An Expert Elicitation of the Effects of Low Salinity Water Exposure on Bottlenose Dolphins
Abstract
1. Introduction
2. Materials and Methods
2.1. Elicitation Approach
2.2. Designing the Elicitation
2.3. Performing the Elicitation
2.3.1. Elicitation Structure
2.3.2. Low Salinity Exposure Scenarios
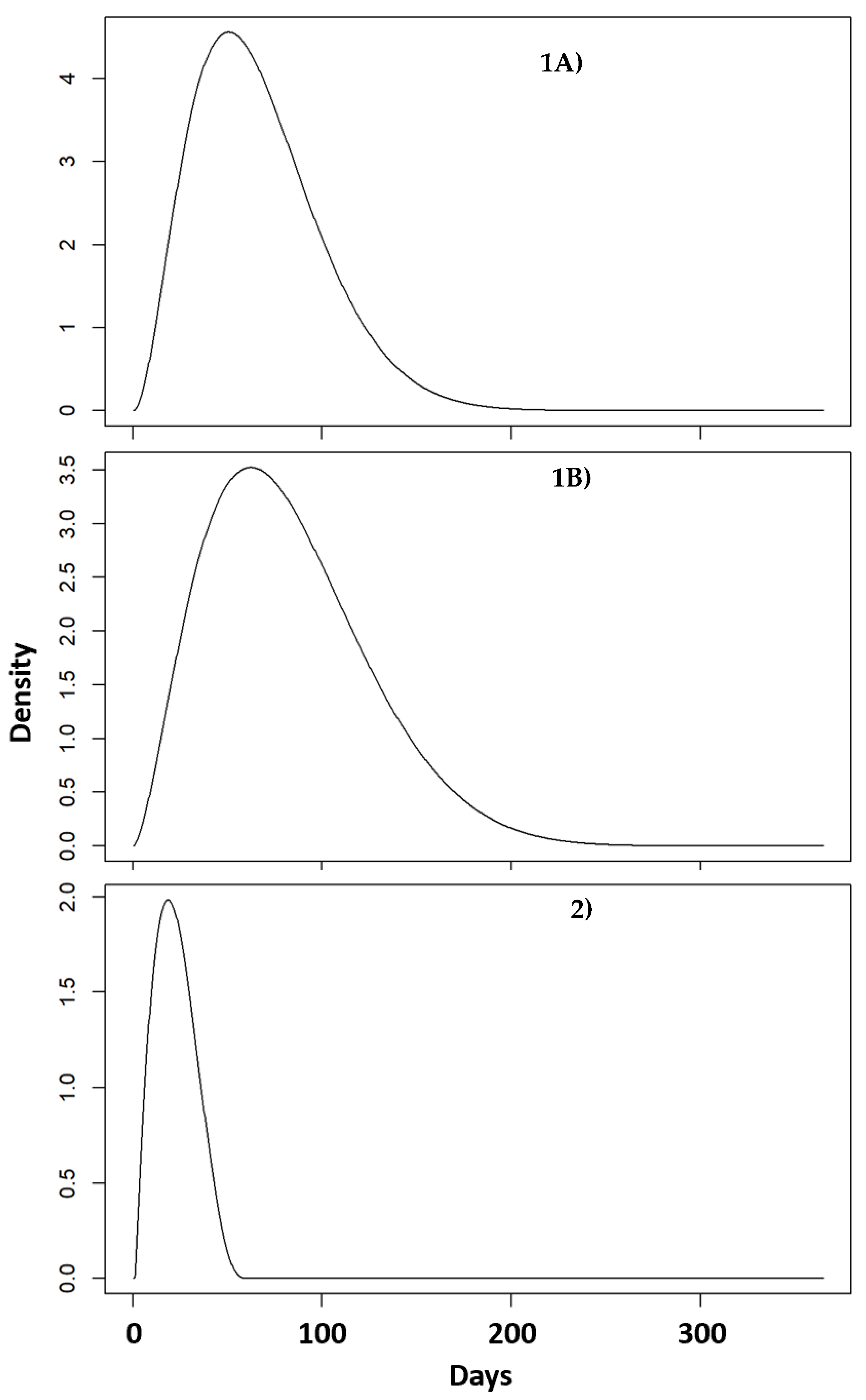
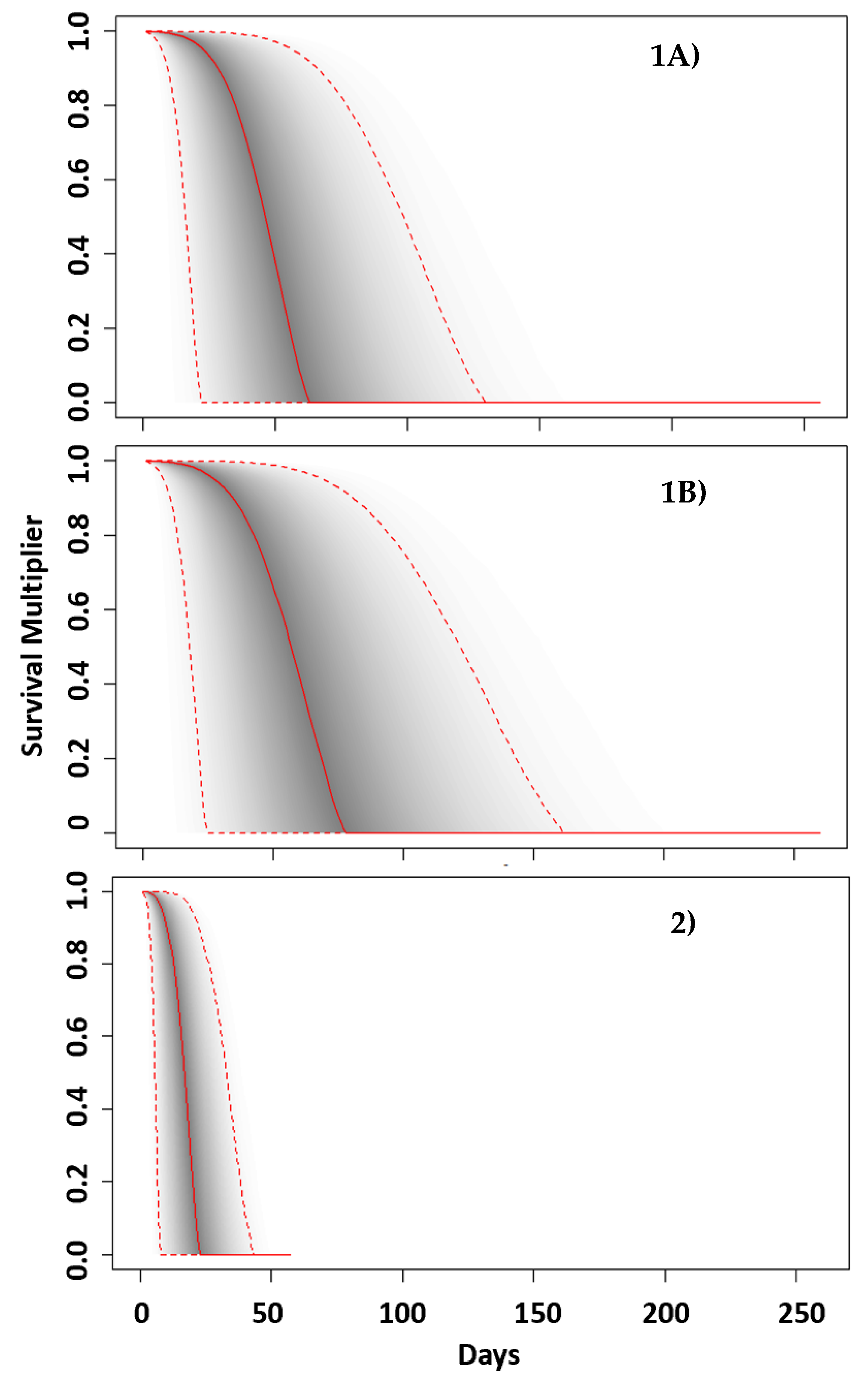
- Scenario 1A: An extended low salinity event. For example, a bay, sound and estuary (BSE) environment (i.e., mean 15–25 ppt) is flooded with fresh or low salinity water until salinity drops (at approx. 0.5 ppt/day—i.e., salinity decreasing over 20–40 days) to below 5 ppt for an extended period. This is an environment in which animals are exposed to other significant stressors (e.g., noise, low quality prey, exposure to contaminants) and are more likely to be in a “compromised health state”.
- Scenario 1B: As in Scenario 1A but in an environment in which there are few other stressors and animals in the population are broadly considered to be “healthy”.
- Scenario 2: “Acute salinity change event”: Bottlenose dolphins experience a change in salinity from typical salinity environment (i.e., mean 15–25 ppt) down to an atypical environment with salinity below 5 ppt for an extended period. This change in salinity occurs within 0–5 days.
