Soil Attributes Mapping with Online Near-Infrared Spectroscopy Requires Spatio-Temporal Local Calibrations
Abstract
1. Introduction
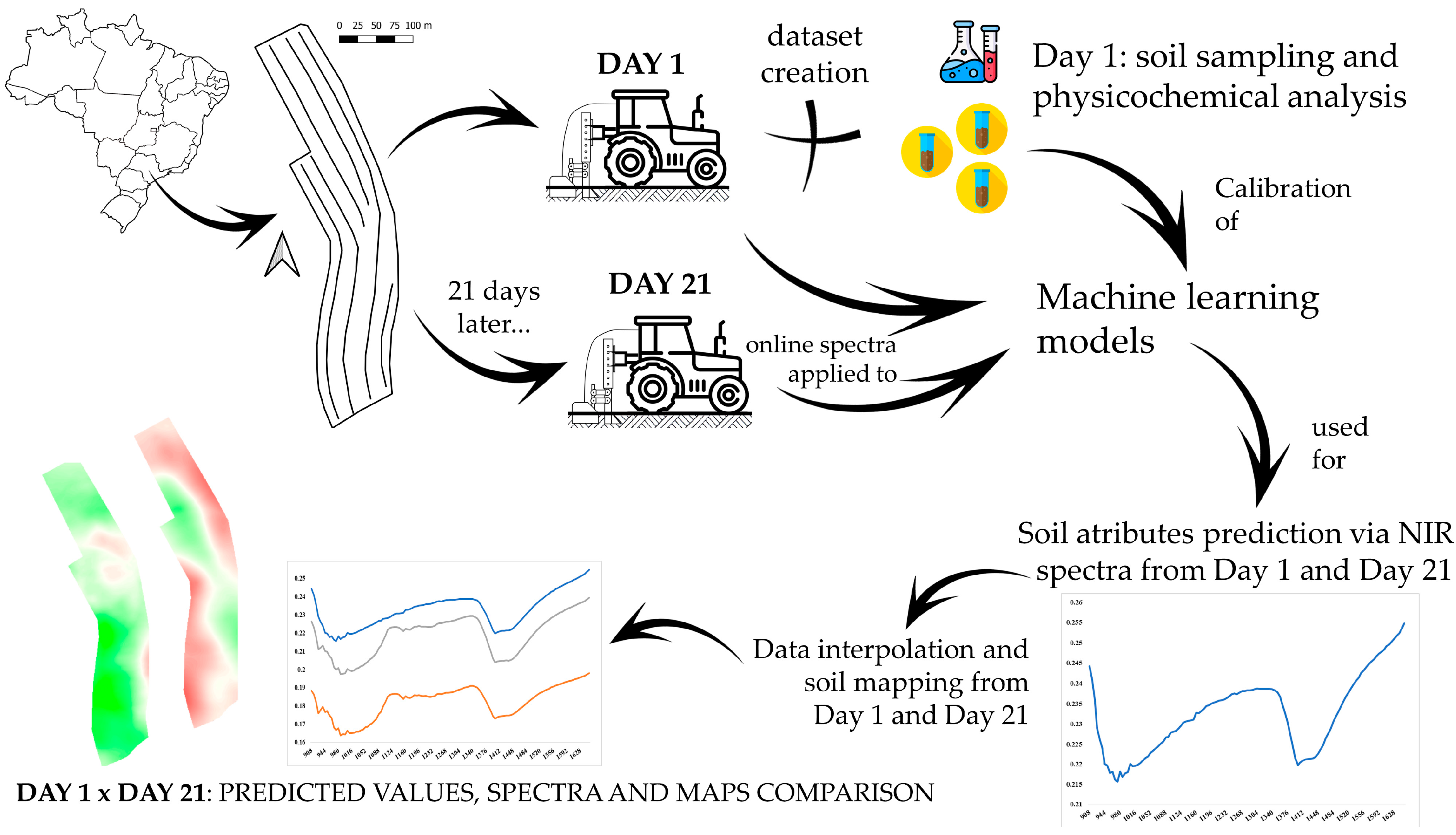
2. Material and Methods
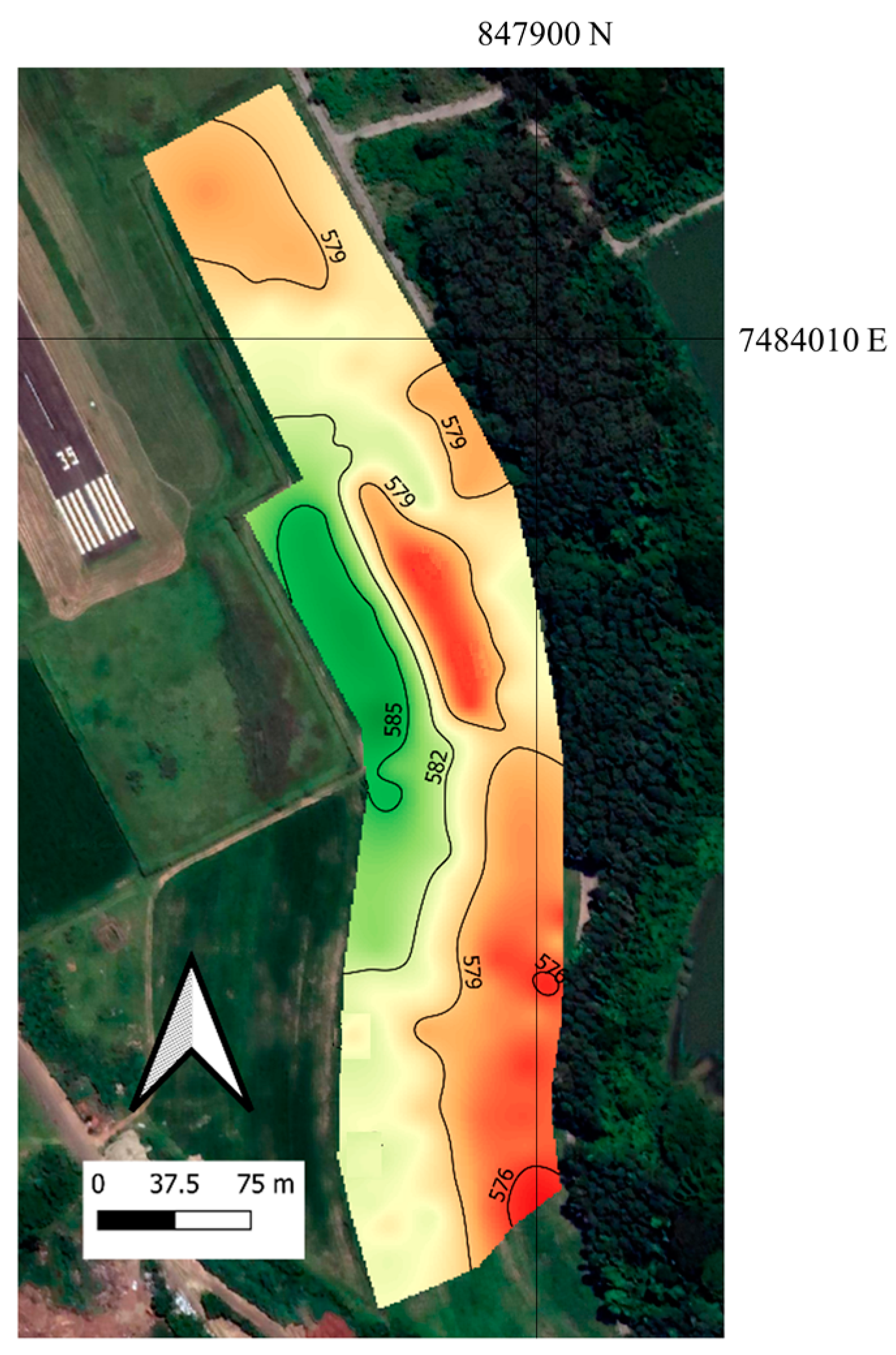
2.1. Study Area
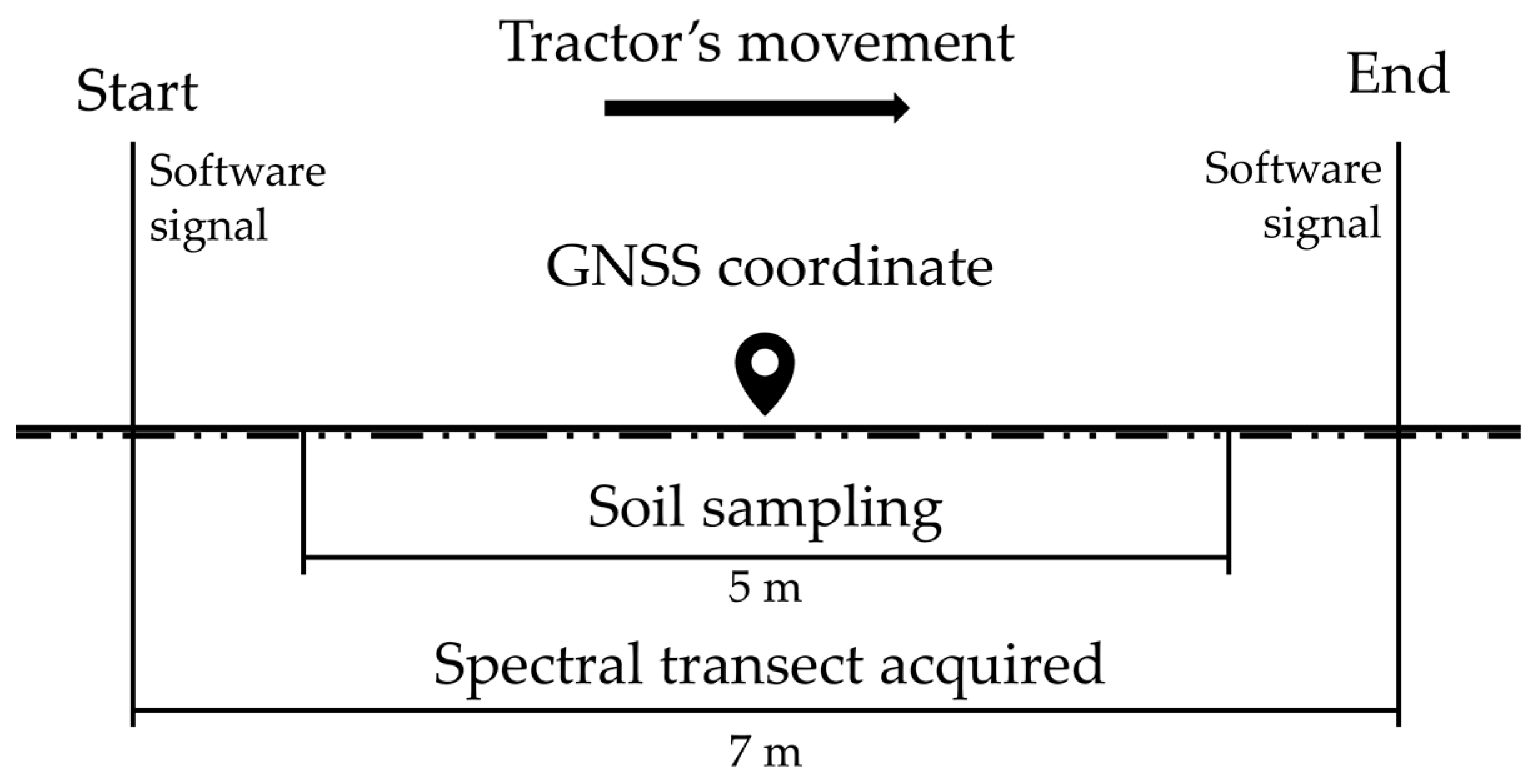
2.2. Online Spectra Acquisition and Soil Sampling
2.3. Machine Learning Models and Data Interpolation
2.4. Prediction Values, Spectra Characteristics, and Maps Analysis
- Overlap population: an ellipse buffer of 2.5 m radius in the direction of the tractor’s movement was created to extract overlap points, as the soil sampling was carried in the length of five meters to match the transect of spectral acquisition (see Section 2.2). This analysis was considered since, at the field operation, two spectra would hardly be acquired at the same point. Although the tractor’s operator, operation speed, and acquisition lines were the same, variations in orientation or border maneuvers could offset the spectra from day 21 from the location of day 1. This would hinder the direct comparison of day 1 vs day 21 as: spectrumday1 1 vs. spectrumday21 1; spectrumday1 n vs. spectrumday21 n; …; spectrumday1 140 vs. spectrumday21 140;
- High correlated population: the 20 highest correlated spectra pairs (day 1 vs. day 21) were identified using Pearson’s correlation analysis. For this purpose, the 125 wavelengths measured were the variables compared for spectra correlation;
- Total population: the nearest neighbors from day 1 and day 21 were joined in pairs, yielding 140 pairs used for correlation analysis. The total population was of higher attention in our study since its analysis can lead to understanding if field management decisions based on the predictive performance of the models over time would be stable and, therefore, reliable—the main goal of this study.
