Comparing the Structure of Fish Assemblage among Natural and Artificial Shallow Rocky Habitats
Abstract
1. Introduction
2. Materials and Methods
Statistical Analysis
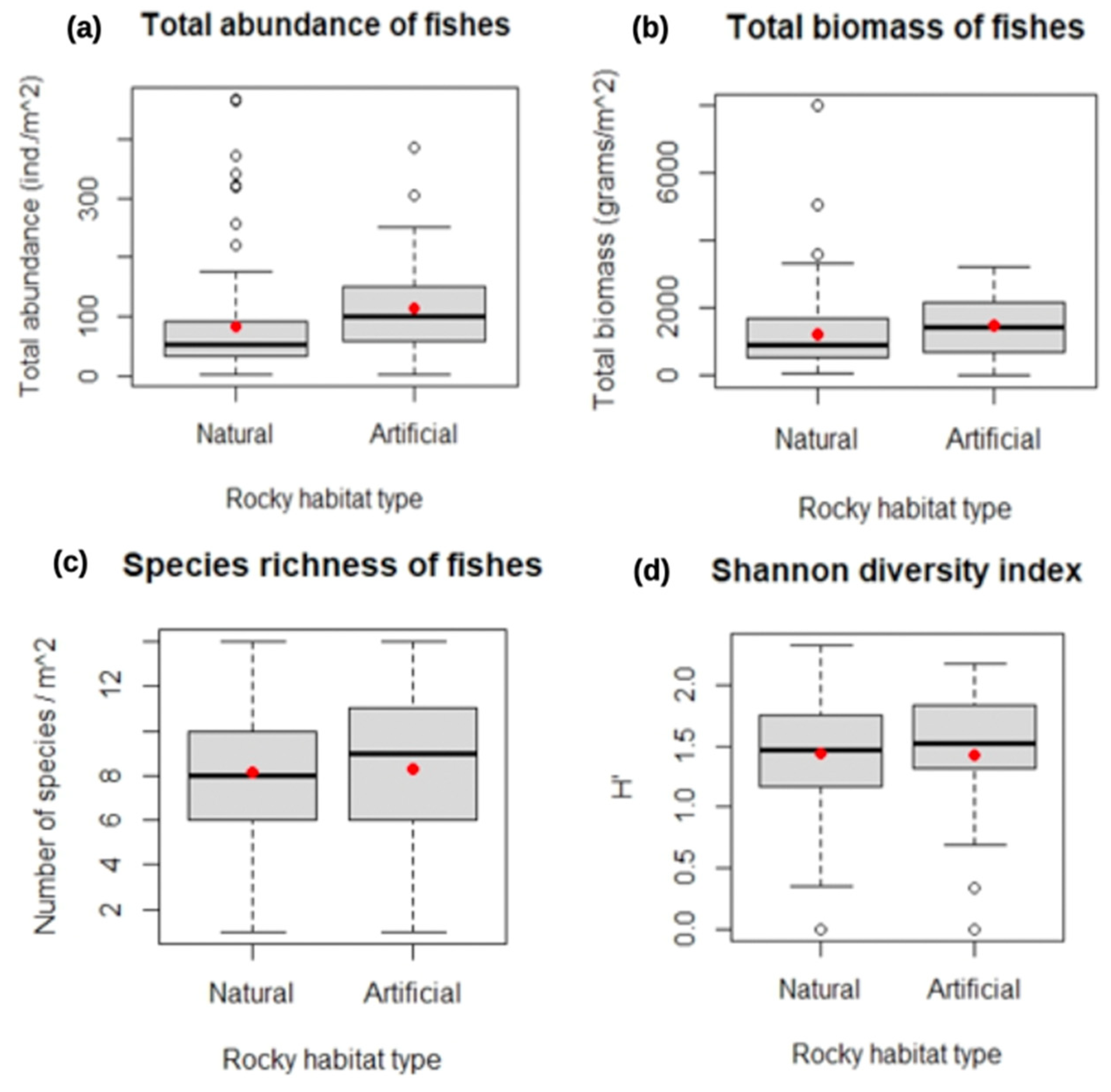
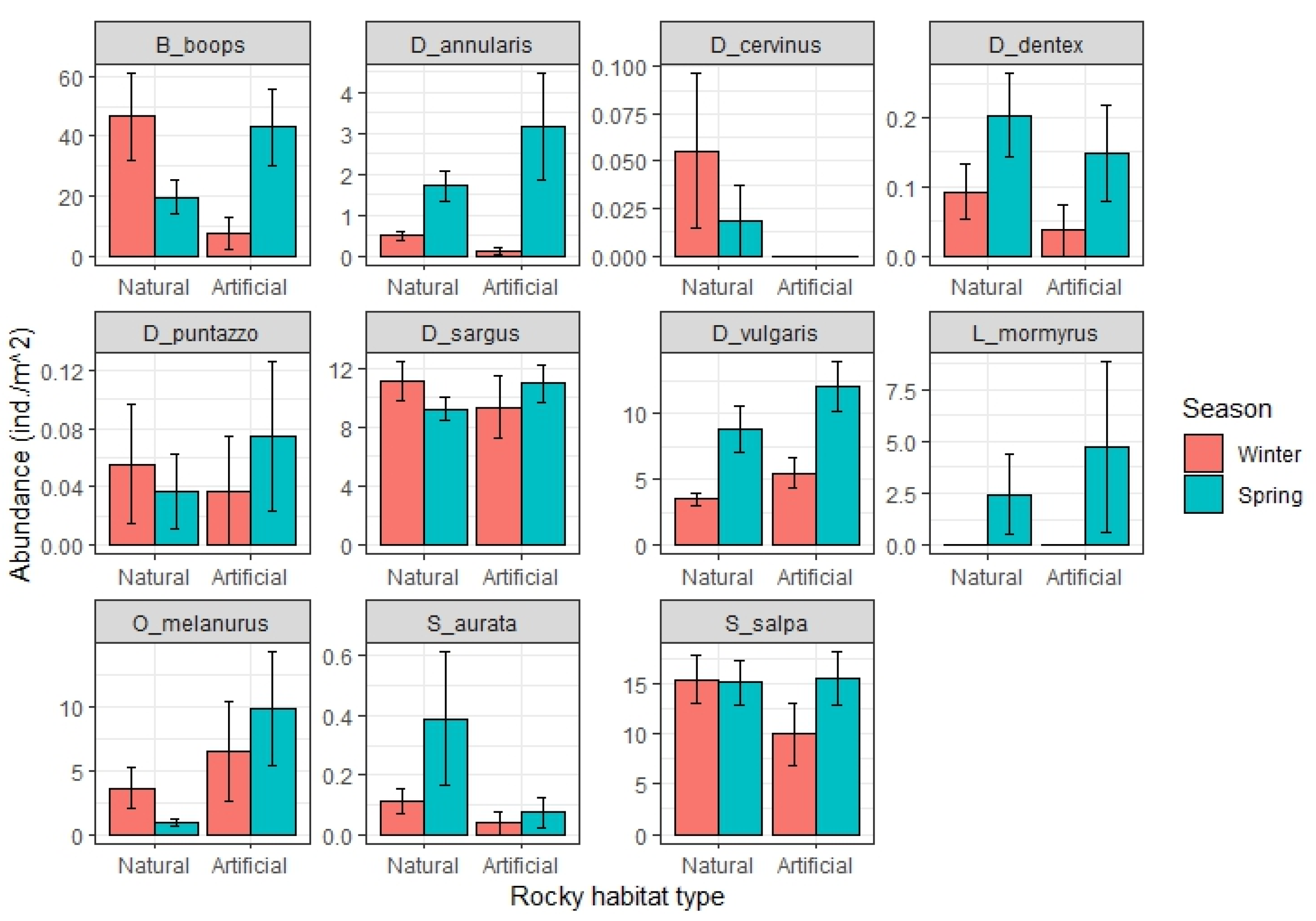
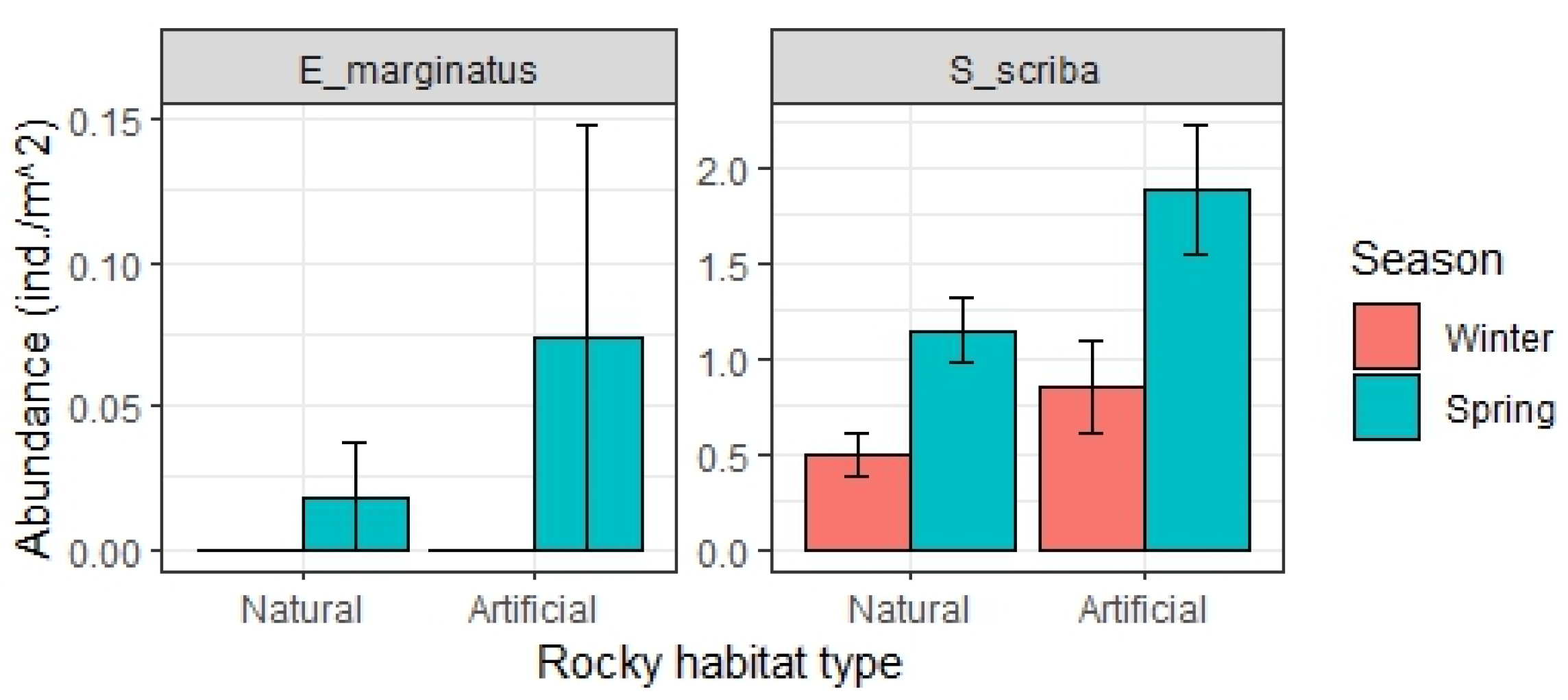
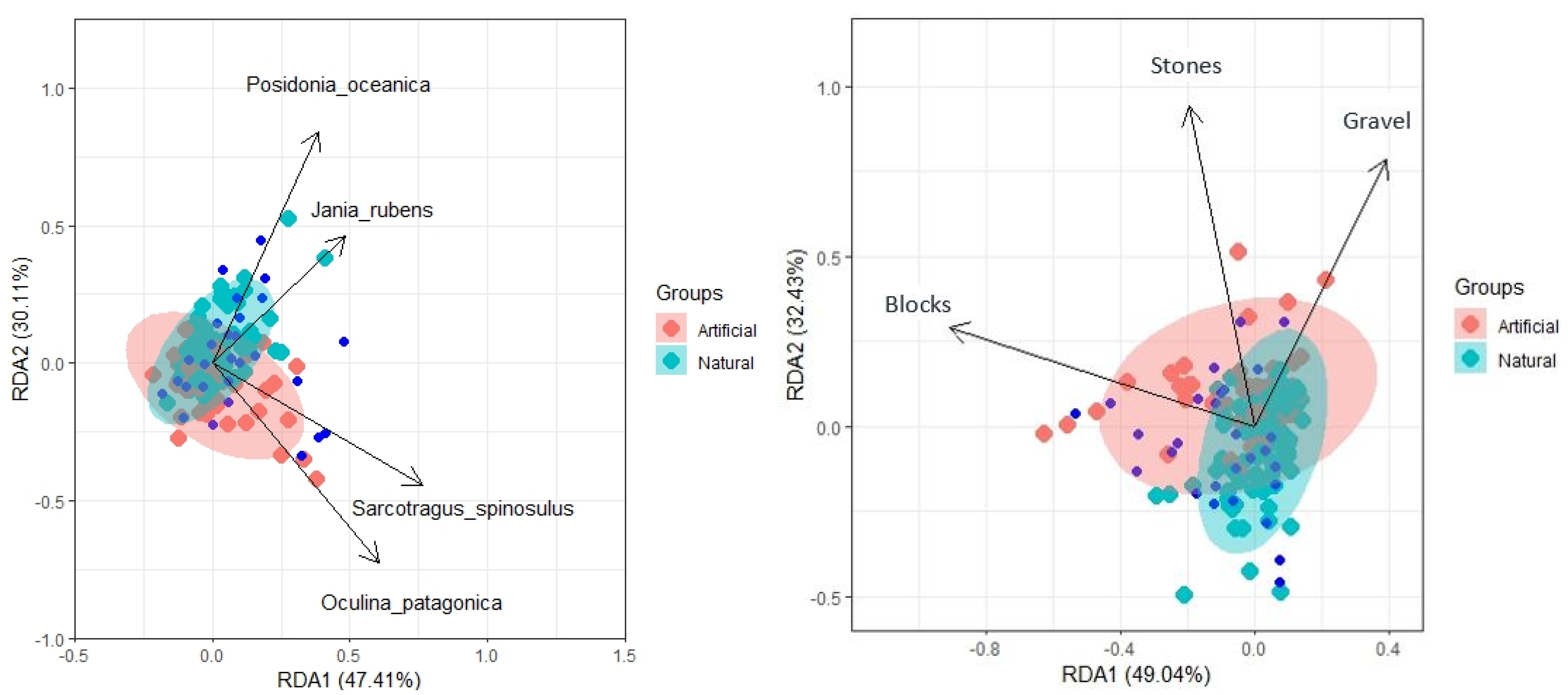
3. Results
3.1. Habitat Features
3.2. Changes in Fish Assemblages
3.3. Relevant Fish Families and Populations
3.4. Multivariate Level Changes of Fish Populations and Relationships with Environmental Variables
4. Discussion
4.1. Changes in Habitat Complexity
4.2. Degradation of Relevant Habitats
4.3. Heterogeneity
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Samourdani, A.; Tzanatos, E. Fish distribution and behavior with regard to the time of day and the anthropogenic structural modification of the shallow littoral. J. Fish Biol. 2022, 100, 820–830. [Google Scholar] [CrossRef] [PubMed]
