Is Sacubitril/Valsartan Able to Change the Timing for Implantation of Cardiac Devices in Heart Failure with Reduced Ejection Fraction?
Abstract
:1. Introduction
2. Methods
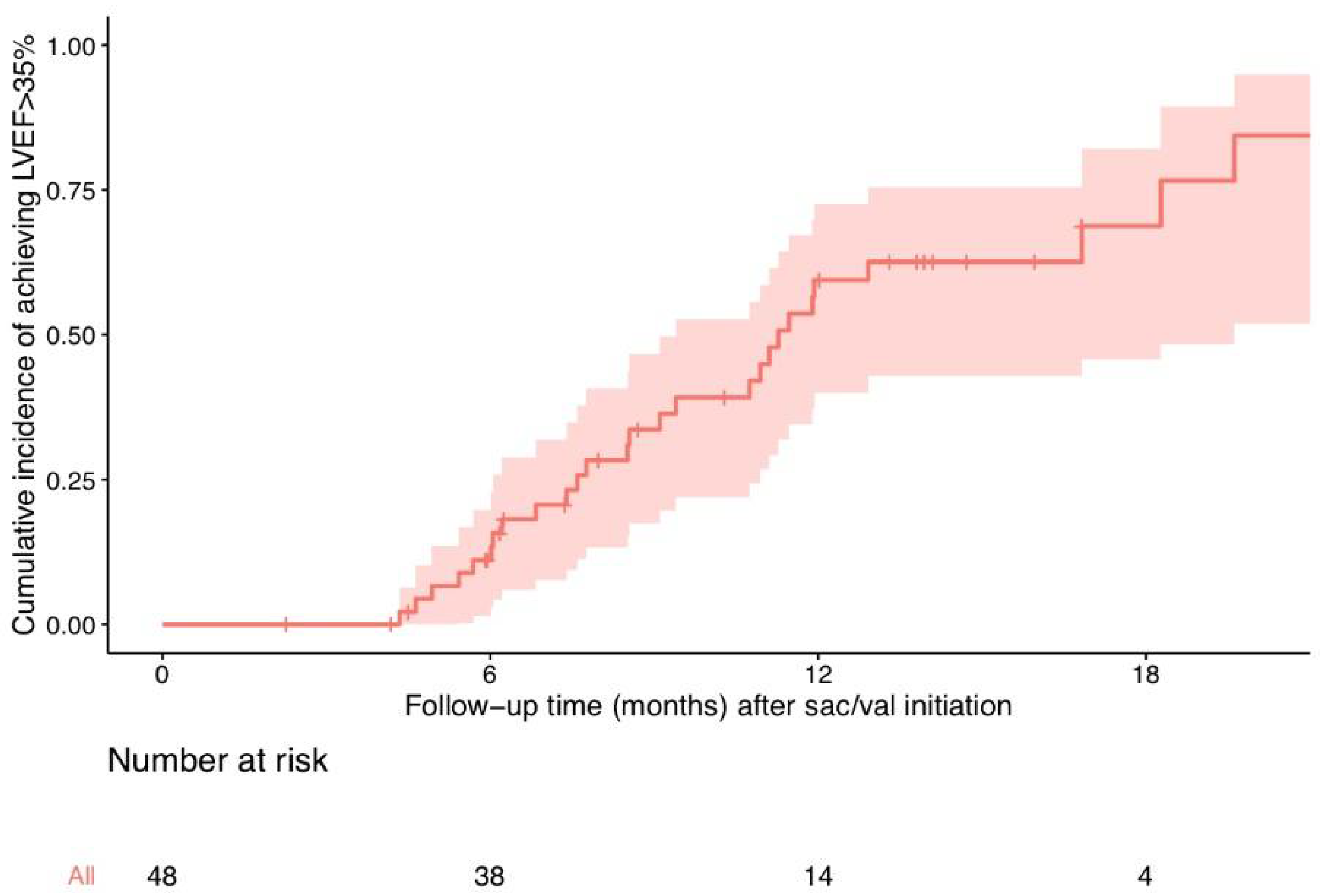
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- McDonagh, T.A.; Metra, M.; Adamo, M.; Gardner, R.S.; Baumbach, A.; Bo¨hm, M.; Burri, H.; Butler, J.; Celutkien, J.; Chioncel, O.; et al. 2021 European Society of Cardiology (ESC) Guidelines for the diagnosis and treatment of acute and chronic heart failure. Eur. Heart J. 2021, 42, 3599–3726. [Google Scholar] [CrossRef] [PubMed]
- Yancy, C.W.; Jessup, M.; Bozkurt, B.; Butler, J.; Casey, D., Jr.; Colvin, M.; Drazner, M.; Filippatos, G.; Fonarow, G.; Givertz, M.; et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. J. Card. Fail. 2017, 23, 628–651. [Google Scholar] [PubMed]
- Ezzat, V.A.; Lee, V.; Ahsan, S.; Chow, A.W.; Segal, O.; Rowland, E.; Lowe, M.D.; Lambiase, P.D. A systematic review of ICD complications in randomised controlled trials versus registries: Is our ‘real-world’ data an underestimation? Open Heart 2015, 2, e000198. [Google Scholar] [CrossRef] [PubMed]
- Connolly, S.J.; Hallstrom, A.P.; Cappato, R.; Schron, E.B.; Kuck, K.H.; Zipes, D.P.; Greene, H.L.; Boczor, S.; Domanski, M.; Follmann, D.; et al. Meta-analysis of the implantable cardioverter defibrillator secondary prevention trials. AVID, CASH and CIDS studies. Antiarrhythmics vs Implantable Defibrillator study. Cardiac Arrest Study Hamburg. Canadian Implantable Defibrillator Study. Eur. Heart J. 2000, 21, 2071–2078. [Google Scholar] [CrossRef]
- Sauer, A.J.; Cole, R.; Jensen, B.C.; Pal, J.; Sharma, N.; Yehya, A.; Vader, J. Practical guidance on the use of sacubitril/valsartan for heart failure. Heart Fail. Rev. 2019, 24, 167–176. [Google Scholar] [CrossRef] [PubMed]
- McMurray, J.; Packer, M.; Desai, A.S.; Gong, J.; Lefkowitz, M.P.; Rizkala, A.R.; Rouleau, J.L.; Shi, V.; Solomon, S.; Swedberg, K.; et al. Angiotensin-neprilysin inhibition versus enalapril in heart failure. N. Engl. J. Med. 2014, 371, 993–1004. [Google Scholar] [CrossRef] [PubMed]
- Januzzi, J., Jr.; Prescott, M.F.; Butler, J.; Felker, G.M.; Maisel, A.S.; McCague, K.; Camacho, A.; Piña, I.L.; Rocha, R.A.; Shah, A.M.; et al. Association of Change in N-Terminal Pro–B-Type Natriuretic Peptide Following Initiation of Sacubitril-Valsartan Treatment With Cardiac Structure and Function in Patients With Heart Failure With Reduced Ejection Fraction. JAMA 2019, 322, 1085–1095. [Google Scholar] [CrossRef] [PubMed]
- Severini, D.; Mboumi, K. Sacubitril/valsartan treatment improved the clinical outcome and reduced the hospitalization rate in three patients with chronic heart failure: A case series. Curr. Med. Res. Opin. 2019, 35 (Suppl. 3), 7–11. [Google Scholar] [CrossRef] [PubMed]