3. Results and Discussion
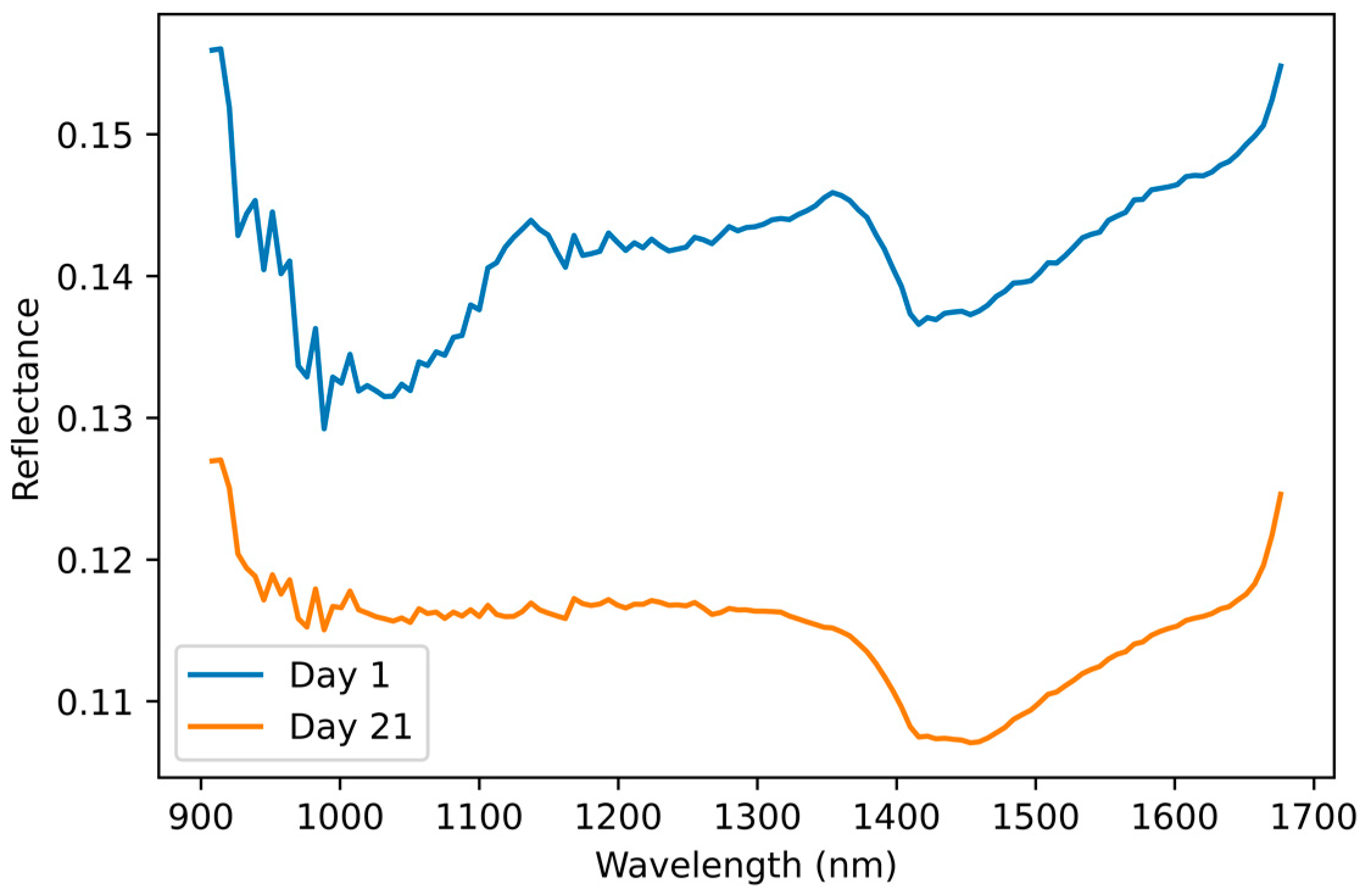
3.1. Prediction Values, Spectra Characteristics, and Maps Analysis
3.1.1. Overlap Population
3.1.2. Highly Correlated Population
3.1.3. Total Population
- Water could intensify the values predicted. However, this may not be the only explanation for the differences in day 21 spectra prediction using ML models calibrated on day 1. The analysis of predicted values, as shown in Table 2, are not intensified but almost independent from each other;
- Other environmental factors must be interfering in spectra acquisition, which can explain the differences between the two days’ predictions. Since it is harder to control the circumstances of a field operation than of a laboratory, any peculiarity can change the aspects of acquired spectra. One possibility to be tested is the measuring chamber designed and validated by Rodionov et al. [61]. No other studies report a methodology to isolate the visible-NIR spectra acquisition during field operations, simulating a laboratory condition, as the usual is the subsoiler shank and spectrophotometer being carried in an open field. This strategy can be tested for acquisitions over time since spectral acquisition will still suffer from topsoil particle size variation, soil moisture, and tractor movement but can isolate other environmental factors.