- Molina, R.; Anfuso, G.; Manno, G.; Gracia-Prieto, F.J. The Mediterranean Coast of Andalusia (Spain): Medium-Term Evolution and Impacts of Coastal Structures. Sustainability 2019, 11, 3539. [Google Scholar] [CrossRef]
- Dahlgren, C.P.; Eggleston, D.B. Ecological processes underlying ontogenetic habitat shifts in a coral reef fish. Ecology 2000, 81, 2227–2240. [Google Scholar] [CrossRef]
- Airoldi, L.; Beck, M.W. Loss, status and trends for coastal marine habitats of Europe. Oceanogr. Mar. Biol. 2007, 45, 345–405. [Google Scholar] [CrossRef]
- Bray, M.J.; Hooke, J.M. Prediction of soft-cliff retreat with accelerating sea-level rise. J. Coast. Res. 1997, 13, 453–467. [Google Scholar]
- Dulvy, N.K.; Jennings, S.; Rogers, S.I.; Maxwell, D.L. Threat and decline in fishes: An indicator of marine biodiversity. Canadian J. Fisher. Aqua. Scien. 2006, 63, 1267–1275. [Google Scholar] [CrossRef]
- García-Charton, J.A.; Pérez-Ruzafa, A.; Sánchez-Jerez, P.; Bayle-Sempere, J.T.; Reñones, O.; Moreno, D. Multi-scale spatial heterogeneity, habitat structure, and the effect of marine reserves on Western Mediterranean rocky reef fish assemblages. Mar. Biol. 2004, 144, 161–182. [Google Scholar] [CrossRef]
- López, I.; Aragonés, L.; Villacampa, Y. Analysis and modelling of cross-shore profile of gravel beaches in the province of Alicante. Ocean Eng. 2016, 118, 173–186. [Google Scholar] [CrossRef]
- Martínez, C. Estudio de Soluciones de las Obras de Abrigo y Ordenación Interior del Puerto Deportivo “Puerto Amor” (TM Alicante). Doctoral Thesis, Polytechnic University of Valencia, Valencia, Spain, 2016. [Google Scholar]
