Dynamics of Oxygen and the Physicochemical Response in Two Comparative Hypoxia Regions
Abstract
1. Introduction
2. Materials and Methods
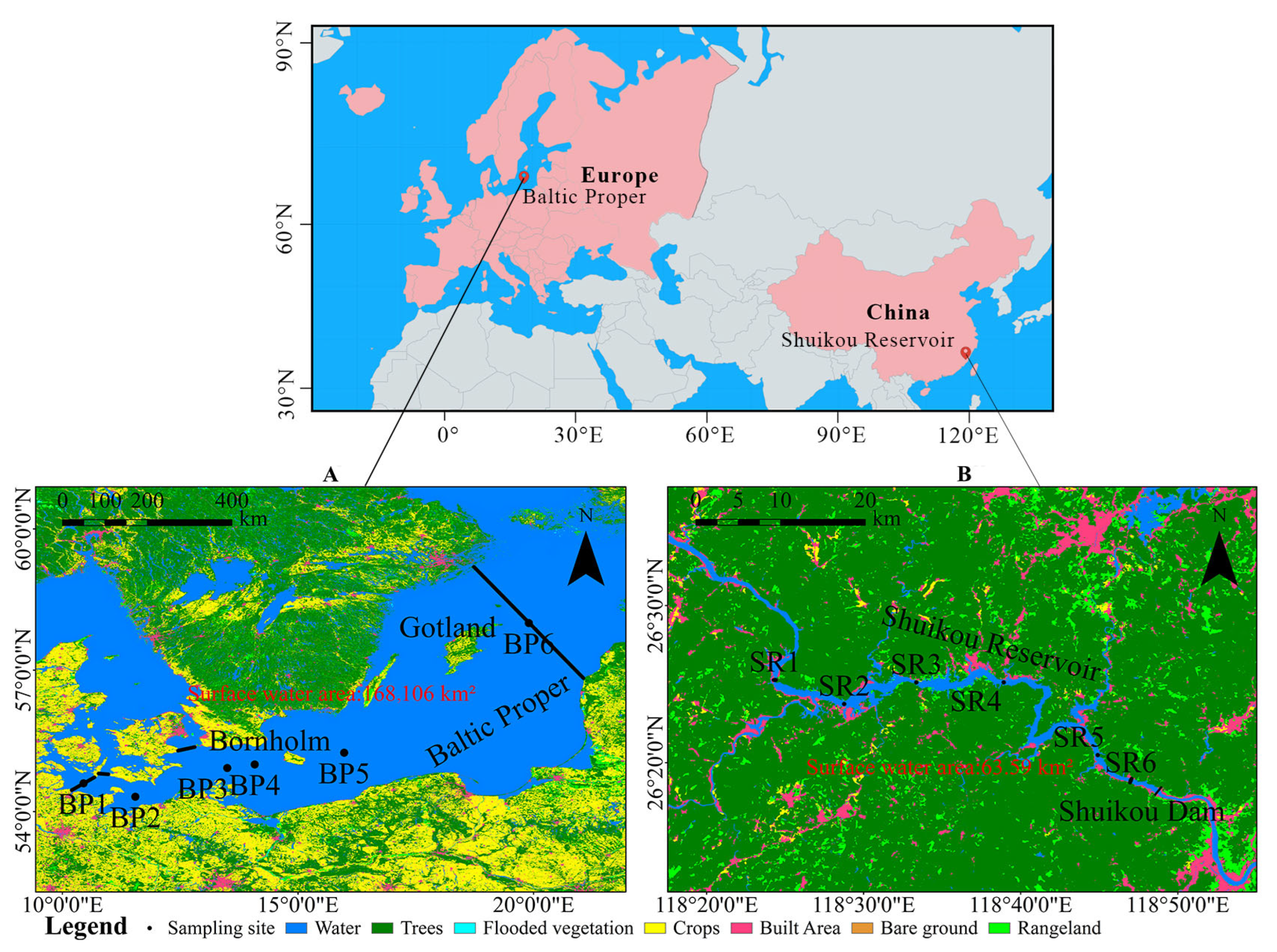
2.1. Study Sites
2.2. Field Surveys
2.3. Hydrological Data Collection
2.4. Data Analysis
2.5. Statistical Analyses and Graphics
3. Results
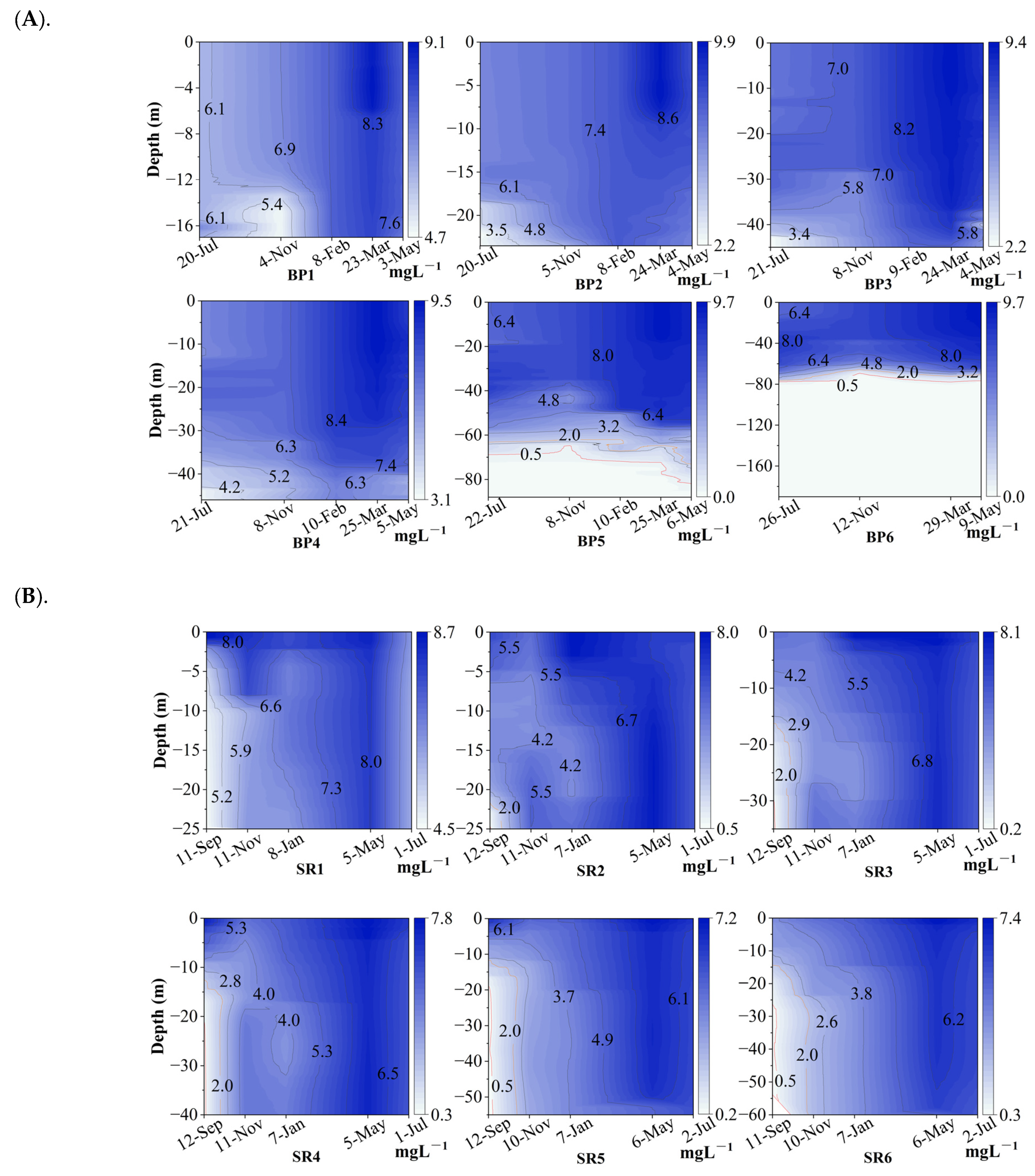
3.1. Changes of DO and the Dynamics of Hypoxia
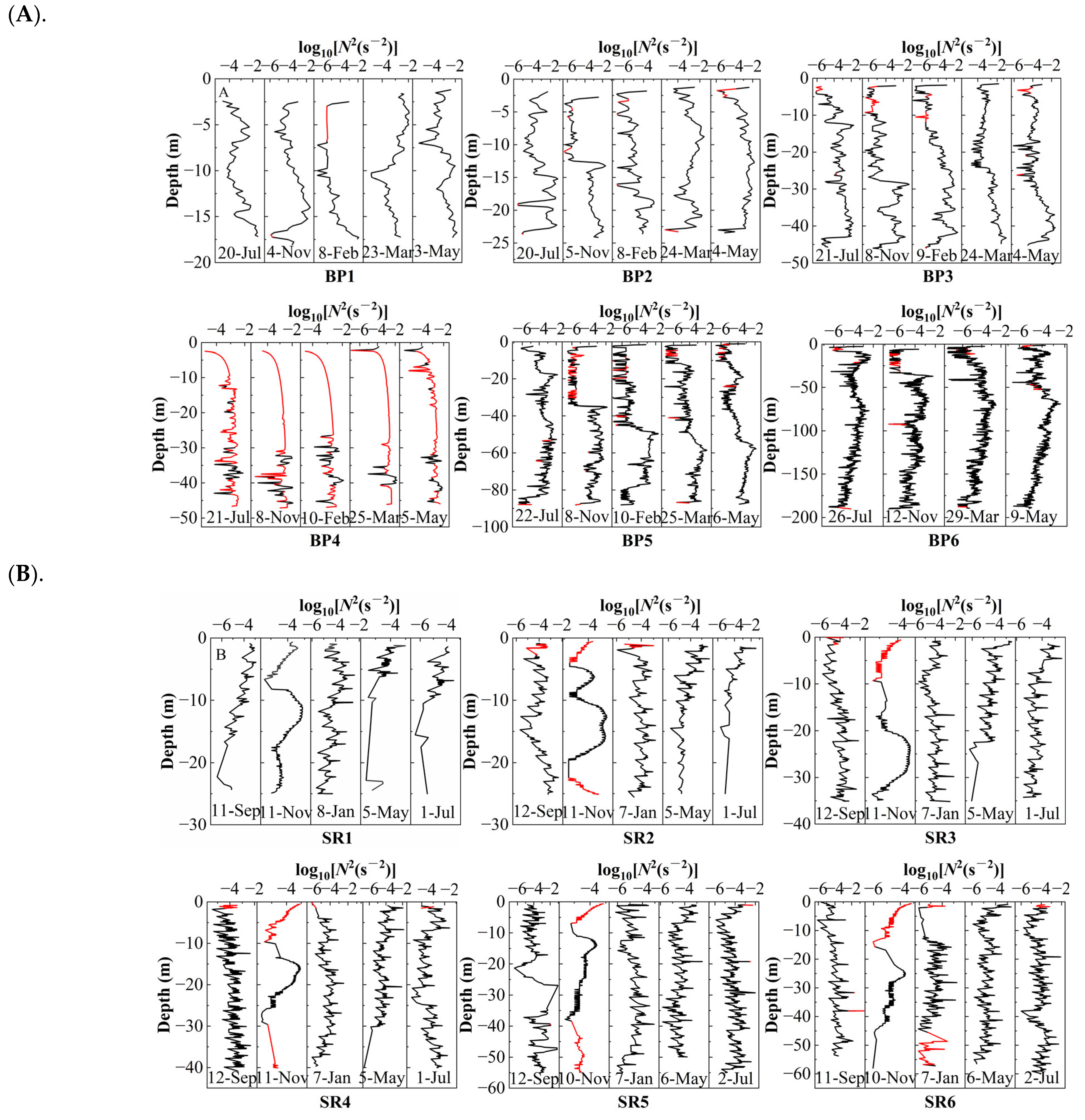
3.2. Contribution of Stratification to the Dynamics of Oxygen
3.3. Changes in Water Quality Parameters
3.3.1. Water Quality Parameters
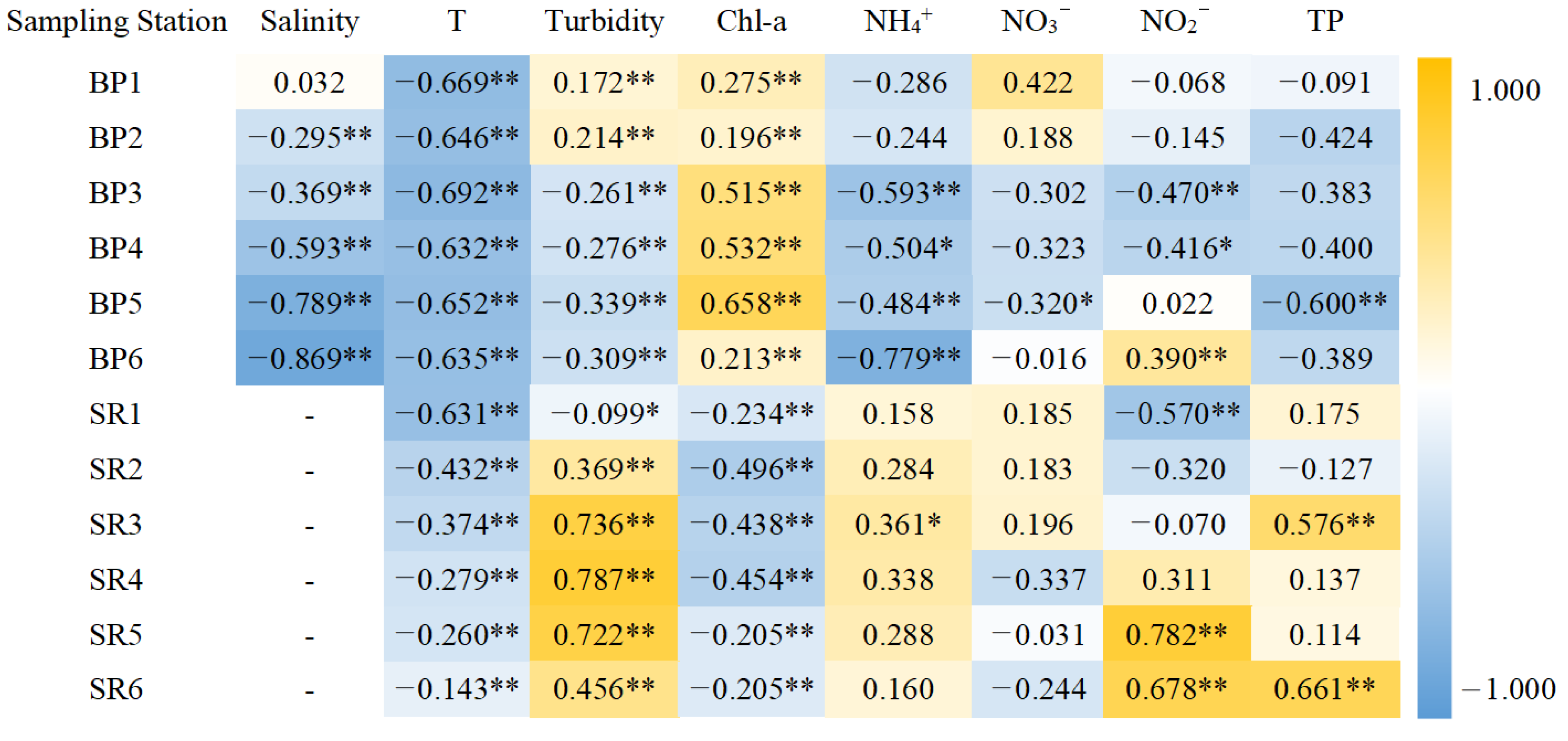
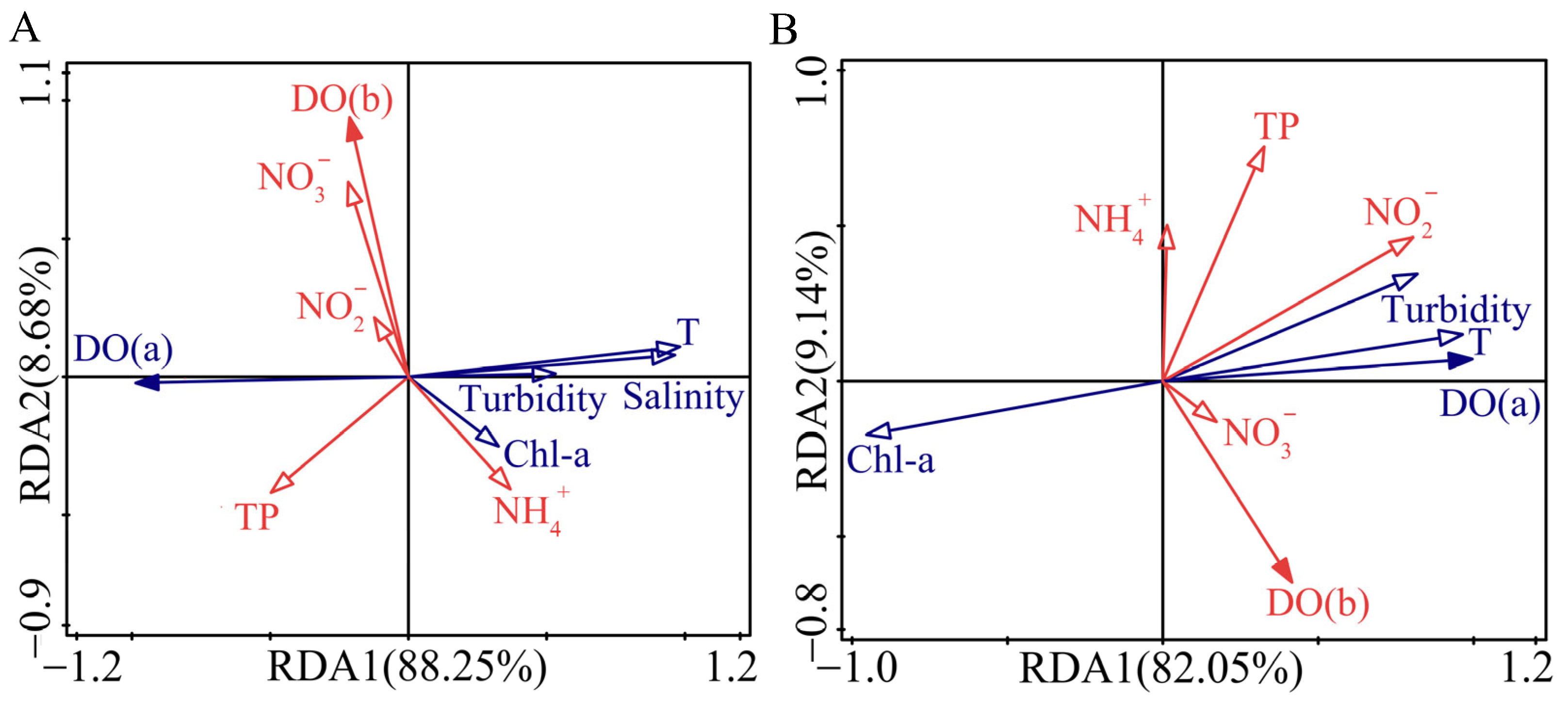
3.3.2. Contribution of Water Quality Parameters to DO
4. Discussion
4.1. Contributing Factors to Seasonal DO Changes
4.2. Contributing Factors for Hypoxia
4.3. Hypoxia Management, Limitations, and Future Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Levin, L.A.; Ekau, W.; Gooday, A.J.; Jorissen, F.; Middelburg, J.J.; Naqvi, S.W.A.; Neira1, C.; Rabalais, N.N.; Zhang, J. Effects of natural and human-induced hypoxia on coastal benthos. Biogeosciences 2009, 6, 2063–2098. [Google Scholar] [CrossRef]
- Rabalais, N.N.; Turner, R.E.; Wiseman, W.J., Jr. Gulf of Mexico hypoxia, a.k.a. “the dead zone”. Annu. Rev. Ecol. Syst. 2002, 33, 235–263. [Google Scholar] [CrossRef]
- Watson, A.J. Oceans on the edge of anoxia. Science 2016, 354, 1529–1530. [Google Scholar] [CrossRef]
- Deeds, J.; Amirbahman, A.; Norton, S.A.; Suitor, D.G.; Bacon, L.C. Predicting anoxia in low-nutrient temperate lakes. Ecol. Appl. 2021, 31, e2361. [Google Scholar] [CrossRef]
- Wei, Q.; Xue, L.; Yao, Q.; Wang, B.; Yu, Z. Oxygen decline in a temperate marginal sea: Contribution of warming and eutrophication. Sci. Total Environ. 2021, 757, 143227. [Google Scholar] [CrossRef] [PubMed]
- Schmidtko, S.; Stramma, L.; Visbeck, M. Decline in global oceanic oxygen content during the past five decades. Nature 2017, 542, 335–339. [Google Scholar] [CrossRef] [PubMed]
- Diaz, R.J.; Rosenberg, R. Marine benthic hypoxia: A review of its ecological effects and the behavioural responses of benthic macrofauna. Oceanogr. Mar. Biol. 1995, 33, 245–303. [Google Scholar]
- Gray, J.S.; Wu, R.S.S.; Or, Y.Y. Effects of hypoxia and organic enrichment on the coastal marine environment. Mar. Ecol. Prog. Ser. 2002, 238, 249–279. [Google Scholar] [CrossRef]
- Vaquer-Suner, R.; Duarte, C.M. Temperature effects on thresholds of hypoxia for marine benthic organisms. Glob. Change Biol. 2011, 17, 1788–1797. [Google Scholar] [CrossRef]
- Lukas, J.; Auer, F.; Goldhammer, T.; Krause, J.; Romanczuk, P.; Klamser, P.P.; Arias-Rodriguez, L.; Bierbach, D. Diurnal changes in hypoxia shape predator-prey interaction in a bird-fish system. Front. Ecol. Evol. 2021, 9, 619193. [Google Scholar] [CrossRef]