- A further investigation into factors such as sunlight, soil temperature, air temperature, etc., needs to be carried out. This means investigating the possibility of providing the models’ auxiliary data to deal with spectra variation and guaranteeing more stability for the ML calibrations which depend on it.
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- International Society of Precision Agriculture. Precision Agriculture Definition; International Society of Precision Agriculture: Monticello, IL, USA, 2021. [Google Scholar]
- Molin, J.P.; Tavares, T.R. Sensor Systems for Mapping Soil Fertility Attributes: Challenges, Advances, and Perspectives in Brazilian Tropical Soils. Eng. Agric. 2019, 39, 126–147. [Google Scholar] [CrossRef]
- Yin, H.; Cao, Y.; Marelli, B.; Zeng, X.; Mason, A.J.; Cao, C. Soil Sensors and Plant Wearables for Smart and Precision Agriculture. Adv. Mater. 2021, 33, 2007764. [Google Scholar] [CrossRef] [PubMed]
- Johnston, A.E.; Poulton, P.R. The Importance of Long-Term Experiments in Agriculture: Their Management to Ensure Continued Crop Production and Soil Fertility; the Rothamsted Experience. Eur. J. Soil Sci. 2018, 69, 113–125. [Google Scholar] [CrossRef] [PubMed]
- Foley, J.A.; DeFries, R.; Asner, G.P.; Barford, C.; Bonan, G.; Carpenter, S.R.; Chapin, F.S.; Coe, M.T.; Daily, G.C.; Gibbs, H.K.; et al. Global Consequences of Land Use. Science 2005, 309, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.; House, J.I.; Bustamante, M.; Sobocká, J.; Harper, R.; Pan, G.; West, P.C.; Clark, J.M.; Adhya, T.; Rumpel, C.; et al. Global Change Pressures on Soils from Land Use and Management. Glob. Chang. Biol. 2016, 22, 1008–1028. [Google Scholar] [CrossRef]
- Vasques, G.M.; Rodrigues, H.M.; Coelho, M.R.; Baca, J.F.M.; Dart, R.O.; Oliveira, R.P.; Teixeira, W.G.; Ceddia, M.B. Field Proximal Soil Sensor Fusion for Improving High-Resolution Soil Property Maps. Soil Syst. 2020, 4, 52. [Google Scholar] [CrossRef]
- Stenberg, B.; Viscarra Rossel, R.A.; Mouazen, A.M.; Wetterlind, J. Visible and Near Infrared Spectroscopy in Soil Science. In Advances in Agronomy; Elsevier: Amsterdam, The Netherlands, 2010; Volume 107. [Google Scholar]
- Nocita, M.; Stevens, A.; Noon, C.; Van Wesemael, B. Prediction of Soil Organic Carbon for Different Levels of Soil Moisture Using Vis-NIR Spectroscopy. Geoderma 2013, 199, 37–42. [Google Scholar] [CrossRef]
- Fang, Q.; Hong, H.; Zhao, L.; Kukolich, S.; Yin, K.; Wang, C. Visible and Near-Infrared Reflectance Spectroscopy for Investigating Soil Mineralogy: A Review. J. Spectrosc. 2018, 2018, 3168974. [Google Scholar] [CrossRef]
- Bönecke, E.; Meyer, S.; Vogel, S.; Schröter, I.; Gebbers, R.; Kling, C.; Kramer, E.; Lück, K.; Nagel, A.; Philipp, G.; et al. Guidelines for Precise Lime Management Based on High-Resolution Soil PH, Texture and SOM Maps Generated from Proximal Soil Sensing Data. Precis. Agric. 2021, 22, 493–523. [Google Scholar] [CrossRef]