2.3.3. Expert Judgements
2.4. Dose–Response Function
3. Results
3.1. Probability Distributions
3.1.1. Extended Low Salinity Events
3.1.2. Acute Salinity Change Events
3.1.3. Expert Rationale
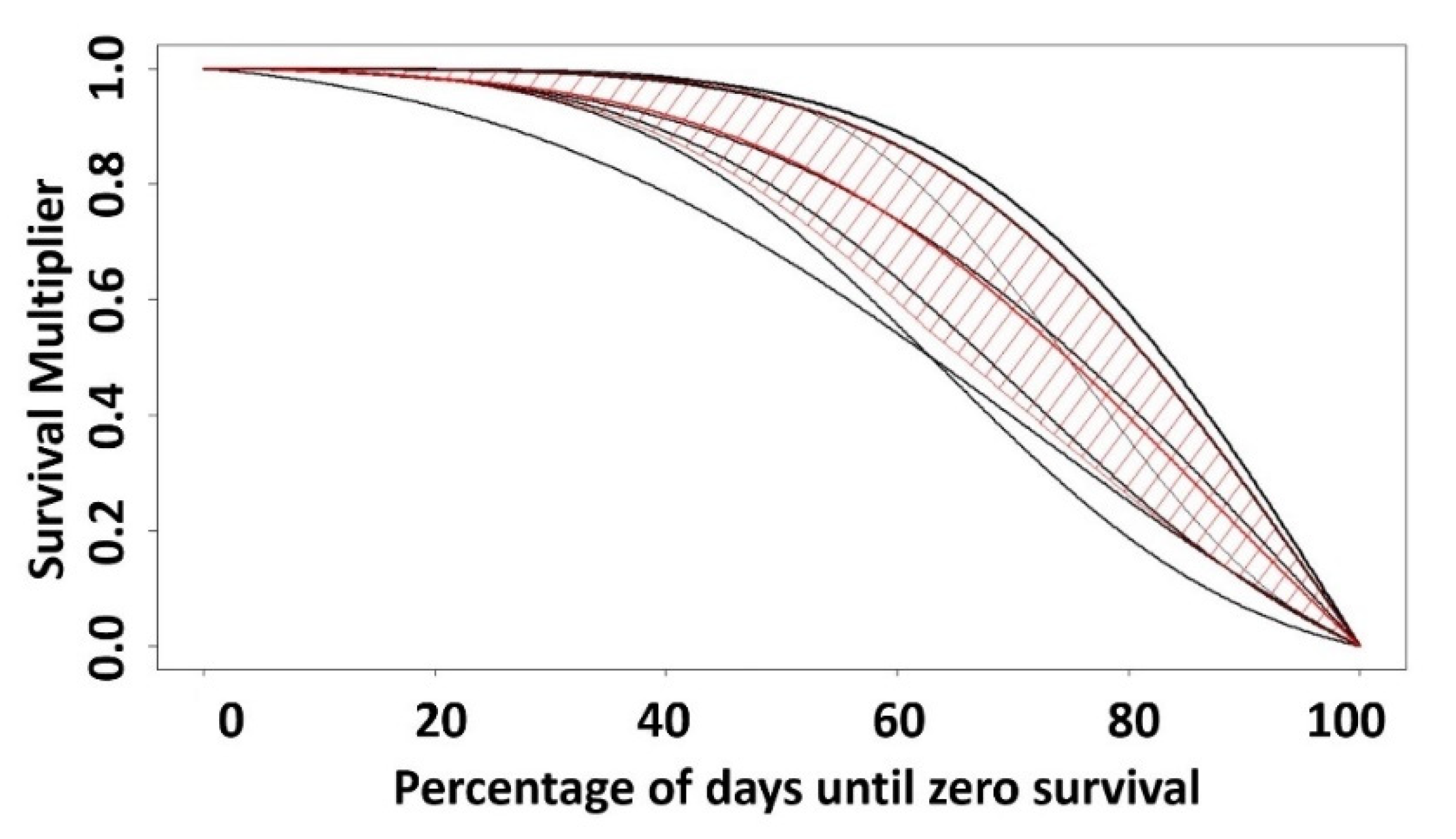
3.2. Dose–Response Function Shape
4. Discussion
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National Academies of Sciences Engineering and Medicine. Approaches to Understanding the Cumulative Effects of Stressors on Marine Mammals; The National Academies Press: Washington, DC, USA, 2017. [Google Scholar]
- Poloczanska, E.S.; Burrows, M.T.; Brown, C.J.; García Molinos, J.; Halpern, B.S.; Hoegh-Guldberg, O.; Kappel, C.V.; Moore, P.J.; Richardson, A.J.; Schoeman, D.S. Responses of marine organisms to climate change across oceans. Front. Mar. Sci. 2016, 3, 62. [Google Scholar] [CrossRef]
- Frölicher, T.L.; Laufkötter, C. Emerging risks from marine heat waves. Nat. Commun. 2018, 9, 650. [Google Scholar] [CrossRef] [PubMed]
- Harvell, C.; Montecino-Latorre, D.; Caldwell, J.; Burt, J.; Bosley, K.; Keller, A.; Heron, S.; Salomon, A.; Lee, L.; Pontier, O. Disease epidemic and a marine heat wave are associated with the continental-scale collapse of a pivotal predator (Pycnopodia helianthoides). Sci. Adv. 2019, 5, eaau7042. [Google Scholar] [CrossRef] [PubMed]
- Bijma, J.; Pörtner, H.-O.; Yesson, C.; Rogers, A.D. Climate change and the oceans–What does the future hold? Mar. Pollut. Bull. 2013, 74, 495–505. [Google Scholar] [CrossRef]
- King, S.L.; Schick, R.S.; Donovan, C.; Booth, C.G.; Burgman, M.; Thomas, L.; Harwood, J. An interim framework for assessing the population consequences of disturbance. Methods Ecol. Evol. 2015, 6, 1150–1158. [Google Scholar] [CrossRef]
- Erbe, C.; Marley, S.A.; Schoeman, R.P.; Smith, J.N.; Trigg, L.E.; Embling, C.B. The Effects of Ship Noise on Marine Mammals—A Review. Front. Mar. Sci. 2019, 6. [Google Scholar] [CrossRef]
