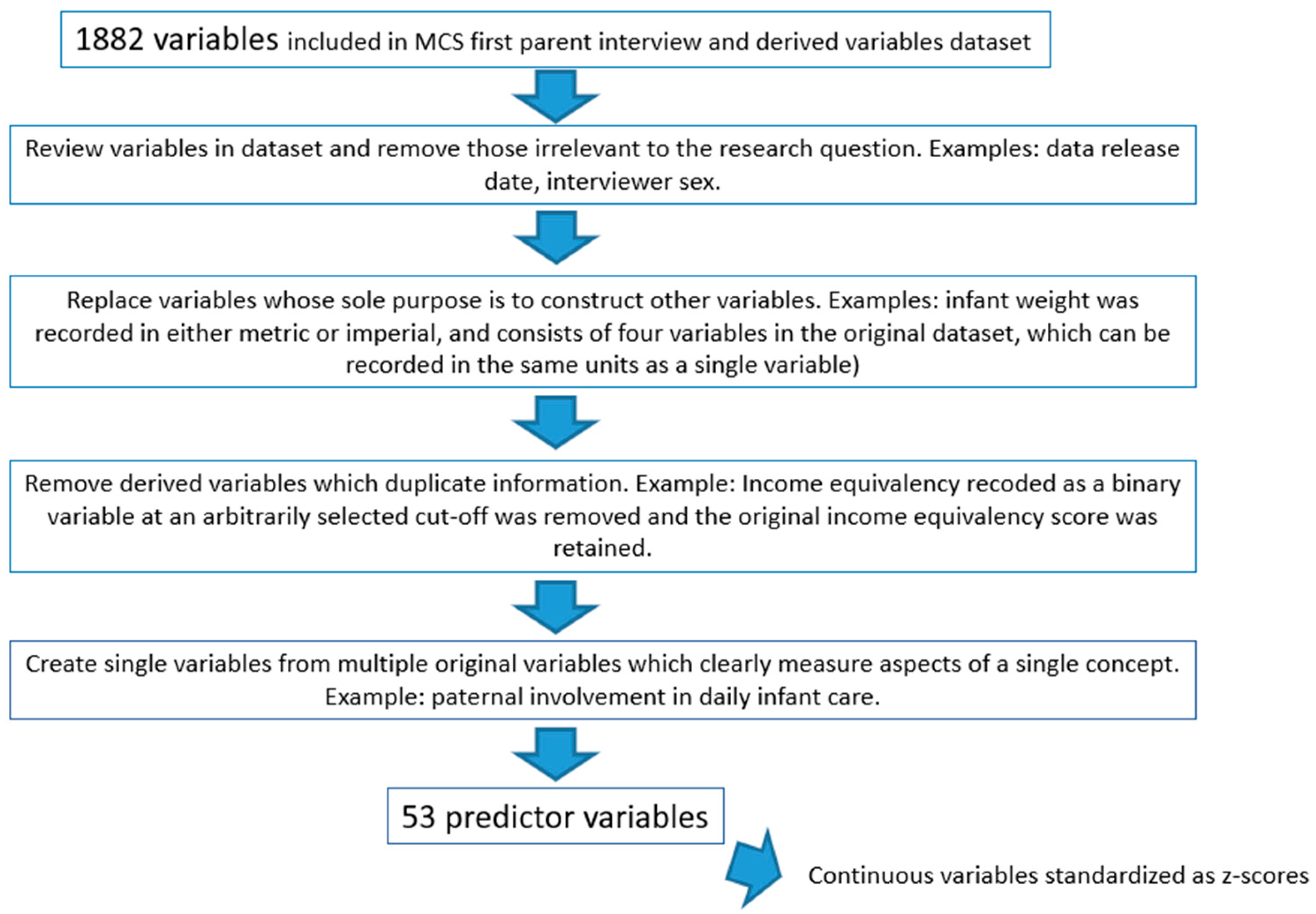
2.3. Predictor Variables (Features)
2.4. Data Analysis
3. Results
3.1. Descriptive Statistics
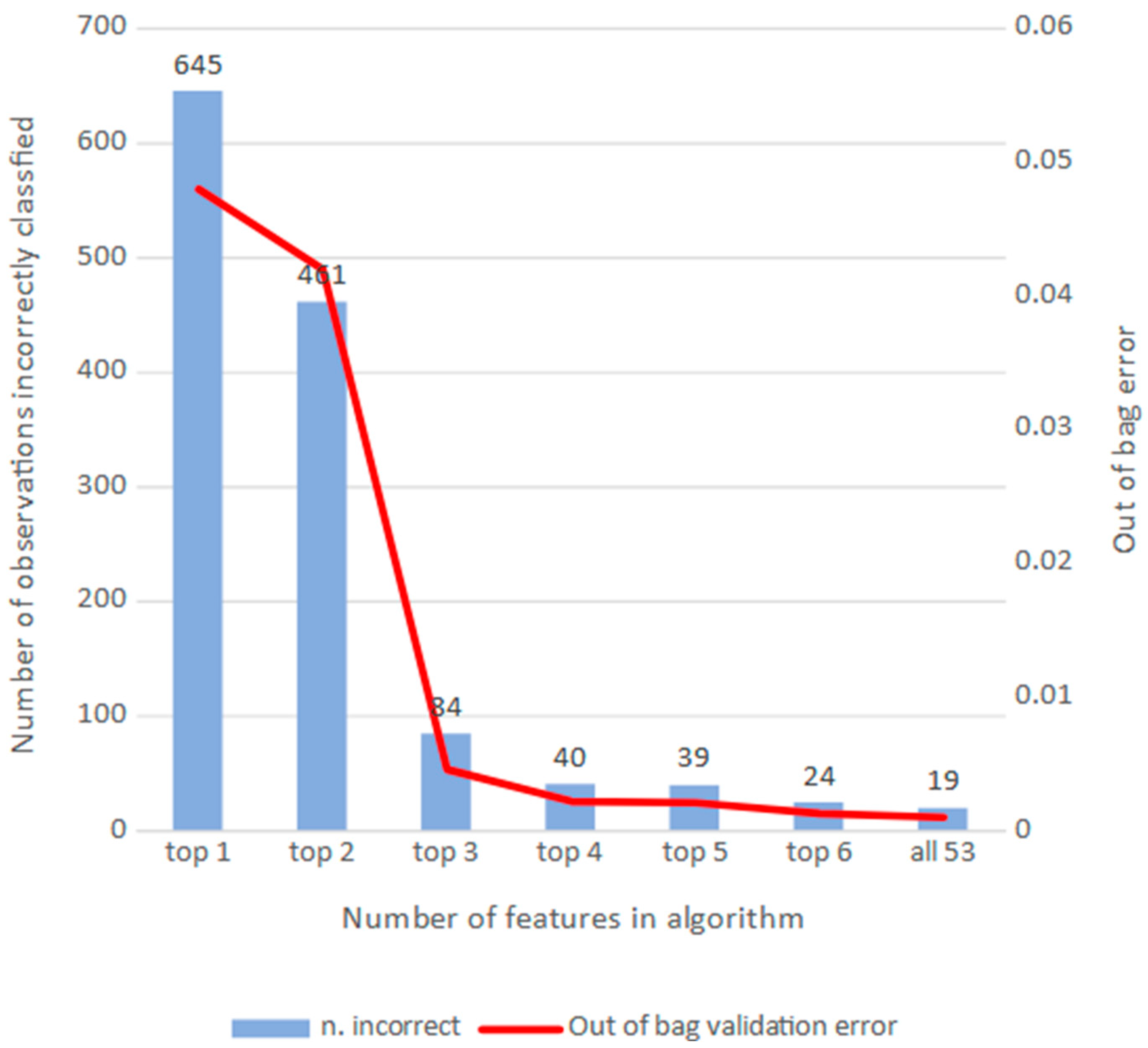
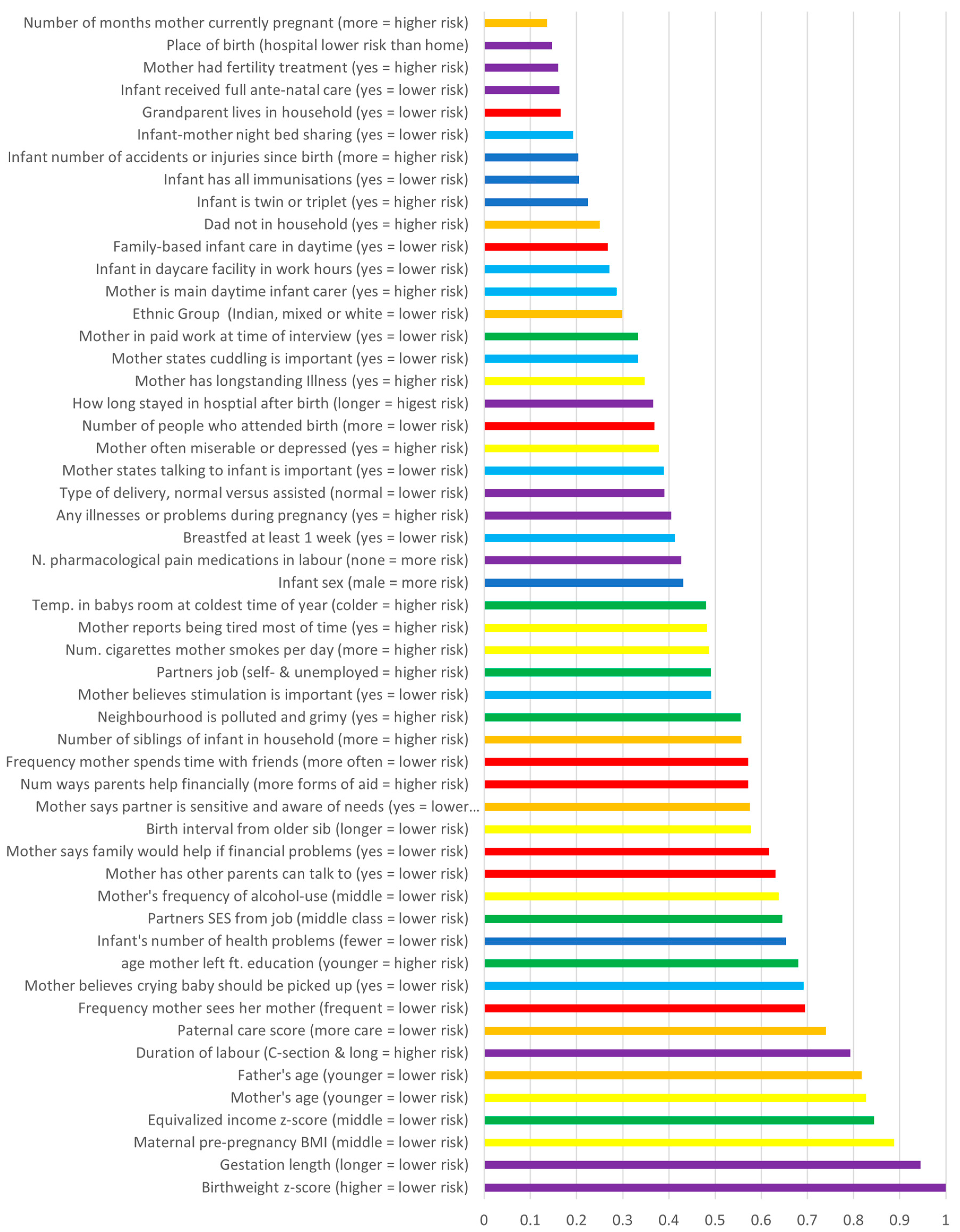
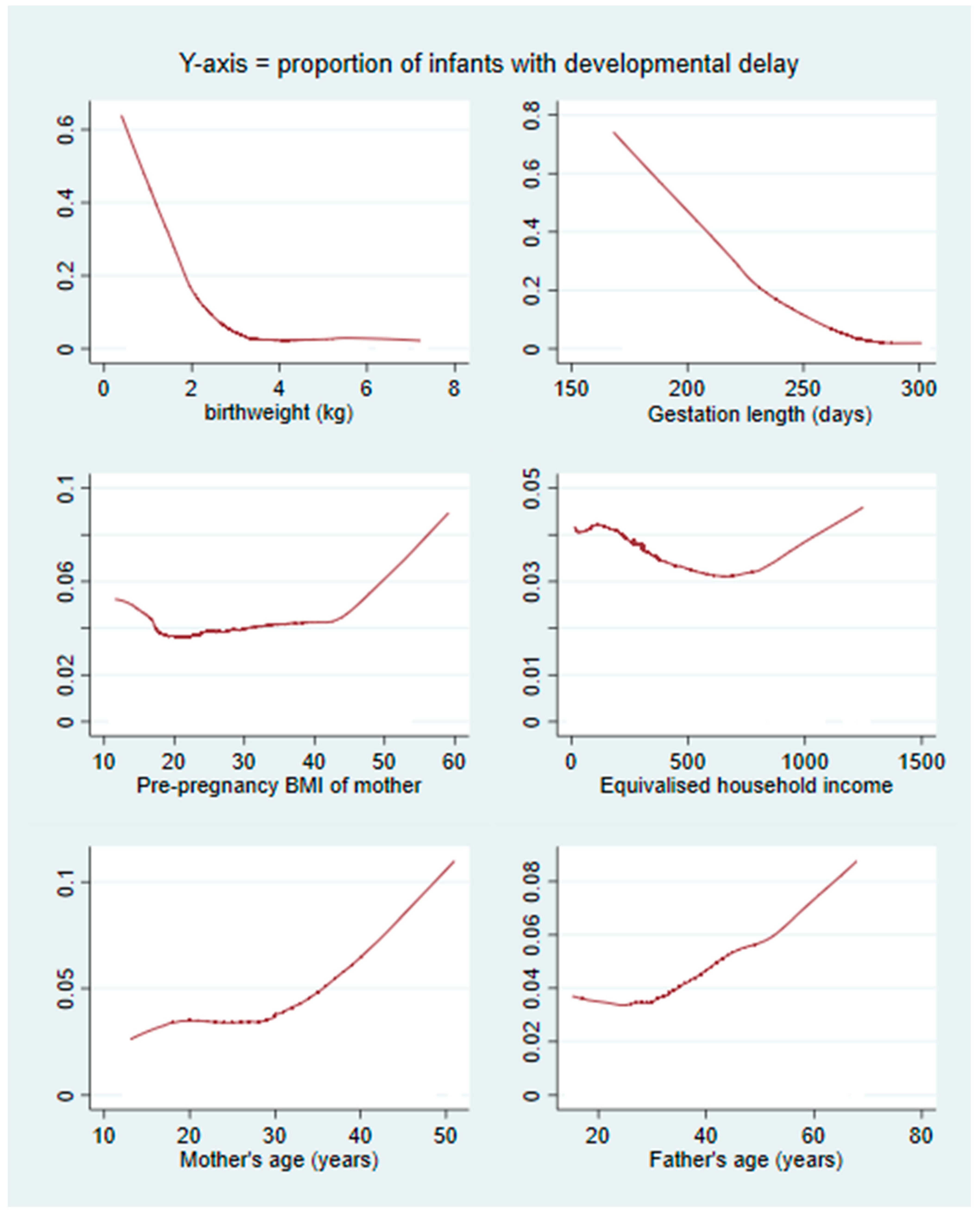
3.2. RF Algorithms
4. Discussion
4.1. Summary
4.2. Comparisons to Prior Studies
4.3. Study Limitations
5. Conclusions
Supplementary Materials
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sadruddin, A.F.; Ponguta, L.A.; Zonderman, A.L.; Wiley, K.S.; Grimshaw, A.; Panter-Brick, C. How do grandparents influence child health and development? A systematic review. Soc. Sci. Med. 2019, 239, 112476. [Google Scholar] [CrossRef]
- Erel, O.; Oberman, Y.; Yirmiya, N. Maternal versus nonmaternal care and seven domains of children’s development. Psychol. Bull. 2000, 126, 727–747. [Google Scholar] [CrossRef] [PubMed]
- Crnic, K.A.; Greenberg, M.T.; Ragozin, A.S.; Robinson, N.M.; Basham, R.B. Effects of Stress and Social Support on Mothers and Premature and Full-Term Infants. Child Dev. 1983, 54, 209. [Google Scholar] [CrossRef]
- Shaver, J.H.; Power, E.A.; Purzycki, B.G.; Watts, J.; Sear, R.; Shenk, M.K.; Sosis, R.; Bulbulia, J.A. Church attendance and alloparenting: An analysis of fertility, social support and child development among English mothers. Philos. Trans. R. Soc. B 2020, 375, 20190428. [Google Scholar] [CrossRef] [PubMed]
- Sacker, A.; Quigley, M.A.; Kelly, Y.J. Breastfeeding and Developmental Delay: Findings from the Millennium Cohort Study. Pediatrics 2006, 118, e682–e689. [Google Scholar] [CrossRef] [PubMed]
- Chiu, W.C.; Liao, H.F.; Chang, P.J.; Chen, P.C.; Chen, Y.C. Duration of breast feeding and risk of developmental delay in Taiwanese children: A nationwide birth cohort study. Paediatr. Perinat. Epidemiol. 2011, 25, 519–527. [Google Scholar] [CrossRef]