- Zillén, L.; Conley, D.J.; Andrén, T.; Andrén, E.; Björck, S. Past occurrences of hypoxia in the Baltic Sea and the role of climate variability, environmental change and human impact. Earth-Sci. Rev. 2008, 91, 77–92. [Google Scholar] [CrossRef]
- Diaz, R.J.; Rosenberg, R. Spreading dead zones and consequences for marine ecosystems. Science 2008, 321, 926–929. [Google Scholar] [CrossRef]
- Rixen, T.; Cowie, G.; Gaye, B.; Goes, J.; do Rosário Gomes, H.; Hood, R.R.; Lachkar, Z.; Schmidt, H.; Segschneider, J.; Singh, A. Reviews and syntheses: Present, past, and future of the oxygen minimum zone in the northern Indian Ocean. Biogeosciences 2020, 17, 6051–6080. [Google Scholar] [CrossRef]
- Zhang, Y.; Fu, H.; Liao, H.; Chen, H.; Liu, Z. Geochemical records of Lake Erhai (South-Western China) reveal the anthropogenically-induced intensification of hypolimnetic anoxia in monomictic lakes. Environ. Pollut. 2022, 299, 118909. [Google Scholar] [CrossRef]
- Diaz, R.J. Overview of Hypoxia around the World. J. Environ. Qual. 2001, 30, 275–281. [Google Scholar] [CrossRef] [PubMed]
- Glenn, S.; Arnone, R.; Bergmann, T.; Bissett, W.P.; Crowley, M.; Cullen, J.; Gryzmski, J.; Haidvogel, D.; Kohut, J.; Moline, M.; et al. Biogeochemical impact of summertime coastal upwelling on the New Jersey Shelf. J. Geophys. Res.-Ocean. 2004, 109, C12S02. [Google Scholar] [CrossRef]
- Turner, R.E.; Rabalais, N.N.; Swenson, E.M.; Kasprzak, M.; Romaire, T. Summer hypoxia in the northern Gulf of Mexico and its prediction from 1978 to 1995. Mar. Environ. Res. 2005, 59, 65–77. [Google Scholar] [CrossRef]
- Kasai, A.; Yamada, T.; Takeda, H. Flow structure and hypoxia in Hiuchi-nada, Seto Inland Sea, Japan. Estuar. Coast. Shelf Sci. 2007, 71, 210–217. [Google Scholar] [CrossRef]
- Liu, J.; Zang, J.; Ran, X.; Zhao, C.; Hua, J.; Jing, X. Sedimentary phosphorus speciation in the coastal hypoxic area of Changjiang estuary and its environmental significance. Huan Jing Ke Xue 2017, 38, 3243–3253. (In Chinese) [Google Scholar] [PubMed]
- Richirt, J.; Riedel, B.; Mouret, A.; Schweizer, M.; Langlet, D.; Seitaj, D.; Meysman, F.J.R.; Slomp, C.P.; Jorissen, F.J. Foraminiferal community response to seasonal anoxia in Lake Grevelingen (the Netherlands). Biogeosciences 2020, 17, 1415–1435. [Google Scholar] [CrossRef]
- Breitburg, D.; Levin, L.A.; Oschlies, A.; Gregoire, M.; Chavez, F.P.; Conley, D.J.; Garcon, V.; Gilbert, D.; Gutierrez, D.; Isensee, K.; et al. Declining oxygen in the global ocean and coastal waters. Science 2018, 359, 46. [Google Scholar] [CrossRef]