- Aragonés, L.; García-Barba, J.; Villacampa, Y.; López, I.; Gómez-Martín, M.E.; Pagán, J.I. Sustainable Development City-Beach in Alicante Sustainable Development City-Beach in Alicante. Int. J. Sustain. Dev. Plan. 2017, 12, 704–712. [Google Scholar] [CrossRef]
- Diario de Alicante. Compromís Pide Rehabilitar la Zona del ‘Fallido’ Puerto Amor. Available online: https://diariodealicante.net/compromis-rehabilitar-puerto-amor/ (accessed on 10 July 2023).
- Chou, L.M.; Lim, G.S.Y.; Leng, C.B. An assessment of fish communities of artificial reef structures in Brunei Darussalam with recommendations for management and development. Res. Manag. Opt. 1991, 1, 15–31. [Google Scholar]
- Lindfield, S.J.; Harvey, E.S.; McIlwain, J.L.; Halford, A.R. Silent fish surveys: Bubble-free diving highlights inaccuracies associated with SCUBA-based surveys in heavily fished areas. Meth. Ecol. Evol. 2014, 5, 1061–1069. [Google Scholar] [CrossRef]
- Europa.eu. N2K ES5213032 Dataforms (s.f.). Available online: https://natura2000.eea.europa.eu/Natura2000/SDF.aspx?site=ES5213032 (accessed on 16 November 2023).
- Harmelin-Vivien, M.L.; Harmelin, J.G.; Chauvet, C.; Duval, C.; Galzin, R.; Lejeune, P.; Barnabe, G.; Blanc, F.; Chevalier, R.; Duclerc, J.; et al. Evaluation visuelle des peuplements et populations de poissons méthodes et problèmes. Rev. D’ecol. Terre Vie 1985, 40, 467–539. [Google Scholar] [CrossRef]
- Valle, C.; Bayle, J.; Ramos, A. Weight-length relationships for selected fish species of the western Mediterranean Sea. J. App. Ichth. 2003, 19, 261–262. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing. In Foundation for Statistical Computing; R Core Team: Vienna, Austria, 2022; Available online: https://www.R-project.org/ (accessed on 1 December 2023).