- Belsky, J. Early child care and early child development: Major findings of the NICHD study of early child care. Eur. J. Dev. Psychol. 2006, 3, 95–110. [Google Scholar] [CrossRef]
- Waynforth, D. Effects of Conception Using Assisted Reproductive Technologies on Infant Health and Development: An Evolutionary Perspective and Analysis Using UK Millennium Cohort Data. Yale J. Biol. Med. 2018, 91, 225–235. [Google Scholar] [PubMed]
- Brown, M.A.; McIntyre, L.L.; Crnic, K.A.; Baker, B.L.; Blacher, J. Preschool Children With and Without Developmental Delay: Risk, Parenting, and Child Demandingness. J. Ment. Health Res. Intellect. Disabil. 2011, 4, 206–226. [Google Scholar] [CrossRef]
- Conde-Agudelo, A.; Castaño, F.; Norton, M.H.; Rosas-Bermudez, A. Effects of Birth Spacing on Maternal, Perinatal, Infant, and Child Health: A Systematic Review of Causal Mechanisms. Stud. Fam. Plan. 2012, 43, 93–114. [Google Scholar] [CrossRef]
- McIntire, D.D.; Bloom, S.L.; Leveno, K.; Casey, B.M. Birth Weight in Relation to Morbidity and Mortality among Newborn Infants. N. Engl. J. Med. 1999, 340, 1234–1238. [Google Scholar] [CrossRef] [PubMed]
- Ketterlinus, R.D.; Henderson, S.H.; Lamb, M.E. Maternal age, sociodemographics, prenatal health and behavior: Influences on neonatal risk status. J. Adolesc. Health Care 1990, 11, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Ozkan, M.; Senel, S.; Arslan, E.A.; Karacan, C.D. The socioeconomic and biological risk factors for developmental delay in early childhood. Eur. J. Pediatr. 2012, 171, 1815–1821. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Sun, Z.; Neiderhiser, J.M.; Uchiyama, M.; Okawa, M. Low birth weight, developmental milestones, and behavioral problems in Chinese children and adolescents. Psychiatry Res. 2001, 101, 115–129. [Google Scholar] [CrossRef] [PubMed]
