Co-Creation Facilitates Translational Research on Upper Limb Prosthetics
Abstract
1. Introduction
2. Background
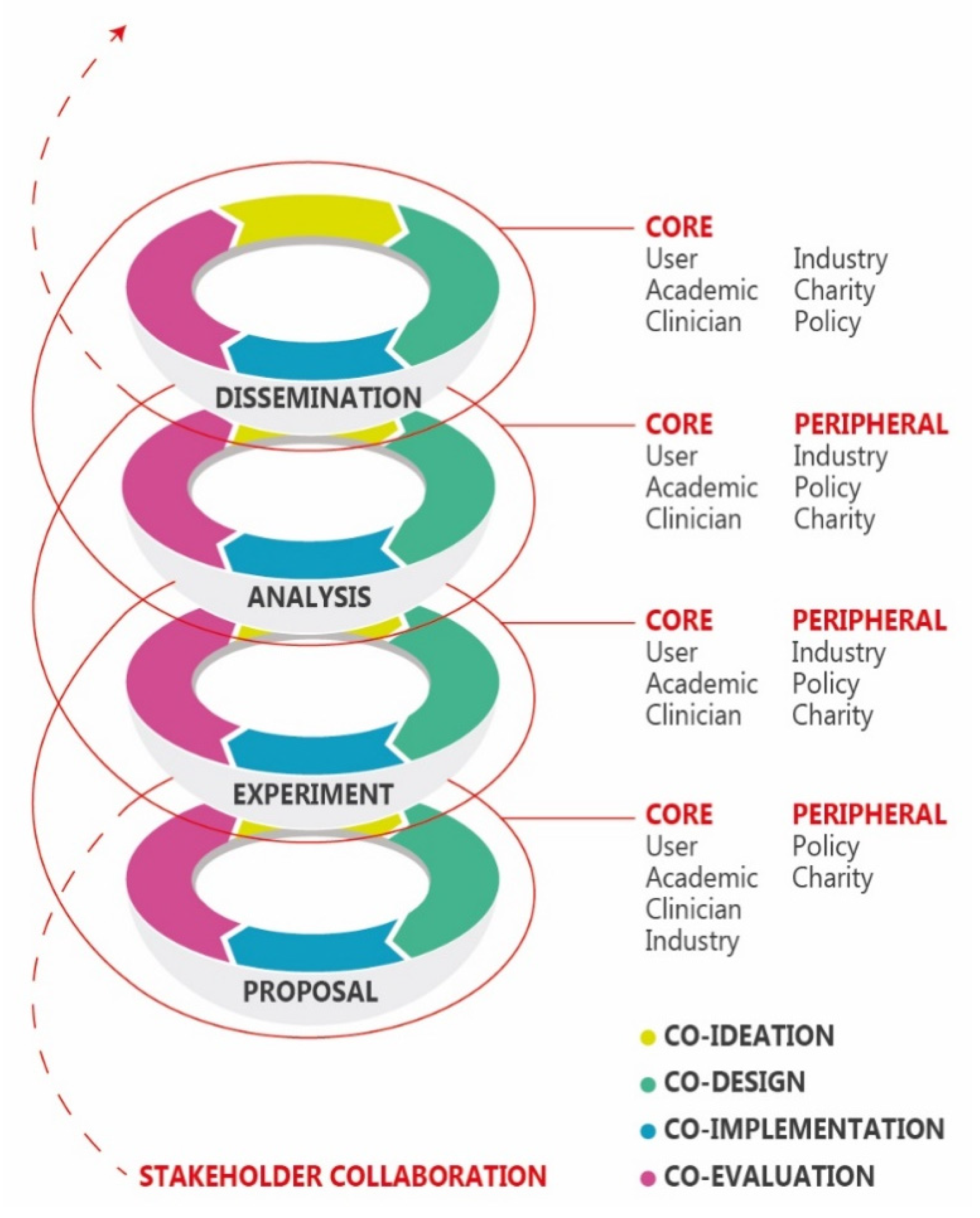
3. Application to Upper Limb Prosthetic Research
4. Challenges and Opportunities
5. Suggestions for the Field
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Dedication
References
- Nazarpour, K. Control of Prosthetic Hands: Challenges and Emerging Avenues, 1st ed.; IET Press: London, UK, 2020. [Google Scholar]
- Jones, H.; Dupan, S.S.G.; Nazarpour, K. The Future of Prosthetics: A User Perspective. In Proceedings of the Trent International Prosthetics Symposium, Manchester, UK, 20–21 March 2019. [Google Scholar]
- Engdahl, S.M.; Christie, B.P.; Kelly, B.M.; Davis, A.; Chestek, C.A.; Gates, D.H. Surveying the interest of individuals with upper limb loss in novel prosthetic control techniques. J. Neuroeng. Rehabil. 2015, 12, 1–11. [Google Scholar] [CrossRef]
- Østlie, K.; Lesjø, I.M.; Franklin, R.J.; Garfelt, B.; Skjeldal, O.H.; Magnus, P. Prosthesis rejection in acquired major upper-limb amputees: A population-based survey. Disabil. Rehabil. Assist. Technol. 2011, 7, 294–303. [Google Scholar] [CrossRef] [PubMed]
- Cordella, F.; Ciancio, A.L.; Sacchetti, R.; Davalli, A.; Cutti, A.G.; Guglielmelli, E.; Zollo, L. Literature Review on Needs of Upper Limb Prosthesis Users. Front. Neurosci. 2016, 10, 209. [Google Scholar] [CrossRef] [PubMed]
- Kumar, D.K.; Jelfs, B.; Sui, X.; Arjunan, S.P. Prosthetic hand control: A multidisciplinary review to identify strengths, shortcomings, and the future. Biomed. Signal Process. Control. 2019, 53, 101588. [Google Scholar] [CrossRef]
- Jang, C.H.; Yang, H.S.; Yang, H.E.; Lee, S.Y.; Kwon, J.W.; Yun, B.D.; Choi, J.Y.; Kim, S.N.; Jeong, H.W. A Survey on Activities of Daily Living and Occupations of Upper Extremity Amputees. Ann. Rehabil. Med. 2011, 35, 907–921. [Google Scholar] [CrossRef]
- Kyberd, P.J.; Wartenberg, C.; Sandsjö, L.; Jönsson, S.; Gow, D.; Frid, J.; Almström, C.; Sperling, L. Survey of upper limb prosthesis users in Sweden and the United Kingdom. J. Prosthet. Orthot. 2007, 19, 55–62. [Google Scholar] [CrossRef]
- Luchetti, M.; Cutti, A.G.; Verni, G.; Sacchetti, R.; Rossi, N. Impact of Michelangelo prosthetic hand: Findings from a crossover longitudinal study. J. Rehabil. Res. Dev. 2015, 52, 605–618. [Google Scholar] [CrossRef]
- Engdahl, S.M.; Chestek, C.A.; Kelly, B.; Davis, A.; Gates, D.H. Factors associated with interest in novel interfaces for upper limb prosthesis control. PLoS ONE 2017, 12, e0182482. [Google Scholar] [CrossRef]
