Abstract
Vascular-free flaps are the most frequently employed method in cases of jaws reconstruction after large tumor removal. This therapeutic choice is due to its intrinsic blood supply, which speeds up recovery and enables healing apart from the injured recipient site. This method is most commonly used in secondary reconstructions brought on by radiation, infection, or prior surgical intervention. In fact, with the help of 3d technology, guides can be designed to perform the grafting very smoothly. The purpose of this review with meta-analysis is to compare the two methods: the one using 3d and the freehand one. Methods: In accordance with the entered keywords, a literature search was conducted on PubMed, Lilacs, and Web of Science. Articles published between 2000 and 4 January 2023 were taken into consideration. Using the Boolean connective AND, the terms “jaw reconstruction”, “cad-cam”, and “cancer” have been joined. Results: After searching the three search motors, 148 articles were selected, and only 7 were chosen for this review. Conclusions: The pre-operative morphological position looks to be accurately restored using the CAD/CAM reconstructive methodology. Reconstruction using CAD-CAM and 3d technology allows for greater accuracy, less operative time, and improved aesthetics.
1. Introduction
Numerous diseases might result in mandibular abnormalities that need to be repaired. Resection may be necessary to remove malignant or benign tumors, and abnormalities of the jaw may affect function. Additionally, sickness or infection may develop in the bone, and trauma may result in fractures. These factors may influence chewing capacity and aesthetics. Jewer et al. established a way of classifying these anomalies depending on whether they affected the central or lateral sections of the mandible [1]. While lateral defects can be repaired with a single straight bone, main faults require repeated osteotomies. The best course of action is to allow for accessible airways while regaining both function and appearance, including chewing, speech, and swallowing.
The usual mandibular reconstruction (MR) method entails moving the bone from a donor site to a recipient site. Autologous bone grafts are fastened with titanium plates. The two main surgical methods available are bone grafts with and without blood vessels. Vascular-free flaps are most frequently employed because they have blood vessels, which speeds up recovery and enables healing apart from the injured recipient site. This method is most commonly used in secondary reconstructions brought on by radiation, infection, or prior surgical intervention. The fibula-free flap (FFF), developed by Hidalgo in 1989, is the most widely used vascularized graft. Due to the fibula’s size and structure, FFF reconstructions offer low donor site morbidity, high caliber blood arteries, and the ability to mold the bone to mimic the natural mandibular contour [2]. The deep circumflex iliac artery (DCIA), developed by Taylor et al. [3], involves harvesting a portion of the iliac bone together with a part of the circumflex iliac artery and the scapular osteocutaneous free flap (SOFF), developed by Swartz et al. [4]. The capacity to replicate the native mandibular shape has been demonstrated by using numerous osteotomies, particularly in abnormalities bigger than 6 cm [3].
Less information is published in the literature about non-vascularized grafts such as iliac bone or costochondral ribs. They are utilized for smaller lesions in which soft tissue is unaffected; trauma is the most frequent cause of these problems. It has been noted that the lack of an intrinsic blood supply slows healing, raising the risk of infection and postoperative problems. Additionally, radiation therapy makes these flaps more vulnerable to osteoradionecrosis when treating malignant tumors [5].
Virtual Planning Using CAD-CAM
New approaches utilizing new technology have been developed to address these difficulties, which are being caused by the higher incidence of preoperative and postoperative complications and the overall increased operation time seen in traditional MR. The usage of 3d printing techniques in MR surgery has increased since Hirsch et al.’s initial report in the literature in 2009 [6]. This technique uses numerous two-dimensional computed tomography (CT) scans of the mandibular region. These scans are converted to 3d models in STL format and saved as DICOM files. Then, using one of seven 3d printing processes, this 3d model is created layer by layer. This technology has been more widely accessible over the past ten years, and hospitals have begun to develop multidisciplinary workflows. For patients treated using CAD/CAM technology and those treated conventionally, Bolzoni et al. [7] discovered that the total cost of care for various treatment programs was comparable. Multiple applications have been developed thanks to little cost differences and improved accessibility. In addition to simulating surgery, 3d models can be fixed with titanium plates around the harvest. Alternatively, dental implants and occlusal splints can be prototyped, and titanium plates can be customized and 3d-printed [8]. With CAD/CAM, options that are not possible with traditional methods are possible, such as the capacity to mirror the unaffected mandible for improved mandibular symmetry, the planning of osteotomy measurements for resection, and the potential to restore proper occlusion. These innovations lessen the need for a surgeon’s specific skill set. When using CAD/CAM techniques, several outcomes have been discussed in the literature, including accuracy, postoperative complications, aesthetics, and cost [9]. This review aims to evaluate the accuracy of the reconstructive process performed by 3d and CAD/CAM technology in terms of surgical precision; in fact, a meta-analysis of the literature regarding the primary studies was performed. In addition, a systematic review of the preliminary studies regarding the operative time and accuracy of the 3d technique compared to freehand technology was performed.
2. Materials and Methods
2.1. Eligibility Criteria
The following Population, Exposure, Comparator, and Outcomes (PICO) criteria were used to determine the admissibility of all documents:
- (P) Participants are patients with oral cancer and operated mandibular/jaw resection who need reconstructive surgeries.
- (I) Interventions are patients with cancer and resection surgery who have undergone reconstruction surgery using 3d technology and CAD/CAM.
- (C) Comparisons are patients with cancer and resection surgery who have undergone reconstruction surgery using a fibula or scapula graft with the conventional technique.
- (O) The outcome is to evaluate the accuracy of the 3d surgical design regarding graft precision. The secondary effect is to assess the surgery’s aesthetic impact and working time. The following inclusion criteria were employed for this meta-analysis: (1) randomized clinical trial (RCT); (2) patients over 18 years of age; (3) comparative studies; (4) diagnosis of oral cancer without the distinction of the stage; (5) need for reconstruction surgery via a vascularized flap; (6) papers published in English.
The exclusion criteria were: (1) studies involving animals; (2) review articles; (3) case reports; (4) lack of practical statistical analysis.
2.2. Search Strategy
A literature search was performed on PubMed, Lilacs, and Web of Science, and articles published from 2000 to 1 April 2023 were considered according to the keywords entered. The terms “jaw reconstruction”, “CAD-CAM”, and “cancer” and the Boolean connection AND has been used to combine [10]. MESH (Medical Subjects Headings) was used to help with the web search (Table 1). We followed the Cochrane Handbook for Systematic Reviews of Interventions and the PRISMA guidelines 2020 for this review and registered the systematic review protocol on PROSPERO with the number CRD42023418080.

Table 1.
Search strategy.
2.3. Data Extraction
Using a specialized data extraction tool on a Microsoft Excel sheet, two reviewers (GM and RF) independently retrieved data from the included papers. A third reviewer assisted to reach a consensus where there was disagreement (MC).
The following data were obtained: (1) First Author; (2) Year; (3) Sample; (4) Nationality; (5) Type of surgery; (6) Results between intergonial distance infra group.