- Munnaf, M.A.; Guerrero, A.; Nawar, S.; Haesaert, G.; Van Meirvenne, M.; Mouazen, A.M. A Combined Data Mining Approach for On-Line Prediction of Key Soil Quality Indicators by Vis-NIR Spectroscopy. Soil Tillage Res. 2021, 205, 104808. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, T.; Liu, J.; Lin, Z.; Li, S.; Wang, R.; Ge, Y. Soil PH Value, Organic Matter and Macronutrients Contents Prediction Using Optical Diffuse Reflectance Spectroscopy. Comput. Electron. Agric. 2015, 111, 69–77. [Google Scholar] [CrossRef]
- Munnaf, M.A.; Nawar, S.; Mouazen, A.M. Estimation of Secondary Soil Properties by Fusion of Laboratory and On-Line Measured Vis-NIR Spectra. Remote Sens. 2019, 11, 2819. [Google Scholar] [CrossRef]
- Munnaf, M.A.; Mouazen, A.M. Development of a Soil Fertility Index Using On-Line Vis-NIR Spectroscopy. Comput. Electron. Agric. 2021, 188, 106341. [Google Scholar] [CrossRef]
- Yang, M.; Mouazen, A.; Zhao, X.; Guo, X. Assessment of a Soil Fertility Index Using Visible and Near-Infrared Spectroscopy in the Rice Paddy Region of Southern China. Eur. J. Soil Sci. 2020, 71, 615–626. [Google Scholar] [CrossRef]
- Viscarra Rossel, R.A.; Adamchuk, V.I.; Sudduth, K.A.; McKenzie, N.J.; Lobsey, C. Proximal Soil Sensing. An Effective Approach for Soil Measurements in Space and Time. Adv. Agron. 2011, 113, 243–291. [Google Scholar] [CrossRef]
- Ben-Dor, E.; Heller, D.; Chudnovsky, A. A Novel Method of Classifying Soil Profiles in the Field Using Optical Means. Soil Sci. Soc. Am. J. 2008, 72, 1113–1123. [Google Scholar] [CrossRef]
- Mouazen, A.M.; Maleki, M.R.; Cockx, L.; Van Meirvenne, M.; Van Holm, L.H.J.; Merckx, R.; De Baerdemaeker, J.; Ramon, H. Optimum Three-Point Linkage Set up for Improving the Quality of Soil Spectra and the Accuracy of Soil Phosphorus Measured Using an on-Line Visible and near Infrared Sensor. Soil Tillage Res. 2009, 103, 144–152. [Google Scholar] [CrossRef]
- Stenberg, B.; Rogstrand, G.; Bölenius, E.; Arvidsson, J. On-Line Soil NIR Spectroscopy: Identification and Treatment of Spectra Influenced by Variable Probe Distance and Residue Contamination. In Proceedings of the 6th European Conference on Precision Agriculture: ECPA 2007, Skiathos, Greece, 3–6 June 2007. [Google Scholar]
- Barra, I.; Haefele, S.M.; Sakrabani, R.; Kebede, F. Soil Spectroscopy with the Use of Chemometrics, Machine Learning and Pre-Processing Techniques in Soil Diagnosis: Recent Advances—A Review. TrAC—Trends Anal. Chem. 2021, 135, 116166. [Google Scholar] [CrossRef]
- Morellos, A.; Pantazi, X.E.; Moshou, D.; Alexandridis, T.; Whetton, R.; Tziotzios, G.; Wiebensohn, J.; Bill, R.; Mouazen, A.M. Machine Learning Based Prediction of Soil Total Nitrogen, Organic Carbon and Moisture Content by Using VIS-NIR Spectroscopy. Biosyst. Eng. 2016, 152, 104–116. [Google Scholar] [CrossRef]
- Kuang, B.; Mahmood, H.S.; Quraishi, M.Z.; Hoogmoed, W.B.; Mouazen, A.M.; van Henten, E.J. Sensing Soil Properties in the Laboratory, in Situ, and on-Line. A Review. In Advances in Agronomy; Elsevier: Amsterdam, The Netherlands, 2012; Volume 114. [Google Scholar]
- Eitelwein, M.T.; Tavares, T.R.; Molin, J.P.; Trevisan, R.G.; de Sousa, R.V.; Demattê, J.A.M. Predictive Performance of Mobile Vis—NIR Spectroscopy for Mapping Key Fertility Attributes in Tropical Soils through Local Models Using PLS and ANN. Automation 2022, 3, 116–131. [Google Scholar] [CrossRef]