- Canadell, J.G.; Monteiro, P.M.S.; Costa, M.H.; Cunha, L.C.; Cox, P.M.; Eliseev, A.V.; Henson, S.; Ishii, M.; Jaccard, S.; Koven, C.; et al. Global Carbon and other Biogeochemical Cycles and Feedbacks. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK; New York, NY, USA, 2021; pp. 673–816. [Google Scholar]
- Rabalais, N.N.; Turner, R.E.; Sen Gupta, B.K.; Boesch, D.F.; Chapman, P.; Murrell, M.C. Characterization and long-term trends of hypoxia in the northern Gulf of Mexico: Does the science support the action plan? Estuaries Coasts 2007, 30, 753–772. [Google Scholar] [CrossRef]
- Fennel, K.; Testa, J.M. Biogeochemical controls on coastal hypoxia. Annu. Rev. Mar. Sci. 2019, 11, 105–130. [Google Scholar] [CrossRef]
- Feely, R.A.; Okazaki, R.R.; Cai, W.J.; Bednarsek, N.; Alin, S.R.; Byrne, R.H.; Fassbender, A. The combined effects of acidification and hypoxia on pH and aragonite saturation in the coastal waters of the California current ecosystem and the northern Gulf of Mexico. Cont. Shelf Res. 2018, 152, 50–60. [Google Scholar] [CrossRef]
- Yang, F.X.; Wang, H.; Bouwman, A.F.; Beusen, A.H.W.; Liu, X.; Wang, J.; Yu, Z.; Yao, Q. Nitrogen from agriculture and temperature as the major drivers of deoxygenation in the central Bohai Sea. Sci. Total Environ. 2023, 893, 164614. [Google Scholar] [CrossRef]
- Lin, J.; Xie, L.; Pietrefesa, L.J.; Xu, H.; Woods, W.; Mallin, M.; Durake, M.J. Water quality responses to simulated flow and nutrient reductions in the Cape Fear estuary and adjacent coastal region, North Carolina. Ecol. Model. 2008, 212, 200–217. [Google Scholar] [CrossRef]
- Testa, J.M.; Kemp, W.M. Oxygen-dynamics and biogeochemical consequences. Treatise Estuar. Coast. Sci. 2011, 5, 163–199. [Google Scholar]
- Zhang, Y.; Wu, Z.; Liu, M.; He, J.; Shi, K.; Zhou, Y.; Wang, M.; Liu, X. Dissolved oxygen stratification and response to thermal structure and long-term climate change in a large and deep subtropical reservoir (Lake Qiandaohu, China). Water Res. 2015, 75, 249–258. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-España, J.; Mata, M.P.; Vegas, J.; Morellón, M.; Rodríguez, J.A.; Salazar, Á.; Yusta, I.; Chaos, A.; Pérez-Martínez, C.; Navas, A. Anthropogenic and climatic factors enhancing hypolimnetic anoxia in a temperate mountain lake. J. Hydrol. 2017, 555, 832–850. [Google Scholar] [CrossRef]
- Du, Y.; Peng, W.; Liu, C. Advances in research on the tempo-spatial characteristics of dissolved oxygen in a stratified reservoir. J. Hydraul. Eng. 2019, 50, 990–998. [Google Scholar]
- Müller, B.; Bryant, L.D.; Matzinger, A.; Wüest, A. Hypolimnetic oxygen depletion in eutrophic lakes. Environ. Sci. Technol. 2012, 46, 9964–9971. [Google Scholar] [CrossRef]
- Mariliis, K.; Ilja, M.; Jüri, E.; Ye, L.; Martin, H.; Lena, V.; Urmas, R. Recent regime of persistent hypoxia in the Baltic Sea. Environ. Res. Commun. 2021, 33, 7. [Google Scholar]