- Van Dokkum, N.H.; Reijneveld, S.A.; Heymans, M.W.; Bos, A.F.; de Kroon, M.L.A. Development of a Prediction Model to Identify Children at Risk of Future Developmental Delay at Age 4 in a Population-Based Setting. Int. J. Environ. Res. Public Health 2020, 17, 8341. [Google Scholar] [CrossRef]
- Abubakar, A.; Holding, P.; Van de Vijver, F.J.R.; Newton, C.; van Baar, A. Children at risk for developmental delay can be recognised by stunting, being underweight, ill health, little maternal schooling or high gravidity. J. Child Psychol. Psychiatry 2009, 51, 652–659. [Google Scholar] [CrossRef]
- Najman, J.M.; Bor, W.; Morrison, J.; Andersen, M.; Williams, G. Child developmental delay and socio-economic disadvantage in Australia: A longitudinal study. Soc. Sci. Med. 1992, 34, 829–835. [Google Scholar] [CrossRef]
- Myszczynska, M.A.; Ojamies, P.N.; Lacoste, A.M.B.; Neil, D.; Saffari, A.; Mead, R.; Hautbergue, G.M.; Holbrook, J.D.; Ferraiuolo, L. Applications of machine learning to diagnosis and treatment of neurodegenerative diseases. Nat. Rev. Neurol. 2020, 16, 440–456. [Google Scholar] [CrossRef]
- Goecks, J.; Jalili, V.; Heiser, L.M.; Gray, J.W. How Machine Learning Will Transform Biomedicine. Cell 2020, 181, 92–101. [Google Scholar] [CrossRef]
- Usta, M.B.; Karabekiroglu, K.; Say, G.N.; Gumus, Y.Y.; Aydın, M.; Sahin, B.; Bozkurt, A.; Karaosman, A.A.; Cobanoglu, C.; Kurt, D.A.; et al. Can We Predict Psychiatric Disorders at the Adolescence Period in Toddlers? A Machine Learning Approach. Psychiatry Behav. Sci. 2020, 10, 7–12. [Google Scholar] [CrossRef]
- Harrison, E.; Syed, S.; Ehsan, L.; Iqbal, N.T.; Sadiq, K.; Umrani, F.; Ahmed, S.; Rahman, N.; Jakhro, S.; Ma, J.Z.; et al. Machine learning model demonstrates stunting at birth and systemic inflammatory biomarkers as predictors of subsequent infant growth—A four-year prospective study. BMC Pediatr. 2020, 20, 498. [Google Scholar] [CrossRef] [PubMed]