2.4. Quality Assessment
Two reviewers assessed the bias risk of the papers using the Cochrane risk-of-bias methodology for randomized trials, Version 2 (RoB 2). Any disagreement was discussed until a consensus was reached with a third reviewer.
2.5. Statistical Analysis
The pooled analysis used the program Review Manager 5.2.8. (Cochrane Collaboration, Copenhagen, Denmark; 2014). In the study, patients with freehand and 3d vascularized flaps were contrasted. The difference in the risk between the two groups was calculated. Using the Higgins Index (I2) and the chi-square test, study heterogeneity was measured and classified as low heterogeneity (30 percent), medium heterogeneity (30–60 percent), and high heterogeneity (>60 percent).
3. Results
3.1. Study Characteristics
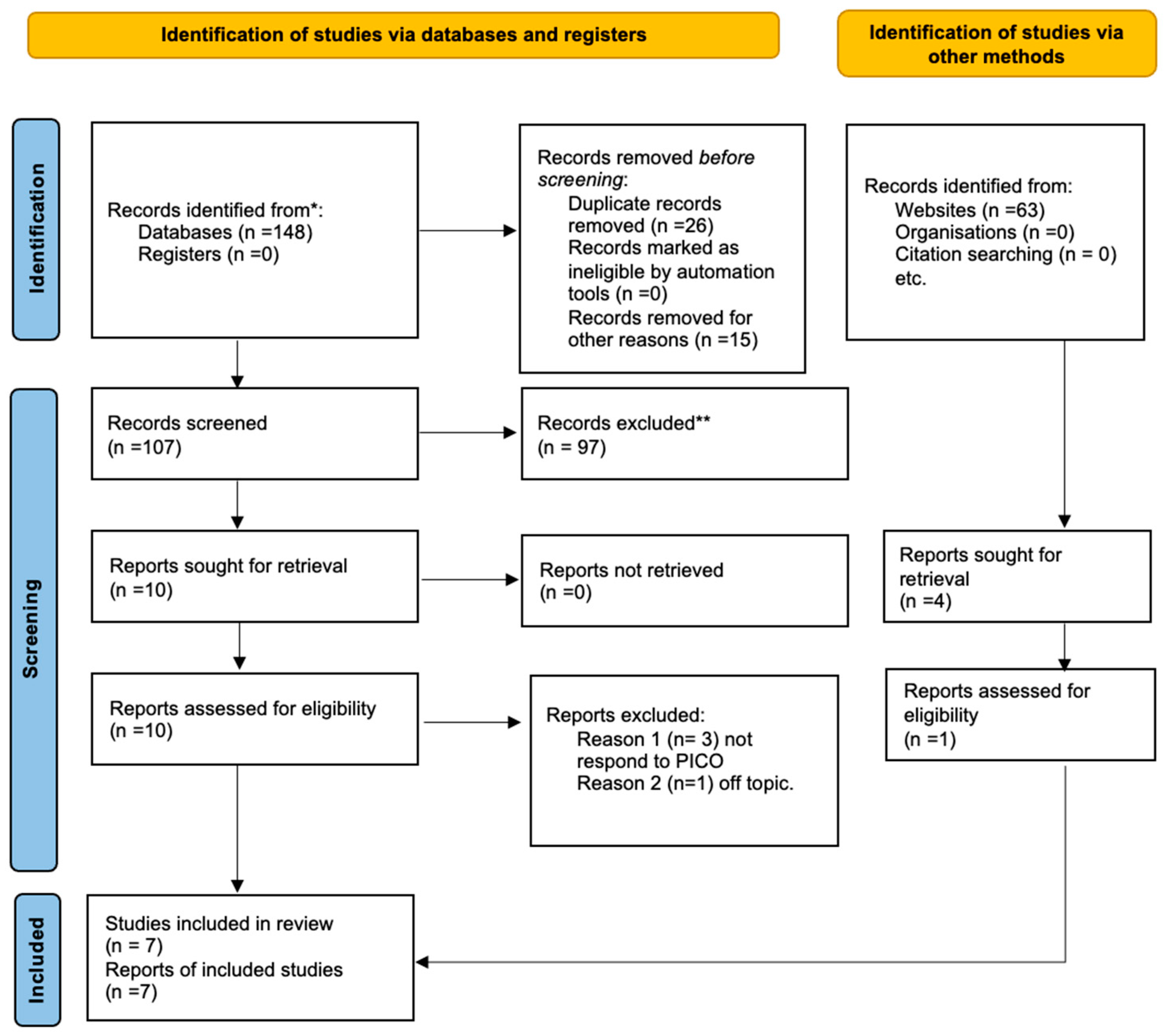
A total of 148 articles were chosen after using all three search engines. Reports that were not in English were eliminated manually. In particular, 15 articles from PubMed were excluded. Due to their duplication, 26 papers were eliminated. A total of 97 publications were eliminated during the first screening phase from the 107 that were taken into consideration based on the inclusion criteria for clinical trials, randomized controlled trials, and comparative studies. Following this initial screening, 10 papers were selected; eligibility was determined by reading the abstracts. Only 6 articles—out of 106—were selected for this review, according the PRISMA 2020 flowchart in Figure 1. One article was deemed off-topic, and three articles failed to reply to PICO. In addition, a manual search of the bibliography, Embase, and Clinical trial.gov was performed, and one article was found that met the inclusion criteria. According to the PICO model, seven papers were chosen for title and abstract screening. A Cochrane library literature search was performed, from which no clinical trials emerged. The studies range from 2016 to 2022 and have been conducted in different parts of the world, including China, Italy, Germany, and the USA. A total of 516 patients were analyzed and divided into clinical study, comparative study, retrospective study, and randomized, double-blind manner in a study group in which maxillary and mandibular reconstruction was performed using 3d reconstruction techniques and in a study group in which surgery was performed freehand. Moreover, in the seven studies analyzed, only three were used for the meta-analysis in which the intergonial distance between the study group and the control group post-surgery was evaluated. In the other studies, complications and functional management were assessed only by comparing the two techniques regarding operative time. All interventions were performed with different software and different methods. However, the rationale and operative procedure remain the same. Therefore, the number of patients analyzed in the meta-analysis that compared the two groups by intergonial distance was 62 in the study group and 59 in the control group.
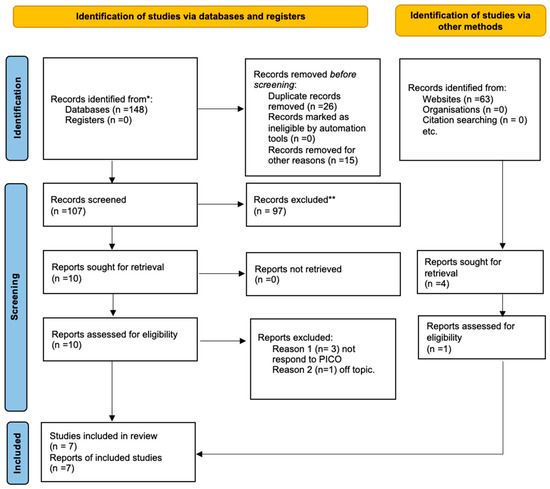
Figure 1.