- Yan, J.; Chen, N.; Wang, F.; Liu, Q.; Wu, Z.; Middelburg, J.J.; Xie, Y.; Guo, W.; Zhang, X. Interaction between oxygen consumption and carbon dioxide emission in a subtropical hypoxic reservoir, southeastern China. J. Geophys. Res. Biogeosci. 2021, 126, e2020JG006133. [Google Scholar] [CrossRef]
- Kullenberg, G.; Jacobsen, T.S. The Baltic Sea: An outline of its physical oceanography. Mar. Pollut. Bull. 1981, 12, 183–186. [Google Scholar] [CrossRef]
- Naumov, L.; Neumann, T.; Radtke, H.; Meier, H.E.M. Limited ventilation of the central Baltic Sea due to elevated oxygen consumption. Front. Mar. Sci. 2023, 10, 1175643. [Google Scholar] [CrossRef]
- Heimbrand, Y.; Limburg, K.; Hüssy, K.; Næraa, T.; Casini, M. Cod otoliths document accelerating climate impacts in the Baltic Sea. Sci. Rep. 2024, 14, 16750. [Google Scholar] [CrossRef]
- HELCOM. Development of tools for assessment of eutrophication in the Baltic Sea. In Baltic Sea Environment Proceedings; Erweko Painotuote Oy: Uusimaa, Finland, 2006; No. 104; p. 62. [Google Scholar]
- Ma, X.; Ma, Y.; Qi, J.; Li, J.; Chen, J.; Liu, J.; Chen, L.; Shi, C.; Xie, R. Changes in perturbation-correlation mov-ing-window two-dimensional correlation spectroscopy of dissolved organic matter induced by dam regulation in a river-type reservoir. Ecotoxicol. Environ. Saf. 2025, 300, 118464. [Google Scholar] [CrossRef] [PubMed]
- Grasshoff, K.; Kremling, K.; Ehrhardt, M. Methods of Seawater Analysis; Wiley-VCH Verlag: Weinheim, Germany, 1999; pp. 188–189. [Google Scholar]
- YSI. EXO User Manual Advanced Water Quality Monitoring Platform; YSI: Yellow Springs, OH, USA, 2019. [Google Scholar]
- Xie, R.; Pang, Y.; Luo, B.; Li, J.; Wu, C.; Zheng, Y.; Sun, Q.; Zhang, P.; Wang, F. Spatiotemporal variability in salinity and hydraulic relationship with salt intrusion in the tidal reaches of the Minjiang River, Fujian province, China. Environ. Sci. Pollut. Res. 2017, 24, 11847–11855. [Google Scholar] [CrossRef] [PubMed]
- HJ 535-2009; Water Quality-Determination of Ammonia Nitrogen-Nesslers Reagent Spectrophotometry. China Environmental Science Press: Beijing, China, 2009. Available online: https://www.mee.gov.cn/ywgz/fgbz/bz/bzwb/jcffbz/201001/W020180319535685015821.pdf (accessed on 1 September 2023). (In Chinese)
- HJ/T 346-2007; Water Quality-Determination of Nitrate-Nitrogen-Ultraviolet Spectrophotometry. China Environmental Science Press: Beijing, China, 2007. Available online: https://www.mee.gov.cn/ywgz/fgbz/bz/bzwb/jcffbz/200703/W020220901375489450689.pdf (accessed on 1 September 2023). (In Chinese)
- GB 7493-1987; Water Quality-Determination of Nitrogen (Nitrite)—Spectrophotometric Method. China Environmental Science Press: Beijing, China, 1987. Available online: https://www.mee.gov.cn/image20010518/3467.pdf (accessed on 1 September 2023). (In Chinese)