- Sugawara, A.T.; Ramos, V.D.; Alfieri, F.M.; Battistella, L.R. Abandonment of assistive products: Assessing abandonment levels and factors that impact on it. Disabil. Rehabil. Assist. Technol. 2018, 13, 716–723. [Google Scholar] [CrossRef]
- Biddiss, E.; Chau, T. Upper-Limb Prosthetics. Am. J. Phys. Med. Rehabil. 2007, 86, 977–987. [Google Scholar] [CrossRef]
- Biddiss, E.A.; Chau, T.T. Upper limb prosthesis use and abandonment. Prosthet. Orthot. Int. 2007, 31, 236–257. [Google Scholar] [CrossRef] [PubMed]
- Postema, K.; Van Der Donk, V.; Van Limbeek, J.; Rijken, R.A.; Poelma, M.J. Prosthesis rejection in children with a unilateral congenital arm defect. Clin. Rehabil. 1999, 13, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Postema, S.G.; Bongers, R.M.; Brouwers, M.A.; Burger, H.; Hermansson, L.M.N.-; Reneman, M.F.; Dijkstra, P.U.; Van Der Sluis, C.K. Upper Limb Absence: Predictors of Work Participation and Work Productivity. Arch. Phys. Med. Rehabil. 2016, 97, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Davidson, J. A survey of the satisfaction of upper limb amputees with their prostheses, their lifestyles, and their abilities. J. Hand Ther. 2002, 15, 62–70. [Google Scholar] [CrossRef]
- Chadwell, A.; Kenney, L.; Granat, M.H.; Thies, S.; Head, J.; Galpin, A.; Baker, R.; Kulkarni, J. Upper limb activity in myoelectric prosthesis users is biased towards the intact limb and appears unrelated to goal-directed task performance. Sci. Rep. 2018, 8, 11084. [Google Scholar] [CrossRef]
- Biddiss, E.; Beaton, D.; Chau, T. Consumer design priorities for upper limb prosthetics. Disabil. Rehabil. Assist. Technol. 2007, 2, 346–357. [Google Scholar] [CrossRef]
- Salminger, S.; Stino, H.; Pichler, L.H.; Gstoettner, C.; Sturma, A.; Mayer, J.A.; Szivak, M.; Aszmann, O.C. Current rates of prosthetic usage in upper-limb amputees—Have innovations had an impact on device acceptance? Disabil. Rehabil. 2020, 1–12. [Google Scholar] [CrossRef]
- Vujaklija, I.; Roche, A.D.; Hasenoehrl, T.; Sturma, A.; Amsuess, S.; Farina, D.; Aszmann, O.C. Translating Research on Myoelectric Control into Clinics—Are the Performance Assessment Methods Adequate? Front. Neurorobotics 2017, 11, 7. [Google Scholar] [CrossRef]
- Simon, A.M.; Turner, K.L.; Miller, L.A.; Hargrove, L.J.; Kuiken, T.A. Pattern recognition and direct control home use of a multi-articulating hand prosthesis. In Proceedings of the IEEE 16th International Conference on Rehabilitation Robotics (ICORR), Toronto, ON, Canada, 24–28 June 2019; pp. 386–391. [Google Scholar] [CrossRef]