Prisma flowchart [10]. * papers identified by search methods; ** papers removed because systematic reviews of the literature.
To start the virtual planning process, a high-resolution CT scan of the craniofacial area and the lower leg or ankle as a potential donor site was acquired; the surgeons were to process the DICOM format data. The surgeon turned the craniofacial skeleton and the donor location into 3d virtual models using this software. The surgeon can plan mandibular and fibular osteotomies thanks to the program. To achieve the optimal aesthetic contour and prevent bone abnormalities on the side afflicted by the tumor, the outer surface of the healthy side of the jaw was thickened to create the bespoke reconstructive titanium plate that supported the bony-free flap. It was secured to the original mandible using a locking mechanism.
The surgeon can then accurately segment the osseous free flap using a specially made cutting guide for the bony free flap. A 3d printer was used to create cutting guides out of polyamide.
3.2. Operative Time and Comfort
At the Department of Otolaryngology and Oncology, ENT Poznan University, eight patients were treated between 2013 and 2015 due to an advanced stage of oral cavity squamous cell carcinoma. Bony-free flaps taken from the scapula (four patients) and fibula (four patients) with skin islands were used to restore hard and soft tissue abnormalities. A physical 3d model was created for four patients needing mandibular reconstruction, consisting of structurally sound and reconstructible sections. High-definition CT scans served as the basis for the 3d models’ creation. All eight treatments resulted in fully healed wounds and were rated effective. Good healing of the complex and soft tissues was achieved in four patients, where a physical model was created using a three-dimensional ABS approach. Free fibular flaps with skin islands were employed in four patients with mandibular and soft tissue abnormalities, and scapular flaps with skin were used in the other four patients. The surgery took an average of 6.5 h in the group using the planned 3d model and 8.5 h in the group using the traditional reconstructive plan. After eight weeks, a comparative examination of both groups’ functionality (joint stability, mandibular mobility, and mastication ability) and cosmetic results was conducted. In contrast to conventional surgery (10.5–12.5 degrees), differential angles were lower in patients with 3d modeling (7.3–9.1 degrees) than in those without. Before and following the surgical intervention, the patients accepted identical mandibular shapes and aesthetics [11].
May et al. identified 264 individuals who received free fibular flap (FFF) reconstructive surgery for mandibular abnormalities between 2000 and 2018 for the study. A total of 32 patients received 3d (3d/VSP) reconstruction, while 230 patients received freehand FFF mandibular reconstruction. Patients under 18 and those without adequate postoperative imaging (CT maxilla/face, PET/CT, or Panorex) within two years of follow-up (n = 4) were excluded from the study. Evaluations were made regarding the patient’s demographics, co-morbidities, pathology, neoadjuvant or adjuvant therapy, overall operating times, and reconstruction timeframes. Secondary outcomes included the surgery time. A total of 28 of the 260 patients that were investigated received FFF with 3d/VSP, compared to 232 who underwent CM FFF reconstruction. Compared to CM, the bony union was considerably higher with 3d/VSP (p = 0.036). In contrast to the 3.6% nonunion rate in the 3D/VSP, the author discovered a 20% nonunion rate (46/232) in the CM. FFF complication rates were 38% (87/232) in the CM group, compared to 11% (3/28) in the 3d/VSP group, which was a statistically significant difference (p = 0.005). Age, surgical indication, sex, and (neo)adjuvant therapy adjustments revealed that patients with 3d and VSP had an estimated risk of FFF complication that was roughly 73% lower than that of patients with CM. In 3d/VSP patients, there was a considerably lower overall risk of fracture and other complications (39% vs. 63%, 11/28 vs. /232, p = 0.023). CM required 562 min (9.4 h) of total operating time compared to 3d/VSP’s 491 min (8.2 h), with reduced variability (118 vs. 124 min). Again with less variability, the total reconstructive time was 250 min as opposed to 294 min (70 min as opposed to 90 min) [12].
Patients who had fibular flap reconstructions of the maxilla and mandible were included in the retrospective study. A radiographic study was assessed to evaluate the ossification status of the jaw. Following virtual surgical planning (VSP), osteosynthesis for jaw reconstruction was carried out either with freehand plates (non-PSI) or 3d virtual surgery (PSI). Laser-melted CAD/CAM-PSI titanium plates were compared to traditional osteosynthesis. The conventional group cut freehand osteotomies to form fibula portions that had been extracted. For the fixation of jaw reconstruction, the patient’s medical records were assessed by the plate system being utilized and divided into two groups: non-PSI and patient-specific (PSI). The ossification states of each junction zone native bone-to-fibula were all collected. The degree of the osseous union was divided into two categories: complete (COU) and incomplete (IOU). A total of 133 patients who met the predetermined inclusion criteria and underwent successful FFF reconstruction of the jaw out of 169 patients were included in the current study. A few cases (n = 36) had to be excluded since the inclusion criteria were not met.
A total of 30 patients (22.6%) had their maxilla reconstructed, while 103 patients (77.3%) had their mandible rebuilt.
All radiographic findings were examined for the condition of the junction zone’s osseous union and classified according to the type of osteosynthesis used. Incomplete bony union was present in both groups at a rate of nearly 90% in the first six months, according to radiographic imaging [13].
A clinical comparison study was conducted to assess the efficacy of the malleolus cap design. The only independent factor was whether a fibula malleolus cap that was 3d-printed for each patient was used to position the harvest guide intraoperatively and precisely. The same chief surgeon performed on every patient in a single facility. Yang et al. reported patient-specific titanium implants used in computer-assisted jaw reconstruction.
The recipient jaw and the donor fibula were modeled in three dimensions using CT data that were collected and segmented. Using the program’s built-in best-fit computation, the postoperative fibula model was superimposed onto the preoperative fibula. The distal remaining fibula’s osteotomy end was chosen as the best place to fit; that plane is the actual osteotomy plane. The postoperatively reconstructed jaw was superimposed on the preoperative plan with the best match of the unoperated portion of the natural mandible [14] (Table 2).

Table 2.
Main characteristics of studies included in the present systematic review.
3.3. Evaluation of Angle
The study by Yang et al. sought to determine whether using surgical plates that were 3d-printed specifically for each patient would increase the accuracy of head and neck reconstruction over traditional plates.
Patients were prospectively enrolled into the comparison trial’s study group using surgical plates that were explicitly 3d-printed for the patients. The patients in the control group with traditional surgical dishes belonged to the same unit’s previous cohort. The precision of head and neck reconstruction was assessed. A total of 33 patients were included in the trial, with 17 assigned to the experimental group and 16 assigned to the control group. The baseline features of the patients were comparable across the two groups. The deviations of the mandible or maxillae were 1.5 mm in the study group and 2.1 mm in the control group (mean difference, −0.7 mm; 95% confidence interval (CI), −1.1 to −0.3; p = 0.003), demonstrating better reconstruction accuracy for 3d patients.