- GB 11893-1989; Water Quality-Determination of Total Phosphorus-Ammonium Molybdate Spectrophotometric Method. China Environmental Science Press: Beijing, China, 1989. Available online: https://www.mee.gov.cn/image20010518/3655.pdf (accessed on 1 September 2023). (In Chinese)
- Sherstyankin, P.P.; Kuimova, L.N. Vertical stability and the Brunt-Vaisala frequency of deep natural waters by the example of Lake Baikal, Lake Tanganyika, and the World Ocean. Dokl. Earth Sci. 2009, 429, 1553–1558. [Google Scholar] [CrossRef]
- Duan, W.; Cheng, X.; Zhu, X.; Ma, T. Variability in upper-ocean salinity stratification in the tropical Pacific Ocean. Acta Oceanol. Sin. 2021, 40, 113–125. [Google Scholar] [CrossRef]
- Gerkema, T.; Zimmerman, J.T.F. An Introduction to Internal Waves; Royal NIOZ: Texel, The Netherlands, 2008; p. 40. [Google Scholar]
- Kang, X.; Xia, M. Stratification variability in a lagoon system in response to a passing storm. Limnol. Oceanogr. 2022, 67, 511–521. [Google Scholar] [CrossRef]
- Boehrer, B.; Schultze, M. Stratification of lakes. Rev. Geophys. 2008, 46, RG2005. [Google Scholar] [CrossRef]
- Jane, S.F.; Mincer, J.L.; Lau, M.P.; Lewis, A.S.L.; Stetler, J.T.; Rose, K.C. Longer duration of season stratification contributes to widespread increases in lake hypoxia and anoxia. Glob. Change Biol. 2023, 29, 1009–1023. [Google Scholar] [CrossRef] [PubMed]
- He, J.; Shen, H.; Xue, R.; Liu, Y.; Zhang, Q.; Li, J. Study on seasonal low dissolved oxygen driving factors and pollution traceability in surface water of XinghuaLixiahe area in Jiangsu Province. Environ. Monit. China 2025, 41, 83–93. (In Chinese) [Google Scholar] [CrossRef] [PubMed]
- Onabule, O.A.; Mitchell, S.B.; Couceiro, F. The effects of freshwater flow and salinity on turbidity and dissolved oxygen in a shallow Macrotidal estuary: A case study of Portsmouth Harbour. Ocean Coast. Manag. 2020, 191, 105179. [Google Scholar] [CrossRef]
- Zang, C.; Huang, S.; Wu, M.; Du, S.; Scholz, M.; Gao, F.; Lin, C.; Guo, Y.; Dong, Y. Comparison of relationships between pH, dissolved oxygen and chlorophyll a for aquaculture and non-aquaculture waters. Water Air Soil Pollut. 2011, 219, 157–174. [Google Scholar] [CrossRef]
- Sutula, M.; Kudela, R.; Hagy, J.D.; Harding, L.W.; Senn, D.; Cloern, J.E.; Bricker, S.; Berg, G.M.; Beck, M. Novel analyses of long-term data provide a scientific basis for chlorophyll-a thresholds in San Francisco Bay. Estuar. Coast. Shelf Sci. 2017, 197, 107–118. [Google Scholar] [CrossRef]
- Katkaew, N.; Chamchoi, N. Threshold amounts of nutrients and the relationship with chlorophyll a during eutrophication phenomenon in small-scale artificial reservoirs. Environ. Sustain. Indic. 2024, 22, 100378. [Google Scholar] [CrossRef]