- Vora, N.; Bianchi, D.W. Genetic considerations in the prenatal diagnosis of overgrowth syndromes. Prenat. Diagn. 2009, 29, 923–929. [Google Scholar] [CrossRef]
- Patel, M.; Waynforth, D. Influences of zero hour contracts and disability—Analysis of the 1970 British Cohort study. SSM-Popul. Health 2022, 19, 101182. [Google Scholar] [CrossRef] [PubMed]
- Grömping, U. Variable Importance Assessment in Regression: Linear Regression versus Random Forest. Am. Stat. 2009, 63, 308–319. [Google Scholar] [CrossRef]
- Breiman, L. Random Forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Ketende, S.; Jones, E. User Guide to Analysing MCS Data Using Stata; Centre for Longitudinal Studies: London, UK, 2011. [Google Scholar]
- Connelly, R.; Platt, L. Cohort Profile: UK Millennium Cohort Study: MCS. Int. J. Epidemiol. 2014, 43, 1719–1725. [Google Scholar] [CrossRef]
- Bricker, D.; Squires, J.; Mounts, L.; Potter, L.; Nickel, R.; Twombly, E.; Farrell, J. Ages and Stages Questionnaire; Paul, H. Brookes: Baltimore, MD, USA, 1999. [Google Scholar]
- Schonlau, M.; Zou, R.Y. The Random Forest Algorithm for Statistical Learning. Stata J. 2020, 20, 3–29. [Google Scholar] [CrossRef]
- Bilano, V.L.; Ota, E.; Ganchimeg, T.; Mori, R.; Souza, J.P. Risk Factors of Pre-Eclampsia/Eclampsia and Its Adverse Outcomes in Low- and Middle-Income Countries: A WHO Secondary Analysis. PLoS ONE 2014, 9, e91198. [Google Scholar] [CrossRef] [PubMed]
| Variable | Coding | Obs | Mean (Std.Dev.) | Min–Max |
|---|---|---|---|---|
| Outcome and its constituent child development measures | ||||
| Development below 2SD (age-adjusted) | 18,432 | 0.038 (0.191) | 0–1 | |
| Smiles | 1 = often, 2 = sometimes, 3 = not yet | 18,432 | 1.006 (0.082) | 1–3 |