- Monzo, L.; Lanzillo, C.; Tota, C.; Lino, S.; Fusco, A.; Minati, M.; Martino, A.; Calò, L. Sacubitril/valsartan effect on left ventricular remodeling: The case of a super-responder. Curr. Med. Res. Opin. 2019, 35 (Suppl. 3), 3–6. [Google Scholar] [CrossRef] [PubMed]
- Díez-Villanueva, P.; Vicent, L.; de la Cuerda, F.; Esteban-Fernández, A.; Gómez-Bueno, M.; Juan-Bagudá, J.; Iniesta, A.M.; Ayesta, A.; Rojas-González, A.; Bover-Freire, R.; et al. Left Ventricular Ejection Fraction Recovery in Patients with Heart Failure and Reduced Ejection Fraction Treated with Sacubitril/Valsartan. Cardiology 2020, 145, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Almufleh, A.; Marbach, J.; Chih, S.; Stadnick, E.; Davies, R.; Liu, P.; Mielniczuk, L. Ejection fraction improvement and reverse remodeling achieved with Sacubitril/Valsartan in heart failure with reduced ejection fraction patients. Am. J. Cardiovasc. Dis. 2017, 7, 108–113. [Google Scholar] [PubMed]
- Martens, P.; Beliën, H.; Dupont, M.; Vandervoort, P.; Mullens, W. The reverse remodeling response to sacubitril/valsartan therapy in heart failure with reduced ejection fraction. Cardiovasc. Ther. 2018, 36, e12435. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, M.; Ferreira, F.; Raposo, A.; Mónica, L.; Cruz, L.; Guimarães, M.; Fandinga, L.; Matias, C.; Proença, G. Impact of a Heart Failure Clinic on Morbidity, Mortality and Quality of Life. Eur. J. Heart Fail. 2020, 22 (Suppl. S1), 71. [Google Scholar]
- R Core Team. A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2020. [Google Scholar]
- Binkley, P.F.; Lesinski, A.; Ferguson, J.P.; Hatton, P.S.; Yamokoski, L.; Hardikar, S.; Cooke, G.E.; Leier, C.V. Recovery of normal ventricular function in patients with dilated cardiomyopathy: Predictors of an increasingly prevalent clinical event. Am. Heart J. 2008, 155, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Merlo, M.; Pyxaras, S.A.; Pinamonti, B.; Barbati, G.; Lenarda, A.D.; Sinagra, G. Prevalence and prognostic significance of left ventricular reverse remodeling in dilated cardiomyopathy receiving tailored medical treatment. J. Am. Coll. Cardiol. 2011, 57, 1468–1476. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, J.E.; Fonarow, G.C.; Yancy, C.W.; Albert, N.M.; Curtis, A.B.; Heywood, J.T.; Inge, P.J.; McBride, M.L.; Mehra, M.R.; O’Connor, C.M.; et al. Factors associated with improvement in ejection fraction in clinical practice among patients with heart failure: Findings from IMPROVE HF. Am. Heart J. 2012, 163, 49–56.e2. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Patients (n = 48) |
|---|---|
| Age (years), mean (SD #) | 72.5 (9.8) |
| Male sex, n (%) | 34 (70.8) |
| Weight (Kg), mean (SD #) | 76.2 (15.0) |
| BMI *, mean (SD #) | 26.8 (4.8) |