Additionally, bone graft precision and bilateral mandibular angles were improved throughout the repair process. The accuracy of the osteotomy was comparable between the two groups regarding the secondary end goals. No differences were discovered in terms of intraoperative blood loss, overall operating time, or hospital stay [15].
The study by Tarsitano et al. evaluated the accuracy in terms of intergonial distance between jaw reconstruction by 3d and by freehand surgery. A preoperative and postoperative CT scan was used to analyze each patient. A CT scan was used to examine several anatomical landmarks to assess the morphological findings: (1) The midline deviation; (2) The amplitude fluctuation of the mandibular angle in grades; (3) The mandibular bi-gonial diameter; and (4) The chin protrusion. The test group considerably outperformed the control group in terms of the mandibular angle (p = 0.034), bi-gonial diameter (p = 0.041), and chin protrusion (p 14 0.05) mean differences between pre-operative and post-operative CT scans. Midline deviation showed no statistically significant variations (p = 0.092) [16]. The study of Chen et al. aimed to assess the precision of fibular flap mandibular repair guided by 3d CAD/CAM and to investigate potential sources of discrepancies. Twenty-eight patients received fibular flap mandibular repair between June 2015 and December 2016. A total of 13 patients underwent traditional freehand procedures, and 15 patients had virtual planning and customized guided templates used. Deviations during mandibulectomy and fibular osteotomy, as well as the overall and triaxial deviation of the appropriate mandibular anatomical landmarks, were measured between virtual planning and post-surgery. The resection line and angle deviations were 1.23–0.98 mm and 4.11 2.60°, respectively. Patients who underwent ramus reconstruction experienced ipsilateral Co. and Go. deviations of 9.79 4.74 mm vs. 3.57 1.62 mm (p 0.05) and 15.17 6.53 mm vs. 4.36 1.68 mm (p 0.05), respectively, in the freehand group and CORPPP group [17] (Table 3).

Table 3.
Characteristic of the studies included in the meta-analysis.
3.4. Meta-Analysis
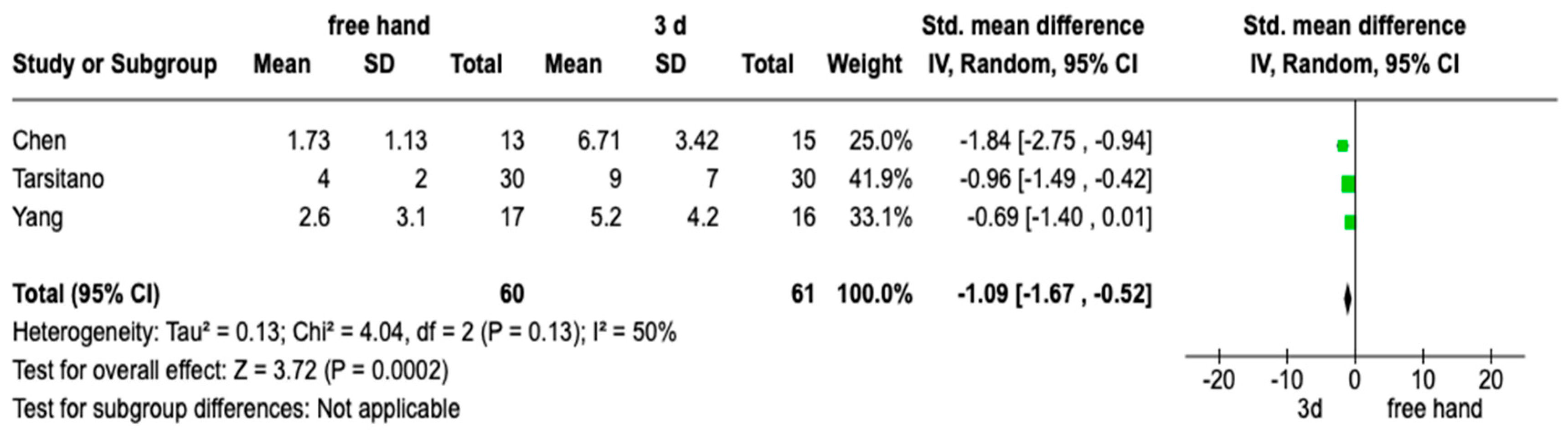
The meta-analysis was conducted by a random model effect because of the high heterogeneity (I2 = 50%) between the three included studies. For the meta-analysis, the intergonial distances between study groups and control groups were considered as common parameters, and therefore, of the seven selected RCTs, only three have these parameters in common in order to conduct a meta-analysis. The overall impact reported in the forest plot (Figure 2) shows that the 3d model technique is an excellent aid for treating maxillary and jaw reconstruction (OR: −1.09; 95% CI: −1.67, −0.52; Z: 3.72; p = 0.0002).
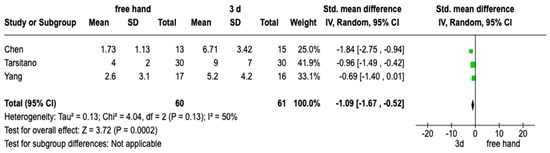
Figure 2.
Forest plot of the meta-analysis performed on the three studies. Random effects model was used; 95% confidence interval was set.