- Davies, R.; Cripps, S.; Nickson, A.; Porter, G. Defining and estimating global marine fisheries bycatch. Mar. Policy 2009, 33, 661–672. [Google Scholar] [CrossRef]
- Trenberth, K.E. Changes in precipitation with climate change. Clim. Res. 2011, 47, 123–138. [Google Scholar] [CrossRef]
- Huizer, S.; Karaoulis, M.; Oude Essink, G.; Bierkens, M. Monitoring and simulation of salinity changes in response to tide and storm surges in a sandy coastal aquifer system. Water Resour. Res. 2017, 53, 6487–6509. [Google Scholar] [CrossRef]
- Holt, T.; Seibert, S.L.; Greskowiak, J.; Freund, H.; Massmann, G. Impact of storm tides and inundation frequency on water table salinity and vegetation on a juvenile barrier island. J. Hydrol. 2017, 554, 666–679. [Google Scholar] [CrossRef]
- Curtis, S. The Atlantic multidecadal oscillation and extreme daily precipitation over the US and Mexico during the hurricane season. Clim. Dyn. 2008, 30, 343–351. [Google Scholar] [CrossRef]
- Nijssen, B.; O’Donnell, G.M.; Lettenmaier, D.P.; Lohmann, D.; Wood, E.F. Predicting the discharge of global rivers. J. Clim. 2001, 14, 3307–3323. [Google Scholar] [CrossRef]
- Vollmer, N.L.; Rosel, P.E. A review of common bottlenose dolphins (Tursiops truncatus truncatus) in the northern Gulf of Mexico: Population biology, potential threats, and management. Southeast Nat. 2013, 12, 1–43. [Google Scholar]
- Hayes, S.; Josephson, E.; Maze-Foley, K.; Rosel, P. US Atlantic and Gulf of Mexico marine mammal stock assessments–2018. NOAA Tech. Memo. NMFS-NE 2019, 258. [Google Scholar] [CrossRef]
- NOAA Fisheries. Common Bottlenose Dolphin (Tursiops Truncatus Truncatus): Northern Gulf of Mexico Bay, Sound, and Estuary Stocks; NOAA Southeast Fisheries Science Center: Miama, FL, USA, 2019; p. 21.
