Atopic Status in Children with Asthma and Respiratory Allergies—Comparative Analysis of Total IgE, ImmunoCAP Phadiatop/fx5 and Euroimmun Pediatric Immunoblot
Abstract
:1. Introduction
2. Objectives
3. Materials and Methods
3.1. Subjects
3.2. Study Design
3.3. Laboratory Immunological Methods
- o
- Determination of serum levels of total IgE by ELISA (enzyme-linked immunosorbent assay), EUROIMMUN Medizinische Labordiagnostica AG
- o
- Euroimmun EUROLINE Pediatric (complex of the most common food and aero-allergens in childhood).
- o
- Specific IgE—multi-screening atopy test—ImmunoCAP, Phadia (Thermo Fischer Scientific Inc, Phadia® AB, Uppsala, Sweden), Phadiatop (aeroallergen complex), and fx5 (MultiCAP food mix)
3.4. Statistical Methods
4. Results
4.1. Demographic Characteristics
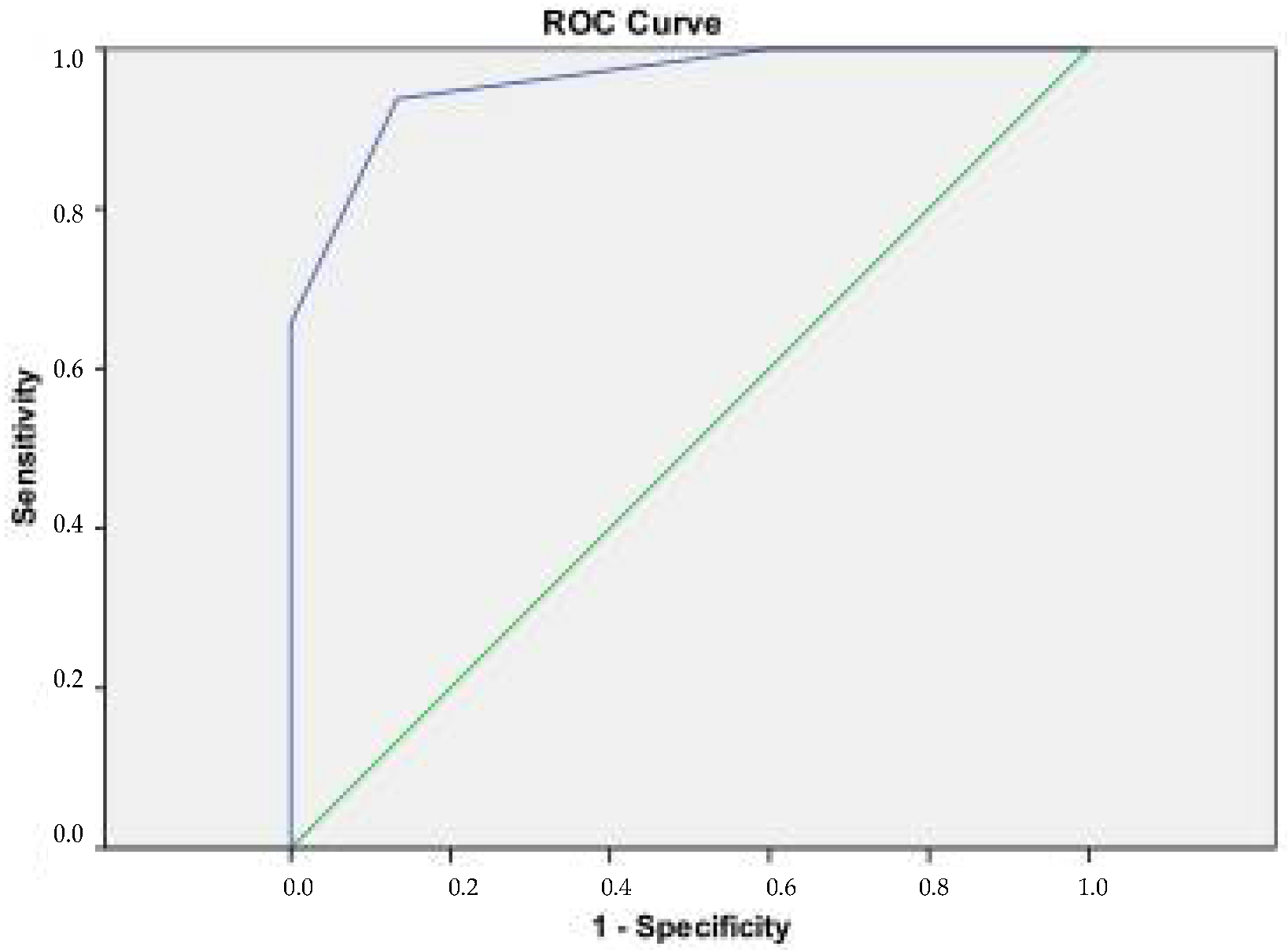
4.2. Total Serum IgE Determination (ELISA)
- Comparative analysis of serum total IgE and sIgE, assessed by two methods (ImmunoCAP Phadiatop/fx5 and Euroimmun pediatric immunoblot)
- Comparative analysis of serum sIgE against aero-allergens, assessed by two methods (ImmunoCAP Phadiatop and Euroimmun aero)
- Comparative analysis of specific IgE against food allergens, assessed by two methods (ImmunoCAP fx5 and Euroimmun food)
- Clinical significance of medical history, total IgE and sIgE, assessed by two methods
5. Discussion
6. Limitation of the Study
7. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pijnenburg, M.W.; Baraldi, E.; Brand, P.L.; Carlsen, K.-H.; Eber, E.; Frischer, T.; Hedlin, G.; Kulkarni, N.; Lex, C.; Mäkelä, M.J.; et al. Monitoring Asthma in Children. Eur. Respir. J. 2015, 25, 178–186. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nelson, H.S.; Szefler, S.J.; Jacobs, J.; Huss, K.; Shapiro, G.; Sternberg, A.L. The relationships among environmental allergen sensitization, allergen exposure, pulmonary function, and bronchial hyperresponsiveness in the Childhood Asthma Management Program. J. Allergy Clin. Immunol. 1999, 104, 775–785. [Google Scholar] [CrossRef]
- Justiz Vaillant, A.A.; Modi, P.; Jan, A. StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK542187/ (accessed on 20 December 2021).
- Thomsen, S.F. Epidemiology and natural history of atopic diseases. Eur. Clin. Respir. J. 2015, 2, 24642. [Google Scholar] [CrossRef] [Green Version]
- Ahmed, H.; Ospina, M.B.; Sideri, K.; Vliagoftis, H. Retrospective analysis of aeroallergen’s sensitization patterns in Edmonton, Canada. Allergy Asthma Clin. Immunol. 2019, 15, 6. [Google Scholar] [CrossRef]