- Chadwell, A.; Diment, L.; Micó-Amigo, M.; Ramírez, D.Z.M.; Dickinson, A.; Granat, M.; Kenney, L.; Kheng, S.; Sobuh, M.; Ssekitoleko, R.; et al. Technology for monitoring everyday prosthesis use: A systematic review. J. Neuroeng. Rehabil. 2020, 17, 1–26. [Google Scholar] [CrossRef]
- Wu, H.; Dyson, M.; Nazarpour, K. Arduino-Based Myoelectric Control: Towards Longitudinal Study of Prosthesis Use. Sensors 2021, 21, 763. [Google Scholar] [CrossRef]
- Van Der Sluis, C.K.; Bongers, R.M. TIPS for Scaling up Research in Upper Limb Prosthetics. Prosthesis 2020, 2, 340–351. [Google Scholar] [CrossRef]
- Woolf, S.H. The Meaning of Translational Research and Why It Matters. JAMA 2008, 299, 211–213. [Google Scholar] [CrossRef] [PubMed]
- Seyhan, A.A. Lost in translation: The valley of death across preclinical and clinical divide—Identification of problems and overcoming obstacles. Transl. Med. Commun. 2019, 4, 1–19. [Google Scholar] [CrossRef]
- Fernandez-Moure, J.S. Lost in Translation: The Gap in Scientific Advancements and Clinical Application. Front. Bioeng. Biotechnol. 2016, 4, 43. [Google Scholar] [CrossRef]
- Chesbrough, H. The Future of Open Innovation. Res. Manag. 2017, 60, 35–38. [Google Scholar] [CrossRef]
- Austin, J.; Drossaert, S.C.H.C.; van Dijk, J.; Mirkovic, J.; Børøsund, E.; Sanderman, R.; Schroevers, M.J.; Bohlmeijer, E.T. Co-creation phases in integrating top-down and bottom-up requirements: Developing a self-compassion app with cancer patients. In Supporting Health by Technology; University of Twente: Enschede, Netherlands, 2020; pp. 13–14. [Google Scholar]
- Webster, A.; Poyade, M.; Rea, P.; Paul, L. The Co-design of Hand Rehabilitation Exercises for Multiple Sclerosis Using Hand Tracking System; Springer: Cham, Switzerland, 2019; Volume 1120, pp. 83–96. [Google Scholar]
- Van den Kieboom, R.C.; Bongers, I.M.; Mark, R.E.; Snaphaan, L.J.; Mulvenna, M.; Kenning, G.; Nomali, M. User-Driven Living Lab for Assistive Technology to Support People With Dementia Living at Home: Protocol for Developing Co-Creation–Based Innovations. JMIR Res. Protoc. 2019, 8, e10952. [Google Scholar] [CrossRef]
- Favela, J.; Kaye, J.; Skubic, M.; Rantz, M.; Tentori, M. Living Labs for Pervasive Healthcare Research. IEEE Pervasive Comput. 2015, 14, 86–89. [Google Scholar] [CrossRef]
- Sanders, E.B.-N.; Stappers, P.J. Co-creation and the new landscapes of design. CoDesign 2008, 4, 5–18. [Google Scholar] [CrossRef]
- De Koning, J.I.J.C.; Crul, M.R.M.; Wever, R. Models of co-creation. In Proceedings of the ServDes 2016, Copenhagen, Denmark, 24–26 May 2016; pp. 266–278. [Google Scholar]
- Pearce, T.; Maple, M.; Shakeshaft, A.; Wayland, S.; McKay, K. What is the Co-Creation of New Knowledge? A Content Analysis and Proposed Definition for Health Interventions. Int. J. Environ. Res. Public Health 2020, 17, 2229. [Google Scholar] [CrossRef]