| Sits up | 18,432 | 1.066 (0.318) | 1–3 | |
| Stands up holding on | 18,432 | 1.475 (0.78) | 1–3 | |
| Puts hands together | 18,432 | 1.209 (0.532) | 1–3 | |
| Grabs objects | 18,432 | 1.01 (0.117) | 1–3 | |
| Holds small objects | 18,432 | 1.147 (0.454) | 1–3 | |
| Passes a toy | 18,432 | 1.065 (0.295) | 1–3 | |
| Walks a few steps | 18,432 | 2.81 (0.519) | 1–3 | |
| Gives a toy | 18,432 | 1.52 (0.717) | 1–3 | |
| Waves bye-bye | 18,432 | 1.912 (0.839) | 1–3 | |
| Extends arms | 18,432 | 1.205 (0.499) | 1–3 | |
| Nods for yes | 18,432 | 2.72 (0.617) | 1–3 | |
| Family and social support | ||||
| Frequency mother sees her mother | 0 = lives with mother, 1 = every day, to 8 = never | 18,544 | 3.277 (2.352) | 0–8 |
| Mother has other parents to talk to | 1 = most, to 5 = least | 17,805 | 2.096 (1.016) | 1–5 |
| Family would help if financial problems | Strongly agree = 1 to strongly disagree = 5 | 17,803 | 1.747 (0.971) | 1–5 |
| Number of types of financial help from grandparents | Gifts, money for daycare, essentials, trust funds, household items, other | 18,547 | 1.235 (1.057) | 0–6 |
| Frequency mother reports spending time with friends | 1 = every day, to 5 = never or no friends | 18,527 | 2.958 (0.974) | 1–5 |
| Number of people who attended birth | 18,432 | 1.12 (0.495) | 0–4 | |
| Family-based infant care in work hours | 1 = no, 2 = yes | 18,387 | 1.17 (0.375) | 1–2 |
| Grandparent lives in household | 1 = yes, 2 = no | 18,432 | 1.921 (0.269) | 1–2 |
| Socioeconomic indicators | ||||
| Equivalised household income | McClement’s equivalised income | 18,432 | 296.833 (217.102) | 14.31–1250.78 |
| Age mother left full time education | 18,341 | 17.578 (2.848) | 5–36 | |
| Partner’s SES from job | NS-SEC 7 classes, 1 = highest, 7 = lowest, 8 = not in work | 18,432 | 5.352 (2.641) | 1–8 |