- Ansotegui, I.J.; Melioli, G.; Canonica, G.W.; Caraballo, L.; Villa, E.; Ebisawa, M.; Passalacqua, G.; Savi, E.; Ebo, D.; Gómez, R.M.; et al. IgE allergy diagnostics and other relevant tests in allergy, a World Allergy Organization position paper. World Allergy Organ. J. 2020, 13, 100080, Erratum in 2021, 14, 100557. [Google Scholar] [CrossRef]
- Szefler, S.J.; Wenzel, S.; Brown, R.; Erzurum, S.C.; Fahy, J.V.; Hamilton, R.G.; Hunt, J.F.; Kita, H.; Liu, A.H.; Panettieri, R.A.; et al. Asthma outcomes: Biomarkers. J. Allergy Clin. Immunol. 2012, 129, S9–S23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sly, P.; Boner, A.L.; Björksten, B.; Bush, A.; Custovic, A.; Eigenmann, P.; Gern, J.E.; Gerritsen, J.; Hamelmann, E.; Helms, P.J.; et al. Early identification of atopy in the prediction of persistent asthma in children. Lancet 2008, 372, 1100–1106. [Google Scholar] [CrossRef] [Green Version]
- Garcia, G.V.; Blake, K. Considerations for the Child with Nonatopic Asthma. Pediatr. Allergy Immunol. Pulmonol. 2020, 33, 39–42. [Google Scholar] [CrossRef] [PubMed]
- Holguin, F. The atopic march: IgE is not the only road. Lancet Respir. Med. 2014, 2, 88–90. [Google Scholar] [CrossRef]
- Sinisgalli, S.; Collins, M.S.; Schramm, C.M. Clinical Features Cannot Distinguish Allergic from Non-allergic Asthma in Children. J. Asthma 2011, 49, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-García, S.; Habernau Mena, A.; Quirce, S. Biomarkers in inflammometry pediatric asthma: Utility in daily clinical practice. Eur. Clin. Respir. J. 2017, 4, 1356160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, D.; Yao, W.; Tiller, C.J.; Kisling, J.; Slaven, J.E.; Yu, Z.; Kaplan, M.H.; Tepper, R.S. Exhaled nitric oxide during infancy as a risk factor for asthma and airway hyperreactivity. Eur. Respir. J. 2015, 45, 98–106. [Google Scholar] [CrossRef] [Green Version]
- Rø, A.D.; Simpson, M.R.; Storrø, O.; Johnsen, R.; Videm, V.; Øien, T. The predictive value of allergen skin prick tests and IgE tests at pre-school age: The PACT study. Pediatr. Allergy Immunol. 2014, 25, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Moeller, A.; Carlsen, K.H.; Sly, P.D.; Baraldi, E.; Piacentini, G.; Pavord, I.; Lex, C.; Saglani, S.; ERS Task Force Monitoring Asthma in Children. Monitoring asthma in childhood: Lung function, bronchial responsiveness and inflammation. Eur. Respir. Rev. 2015, 136, 204–215. [Google Scholar] [CrossRef] [Green Version]