3.5. Assessed the Risk of Bias
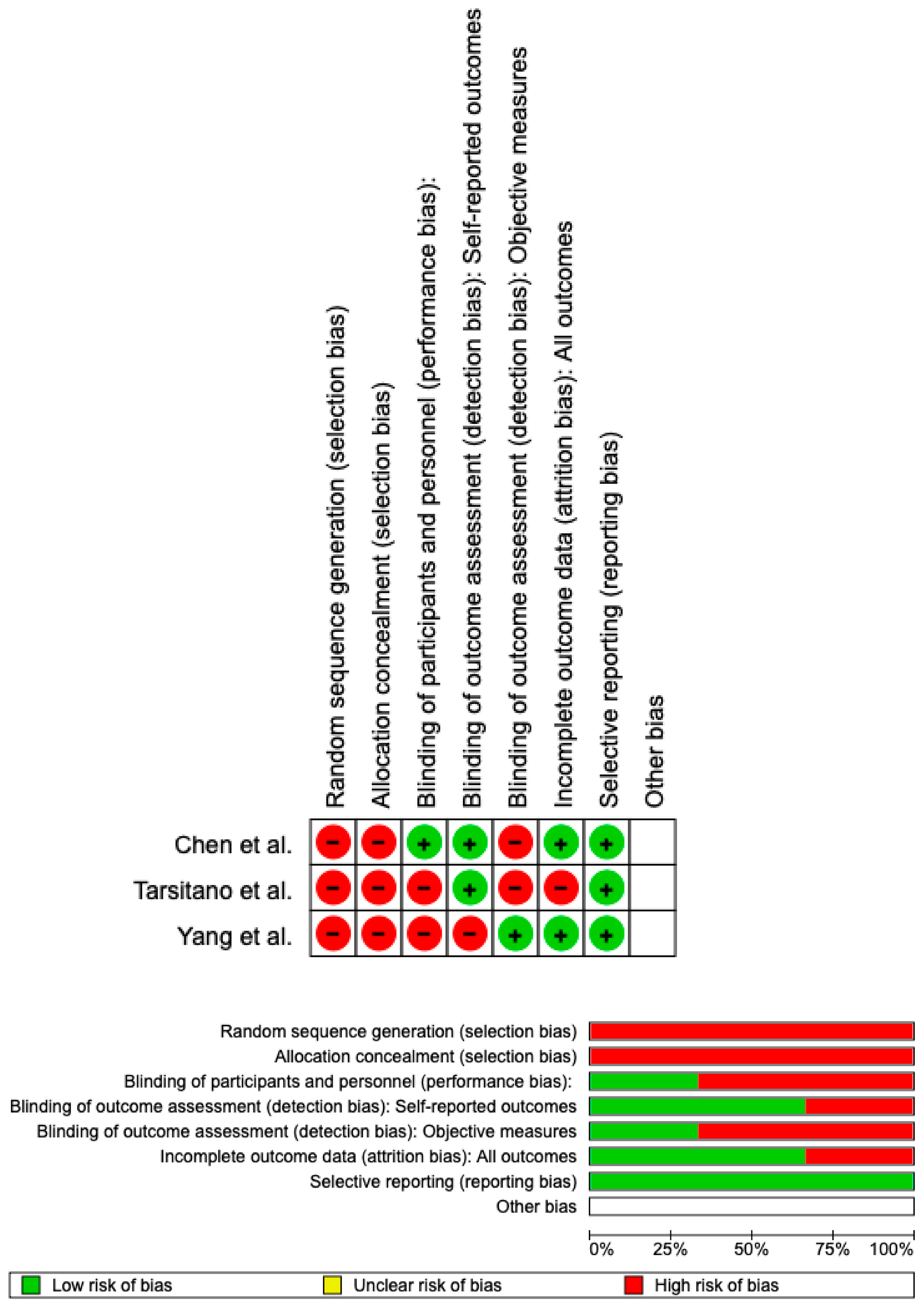
The risk of bias was calculated using RoB 2 and displayed in Figure 3. A low risk of bias was guaranteed with respect to the randomization procedure in all of the studies. Although all the studies included in the analysis properly removed bias in the choice of reported outcomes, only 25% of the studies excluded bias in self-reported outcomes. However, all studies included in the analysis reported full outcome data. Overall, it was determined that there was little chance of prejudice for all three.
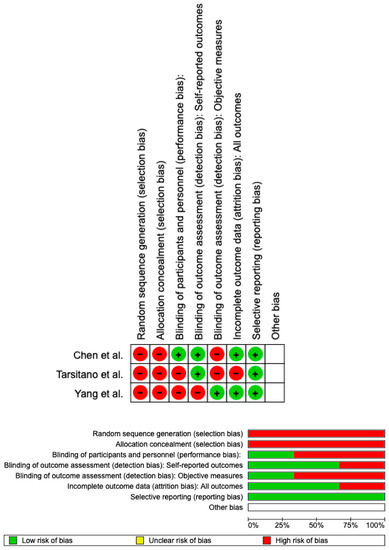
Figure 3.
Risk of bias; green is a low risk of bias; red is a high risk of bias.
4. Discussion
Reconstructive surgery aims to preserve chewing function, restore the lower third of the face’s features, maintain proper occlusion, and open up the potential of placing dental implants. A good quality of life can be maintained with appropriate temporomandibular joint mobility [18]. Complication rates for the fibular free flaps are significant, ranging from 7 to 69% (plate exposure, fistulas). Contrarily, nonvascular bone grafts in reconstructive surgery have substantial drawbacks; they are only used in small defects (less than 5 cm). Currently, in cases with substantial mandibular excision, free vascularized flaps continue to be the gold standard. The reconstruction of significant segmental mandibular deficits is possible. Free flaps had a rapid osteointegration and a low risk of fracture [19].
Additionally, if additional radiotherapy becomes necessary, flaps provide no limitations to the patient. The literature extensively describes how mandibular reconstruction procedures can be planned accurately utilizing 3d printing to produce good aesthetic and functional results. Modern reconstructive surgical methods, however, necessitate close coordination between the surgeons, a team of engineers creating 3d-printed models, and the radiologists. The group in which the 3d model was used has excellent mandibular symmetry, according to the results of the measured angles. Different authors give remarkably similar findings [20,21,22]. In all operated patients who used 3d model printing, the complete stability of the mandibular connection with the preservation of osseointegration was established during the follow-up examination, performed eight weeks after the surgical operation [23,24]. It permitted entire temporomandibular joint movement and the conservation of masticatory function. Compared to conventional mandibular osteotomies and reconstructions, the 3d model printing technology allowed for a surgical procedure time reduction of roughly 2.0 h [25]. Compared to the conventional method, it may result in a longer pre-surgical approach (an additional three days). Additionally, compared to traditional reconstructive techniques, therapy costs are much higher when new technological solutions are used [26,27]. However, as technology advances quickly, it becomes less expensive to use new software, 3d-printing hardware, and materials, making it easier to employ these methods [28].
Compared to conventional methods of freehand contouring, 3d model printing and VSP (3D/VSP) had an essential osteointegration of the fibular flap. Significantly less time was spent on the entire surgical procedure and the rebuilding of 3d/VSP patients [29,30]. The time to the bony union also considerably decreased, statistically, from 1.4 years with conventional methods to 0.8 years with the 3D/VSP. Precision osteotomies using the stereo lithic cutting guides increase the amount of bone-to-bone contact between the mandible and fibular bone segments, likely improving the bony union [31].
The mandibular union typically takes 4 to 8 weeks [32]. Previous research on craniofacial trauma has shown that nonunion can worsen the life of patients.
With 3D models, the surgeon can better plan the resection, predict and see the defect, and tailor the soft tissue and bone flap components to the fault before inserting the flap into the patient. Less anaesthesia time, less surgeon fatigue, and fewer complications might result from the shorter operating and reconstructive times. 3d/VSP increased inoperative effectiveness, accuracy, and precision [33]. In patients with complicated anatomy or pathologies, this might be helpful. The potential for the degree of resection to change is a drawback of custom reconstruction plates. This can change the osteotomy location, making a specially made plate useless. The osseous union indicates ideal healing at the neo-jaw location after the fibula-free flap repair of the jaws. Bone union is important to prevent fractures [34].
Osteosynthesis plates’ artifacts may hurt quality. CBCT has been explicitly used in our patient pool to plan dental rehabilitation. For oncological follow-up, CT scans with slice thicknesses of up to 3 mm were obtained in the past.
Meanwhile, the slice thickness has dramatically decreased, enabling a more accurate evaluation of the segmental gaps. The rationale for CT exams following reconstruction in benign conditions is extremely rigorous for radiation protection concerns. Hence, they were not commonly performed.