- Wickham, H.; Hester, J.; Bryan, J. Readr: Read Rectangular Text Data. R Package Version 2.1.4. Available online: https://CRAN.R-project.org/package=readr (accessed on 1 December 2023).
- Morales, M. Team wcdbtRDC, Community wgaftRl, Murdoch. eD. sciplot: Scientific Graphing Functions for Factorial Designs. R Package Version 1.2-0. Available online: https://CRAN.R-project.org/package=sciplot (accessed on 1 December 2020).
- Wickham, H.; Averick, M.; Bryan, J.; Chang, W.; McGowan, L.D.; François, R.; Grolemund, G.; Hayes, A.; Henry, L.; Hester, J.; et al. Welcome to the tidyverse. J. Open Source Softw. 2019, 4, 1686. [Google Scholar] [CrossRef]
- Oksanen, J.; Simpson, G.; Blanchet, F.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, R.; Solymos, P.; Stevens, M.; Szoecs, E.; et al. Vegan: Community Ecology Package. R Package Version 2.6-4. Available online: https://CRAN.R-project.org/package=vegan (accessed on 1 December 2020).
- Slowikowski, K. Ggrepel: Automatically Position Non-Overlapping Text Labels with ‘ggplot2’. R Package Version 0.9.3. Available online: https://CRAN.R-project.org/package=ggrepel (accessed on 1 December 2023).
- Beck, M. Ggord: Ordination Plots with ggplot2. R package version 1.1.7. 2022. Available online: https://fawda123.github.io/ggord/ (accessed on 1 December 2023).
- Heine, J. Pairwise: Rasch Model Parameters by Pairwise Algorithm. R Package Version 0.6.1-0. Available online: https://CRAN.R-project.org/package=pairwise (accessed on 1 December 2023).
- Wei, T.; Simko, V. R package ‘corrplot’: Visualization of a Correlation Matrix (Version 0.92). Available online: https://github.com/taiyun/corrplot (accessed on 1 December 2021).
- Waring, E.; Quinn, M.; McNamara, A.; Arino de la Rubia, E.; Zhu, H.; Ellis, S. Skimr: Compact and Flexible Summaries of Data. R Package Version 2.1.5. Available online: https://CRAN.R-project.org/package=skimr (accessed on 1 December 2022).
- Sjoberg, D.D.; Whiting, K.; Curry, M.; Lavery, J.A.; Larmarange, J. Reproducible summary tables with the gtsummary package. R J. 2021, 13, 570–580. [Google Scholar] [CrossRef]
- Dowle, M.; Srinivasan, A. Data.table: Extension of ‘data.frame’. R Package Version 1.14.8. Available online: https://CRAN.R-project.org/package=data.table (accessed on 1 December 2023).
- Xie, Y. Knitr: A General-Purpose Package for Dynamic Report Generation in R. R Package Version 1.41. 2022. Available online: https://rdrr.io/cran/knitr/ (accessed on 1 December 2023).
- Lüdecke, D. SjPlot: Data Visualization for Statistics in Social Science. R Package Version 2.8.14. Available online: https://CRAN.R-project.org/package=sjPlot (accessed on 1 December 2023).
- Zhu, H. KableExtra: Construct Complex Table with ‘kable’ and Pipe Syntax. R Package Version 1.3.4. Available online: https://CRAN.R-project.org/package=kableExtra (accessed on 1 December 2021).
- Hugh-Jones, D. Huxtable: Easily Create and Style Tables for LaTeX, HTML and Other Formats. R Package Version 5.5.2. Available online: https://CRAN.R-project.org/package=huxtable (accessed on 1 December 2022).