- Fasce, L.; Tosca, M.A.; Olcese, R.; Ciprandi, G.; Baroffio, M. Atopy in wheezing infants always starts with monosensitization. Allergy Asthma Proc. 2007, 28, 449–453. [Google Scholar] [CrossRef] [PubMed]
- Migueres, M.; Dávila, I.; Frati, F.; Azpeitia, A.; Jeanpetit, Y.; Lhéritier-Barrand, M.; Incorvaia, C.; Ciprandi, G.; PlurAL Study Group. Types of sensitization to aeroallergens: Definitions, prevalences and impact on the diagnosis and treatment of allergic respiratory disease. Clin. Transl. Allergy 2014, 4, 16. [Google Scholar] [CrossRef] [Green Version]
- Burrows, B.; Martinez, F.D.; Cline, M.G.; Lebowitz, M.D. The relationship between parental and children’s serum IgE and asthma. Am. J. Respir. Crit. Care Med. 1995, 152, 1497–1500. [Google Scholar] [CrossRef]
- Hamilton, R.G. Microarray Technology Applied to Human Allergic Disease. Microarrays 2017, 6, 3. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paganelli, R.; Ansotegui, I.J.; Sastre, J.; Lange, C.-E.; Roovers, M.H.W.M.; de Groot, H.; Lindholm, N.B.; Ewan, P.W. Specific IgE antibodies in the diagnosis of atopic disease: Clinical evaluation of a new in vitro test system, UniCAPTM, in six European allergy clinics. Allergy 2008, 53, 763–768. [Google Scholar] [CrossRef]
- Wever, A.M.J.; Wever-Hess, J.; Van Schayck, C.P.; Van Weel, C. Evaluation of the Phadiatop® test in an epidemiological study. Allergy 1990, 45, 92–97. [Google Scholar] [CrossRef]
- Liu, Y.-H.; Chou, H.-H.; Jan, R.-L.; Lin, H.-J.; Liang, C.-C.; Wang, J.-Y.; Wu, Y.-C.; Shieh, C.-C. Comparison of two specific allergen screening tests in different patient groups. Acta Paediatr. Taiwan 2006, 47, 116–122. [Google Scholar]
- Khasawneh, R.; Hiary, M.; Abadi, B.; Salameh, A.; Moman, S. Total and Specific Immunoglobulin E for Detection of Most Prevalent Aeroallergens in a Jordanian Cohort. Med. Arch. 2019, 73, 272–275. [Google Scholar] [CrossRef]
- Wickman, M.; Ahlstedt, S.; Lilja, G.; Hamsten, M.V.H. Quantification of IgE antibodies simplifies the classification of allergic diseases in 4-year-old children. A report from the prospective birth cohort study—BAMSE. Pediatr. Allergy Immunol. 2003, 14, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Zeng, G.; Hu, H.; Zheng, P.; Wu, G.; Wei, N.; Liang, X.; Sun, B.; Zhang, X. The practical benefit of Phadiatop test as the first-line in vitro allergen-specific immunoglobulin E (sIgE) screening of aeroallergens among Chinese asthmatics: A validation study. Ann. Transl. Med. 2018, 6, 151. [Google Scholar] [CrossRef] [PubMed]