| Partner’s employment status | 1 = employed, 2 = self-employed, 3 = looking for work, 4 = not seeking work due to health, 5 = New Deal/apprenticeship, 6 = student, 7 = no partner/unknown | 18,432 | 3.388 (3.084) | 1–8 |
| Mother employed | Mother in paid work at 9 month interview = 1, else = 2 | 18,399 | 1.448 (0.497) | 1–2 |
| Winter temperature in room where baby sleeps | 5-point scale where 1 = warmest and 5 = cold | 18,310 | 2.301 (0.745) | 1–5 |
| Mother’s report of pollution & grime in neighbourhood | Reported on a 4-point scale, 1 = most, to 4 = least pollution | 18,218 | 3.089 (0.892) | 1–4 |
| Infant characteristics | ||||
| Infant’s sex | 1 = male, 2 = female | 18,432 | 1.487 (0.5) | 1–2 |
| Infant has all immunisations | 1 = yes, 2 = no | 18,175 | 1.039 (0.194) | 1–2 |
| Infant’s age in days when mother was interviewed | 18,432 | 295.487 (15.23) | 243–382 | |
| Infant’s number of reported illness | 18,422 | 1.633 (1.992) | 0–50 | |
| Infant’s number of accidents | 18,430 | 0.083 (0.296) | 0–5 | |
| Beliefs about parenting & parenting practices | ||||
| Beliefs: Baby should be picked up when cries | 1 = strongly agree, to 5 = strongly disagree | 17,810 | 2.966 (1.045) | 1–5 |
| Beliefs: Stimulation is important for infant development | 1 = strongly agree, to 5 = strongly disagree | 17,806 | 1.431 (0.626) | 1–5 |
| Beliefs: Talking to infants is important | 1 = strongly agree, to 5 = strongly disagree | 17,814 | 1.200 (0.448) | 1–5 |
| Beliefs: cuddling infants is important | 1 = strongly agree, to 5 = strongly disagree | 17,815 | 1.191 (0.452) | 1–5 |
| Bed co-sleeping main sleeping arrangement in first 9 months | 1 = no, 2 = yes | 18,431 | 1.089 (0.285) | 1–2 |
| Breastfed at least 1 week | 1 = no, 2 = yes | 18,431 | 1.536 (0.499) | 1–2 |