- Paerl, H.W.; Havens, K.E.; Xu, H.; Zhu, G.; McCarthy, M.J.; Newell, S.E.; Scott, J.T.; Hall, N.S.; Otten, T.G.; Qin, B. Mitigating eutrophication and toxic cyanobacterial blooms in large lakes: The evolution of a dual nutrient (N and P) reduction paradigm. Hydrobiologia 2020, 847, 4359–4375. [Google Scholar] [CrossRef]
- Paerl, H.W.; Hall, N.S.; Calandrino, E.S. Controlling harmful cyanobacterial blooms in a world experiencing anthropogenic and climatic-induced change. Sci. Total Environ. 2011, 409, 1739–1745. [Google Scholar] [CrossRef]
- Jilbert, T.; Gustafsson, B.G.; Veldhuijzen, S.; Reed, D.C.; van Helmond, N.A.G.M.; Hermans, M.; Slomp, C.P. Iron-phosphorus feedbacks drive multidecadal oscillations in Baltic Sea hypoxia. Geophys. Res. Lett. 2021, 48, e2021GL095908. [Google Scholar] [CrossRef]
- Li, G.; Cheng, L.; Zhu, J.; Trenberth, K.; Mann, M.E.; Abraham, J.P. Increasing ocean stratification over the past half-century. Nat. Clim. Change 2020, 10, 1116–1123. [Google Scholar] [CrossRef]
- Rolff, C.; Walve, J.; Larsson, U.; Elmgren, R. How oxygen deficiency in the Baltic Sea proper has spread and worsened: The role of ammonium and hydrogen sulphide. Ambio 2022, 51, 2308–2324. [Google Scholar] [CrossRef]
- Taavi, L.; Urmas, L. Stratification has strengthened in the Baltic sea—An analysis of 35 years of observational data. Front. Earth Sci. 2019, 7, 174. [Google Scholar]
- Löptien, U.; Renz, M.; Dietze, H. Major Baltic Inflows come in different flavours. Commun. Earth Environ. 2025, 6, 232. [Google Scholar] [CrossRef]
- Ahlgren, J.; Grimvall, A.; Omstedt, A.; Rolff, C.; Wikner, J. Temperature, DOC level and basin interactions explain the declining oxygen concentrations in the Bothnian Sea. J. Mar. Syst. 2017, 170, 22–30. [Google Scholar] [CrossRef]
- Hermans, M.; Lenstra, W.K.; van Helmond, N.A.G.M.; Behrends, T.; Egger, M.; Séguret, M.J.M.; Gustafsson, E.; Gustafsson, B.G.; Slomp, C.P. Impact of natural re-oxygenation on the sediment dynamics of manganese, iron and phosphorus in a euxinic Baltic Sea basin. Geochim. Et Cosmochim. Acta 2019, 246, 174–196. [Google Scholar] [CrossRef]
- Yakushev, E.V.; Pollehne, F.; Jost, G.; Kuznetsov, I.; Schneider, B.; Umlauf, L. Analysis of the water column oxic/anoxic interface in the Black and Baltic seas with a numerical model. Mar. Chem. 2007, 107, 388–410. [Google Scholar] [CrossRef]
- Jane, S.F.; Hansen, G.J.A.; Kraemer, B.M.; Leavitt, P.R.; Mincer, J.L.; North, R.L.; Pilla, R.M.; Stetler, J.T.; Williamson, C.E.; Woolway, R.I.; et al. Widespread deoxygenation of temperate lakes. Nature 2021, 594, 66–70. [Google Scholar] [CrossRef]