- Park, K.H.; Lee, J.; Sim, D.W.; Lee, S.C. Comparison of Singleplex Specific IgE Detection Immunoassays: ImmunoCAP Phadia 250 and Immulite 2000 3gAllergy. Ann. Lab. Med. 2018, 38, 23–31. [Google Scholar] [CrossRef]
- Baldacci, S.; Omenaas, E.; Oryszczyn, M. Allergy markers in respiratory epidemiology. Eur. Respir. J. 2001, 17, 773–790. [Google Scholar] [CrossRef] [Green Version]
- Hamilton, R.G.; Adkinson, N.F. In vitro assays for the diagnosis of IgE-mediated disorders. J. Allergy Clin. Immunol. 2004, 114, 213–225. [Google Scholar] [CrossRef]
- Frith, J.; Fleming, L.; Bossley, C.; Ullmann, N.; Bush, A. The complexities of defining atopy in severe childhood asthma. Clin. Exp. Allergy 2011, 41, 948–953. [Google Scholar] [CrossRef]
- Hamilton, R.G.; Matsson, P.N.; Hovanec-Burns, D.L.; Van Cleve, M.; Chan, S.; Kober, A.; Kleine-Tebbe, J.R.; Renz, H.; Magnusson, C.; Quicho, R.; et al. Analytical Performance Characteristics, Quality Assurance and Clinical Utility of Immunological Assays for Human IgE Antibodies of Defined Allergen Specificities. (CLSI-ILA20-A3). J. Allergy Clin. Immunol. 2015, 135, AB8. [Google Scholar] [CrossRef]
- Ferraro, V.; Zanconato, S.; Carraro, S. Timing of Food Introduction and the Risk of Food Allergy. Nutrients 2019, 11, 1131. [Google Scholar] [CrossRef] [Green Version]
- Dhami, S.; Sheikh, A. Estimating the prevalence of aero-allergy and/or food allergy in infants, children and young people with moderate-to-severe atopic eczema/dermatitis in primary care: Multi-centre, cross-sectional study. J. R. Soc. Med. 2015, 108, 229–236. [Google Scholar] [CrossRef] [Green Version]
- Chang, Y.-C.; Lee, T.-J.; Huang, C.-C.; Chang, P.-H.; Chen, Y.-W.; Fu, C.-H. The Role of Phadiatop Tests and Total Immunoglobulin E Levels in Screening Aeroallergens: A Hospital-Based Cohort Study. J. Asthma Allergy 2021, 14, 135–140. [Google Scholar] [CrossRef]
- Pierotti, F.F.; Aranda, C.S.; Cocco, R.R.; Sarinho, E.; Sano, F.; Porto, A.; Rosário, N.; Neto, H.J.C.; Goudouris, E.; Moraes, L.S.; et al. Phadiatop, Phadiatop Infant and total IgE evaluated in allergic Brazilian children and adolescents. Allergol. Immunopathol. 2020, 48, 259–264. [Google Scholar] [CrossRef]
- EUROLINE Pediatric (IgE) Test Instruction. Available online: http://shop.tinyteria.com/index.php?route=extension/module/free_downloads/download&did=1419 (accessed on 20 December 2021).