- Groffman, P.M.; Bain, D.J.; Band, L.E.; Belt, K.T.; Brush, G.S.; Grove, J.M.; Pouyat, R.V.; Yesilonis, I.C.; Zipperer, W.C. Down by the Riverside: Urban Riparian Ecology. Front. Ecol. Env. 2003, 1, 315–321. [Google Scholar] [CrossRef]
- Thompson, K.; Austin, K.C.; Smith, R.M.; Warren, P.H.; Angold, P.G.; Gaston, K.J. Urban domestic gardens (I): Putting small-scale plant diversity in context. J. Veg. Sci. 2003, 14, 71–78. [Google Scholar] [CrossRef]
- Charton, J.A.G.; Ruzafa, A.P. Correlation between habitat structure and a rocky reef fish assemblage in the Southwest Mediterranean. Mar. Ecol. 1998, 19, 111–128. [Google Scholar] [CrossRef]
- Aguilera, M.A.; Broitman, B.R.; Thiel, M. Spatial variability in community composition on a granite breakwater versus natural rocky shores: Lack of microhabitats suppresses intertidal biodiversity. Mar. Poll. Bull. 2014, 87, 257–268. [Google Scholar] [CrossRef]
- Moschella, P.S.; Abbiati, M.; Åberg, P.; Airoldi, L.; Anderson, J.M.; Bacchiocchi, F.; Bulleri, F.; Dinesen, G.E.; Frost, M.; Gacia, E.; et al. Low-crested coastal defence structures as artificial habitats for marine life: Using ecological criteria in design. Coast. Eng. 2005, 52, 1053–1071. [Google Scholar] [CrossRef]
- Pledger, A.G.; Brewin, P.; Mathers, K.L.; Phillips, J.; Wood, P.J.; Yu, D. The effects of water injection dredging on low-salinity estuarine ecosystems: Implications for fish and macroinvertebrate communities. Ecol. Indic. 2021, 122, 107244. [Google Scholar] [CrossRef]
- Cruz, L.C.; Pompeu, P.S. Drivers of fish assemblage structures in a Neotropical urban watershed. Urb. Ecosys. 2020, 23, 819–829. [Google Scholar] [CrossRef]
- Ruiz, J.M.; Romero, J. Effects of disturbances caused by coastal constructions on spatial structure, growth dynamics and photosynthesis of the seagrass Posidonia oceanica. Mar. Poll. Bull. 2003, 46, 1523–1533. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Torquemada, Y.; González-Correa, J.M.; Martínez, J.E.; Sánchez-Lizaso, J.L. Evaluation of the effects produced by the construction and expansion of marinas on Posidonia oceanica (L.) Delile meadows. J. Coast. Res. 2005, 49, 94–99. [Google Scholar]
- Sánchez-Jerez, P.; Ramos Esplá, A. Detection of environmental impacts by bottom trawling on Posidonia oceanica (L.) Delile meadows: Sensitivity of fish and macroinvertebrate communities. J. Aqua. Ecosys. Health 1996, 5, 239–253. [Google Scholar] [CrossRef]
- Levin, L.A.; Sibuet, M.; Gooday, A.J.; Smith, C.R.; Vanreusel, A. The roles of habitat heterogeneity in generating and maintaining biodiversity on continental margins: An introduction. Mar. Ecol. 2010, 31, 1–5. [Google Scholar] [CrossRef]
- Ferrario, F.; Iveša, L.; Jaklin, A.; Perkol-Finkel, S.; Airoldi, L. The overlooked role of biotic factors in controlling the ecological performance of artificial marine habitats. J. App. Ecol. 2016, 53, 16–24. [Google Scholar] [CrossRef]
- Clavel, J.; Julliard, R.; Devictor, V. Worldwide decline of specialist species: Toward a global functional homogenization? Front. Ecol. Env. 2011, 9, 222–228. [Google Scholar] [CrossRef]