Despite the constraints of the retrospective study design, our findings show that poly-segmental reconstructions using CAD/CAM-PSI had a greater rate of inadequate bony fusion between the flap bone segments than those with native bone alone (p = 0.003). After jaw reconstruction with FFF, Hashemi et al. [35] examined 38 osteotomy sites in n = 13 patients and discovered comparable outcomes. They found that neither the internal nor the intermediate gap sites approached statistical significance independently. Accordingly, the study of Yang et al. compared conventional surgical plates, and 3d-printed patient-specific plates reduce the distance and imprecision in terms of the distance and angulation of the reconstruction [36]. This is the first study to compare the accuracy of outcomes for head and neck reconstruction using 3d-printed patient-specific versus traditional surgical plates [37].
Al Maruf can examine head and neck reconstruction accuracy with a 3d model by contrasting postoperative reconstruction results with preoperative virtual designs thanks to computer technology. The correctness of the reconstruction was rigorously evaluated in the current study using several metrics.
To improve precision, decrease morbidity, and shorten recovery times during mandibular reconstruction, CAD/CAM technologies are applied [38,39]. Planning and carrying out mandibular reconstruction now have many new prospects because of the design and production of patient-specific reconstruction plates, especially when combined with cutting guides. A straight fibular flap can be shaped using specialized surgical cutting guides to mimic a neo-mandible. To make the insertion of an implant-supported prosthesis easier, the fibular osseous fragment is virtually selected and oriented optimally [40]. It is possible to precisely estimate the length of the free flap’s bony component and the vascular pedicle’s width if mandible-wide restoration is necessary.
The precision of bone segmentation can be increased by utilizing a piezoelectric saw, which provides high procedural control. A special laser-sintering plate can accurately reproduce the natural mandibular contour. Despite the availability of numerous plate designs, it is crucial to precisely move the intended plate and bony graft to the surgical site. This calls for CAD/CAM-produced guidance. The vital connections between virtual planning and surgery are guides. Guide design and precision are essential to make a reconstructive plan a reality [20,41].
The study of Chen et al. indicated that the CAD/CAM group had a sizable advantage in restoring the mandibular arch. Every patient in the control group showed a decreased bone diameter, indicating a facial contraction of the mandibular external profile. This results in an apparent cosmetic change and variance in the occlusal arch, which makes oral rehabilitation more challenging. Personalized plates can significantly aid the surgeon in restoring the mandible’s transverse diameter. The bony surface can be fitted precisely using a special laser-sintering plate, replicating the natural mandibular contour.
Our microvascular success rate of 100% demonstrated the procedure’s safety. Clinical outcomes in tailored functional mandibular repair had been achieved with digital surgical technologies [42].
According to Xu, virtual surgery enables the modelling of all crucial preoperative steps, such as establishing the lesion resection range, planning the fibula osteotomy, and positioning and shaping the fibular segments. Models and templates for surgical guidance are designed using 3d printing technology. Additionally, it improves the predictability and efficiency of operations and aids the training of young surgeons. The preoperative virtual models were superimposed onto the reconstructed mandibles to calculate conventional absolute deviations [43]. The repaired mandible, however, was diverted by muscles and adapted to occlusion in the physiological state, resulting in physiological abnormalities on both the lesion side and the healthy side. Yang et al. talked about a unique approach to assessing physiological deviations. The temporomandibular joint (TMJ) variations substantially affected the lesion more than the healthy side in the 3d-printed plate group. Patients without ramus and condyle preservation also had considerably larger condyle and joint space deviations [43]. This outcome matched what we found in our research. The CORPPP group demonstrated significantly lower ipsilateral mandibular anatomical landmark deviations than the freehand group when we evaluated the physiological variation, indicating that the CORPPP technique enhanced the physiological position of the reconstructed mandible [44,45].
Key factors may have changed the results and are probably due to categorizations. To reduce selection error, anatomical landmarks on the residual mandible should be chosen in advance for measurement due to the significant shape differences between the mandible and fibula. As a result, the presence or absence of the ramus on the ipsilateral side may affect the deviation. Second, the resection range was thought to impact the variations of the reconstructed mandible, particularly the posterior portion. Therefore, we can say that reconstructive surgery using 3d technology has greater predictability, greater accuracy, and faster ossification between areas.
5. Conclusions
The CAD/CAM reconstructive technique, as shown in the meta-analysis, has greater accuracy and precision than the freehand technique. Virtual programming facilitates the creation of fibula models. This method also reduces problems and gives the surgeon greater procedural control. Although one study disagrees regarding the accuracy of 3d, this method and the literature review affirm that the use of 3d and CAD/CAM technology is beneficial in surgical treatment in terms of the effectiveness and duration of surgery. The only study in disagreement (Yang et al. [15]) points out that, regarding the intergonial distance, 3d technology is not highly accurate. However, we can conclude that the 3d technique is highly accurate for mandibular reconstruction.
Author Contributions