- Singh, S. Asthma diagnosis and treatment—1001. Identification of prevalent sensitizing allergens in India. World Allergy Organ. J. 2013, 6, P1. [Google Scholar] [CrossRef] [Green Version]
- Thermo. Available online: https://www.thermofisher.com/phadia/wo/en/our-solutions/immunocap-allergy-solutions/specific-ige-single-allergens/atopy.html (accessed on 20 December 2021).
- Skevaki, C.; Tafo, P.; Eiringhaus, K.; Timmesfeld, N.; Weckmann, M.; Happle, C.; Nelson, P.P.; Maison, N.; Schaub, B.; Ricklefs, I.; et al. Allergen extract- and component-based diagnostics in children of the ALLIANCE asthma cohort. Clin. Exp. Allergy 2021, 51, 1331–1345. [Google Scholar] [CrossRef]






| Аccording to the Medical History | Girls N 28 | Boys N 58 | Total N 86 |
|---|---|---|---|
| Prior allergy symptoms | 14 (50.0%) | 28 (48.2%) | 42 (48.8%) |
| Concomitant AR | 13 (46.4%) | 34 (58,6%) | 47 (54.7%) |
| Tested Allergen | AUC | SE | р | 95% CI | |
|---|---|---|---|---|---|
| Lower Limit | Upper Limit | ||||
| f2,f76,f77,f78 | 0.620 | 0.095 | 0.196 | 0.434 | 0.805 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lazova, S.; Baleva, M.; Priftis, S.; Naseva, E.; Velikova, T. Atopic Status in Children with Asthma and Respiratory Allergies—Comparative Analysis of Total IgE, ImmunoCAP Phadiatop/fx5 and Euroimmun Pediatric Immunoblot. Sinusitis 2022, 6, 1-14. https://doi.org/10.3390/sinusitis6010001
Lazova S, Baleva M, Priftis S, Naseva E, Velikova T. Atopic Status in Children with Asthma and Respiratory Allergies—Comparative Analysis of Total IgE, ImmunoCAP Phadiatop/fx5 and Euroimmun Pediatric Immunoblot. Sinusitis. 2022; 6(1):1-14. https://doi.org/10.3390/sinusitis6010001
Chicago/Turabian StyleLazova, Snezhina, Marta Baleva, Stamatios Priftis, Emilia Naseva, and Tsvetelina Velikova. 2022. "Atopic Status in Children with Asthma and Respiratory Allergies—Comparative Analysis of Total IgE, ImmunoCAP Phadiatop/fx5 and Euroimmun Pediatric Immunoblot" Sinusitis 6, no. 1: 1-14. https://doi.org/10.3390/sinusitis6010001
APA StyleLazova, S., Baleva, M., Priftis, S., Naseva, E., & Velikova, T. (2022). Atopic Status in Children with Asthma and Respiratory Allergies—Comparative Analysis of Total IgE, ImmunoCAP Phadiatop/fx5 and Euroimmun Pediatric Immunoblot. Sinusitis, 6(1), 1-14. https://doi.org/10.3390/sinusitis6010001