- Monnard, K.; Benjamins, M.R.; Hirschtick, J.L.; Castro, M.; Roesch, P.T. Co-Creation of Knowledge: A Community-Based Approach to Multilevel Dissemination of Health Information. Health Promot. Pract. 2021, 22, 215–223. [Google Scholar] [CrossRef]
- Van Der Scheer, L.; Garcia, E.; Van Der Laan, A.L.; Van Der Burg, S.; Boenink, M. The Benefits of Patient Involvement for Translational Research. Health Care Anal. 2014, 25, 225–241. [Google Scholar] [CrossRef] [PubMed]
- Oliver, K.; Kothari, A.; Mays, N. The dark side of coproduction: Do the costs outweigh the benefits for health research? Health Res. Policy Syst. 2019, 17, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.L.; Harnett, J. Are Real-World Data and Evidence Good Enough to Inform Health Care and Health Policy Decision-Making? In Decision Making in a World of Comparative Effectiveness Research; Birnbaum, H., Greenberg, P., Eds.; ADIS: Singapore, 2017; pp. 97–103. [Google Scholar] [CrossRef]
- Langley, J.; Wheeler, G.; Mills, N.; Heron, N. Starworks: Politics, power and expertise in co-producing a research, patient, practice and industry partnership for child prosthetics. In Proceedings of the Design4Health Conference, Amsterdam, The Netherlands, 1–3 July 2020; Volume 2, pp. 108–115. [Google Scholar]
- Hargrove, L.J.; Miller, L.A.; Turner, K.; Kuiken, T.A. Myoelectric Pattern Recognition Outperforms Direct Control for Transhumeral Amputees with Targeted Muscle Reinnervation: A Randomized Clinical Trial. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Brinton, M.R.; Barcikowski, E.; Davis, T.; Paskett, M.; George, J.A.; Clark, G.A. Portable Take-Home System Enables Proportional Control and High-Resolution Data Logging With a Multi-Degree-of-Freedom Bionic Arm. Front. Robot. AI 2020, 7, 559034. [Google Scholar] [CrossRef]
- Garske, C.A.; Dyson, M.; Dupan, S.; Nazarpour, K. Perception of Game-Based Rehabilitation in Upper Limb Prosthetic Training: Survey of Users and Researchers. JMIR Serious Games 2021, 9, e23710. [Google Scholar] [CrossRef]
- Grant, A.D.; Wolf, G.I.; Nebeker, C. Approaches to governance of participant-led research: A qualitative case study. BMJ Open 2019, 9, e025633. [Google Scholar] [CrossRef]
- Hickey, G.; Brearley, S.; Coldham, T.; Denegri, S.; Green, G.; Staniszewska, S.; Tembo, D.; Torok, K.; Turner, K. Guidance on Co-producing a Research Project; NIHR INVOLVE: Southampton, UK, 2018. [Google Scholar]
- The Ethical Roadmap. Available online: https://ethicalroadmap.org/en/ (accessed on 25 February 2021).