- Franceschini, M.H.D.; Demattê, J.A.M.; Kooistra, L.; Bartholomeus, H.; Rizzo, R.; Fongaro, C.T.; Molin, J.P. Effects of External Factors on Soil Reflectance Measured On-the-Go and Assessment of Potential Spectral Correction through Orthogonalisation and Standardisation Procedures. Soil Tillage Res. 2018, 177, 19–36. [Google Scholar] [CrossRef]
- Nawar, S.; Mouazen, A.M. Optimal Sample Selection for Measurement of Soil Organic Carbon Using On-Line Vis-NIR Spectroscopy. Comput. Electron. Agric. 2018, 151, 469–477. [Google Scholar] [CrossRef]
- Brown, D.J. Using a Global VNIR Soil-Spectral Library for Local Soil Characterization and Landscape Modeling in a 2nd-Order Uganda Watershed. Geoderma 2007, 140, 444–453. [Google Scholar] [CrossRef]
- Canal Filho, R.; Molin, J.P. Spatial Distribution as a Key Factor for Evaluation of Soil Attributes Prediction at Field Level Using Online Near-Infrared Spectroscopy. Front. Soil Sci. 2022, 2, 984963. [Google Scholar] [CrossRef]
- Wetterlind, J.; Stenberg, B.; Söderström, M. Increased Sample Point Density in Farm Soil Mapping by Local Calibration of Visible and near Infrared Prediction Models. Geoderma 2010, 156, 152–160. [Google Scholar] [CrossRef]
- Gogé, F.; Gomez, C.; Jolivet, C.; Joffre, R. Which Strategy Is Best to Predict Soil Properties of a Local Site from a National Vis-NIR Database? Geoderma 2014, 213, 1–9. [Google Scholar] [CrossRef]
- Stevens, A.; Nocita, M.; Tóth, G.; Montanarella, L.; van Wesemael, B. Prediction of Soil Organic Carbon at the European Scale by Visible and Near InfraRed Reflectance Spectroscopy. PLoS ONE 2013, 8, e66409. [Google Scholar] [CrossRef]
- Lee, H.; Beighley, R.E.; Alsdorf, D.; Jung, H.C.; Shum, C.K.; Duan, J.; Guo, J.; Yamazaki, D.; Andreadis, K. Characterization of Terrestrial Water Dynamics in the Congo Basin Using GRACE and Satellite Radar Altimetry. Remote Sens Env. 2011, 115, 3530–3538. [Google Scholar] [CrossRef]
- Uebbing, B.; Forootan, E.; Braakmann-Folgmann, A.; Kusche, J. Inverting Surface Soil Moisture Information from Satellite Altimetry over Arid and Semi-Arid Regions. Remote Sens. Environ. 2017, 196, 205–223. [Google Scholar] [CrossRef]
- Orth, R.; Koster, R.D.; Seneviratne, S.I. Inferring Soil Moisture Memory from Streamflow Observations Using a Simple Water Balance Model. J. Hydrometeorol. 2013, 14, 1773–1790. [Google Scholar] [CrossRef]
- Pasquini, C. Near Infrared Spectroscopy: A Mature Analytical Technique with New Perspectives—A Review. Anal. Chim. Acta 2018, 1026, 8–36. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.P.; Lee, C.K.; Dai, Y.H.; Shen, Y. Effect of Wetting on the Determination of Soil Organic Matter Content Using Visible and Near-Infrared Spectrometer. Geoderma 2020, 376, 114528. [Google Scholar] [CrossRef]
- Teixeira, P.C.; Donagemma, G.K.; Fontana, A.; Teixeira, W.G. Manual de Métodos de Análise de Solo; Embrapa: Brasilia, Brazil, 2017; Volume 3. [Google Scholar]