| Systolic blood pressure (mm Hg), mean (SD #) | 128.4 (20.6) |
| Heart rate (bpm), mean (SD #) | 71.9 (15.8) |
| Ischemic etiology, n (%) | 28 (58.3) |
| LVEF $ (%), mean (SD #) | 27.9 (5.6) |
| NT-proBNP + (pg/mL), median (IQR) | 3714 (3637) |
| NYHA § functional class, n (%) | |
| II | 29 (60.4) |
| III | 19 (39.6) |
| Valvular prosthesis | 5 (10.4) |
| Stages of CKD † according to eGFR †, n (%) | |
| Stage 1 (≥90 mL/min/1.73 m2) | 5 (10.4) |
| Stage 2 (≥60 and <90 mL/min/1.73 m2) | 16 (33.3) |
| Stage 3 (≥30 and <60 mL/min/1.73 m2) | 18 (37.5) |
| Stage 4 (≥15 and <30 mL/min/1.73 m2) | 3 (6.3) |
| Other comorbidities, n (%) | |
| Atrial fibrillation | 23 (47.9) |
| Anemia | 10 (20.8) |
| COPD ‡ | 5 (10.4) |
| Diabetes mellitus | 16 (33.3) |
| Dyslipidemia | 24 (50.0) |
| Hypertension | 34 (70.8) |
| Treatment at baseline, n (%) | |
| ACEi ¶ | 39 (81.3) |
| ARB ¶ | 5 (10.4) |
| Beta-blockers | 46 (95.8) |
| Digitalis glycosides | 1 (2.1) |
| Ivabradine | 8 (16.7) |
| Loop diuretics | 39 (81.3) |
| MRA ¶ | 26 (54.2) |
| Thiazides | 1 (2.1) |
| Characteristics | Crude Analysis | Adjusted Analysis | ||
|---|---|---|---|---|
| IRR * | 95%CI | IRR * | 95%CI | |
| Sex: female vs. male | 2.4 | 1.1–5.5 | 2.4 | 0.8–8.4 |
| Age: ≥70 years vs. <70 years | 1.0 | 0.4–2.1 | 1.1 | 0.4–3.1 |
| NYHA # class: III vs. II | 0.8 | 0.4–1.7 | 0.8 | 0.3–2.2 |
| Ischemic etiology: yes vs. no | 0.2 | 0.4–1.8 | 0.8 | 0.3–2.2 |
| CKD †: yes vs. no | 0.7 | 0.3–1.6 | 0.5 | 0.2–1.4 |
| Diabetes mellitus: yes vs. no | 0.8 | 0.3–1.8 | 0.9 | 0.3–2.7 |
| LVEF §: 30–35% vs. 15–30% | 4.5 | 1.8–11.1 | 3.7 | 1.4–10.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nogueira, M.A.; Brochado, M.; Nabais, I.; Batista, É.; Matias, C.; Proença, G. Is Sacubitril/Valsartan Able to Change the Timing for Implantation of Cardiac Devices in Heart Failure with Reduced Ejection Fraction? Hearts 2022, 3, 88-95. https://doi.org/10.3390/hearts3030012
Nogueira MA, Brochado M, Nabais I, Batista É, Matias C, Proença G. Is Sacubitril/Valsartan Able to Change the Timing for Implantation of Cardiac Devices in Heart Failure with Reduced Ejection Fraction? Hearts. 2022; 3(3):88-95. https://doi.org/10.3390/hearts3030012
Chicago/Turabian StyleNogueira, Marta Afonso, Marisa Brochado, Inês Nabais, Élia Batista, Carla Matias, and Gonçalo Proença. 2022. "Is Sacubitril/Valsartan Able to Change the Timing for Implantation of Cardiac Devices in Heart Failure with Reduced Ejection Fraction?" Hearts 3, no. 3: 88-95. https://doi.org/10.3390/hearts3030012
APA StyleNogueira, M. A., Brochado, M., Nabais, I., Batista, É., Matias, C., & Proença, G. (2022). Is Sacubitril/Valsartan Able to Change the Timing for Implantation of Cardiac Devices in Heart Failure with Reduced Ejection Fraction? Hearts, 3(3), 88-95. https://doi.org/10.3390/hearts3030012