- Rosel, P.; Wilcox, L.; Sinclair, C.; Speakman, T.; Tumlin, M.; Litz, J.; Zolman, E. Genetic assignment to stock of stranded common bottlenose dolphins in southeastern Louisiana after the Deepwater Horizon oil spill. Endanger. Species Res. 2017, 33, 221–234. [Google Scholar] [CrossRef]
- Balmer, B.C.; Schwacke, L.H.; Wells, R.S.; George, R.C.; Hoguet, J.; Kucklick, J.R.; Lane, S.M.; Martinez, A.; McLellan, W.A.; Rosel, P.E.; et al. Relationship between persistent organic pollutants (POPs) and ranging patterns in common bottlenose dolphins (Tursiops truncatus) from coastal Georgia, USA. Sci. Total Environ. 2011, 409, 2094–2101. [Google Scholar] [CrossRef]
- Rowles, T.K.; Schwacke, L.S.; Wells, R.S.; Saliki, J.T.; Hansen, L.; Hohn, A.; Townsend, F.; Sayre, R.A.; Hall, A.J. Evidence of susceptibility to morbillivirus infection in cetaceans from the United States. Mar. Mammal Sci. 2011, 27, 1–19. [Google Scholar] [CrossRef]
- Schwacke, L.H.; Thomas, L.; Wells, R.S.; McFee, W.E.; Hohn, A.A.; Mullin, K.D.; Zolman, E.S.; Quigley, B.M.; Rowles, T.K.; Schwacke, J.H. Quantifying injury to common bottlenose dolphins from the Deepwater Horizon oil spill using an age-, sexand class-structured population model. Endang. Species Res. 2017, 33, 265–279. [Google Scholar] [CrossRef]
- Takeshita, R.; Sullivan, L.; Smith, C.; Collier, T.; Hall, A.; Brosnan, T.; Rowles, T.; Schwacke, L. The Deepwater Horizon oil spill marine mammal injury assessment. Endang. Species Res. 2017, 33, 95–106. [Google Scholar] [CrossRef]
- Venn-Watson, S.K.; Townsend, F.I.; Daniels, R.L.; Sweeney, J.C.; Mcbain, J.W.; Klatsky, L.J.; Hicks, C.L.; Staggs, L.A.; Rowles, T.K.; Schwacke, L.H.; et al. Hypocitraturia in Common Bottlenose Dolphins (Tursiops truncatus): Assessing a Potential Risk Factor for Urate Nephrolithiasis. Comp. Med. 2010, 60, 149–153. [Google Scholar]
- Mchugh, K.A.; Allen, J.B.; Barleycorn, A.A.; Wells, R.S. Severe Karenia brevis red tides influence juvenile bottlenose dolphin (Tursiops truncatus) behavior in Sarasota Bay, Florida. Mar. Mammal Sci. 2011, 27, 622–643. [Google Scholar] [CrossRef]
- Litz, J.A.; Baran, M.A.; Bowen-Stevens, S.R.; Carmichael, R.H.; Colegrove, K.M.; Garrison, L.P.; Fire, S.E.; Fougeres, E.M.; Hardy, R.; Holmes, S. Review of historical unusual mortality events (UMEs) in the Gulf of Mexico (1990–2009): Providing context for the multi-year northern Gulf of Mexico cetacean UME declared in 2010. Dis. Aquat. Org. 2014, 112, 161–175. [Google Scholar] [CrossRef]
- Carmichael, R.H.; Graham, W.M.; Aven, A.; Worthy, G.; Howden, S. Were Multiple Stressors a ‘Perfect Storm’ for Northern Gulf of Mexico Bottlenose Dolphins (Tursiops truncatus) in 2011? PLoS ONE 2012, 7. [Google Scholar] [CrossRef]
- Rosel, P.E.; Watts, H. Hurricane impacts on bottlenose dolphins in the northern Gulf of Mexico. Gulf Mex. Sci. 2007, 25, 88. [Google Scholar] [CrossRef][Green Version]
- Andersen, S. Treatment of water in dolphinaria. Aquat. Mamm 1973, 1, 1–18. [Google Scholar]
- Wells, R.S.; Rhinehart, H.L.; Hansen, L.J.; Sweeney, J.C.; Townsend, F.I.; Stone, R.; Casper, D.R.; Scott, M.D.; Hohn, A.A.; Rowles, T.K. Bottlenose dolphins as marine ecosystem sentinels: Developing a health monitoring system. EcoHealth 2004, 1, 246–254. [Google Scholar] [CrossRef]
- Hart, L.B.; Rotstein, D.S.; Wells, R.S.; Allen, J.; Barleycorn, A.; Balmer, B.C.; Lane, S.M.; Speakman, T.; Zolman, E.S.; Stolen, M. Skin lesions on common bottlenose dolphins (Tursiops truncatus) from three sites in the Northwest Atlantic, USA. PLoS ONE 2012, 7, e33081. [Google Scholar] [CrossRef]