| Work hours infant care is daycare centre | 1 = no, 2 = yes | 18,432 | 1.115 (0.319) | 1–2 |
| Main work hours infant care is mother | 1 = no, 2 = yes | 18,432 | 1.691 (0.462) | 1–2 |
| Factors in pregnancy & birth | ||||
| Birthweight (kg) | 18,382 | 3.344 (0.589) | 0.39–7.23 | |
| Estimated gestational age at birth (days) | 18,201 | 275.727 (14.056) | 168–301 | |
| Number of pharmacological pain interventions in labour | 18,293 | 0.731 (0.667) | 0–4 | |
| Infant conceived using fertility treatment | 1 = no, 2 = yes | 18,425 | 1.974 (0.159) | 1–2 |
| Duration of labour | In hours, C-section = 0 | 17,680 | 9.160 (11.145) | 0–100 |
| Type of delivery | 1 = normal, C-section & emergency = 2 | 18,398 | 1.313 (0.464) | 1–2 |
| Singleton birth | 1 = singleton, 2 = twin, 3 = triplet | 18,432 | 1.014 (0.123) | 1–3 |
| Pregnancy illnesses (e.g., preeclampsia) | 1 = yes, 2 = no | 18,396 | 1.623 (0.485) | 1–2 |
| Place of birth | Hospital = 1, else 2 | 18,401 | 1.020 (0.142) | 1–2 |
| How long mother and infant stayed in hospital after birth | 1 = weeks, 2 = days, 3 = hours | 18,020 | 2.046 (0.421) | 1–3 |
| Received full ante-natal care | 1 = yes, 2 = no | 18,391 | 1.038 (0.192) | 1–2 |
| Maternal factors | ||||
| Mother’s pre-pregnancy body mass index | 16,813 | 23.649 (4.451) | 11.65–59.18 | |
| Mother’s birth year | 18,426 | 1972 (5.95) | 1949–1987 | |
| Mother reports being tired all the time | 1 = yes, 2 = no | 17,805 | 1.509 (0.5) | 1–2 |
| Mother reports being depressed | 1 = yes, 2 = no | 17,802 | 1.849 (0.358) | 1–2 |
| Average number of cigarettes mother smokes per day | 18,420 | 3.315 (6.271) | 0–60 | |
| Frequency mother drinks alcohol | Every day = 1 to never = 7 | 18,429 | 5.134 (1.49) | 1–7 |
| Mother has longstanding illness | 1 = yes, 2 = no | 18,425 | 1.789 (0.408) | 1–2 |
| Number of months pregnant at interview | 18,423 | 0.196 (1.013) | 0–10 | |