Conceptualization, S.C. and A.B.; methodology, R.F.; software, R.F.; validation, G.M., M.C. and R.F.; formal analysis, S.C.; investigation, R.F.; resources, M.C.; data curation, R.F.; writing—original draft preparation, G.M.; writing—review and editing, M.C.; visualization, M.C.; supervision, M.C.; project administration, R.F.; funding acquisition, M.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
All data described in the study are presented in the manuscript. The datasets analyzed are available from the corresponding author on reasonable request.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Jewer, D.D.; Boyd, J.B.; Manktelow, R.T.; Zuker, R.M.; Rosen, I.B.; Gullane, P.J.; Rotstein, L.E.; Freeman, J.E. Orofacial and Mandibular Reconstruction with the Iliac Crest Free Flap: A Review of 60 Cases and a New Method of Classification. Plast. Reconstr. Surg. 1989, 84, 391–403, discussion 404–405. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, D.A. Fibula Free Flap Mandible Reconstruction. Microsurgery 1994, 15, 238–244. [Google Scholar] [CrossRef]
- Taylor, G.I. Reconstruction of the Mandible with Free Composite Iliac Bone Grafts. Ann. Plast. Surg. 1982, 9, 361–376. [Google Scholar] [CrossRef] [PubMed]
- Swartz, W.M.; Banis, J.C.; Newton, E.D.; Ramasastry, S.S.; Jones, N.F.; Acland, R. The Osteocutaneous Scapular Flap for Mandibular and Maxillary Reconstruction. Plast. Reconstr. Surg. 1986, 77, 530–545. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.H.; Zhu, J.; Deng, J.Y.; Xia, B.; Xu, B. Three-Dimensional Virtual Technology in Reconstruction of Mandibular Defect Including Condyle Using Double-Barrel Vascularized Fibula Flap. J. Cranio-Maxillofac. Surg. 2013, 41, 417–422. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, D.L.; Garfein, E.S.; Christensen, A.M.; Weimer, K.A.; Saddeh, P.B.; Levine, J.P. Use of Computer-Aided Design and Computer-Aided Manufacturing to Produce Orthognathically Ideal Surgical Outcomes: A Paradigm Shift in Head and Neck Reconstruction. J. Oral Maxillofac. Surg. 2009, 67, 2115–2122. [Google Scholar] [CrossRef]
- Bolzoni, A.; Mapelli, A.; Baj, A.; Sidequersky, F.V.; Giannì, A.B.; Sforza, C. Evaluation of Three-Dimensional Mandibular Movements after Reconstruction with Free Fibula Flap. Acta Otorhinolaryngol. Italy 2015, 35, 371–378. [Google Scholar] [CrossRef]
- Morrison, S.D.; Satterwhite, T. Lower Jaw Recontouring in Facial Gender-Affirming Surgery. Facial Plast. Surg. Clin. N. Am. 2019, 27, 233–242. [Google Scholar] [CrossRef]
- Kasper, R.; Scheurer, M.; Pietzka, S.; Sakkas, A.; Schramm, A.; Wilde, F.; Ebeling, M. MRONJ of the Mandible-From Decortication to a Complex Jaw Reconstruction Using a CAD/CAM-Guided Bilateral Scapula Flap. Medicina 2023, 59, 535. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. J. Clin. Epidemiol. 2021, 134, 178–189. [Google Scholar] [CrossRef]
- Jacek, B.; Maciej, P.; Tomasz, P.; Agata, B.; Wiesław, K.; Radosław, W.; Filip, G. 3D Printed Models in Mandibular Reconstruction with Bony Free Flaps. J. Mater. Sci. Mater. Med. 2018, 29, 23. [Google Scholar] [CrossRef] [PubMed]
- May, M.M.; Howe, B.M.; O’Byrne, T.J.; Alexander, A.E.; Morris, J.M.; Moore, E.J.; Kasperbauer, J.L.; Janus, J.R.; Van Abel, K.M.; Dickens, H.J.; et al. Short and Long-term Outcomes of three-dimensional Printed Surgical Guides and Virtual Surgical Planning versus Conventional Methods for Fibula Free Flap Reconstruction of the Mandible: Decreased Nonunion and Complication Rates. Head Neck 2021, 43, 2342–2352. [Google Scholar] [CrossRef] [PubMed]
- Knitschke, M.; Yonan, M.; Roller, F.C.; Pons-Kühnemann, J.; Attia, S.; Howaldt, H.-P.; Streckbein, P.; Böttger, S. Osseous Union after Jaw Reconstruction with Fibula-Free Flap: Conventional vs. CAD/CAM Patient-Specific Implants. Cancers 2022, 14, 5774. [Google Scholar] [CrossRef] [PubMed]
- Pu, J.J.; Choi, W.S.; Yeung, W.K.; Yang, W.-F.; Zhu, W.-Y.; Su, Y.-X. A Comparative Study on a Novel Fibula Malleolus Cap to Increase the Accuracy of Oncologic Jaw Reconstruction. Front. Oncol. 2022, 11, 743389. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Choi, W.S.; Wong, M.C.-M.; Powcharoen, W.; Zhu, W.; Tsoi, J.K.-H.; Chow, M.; Kwok, K.-W.; Su, Y. Three-Dimensionally Printed Patient-Specific Surgical Plates Increase Accuracy of Oncologic Head and Neck Reconstruction Versus Conventional Surgical Plates: A Comparative Study. Ann. Surg. Oncol. 2021, 28, 363–375. [Google Scholar] [CrossRef]
- Tarsitano, A.; Ciocca, L.; Scotti, R.; Marchetti, C. Morphological Results of Customized Microvascular Mandibular Reconstruction: A Comparative Study. J. Cranio-Maxillofac. Surg. 2016, 44, 697–702. [Google Scholar] [CrossRef]
- Chen, J.; Zhang, R.; Liang, Y.; Ma, Y.; Song, S.; Jiang, C. Deviation Analyses of Computer-Assisted, Template-Guided Mandibular Reconstruction With Combined Osteotomy and Reconstruction Pre-Shaped Plate Position Technology: A Comparative Study. Front. Oncol. 2021, 11, 719466. [Google Scholar] [CrossRef]
- Dai, J.; Wang, X.; Dong, Y.; Yu, H.; Yang, D.; Shen, G. Two- and Three-Dimensional Models for the Visualization of Jaw Tumors Based on CT-MRI Image Fusion. J. Craniofac. Surg. 2012, 23, 502–508. [Google Scholar] [CrossRef]
- Byun, S.H.; Lim, H.K.; Yang, B.E.; Kim, S.M.; Lee, J.H. Delayed Reconstruction of Palatomaxillary Defect Using Fibula Free Flap. J. Clin. Med. 2020, 9, 884. [Google Scholar] [CrossRef]
- Bouchet, B.; Raoul, G.; Julieron, B.; Wojcik, T. Functional and Morphologic Outcomes of CAD/CAM-Assisted versus Conventional Microvascular Fibular Free Flap Reconstruction of the Mandible: A Retrospective Study of 25 Cases. J. Stomatol. Oral Maxillofac. Surg. 2018, 119, 455–460. [Google Scholar] [CrossRef]
- Numajiri, T.; Morita, D.; Nakamura, H.; Yamochi, R.; Tsujiko, S.; Sowa, Y. Designing CAD/CAM Surgical Guides for Maxillary Reconstruction Using an In-House Approach. J. Vis. Exp. 2018, 138, e58015. [Google Scholar] [CrossRef]
- Bolzoni, A.R.; Pollice, A.; Nuti, M.; Baj, A.; Rossi, D.S.; Beltramini, G.A. Clinical and Functional Outcomes of Cad/Cam Mandibular Reconstruction with Free Fibular Flap Comparing Traditional versus Micro-Invasive Intraoral Surgical Approaches. J. Biol. Regul. Homeost. Agents 2020, 34, 175–184, Technology in Medicine. [Google Scholar] [PubMed]