- McDonald, T.L.; Hornsby, F.E.; Speakman, T.R.; Zolman, E.S.; Mullin, K.D.; Sinclair, C.; Rosel, P.E.; Thomas, L.; Schwacke, L.H. Survival, density, and abundance of common bottlenose dolphins in Barataria Bay (USA) following the Deepwater Horizon oil spill. Endanger. Species Res. 2017, 33, 193–209. [Google Scholar] [CrossRef]
- Hornsby, F.E.; McDonald, T.L.; Balmer, B.C.; Speakman, T.R.; Mullin, K.D.; Rosel, P.E.; Wells, R.S.; Telander, A.C.; Marcy, P.W.; Klaphake, K.C. Using salinity to identify common bottlenose dolphin habitat in Barataria Bay, Louisiana, USA. Endanger. Species Res. 2017, 33, 181–192. [Google Scholar] [CrossRef]
- McClain, A.M.; Daniels, R.; Gomez, F.M.; Ridgway, S.H.; Takeshita, R.; Jensen, E.D.; Smith, C.R. Physiological Effects of Low Salinity Exposure on Bottlenose Dolphins (Tursiops truncatus). J. Zool. Bot. Gard. 2020, 1, 61–75. [Google Scholar] [CrossRef]
- Colbert, A.A.; Scott, G.; Fulton, M.; Wirth, E.; Daugomah, J.; Key, P.; Strozier, E.; Galloway, S. Investigation of Unusual Mortalities of Bottlenose Dolphins Along the Mid-Texas Coastal Bay Ecosystem During 1992; US Department of Commerce: Washington, DC, USA, 1999.
- Mullin, K.; Barry, K.P.; Sinclair, C.; Litz, J.A.; Maze-Foley, K.; Fougères, E.M.; Ewing, R.; Gorgone, A.M.; Adams, J.; Tumlin, M. Common Bottlenose Dolphins (Tursiops Truncatus) in Lake Pontchartrain, Louisiana, 2007 to Mid-2014; NOAA Southeast Fisheries Science Center: Miama, FL, USA, 2015.
- Barry, K.P.; Gorgone, A.M.; Mase, B. Lake Pontchartrain, Louisiana Bottlenose Dolphin Survey Summary 28 April 2008–10 May 2008. In Southeast Fisheries Science Center, National Marine Fisheries Service, NOAA Protected Resources and Biodiversity Division, PRBD Contribution: PRBD-08/09-01; NOAA Southeast Fisheries Science Center: Miama, FL, USA, 2008. [Google Scholar]
- Holyoake, C.; Finn, H.; Stephens, N.; Duignan, P.; Salgado, C.; Smith, H.; Bejder, L.; Linke, T.; Daniel, C.; Lo, H. Technical Report on the Bottlenose Dolphin (Tursiops Aduncus) Unusual Mortality Event within the Swan Canning Riverpark, June–October 2009; Murdoch University: Perth, Australia, 2010. [Google Scholar]
- Rowe, L.E.; Currey, R.J.; Dawson, S.M.; Johnson, D. Assessment of epidermal condition and calf size of Fiordland bottlenose dolphin Tursiops truncatus populations using dorsal fin photographs and photogrammetry. Endanger. Species Res. 2010, 11, 83–89. [Google Scholar] [CrossRef]
- Duignan, P.J.; Stephens, N.S.; Robb, K. Fresh water skin disease in dolphins: A case definition based on pathology and environmental factors in Australia. Sci. Rep. 2020, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.E.; Hurley, B.J.; Toms, C.N.; Mackey, A.D.; Solangi, M.; Kuczaj, S.A. Hurricane impacts on the foraging patterns of bottlenose dolphins Tursiops truncatus in Mississippi Sound. Mar. Ecol. Prog. Ser. 2013, 487, 231–244. [Google Scholar] [CrossRef]
- Fandel, A.D.; Garrod, A.; Hoover, A.; Wingfield, J.; Lyubchich, V.; Secor, D.; Hodge, K.; Rice, A.; Bailey, H. Effects of intense storm events on dolphin occurrence and foraging behavior. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- O’Hagan, A. Expert knowledge elicitation: Subjective but scientific. Am. Stat. 2019, 73, 69–81. [Google Scholar] [CrossRef]
- Brown, B.B. Delphi Process: A Methodology Used for the Elicitation of Opinions of Experts; Rand Corp.: Santa Monica, CA, USA, 1968. [Google Scholar]
- O’Hagan, A.; Buck, C.E.; Daneshkhah, A.; Eiser, J.R.; Garthwaite, P.H.; Jenkinson, D.J.; Oakley, J.E.; Rakow, T. Uncertain Judgements: Eliciting Experts’ Probabilities; John Wiley & Sons: Hoboken, NJ, USA, 2006. [Google Scholar]
- European Food Safety Authority. Guidance on Expert Knowledge Elicitation in Food and Feed Safety Risk Assessment; European Food Safety Authority (EFSA): Parma, Italy, 2014.