| Paternal & family factors | ||||
| Ethnicity | 1 = white, 2 = mixed, 3 = India, 4 = Pakistani, 5 = Bangladeshi, 6 = Caribbean, 7 = African, 8 = East Asian & others | 18,402 | 1.627 (1.609) | 1–8 |
| Father present in household | 0 = yes, 1 = no | 18,403 | 0.172 (0.378) | 0–1 |
| Father’s age when infant was born | 18,395 | 31.91 (5.713) | 15–68 | |
| Paternal involvement score: how much help father is | Summed score of how often father does: general childcare, feeding, getting up in night, changing nappies. 1 = least, to 21 = most | 16,255 | 10.205 (5.868) | 1–21 |
| Birth interval in months from older sibling | 8997 | 42.803 (27.86) | 9–318 | |
| Number of siblings in household | 18,432 | 0.938 (1.081) | 0–9 | |
| Mother reports partner sensitive and aware of her needs | Strongly agree = 1 to strongly disagree = 5 | 14,358 | 1.986 (0.929) | 1–5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Waynforth, D. A Machine Learning Algorithm Predicting Infant Psychomotor Developmental Delay Using Medical and Social Determinants. Reprod. Med. 2023, 4, 106-117. https://doi.org/10.3390/reprodmed4020012
Waynforth D. A Machine Learning Algorithm Predicting Infant Psychomotor Developmental Delay Using Medical and Social Determinants. Reproductive Medicine. 2023; 4(2):106-117. https://doi.org/10.3390/reprodmed4020012
Chicago/Turabian StyleWaynforth, David. 2023. "A Machine Learning Algorithm Predicting Infant Psychomotor Developmental Delay Using Medical and Social Determinants" Reproductive Medicine 4, no. 2: 106-117. https://doi.org/10.3390/reprodmed4020012
APA StyleWaynforth, D. (2023). A Machine Learning Algorithm Predicting Infant Psychomotor Developmental Delay Using Medical and Social Determinants. Reproductive Medicine, 4(2), 106-117. https://doi.org/10.3390/reprodmed4020012