- Dala-Corte, R.B.; Melo, A.S.; Siqueira, T.; Bini, L.M.; Martins, R.T.; Cunico, A.M.; Pes, A.M.; Magalhães, A.L.B.; Godoy, B.S.; Leal, C.G.; et al. Thresholds of freshwater biodiversity in response to riparian vegetation loss in the Neotropical region. J. App. Ecol. 2020, 57, 1391–1402. [Google Scholar] [CrossRef]
- Evans, A.J.; Firth, L.B.; Hawkins, S.J.; Morris, E.S.; Goudge, H.; Moore, P. Drill-cored rock pools: An effective method of ecological enhancement on artificial structures. Mar. Freshwat. Res. 2015, 67, 123–130. [Google Scholar] [CrossRef]
- Leydet, K.P.; Hellberg, M.E. The invasive coral Oculina patagonica has not been recently introduced to the Mediterranean from the western Atlantic. BMC Evol. Biol. 2015, 15, 79. [Google Scholar] [CrossRef] [PubMed]
- FAO; SER; IUCN CEM. Standards of Practice to Guide Ecosystem Restoration. A Contribution to the United Nations Decade on Ecosystem Restoration; FAO: Rome, Italy, 2023. [Google Scholar] [CrossRef]



| Total Abundance | Total Biomass | Species Richness | Shannon Diversity | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| df | SC | Pr > (F) | df | SC | Pr > (F) | df | SC | Pr > (F) | df | SC | Pr > (F) | |
| Lo | 2 | 0.1268 | 0.004 * | 1 | 0.0517 | 0.027 * | 2 | 0.005 | 0.350 | 2 | 0.009 | 0.490 |
| Se | 1 | 0.0522 | 0.031 * | 1 | 0.0389 | 0.048 * | 1 | 0.038 | 0.001 * | 1 | 0.059 | 0.001 * |
| LoxSe | 2 | 0.0914 | 0.010 * | 1 | 0.1339 | 0.001 * | 2 | 0.025 | 0.003 * | 2 | 0.021 | 0.166 |
| Ti(Se) | 4 | 0.1529 | 0.010 * | 4 | 0.0352 | 0.548 | 4 | 0.028 | 0.005 * | 4 | 0.043 | 0.800 * |
| LoxTi(Se) | 8 | 0.0889 | 0.382 | 4 | 0.0293 | 0.639 | 8 | 0.019 | 0.441 | 8 | 0.077 | 0.073 * |
| Si(LoxTi(Se)) | 26 | 0.8538 | 0.002 * | 24 | 0.1506 | 0.968 | 26 | 0.087 | 0.459 | 36 | 0.243 | 0.199 |
| Residual | 108 | 1.1015 | - | 126 | 1.3616 | - | 108 | 0.256 | - | 108 | 0.605 | - |
| Pairwise Lo | A ≠ N2 | - | - | - | ||||||||
| Pairwise Se | - | - | W ≠ S | W ≠ S | ||||||||
| Pairwise LoxEp | A:W ≠ A:S, N1:W ≠ A:S, A:S ≠ N1:S, A:S ≠ N2:S | A:W ≠ N1:S, A:S ≠ N2:S | A:W ≠ A:S,A:W ≠ N1:S, N1:W ≠ A:S,N1:W ≠ N1:S, A:S ≠ N2:S, N1:S ≠ N2:S | A:W ≠ N1:S, N1:W ≠ N1:S | ||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Salines, L.; Sanchez-Jerez, P. Comparing the Structure of Fish Assemblage among Natural and Artificial Shallow Rocky Habitats. Oceans 2024, 5, 244-256. https://doi.org/10.3390/oceans5020015
García-Salines L, Sanchez-Jerez P. Comparing the Structure of Fish Assemblage among Natural and Artificial Shallow Rocky Habitats. Oceans. 2024; 5(2):244-256. https://doi.org/10.3390/oceans5020015
Chicago/Turabian StyleGarcía-Salines, Laura, and Pablo Sanchez-Jerez. 2024. "Comparing the Structure of Fish Assemblage among Natural and Artificial Shallow Rocky Habitats" Oceans 5, no. 2: 244-256. https://doi.org/10.3390/oceans5020015
APA StyleGarcía-Salines, L., & Sanchez-Jerez, P. (2024). Comparing the Structure of Fish Assemblage among Natural and Artificial Shallow Rocky Habitats. Oceans, 5(2), 244-256. https://doi.org/10.3390/oceans5020015