- Tarsitano, A.; Battaglia, S.; Ramieri, V.; Cascone, P.; Ciocca, L.; Scotti, R.; Marchetti, C. Short-Term Outcomes of Mandibular Reconstruction in Oncological Patients Using a CAD/CAM Prosthesis Including a Condyle Supporting a Fibular Free Flap. J. Cranio-Maxillofac. Surg. 2017, 45, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Chauvel-Picard, J.; Kreutzer, K.; Heiland, M.; Kreusch, T.; Ebker, T.; Beck-Broichsitter, B. One Stage Microvascular Mandible Reconstruction by Using Scapula Chimeric Flap Combined with Computer-aided-design and Computer-aided-manufacturing Plate Including Bilateral Alloplastic TMJ Prosthesis: A Case Report. Microsurgery 2021, 41, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Kraeima, J.; Glas, H.H.; Merema, B.B.J.; Vissink, A.; Spijkervet, F.K.L.; Witjes, M.J.H. Three-Dimensional Virtual Surgical Planning in the Oncologic Treatment of the Mandible. Oral Dis. 2021, 27, 14–20. [Google Scholar] [CrossRef]
- Neto, T.; Horta, R.; Balhau, R.; Coelho, L.; Silva, P.; Correia-Sá, I.; Silva, Á. Resection and Microvascular Reconstruction of Bisphosphonate-Related Osteonecrosis of the Jaw: The Role of Microvascular Reconstruction. Head Neck 2016, 38, 1278–1285. [Google Scholar] [CrossRef]
- Ricotta, F.; Battaglia, S.; Bolognesi, F.; Ceccariglia, F.; Marchetti, C.; Tarsitano, A. Use of CAD-CAM Bridging Mandibular Prosthesis in Osteonecrosis of the Jaw: The Experience of Our School. J. Clin. Med. 2020, 9, 3516. [Google Scholar] [CrossRef]
- Piotrowska-Seweryn, A.; Szymczyk, C.; Walczak, D.A.; Krakowczyk, Ł.; Maciejewski, A.; Hadasik, G.; Wierzgoń, J.; Szumniak, R.; Drozdowski, P.; Paul, P.; et al. Fibular Free Flap and Iliac Crest Free Flap Mandibular Reconstruction In Patients With Mandibular Ameloblastomas. J. Craniofac. Surg. 2022, 33, 1962–1970. [Google Scholar] [CrossRef]
- Ritschl, L.M.; Kilbertus, P.; Grill, F.D.; Schwarz, M.; Weitz, J.; Nieberler, M.; Wolff, K.-D.; Fichter, A.M. In-House, Open-Source 3D-Software-Based, CAD/CAM-Planned Mandibular Reconstructions in 20 Consecutive Free Fibula Flap Cases: An Explorative Cross-Sectional Study With Three-Dimensional Performance Analysis. Front. Oncol. 2021, 11, 731336. [Google Scholar] [CrossRef]
- Alwadeai, M.S.; Al-aroomy, L.A.; Shindy, M.I.; Amin, A.A.-W.; Zedan, M.H. Aesthetic Reconstruction of Onco-Surgical Maxillary Defects Using Free Scapular Flap with and without CAD/CAM Customized Osteotomy Guide. BMC Surg. 2022, 22, 362. [Google Scholar] [CrossRef]
- Liu, S.; Mazur, T.R.; Li, H.; Curcuru, A.; Green, O.L.; Sun, B.; Mutic, S.; Yang, D. A Method to Reconstruct and Apply 3D Primary Fluence for Treatment Delivery Verification. J. Appl. Clin. Med. Phys. 2017, 18, 128–138. [Google Scholar] [CrossRef] [PubMed]
- Le Clerc, N.; Baudouin, R.; Carlevan, M.; Khoueir, N.; Verillaud, B.; Herman, P. 3D Titanium Implant for Orbital Reconstruction after Maxillectomy. J. Plast. Reconstr. Aesthet. Surg. 2020, 73, 732–739. [Google Scholar] [CrossRef] [PubMed]
- Duchscherer, J.; Aalto, D.; Westover, L. Evaluation of Facial Symmetry after Jaw Reconstruction Surgery. Comput. Methods Biomech. Biomed. Eng. 2021, 24, 1212–1220. [Google Scholar] [CrossRef]
- Mahdian, N.; Dostálová, T.; Danĕk, J.; Nedoma, J.; Kohout, J.; Hubáček, M.; Hliňáková, P. 3D Reconstruction of TMJ after Resection of the Cyst and the Stress-Strain Analyses. Comput. Methods Programs Biomed. 2013, 110, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, S.; Oda, M.; Onoue, K.; Basa, K.; Rubin, S.J.; Sakai, O.; Salama, A.; Ezzat, W.H. Determining the Optimal Osteotomy Distance with the Fibula Free Flap in Mandibular Reconstruction. Am. J. Otolaryngol. 2020, 41, 102436. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.-F.; Choi, W.S.; Zhu, W.-Y.; Zhang, C.-Y.; Li, D.T.S.; Tsoi, J.K.-H.; Tang, A.W.-L.; Kwok, K.-W.; Su, Y.-X. Spatial Deviations of the Temporomandibular Joint after Oncological Mandibular Reconstruction. Int. J. Oral Maxillofac. Surg. 2022, 51, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Al Maruf, D.S.A.; Ghosh, Y.A.; Xin, H.; Cheng, K.; Mukherjee, P.; Crook, J.M.; Wallace, G.G.; Klein, T.J.; Clark, J.R. Hydrogel: A Potential Material for Bone Tissue Engineering Repairing the Segmental Mandibular Defect. Polymers 2022, 14, 4186. [Google Scholar] [CrossRef]
- Ciocca, L.; Mazzoni, S.; Fantini, M.; Persiani, F.; Baldissara, P.; Marchetti, C.; Scotti, R. A CAD/CAM-Prototyped Anatomical Condylar Prosthesis Connected to a Custom-Made Bone Plate to Support a Fibula Free Flap. Med. Biol. Eng. Comput. 2012, 50, 743–749. [Google Scholar] [CrossRef]
- Wilde, F.; Winter, K.; Kletsch, K.; Lorenz, K.; Schramm, A. Mandible Reconstruction Using Patient-Specific Pre-Bent Reconstruction Plates: Comparison of Standard and Transfer Key Methods. Int. J. Comput. Assist. Radiol. Surg. 2015, 10, 129–140. [Google Scholar] [CrossRef]
- Ciocca, L.; Mazzoni, S.; Fantini, M.; Persiani, F.; Marchetti, C.; Scotti, R. CAD/CAM Guided Secondary Mandibular Reconstruction of a Discontinuity Defect after Ablative Cancer Surgery. J. Cranio-Maxillofac. Surg. 2012, 40, e511-5. [Google Scholar] [CrossRef]
- Ritschl, L.; Mücke, T.; Fichter, A.; Güll, F.; Schmid, C.; Duc, J.; Kesting, M.; Wolff, K.-D.; Loeffelbein, D. Functional Outcome of CAD/CAM-Assisted versus Conventional Microvascular, Fibular Free Flap Reconstruction of the Mandible: A Retrospective Study of 30 Cases. J. Reconstr. Microsurg. 2017, 33, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Moore, E.J.; Hinni, M.L.; Arce, K.; Salinas, T. Mandibular Alveolar Reconstruction Using Three-Dimensional Planning. Curr. Opin. Otolaryngol. Head Neck Surg. 2013, 21, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Liu, H.; Du, W.; Wang, Y.; Hu, J.; Luo, E. Computer-Aided Design and Computer-Aided Manufacturing Cutting Guides in Eminoplasty for the Treatment of Temporomandibular Joint Dislocation. J. Craniofac. Surg. 2019, 30, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Valentini, V.; Gennaro, P.; Torroni, A.; Longo, G.; Aboh, I.V.; Cassoni, A.; Battisti, A.; Anelli, A. Scapula Free Flap for Complex Maxillofacial Reconstruction. J. Craniofac. Surg. 2009, 20, 1125–1131. [Google Scholar] [CrossRef] [PubMed]
- Voss, P.J.; Steybe, D.; Fuessinger, M.A.; Semper-Hogg, W.; Metzger, M.; Schmelzeisen, R.; Poxleitner, P. Vascularized Scapula and Latissimus Dorsi Flap for CAD/CAM Assisted Reconstruction of Mandibular Defects Including the Mandibular Condyle: Technical Report and Clinical Results. BMC Surg. 2019, 19, 67. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).