- Knol, A.B.; Slottje, P.; van der Sluijs, J.P.; Lebret, E. The use of expert elicitation in environmental health impact assessment: A seven step procedure. Environ. Health 2010, 9, 19. [Google Scholar] [CrossRef]
- MacMillan, D.C.; Marshall, K. The Delphi process–an expert-based approach to ecological modelling in data-poor environments. Anim. Conserv. 2006, 9, 11–19. [Google Scholar] [CrossRef]
- Sivle, L.D.; Kvadsheim, P.H.; Curé, C.; Isojunno, S.; Wensveen, P.J.; Lam, F.-P.A.; Visser, F.; Kleivane, L.; Tyack, P.L.; Harris, C.M. Severity of expert-identified behavioural responses of humpback whale, minke whale, and northern bottlenose whale to naval sonar. Aquat. Mamm. 2015, 41, 469. [Google Scholar] [CrossRef]
- Aspinall, W. A route to more tractable expert advice. Nature 2010, 463, 294. [Google Scholar] [CrossRef] [PubMed]
- Lenton, T.M.; Held, H.; Kriegler, E.; Hall, J.W.; Lucht, W.; Rahmstorf, S.; Schellnhuber, H.J. Tipping elements in the Earth’s climate system. Proc. Natl. Acad. Sci. USA 2008, 105, 1786–1793. [Google Scholar] [CrossRef]
- Bamber, J.L.; Aspinall, W. An expert judgement assessment of future sea level rise from the ice sheets. Nat. Clim. Chang. 2013, 3, 424. [Google Scholar] [CrossRef]
- Albert, I.; Donnet, S.; Guihenneuc-Jouyaux, C.; Low-Choy, S.; Mengersen, K.; Rousseau, J. Combining expert opinions in prior elicitation. Bayesian Anal. 2012, 7, 503–532. [Google Scholar] [CrossRef]
- Boobis, A.; Flari, V.; Gosling, J.P.; Hart, A.; Craig, P.; Rushton, L.; Idahosa-Taylor, E. Interpretation of the margin of exposure for genotoxic carcinogens–Elicitation of expert knowledge about the form of the dose response curve at human relevant exposures. Food Chem. Toxicol. 2013, 57, 106–118. [Google Scholar] [CrossRef]
- Booth, C.; Burgman, M.; Donovan, C.; Harwood, J.; Thomas, L.; Schick, R.; Wood, J. PCoD Lite-Using an Interim PCoD Protocol to Assess the Effects of Disturbance Associated with US Navy Exercises on Marine Mammal Populations; DTIC Document: St Andrews, UK, 2014. [Google Scholar]
- Tollit, D.; Harwood, J.; Booth, C.; Thomas, L.; New, L.F.; Wood, J. Cook Inlet Beluga Whale PCoD Expert Elicitation Workshop Report. Prepared by SMRU Consulting North America for NOAA Fisheries; SMRU Consulting: Friday Harbor, WA, USA, 2016. [Google Scholar]
- Martin, T.G.; Burgman, M.A.; Fidler, F.; Kuhnert, P.M.; Low-Choy, S.; McBride, M.; Mengersen, K. Eliciting expert knowledge in conservation science. Conserv. Biol. 2012, 26, 29–38. [Google Scholar] [CrossRef]
- Runge, M.C.; Converse, S.J.; Lyons, J.E. Which uncertainty? Using expert elicitation and expected value of information to design an adaptive program. Biol. Conserv. 2011, 144, 1214–1223. [Google Scholar] [CrossRef]
- Morgan, M.G. Use (and abuse) of expert elicitation in support of decision making for public policy. Proc. Natl. Acad. Sci. USA 2014, 111, 7176–7184. [Google Scholar] [CrossRef] [PubMed]
- Kynn, M. The ‘heuristics and biases’ bias in expert elicitation. J. R. Stat. Soc. Ser. Stat. Soc. 2008, 171, 239–264. [Google Scholar] [CrossRef]
- Kahneman, D. Thinking, Fast and Slow; Macmillan: London, UK, 2011. [Google Scholar]