- Pilla, R.M.; Williamson, C.E.; Adamovich, B.V.; Adrian, R.; Anneville, O.; Chandra, S.; Colom-Montero, W.; Devlin, S.P.; Dix, M.A.; Dokulil, M.T.; et al. Deeper waters are changing less consistently than surface waters in a global analysis of 102 lakes. Sci. Rep. 2020, 10, 20514. [Google Scholar] [CrossRef]
- Alexander, J.E.; Thorp, J.H.; Fell, R.D. Turbidity and temperature effects on oxygen consumption in the Zebra mussel (Dreissena polymorpha). Can. J. Fish. Aquat. Sci. 1994, 51, 179–184. [Google Scholar] [CrossRef]
- Zhu, X.; Jia, G.; Tian, Y.; Mo, A.; Xu, W.; Miao, L.; Xu, S.; Yan, W. Photosynthetic Production Determines bottom water oxygen variations in the upwelling coastal south China sea over recent decades. Front. Earth Sci. 2021, 9, 759317. [Google Scholar] [CrossRef]
- Sarkar, A.; Naqvi, S.W.A.; Lavik, G.; Pratihary, A.; Naik, H.; Shirodkar, G.; Kuypers, M.M.M. Massive nitrogen loss over the western Indian continental shelf during seasonal anoxia: Evidence from isotope pairing technique. Front. Mar. Sci. 2020, 7, 678. [Google Scholar] [CrossRef]
- Liu, C.; Wang, S.; Liu, X.; Zhou, H.; Li, B.; Du, Y.; Wang, L. Characteristics of water quality response to hypolimnetic anoxia in Daheiting Reservoir. Water Sci. Technol. 2022, 85, 2065–2075. [Google Scholar] [CrossRef]
- Horne, A.J.; Jung, R.; Lai, H.; Faisst, B.; Beutel, M. Hypolimnetic oxygenation 2: Oxygen dynamics in a large reservoir with submerged down-flow contact oxygenation (Speece cone). Lake Reserv. Manag. 2019, 35, 323–337. [Google Scholar] [CrossRef]
- Holtermann, P.; Prien, R.; Naumann, M.; Umlauf, L. Interleaving of oxygenized intrusions into the Baltic Sea redoxcline. Limnol. Oceanogr. 2020, 65, 482–503. [Google Scholar] [CrossRef]
- Stigebrandt, A.; Kalén, O. Improving oxygen conditions in the deeper parts of Bornholm sea by pumped injection of winter water. Ambio 2013, 42, 587–595. [Google Scholar] [CrossRef] [PubMed]
- Salmi, P.; Malin, I.; Salonen, K. Pumping of epilimnetic water into hypolimnion improves oxygen but not necessarily nutrient conditions in a lake recovering from eutrophication. Inland Waters 2014, 4, 425–434. [Google Scholar] [CrossRef]
- Prashanth, K.S.; Ashish, K. Enhancement of dissolved oxygen level in the reservoir downstream using air injection techniques in hydroturbines: A review. Energy Sources Part A Recovery Util. Environ. Eff. 2024, 46, 9935–9954. [Google Scholar]
| Sampling Station | Salinity | DO/ (mg/L) | T/ (°C) | Turbidity (NTU) | Chl-a/ (μg/L) | NH4+/ (μmol/L) | NO3−/(μmol/L) | NO2−/(μmol/L) | TP/ (μmol/L) |
|---|---|---|---|---|---|---|---|---|---|
| BP1 | 11.99~23.18, 313 | 4.69~9.07, 313 | 4.46~20.90, 313 | 0.09~1.17, 313 | 0.59~4.17, 313 | 0.18~2.50, 15 | LD~6.33, 15 | 0.02~0.73, 15 | 0.37~1.19, 15 |
| BP2 | 9.05~25.61, 443 | 2.21~9.94, 443 | 4.33~20.04, 443 | 0.05~2.43, 443 | 0.43~5.17, 443 | 0.16~5.21, 20 | LD~6.90, 20 | 0.01~0.60, 20 | 0.42~1.66, 20 |