- Agarwal, A.; Shah, D.; Shen, D.; Song, D. On Robustness of Principal Component Regression. In Advances in Neural Information Processing Systems; NeurIPS: New Orleans, LA, USA, 2021; Volume 116. [Google Scholar] [CrossRef]
- Chang, C.W.; Laird, D.A.; Mausbach, M.J.; Hurburgh, C.R. Near-infrared reflectance spectroscopy–principal components regression analyses of soil properties. Soil Sci. Soc. Am. J. 2001, 65, 480–490. [Google Scholar] [CrossRef]
- Seasholtz, M.B.; Kowalski, B. The Parsimony Principle Applied to Multivariate Calibration. Anal Chim. Acta 1993, 277, 165–177. [Google Scholar] [CrossRef]
- Tracy, T.; Fu, Y.; Roy, I.; Jonas, E.; Glendenning, P. Towards Machine Learning on the Automata Processor. In High Performance Computing, Proceedings of the 31st International Conference, ISC High Performance 2016, Frankfurt, Germany, 19–23 June 2016; Springer: Berlin/Heidelberg, Germany, 2016; Volume 9697, p. 9697. [Google Scholar]
- Kluyver, T.; Ragan-Kelley, B.; Pérez, F.; Granger, B.; Bussonnier, M.; Frederic, J.; Kelley, K.; Hamrick, J.; Grout, J.; Corlay, S.; et al. Jupyter Notebooks—A Publishing Format for Reproducible Computational Workflows. In Positioning and Power in Academic Publishing: Players, Agents and Agendas, Proceedings of the 20th International Conference on Electronic Publishing, Göttingen, Germany, 7–9 June 2016; ELPUB: Stuttgart, Germany, 2016. [Google Scholar]
- Python Software 3.9.12; Foundation Python: Wilmington, DE, USA, 2022.
- Jung, Y. Multiple Predicting K-Fold Cross-Validation for Model Selection. J. Nonparametr. Stat. 2018, 30, 197–215. [Google Scholar] [CrossRef]
- Minasny, B.; McBratney, A.B.; Whelan, B.M. VESPER, Version 1.62; Australian Centre for Precision Agriculture, The University of Sydney: Sydney, Australia, 2006. [Google Scholar]
- QGIS Development Team. QGIS Geographic Information System; QGIS: London, UK, 2022. [Google Scholar]
- Nocita, M.; Stevens, A.; van Wesemael, B.; Aitkenhead, M.; Bachmann, M.; Barthès, B.; Ben-Dor, E.; Brown, D.J.; Clairotte, M.; Csorba, A.; et al. Soil Spectroscopy: An Alternative to Wet Chemistry for Soil Monitoring. Adv. Agron. 2015, 132, 139–159. [Google Scholar] [CrossRef]
- Pavinato, P.S.; Cherubin, M.R.; Soltangheisi, A.; Rocha, G.C.; Chadwick, D.R.; Jones, D.L. Revealing Soil Legacy Phosphorus to Promote Sustainable Agriculture in Brazil. Sci. Rep. 2020, 10, 15615. [Google Scholar] [CrossRef] [PubMed]
- Mouazen, A.M.; Kuang, B. On-Line Visible and near Infrared Spectroscopy for in-Field Phosphorous Management. Soil Tillage Res. 2016, 155, 471–477. [Google Scholar] [CrossRef]
- Jackson, M.L.; Sherman, G.D. Chemical Weathering of Minerals in Soils. Adv. Agron. 1953, 5, 219–318. [Google Scholar] [CrossRef]
- Lu, Y.; Gao, Y.; Nie, J.; Liao, Y.; Zhu, Q. Substituting Chemical P Fertilizer with Organic Manure: Effects on Double-Rice Yield, Phosphorus Use Efficiency and Balance in Subtropical China. Sci. Rep. 2021, 11, 8629. [Google Scholar] [CrossRef]
- Wang, H.; Xu, J.; Liu, X.; Zhang, D.; Li, L.; Li, W.; Sheng, L. Effects of Long-Term Application of Organic Fertilizer on Improving Organic Matter Content and Retarding Acidity in Red Soil from China. Soil Tillage Res. 2019, 195, 104382. [Google Scholar] [CrossRef]