- Gosling, J.P. SHELF: The Sheffield elicitation framework. In Elicitation; Springer: Berlin/Heidelberg, Germany, 2018; pp. 61–93. [Google Scholar]
- Astfalck, L.; Cripps, E.; Gosling, J.; Hodkiewicz, M.; Milne, I. Expert elicitation of directional metocean parameters. Ocean. Eng. 2018, 161, 268–276. [Google Scholar] [CrossRef]
- Pasanisi, A.; Keller, M.; Parent, E. Estimation of a quantity of interest in uncertainty analysis: Some help from Bayesian decision theory. Reliab. Eng. Syst. Saf. 2012, 100, 93–101. [Google Scholar] [CrossRef]
- Hart, A.; Gosling, J.P.; Quigley, J.; Revie, M.; Thulke, H.H.; Brock, T.; O’Hagan, T.; Charlton, C. Training courses on “Steering an Expert Knowledge Elicitation” and “Use of the Expert Knowledge Elicitation Guidance in Risk Assessments for EFSA Management” and “Conduct of the Sheffield protocol for an Expert Knowledge Elicitation”. EFSA Supporting Publ. 2018, 15, 1450E. [Google Scholar] [CrossRef]
- Renooij, S. Probability elicitation for belief networks: Issues to consider. Knowl. Eng. Rev. 2001, 16, 255. [Google Scholar] [CrossRef]
- Carey, J.M.; Burgman, M.A. Linguistic uncertainty in qualitative risk analysis and how to minimize it. Ann. N. Y. Acad. Sci. 2008, 1128, 13–17. [Google Scholar] [CrossRef]
- Hora, S.C. Acquisition of expert judgment: Examples from risk assessment. J. Energy Eng. 1992, 118, 136–148. [Google Scholar] [CrossRef]
- R Core Team R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016.
- Fazioli, K.L.; Hofmann, S.; Wells, R.S. Use of Gulf of Mexico coastal waters by distinct assemblages of bottlenose dolphins (Tursiops truncatus). Aquat. Mamm. 2006, 32, 212. [Google Scholar] [CrossRef]
- Fury, C.A.; Harrison, P.L. Impact of flood events on dolphin occupancy patterns. Mar. Mammal Sci. 2011, 27, E185–E205. [Google Scholar] [CrossRef]
- Hoffland, T.; Yeater, D.B.; Kuczaj II, S.A.; Solangi, M. Importance of social relationships in a group of Bottlenose dolphins (Tursiops truncatus) during a natural disaster. Aquat. Mamm. 2017, 43, 391. [Google Scholar] [CrossRef]
- Hickey, B.; MacCready, P.; Elliott, E.; Kachel, N. Dense saline plumes in Exuma sound, Bahamas. J. Geophys. Res. Ocean. 2000, 105, 11471–11488. [Google Scholar] [CrossRef]
- Manzello, D.P.; Enochs, I.C.; Melo, N.; Gledhill, D.K.; Johns, E.M. Ocean acidification refugia of the Florida Reef Tract. PLoS ONE 2012, 7, e41715. [Google Scholar]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Booth, C.; Thomas, L. An Expert Elicitation of the Effects of Low Salinity Water Exposure on Bottlenose Dolphins. Oceans 2021, 2, 179-192. https://doi.org/10.3390/oceans2010011
Booth C, Thomas L. An Expert Elicitation of the Effects of Low Salinity Water Exposure on Bottlenose Dolphins. Oceans. 2021; 2(1):179-192. https://doi.org/10.3390/oceans2010011
Chicago/Turabian StyleBooth, Cormac, and Len Thomas. 2021. "An Expert Elicitation of the Effects of Low Salinity Water Exposure on Bottlenose Dolphins" Oceans 2, no. 1: 179-192. https://doi.org/10.3390/oceans2010011
APA StyleBooth, C., & Thomas, L. (2021). An Expert Elicitation of the Effects of Low Salinity Water Exposure on Bottlenose Dolphins. Oceans, 2(1), 179-192. https://doi.org/10.3390/oceans2010011