| BP3 | 7.22~18.05, 881 | 2.20~9.39, 881 | 3.81~20.04, 881 | LD~1.38, 881 | 0.20~2.24, 881 | 0.16~5.38, 35 | LD~7.01, 35 | 0.02~0.73, 35 | 0.54~1.93, 20 |
| BP4 | 7.32~18.15, 900 | 3.08~9.50, 900 | 3.99~19.68, 900 | LD~1.59, 900 | 0.16~2.94, 900 | 0.16~3.51, 25 | LD~8.52, 25 | LD~1.02, 25 | 0.52~1.42, 20 |
| BP5 | 7.30~16.17, 1726 | 0.03~9.61, 1726 | 3.73~20.95, 1726 | LD~1.32, 1726 | 0.15~2.85, 1726 | 0.10~14.78, 50 | LD~10.21, 50 | LD~0.36, 50 | 0.47~9.21, 30 |
| BP6 | 6.98~12.43, 2992 | 0.0076~9.65, 2992 | 3.04~21.26, 2992 | LD~2.86, 2992 | 0.16~4.05, 2992 | LD~23.04, 52 | LD~5.64, 52 | LD~0.63, 52 | 0.44~5.42, 22 |
| SR1 | 0.02~0.04 | 4.54~8.74, 869 | 14.52~32.58, 869 | 1.29~26.79, 664 | 0.00~42.32, 869 | 0.26~12.86, 29 | 63.14~107.84, 29 | 1.77~9.17, 29 | 0.48~2.74, 29 |
| SR2 | 0.58~8.00, 927 | 14.79~32.39, 927 | 0.86~29.65, 653 | 0.04~36.57, 927 | 0.62~24.14, 25 | 57.44~103.62, 25 | 0.21~8.58, 25 | 0.67~3.64, 25 | |
| SR3 | 0.29~8.09, 1032 | 14.96~31.95, 1032 | 0.51~19.02, 737 | 0.03~30.48, 1032 | 0.82~14.17, 30 | 63.41~105.66, 30 | 0.21~8.86, 30 | 0.32~4.35, 30 | |
| SR4 | 0.31~7.75, 1482 | 15.15~31.11, 1482 | 0.24~10.50, 1032 | 0.08~15.60, 1482 | 1.02~28.08, 30 | 64.36~103.90, 30 | 0.21~10.15, 30 | 0.41~5.25, 30 | |
| SR5 | 0.23~7.23, 1787 | 15.74~31.95, 1787 | 0.05~25.70, 1350 | 0.04~12.03, 1787 | 1.20~33.16, 35 | 59.75~106.07, 35 | 0.21~6.81, 35 | 0.34~2.80, 35 | |
| SR6 | 0.27~7.36, 1881 | 15.98~31.61, 1881 | 0.20~24.26, 869 | 0.10~5.78, 1881 | 1.79~14.36, 39 | 5.71~118.57, 39 | 0.21~5.86, 39 | 0.48~2.15, 39 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, J.; Waniek, J.J.; Kuss, J.; Beltran-Perez, O.; Peng, J.-P.; Shi, C.; Chen, J.; Liu, J.; Chen, L.; Xie, R. Dynamics of Oxygen and the Physicochemical Response in Two Comparative Hypoxia Regions. Oceans 2025, 6, 54. https://doi.org/10.3390/oceans6030054
Wei J, Waniek JJ, Kuss J, Beltran-Perez O, Peng J-P, Shi C, Chen J, Liu J, Chen L, Xie R. Dynamics of Oxygen and the Physicochemical Response in Two Comparative Hypoxia Regions. Oceans. 2025; 6(3):54. https://doi.org/10.3390/oceans6030054
Chicago/Turabian StyleWei, Jian, Joanna J. Waniek, Joachim Kuss, Oscar Beltran-Perez, Jen-Ping Peng, Chengchun Shi, Jin Chen, Jihui Liu, Lili Chen, and Rongrong Xie. 2025. "Dynamics of Oxygen and the Physicochemical Response in Two Comparative Hypoxia Regions" Oceans 6, no. 3: 54. https://doi.org/10.3390/oceans6030054
APA StyleWei, J., Waniek, J. J., Kuss, J., Beltran-Perez, O., Peng, J.-P., Shi, C., Chen, J., Liu, J., Chen, L., & Xie, R. (2025). Dynamics of Oxygen and the Physicochemical Response in Two Comparative Hypoxia Regions. Oceans, 6(3), 54. https://doi.org/10.3390/oceans6030054