- Langley, J.; Wolstenholme, D.; Cooke, J. ’Collective making’ as knowledge mobilisation: The contribution of participatory design in the co-creation of knowledge in healthcare. BMC Health Serv. Res. 2018, 18, 585. [Google Scholar] [CrossRef]
- National Institute for Health Research. What is Public Involvement in Research? Available online: https://www.invo.org.uk/find-out-more/what-is-public-involvement-in-research-2/ (accessed on 3 February 2021).
- Jones, H.; Nazarpour, K. Gaining NHS ethical approval from the perspective of a biomedical engineering team. Br. J. Healthc. Manag. 2018, 24, 71–76. [Google Scholar] [CrossRef]
- The Royal Society, Pairing Scheme. Available online: https://royalsociety.org/grants-schemes-awards/pairing-scheme/ (accessed on 3 February 2021).
- Williams, O.; Sarre, S.; Papoulias, S.C.; Knowles, S.; Robert, G.; Beresford, P.; Rose, D.; Carr, S.; Kaur, M.; Palmer, V.J. Lost in the shadows: Reflections on the dark side of co-production. Health Res. Policy Syst. 2020, 18, 1–10. [Google Scholar] [CrossRef]
- Burns, J.A.; Korzec, K.; Dorris, E.R. From intent to implementation: Factors affecting public involvement in life science research. BioRxiv 2019, 748889. [Google Scholar] [CrossRef]
- Gradim, L.C.; Jose, M.A.; Da Cruz, D.M.C.; Lopes, R.D.D. IoT services and applications in rehabilitation: An interdisciplinary and meta-analysis review. IEEE Trans. Neural Syst. Rehabil. Eng. 2020, 28, 1. [Google Scholar] [CrossRef]
- Seah, K.M. COVID-19: Exposing digital poverty in a pandemic. Int. J. Surg. 2020, 79, 127–128. [Google Scholar] [CrossRef] [PubMed]
- Fiorini, L.; Tabeau, K.; D’Onofrio, G.; Coviello, L.; De Mul, M.; Sancarlo, D.; Fabbricotti, I.; Cavallo, F. Co-creation of an assistive robot for independent living: Lessons learned on robot design. Int. J. Interact. Des. Manuf. 2019, 14, 491–502. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jones, H.; Dupan, S.; Coutinho, M.; Day, S.; Desmond, D.; Donovan-Hall, M.; Dyson, M.; Ekins-Coward, T.; Kenney, L.; Krasoulis, A.; et al. Co-Creation Facilitates Translational Research on Upper Limb Prosthetics. Prosthesis 2021, 3, 110-118. https://doi.org/10.3390/prosthesis3020012
Jones H, Dupan S, Coutinho M, Day S, Desmond D, Donovan-Hall M, Dyson M, Ekins-Coward T, Kenney L, Krasoulis A, et al. Co-Creation Facilitates Translational Research on Upper Limb Prosthetics. Prosthesis. 2021; 3(2):110-118. https://doi.org/10.3390/prosthesis3020012
Chicago/Turabian StyleJones, Hannah, Sigrid Dupan, Maxford Coutinho, Sarah Day, Deirdre Desmond, Margaret Donovan-Hall, Matthew Dyson, Thea Ekins-Coward, Laurence Kenney, Agamemnon Krasoulis, and et al. 2021. "Co-Creation Facilitates Translational Research on Upper Limb Prosthetics" Prosthesis 3, no. 2: 110-118. https://doi.org/10.3390/prosthesis3020012
APA StyleJones, H., Dupan, S., Coutinho, M., Day, S., Desmond, D., Donovan-Hall, M., Dyson, M., Ekins-Coward, T., Kenney, L., Krasoulis, A., McIntosh, D., Memarzadeh, K., Small, E., Wheeler, G., Wu, H., & Nazarpour, K. (2021). Co-Creation Facilitates Translational Research on Upper Limb Prosthetics. Prosthesis, 3(2), 110-118. https://doi.org/10.3390/prosthesis3020012