- Kortüm, G.; Braun, W.; Herzog, G. Principles and Techniques of Diffuse-Reflectance Spectroscopy. Angew. Chem. Int. Ed. Engl. 1963, 2, 333–341. [Google Scholar] [CrossRef]
- Terra, F.S.; Demattê, J.A.M.; Viscarra Rossel, R.A. Proximal Spectral Sensing in Pedological Assessments: Vis-NIR Spectra for Soil Classification Based on Weathering and Pedogenesis. Geoderma 2018, 318, 123–136. [Google Scholar] [CrossRef]
- Rasa, K.; Heikkinen, J.; Hannula, M.; Arstila, K.; Kulju, S.; Hyväluoma, J. How and Why Does Willow Biochar Increase a Clay Soil Water Retention Capacity? Biomass Bioenergy 2018, 119, 346–353. [Google Scholar] [CrossRef]
- Shepherd, M.A.; Harrison, R.; Webb, J. Managing Soil Organic Matter—Implications for Soil Structure on Organic Farms. Soil Use Manag. 2002, 18, 284–292. [Google Scholar] [CrossRef]
- Soane, B.D. The Role of Organic Matter in Soil Compactibility: A Review of Some Practical Aspects. Soil Tillage Res. 1990, 16, 179–201. [Google Scholar] [CrossRef]
- Huang, J.; Zare, E.; Malik, R.S.; Triantafilis, J. An Error Budget for Soil Salinity Mapping Using Different Ancillary Data. Soil Res. 2015, 53, 561–575. [Google Scholar] [CrossRef]
- Vishwakarma, G.; Sonpal, A.; Hachmann, J. Metrics for Benchmarking and Uncertainty Quantification: Quality, Applicability, and Best Practices for Machine Learning in Chemistry. Trends Chem. 2021, 3, 146–156. [Google Scholar] [CrossRef]
- Wijewardane, N.K.; Ge, Y.; Morgan, C.L.S. Moisture Insensitive Prediction of Soil Properties from VNIR Reflectance Spectra Based on External Parameter Orthogonalization. Geoderma 2016, 267, 92–101. [Google Scholar] [CrossRef]
- Rodionov, A.; Welp, G.; Damerow, L.; Berg, T.; Amelung, W.; Pätzold, S. Towards on-the-go field assessment of soil organic carbon using Vis-NIR diffuse reflectance spectroscopy: Developing and testing a novel tractor-driven measuring chamber. Soil Tillage Res. 2015, 145, 93–102. [Google Scholar] [CrossRef]



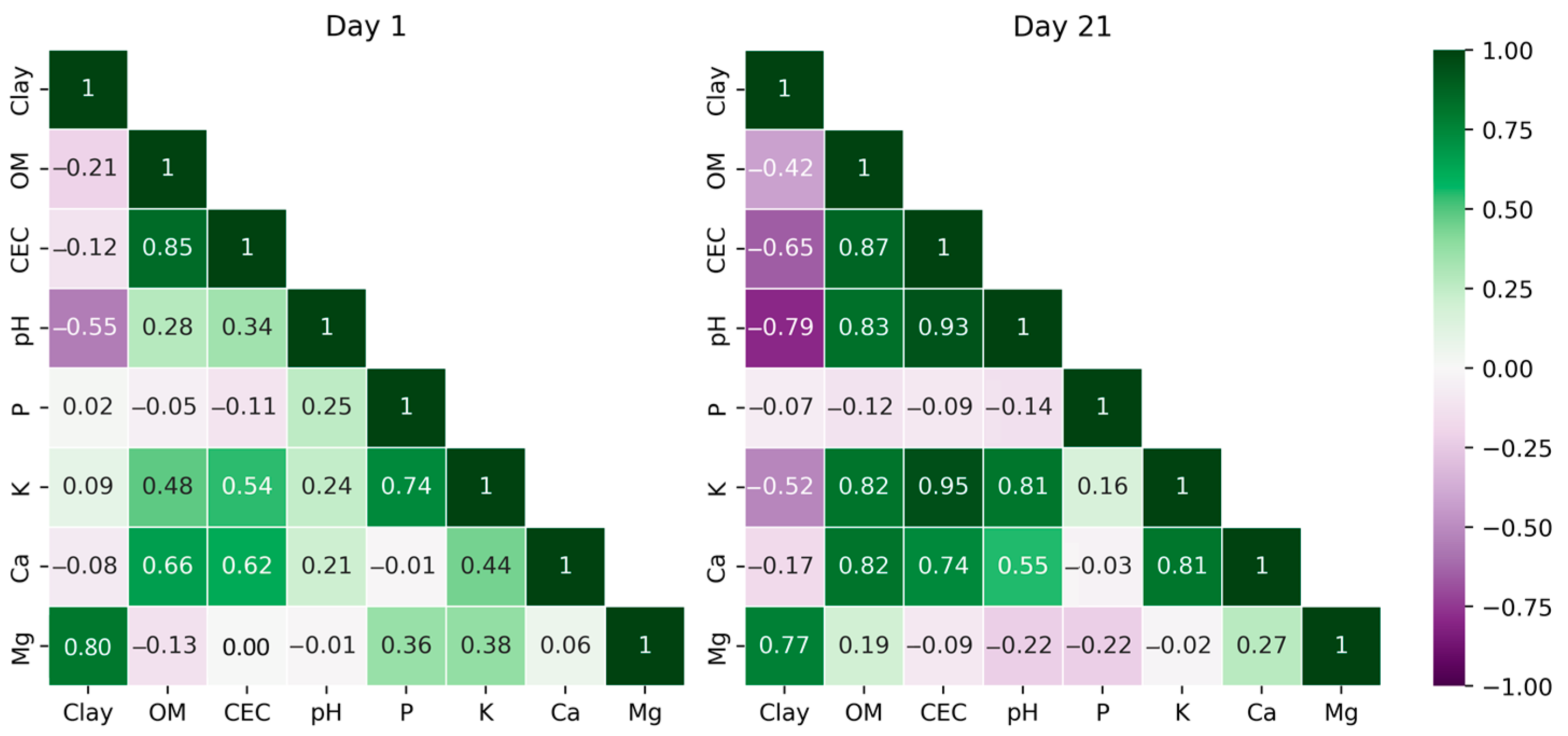
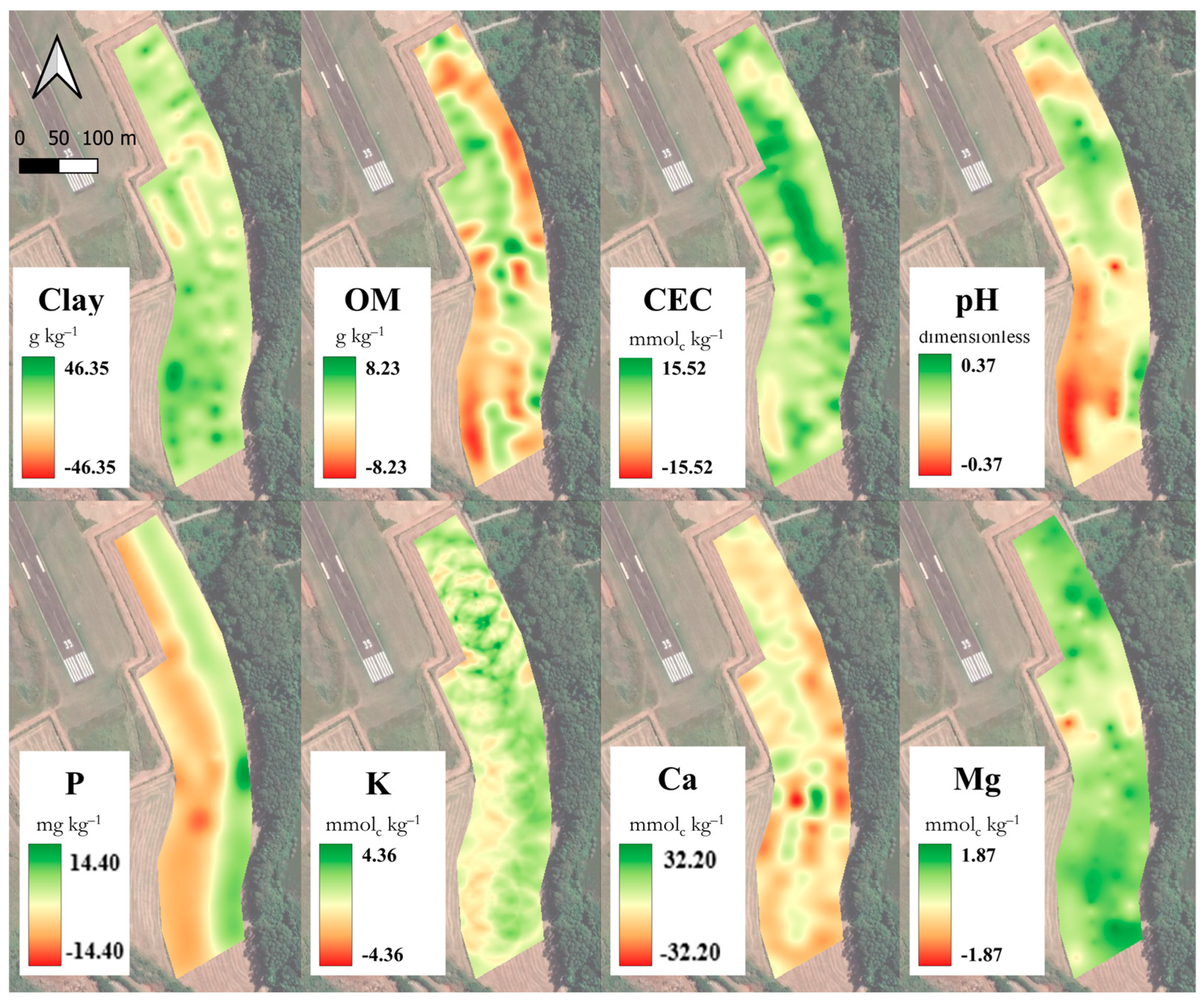
| Unit | Min | Max | Range | R2 | RMSE | MAE | NC | % Var | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Clay | g kg−1 | 51 | - | 183 | 132 | 0.17 | 19.88 | 15.08 | 4 | 24.01 |
| OM | 12 | - | 35 | 23 | 0.75 | 3.11 | 2.28 | 9 | 40.69 | |
| CEC | mmolc kg−1 | 45 | - | 68 | 24 | 0.60 | 3.51 | 2.78 | 6 | 26.70 |
| pH | - | 4.1 | - | 6.8 | 2.7 | 0.03 | 0.32 | 0.27 | 4 | 12.09 |
| P | mg kg−1 | 5 | - | 68 | 63 | 0.02 | 9.39 | 8.59 | 3 | 18.42 |
| K | mmolc kg−1 | 0.4 | - | 5.0 | 4.6 | 0.14 | 0.93 | 0.77 | 6 | 32.68 |
| Ca | 10 | - | 34 | 24 | 0.39 | 2.54 | 2.08 | 10 | 58.62 | |
| Mg | 5 | - | 22 | 17 | 0.01 | 1.83 | 1.41 | 1 | 5.85 |
| Clay | OM | CEC | pH | P | K | Ca | Mg | |
|---|---|---|---|---|---|---|---|---|
| Overlap | −0.10 | 0.09 | −0.11 | −0.19 | −0.73 ** | −0.22 | 0.33 | 0.13 |
| High correlated | −0.06 | 0.19 | 0.32 | 0.04 | −0.80 ** | −0.03 | 0.24 | 0.19 |
| Total | −0.02 | −0.03 | −0.14 | −0.26 ** | −0.56 ** | −0.01 | 0.22 ** | 0.20 |
| Day 1 | Day 21 | |||||||
|---|---|---|---|---|---|---|---|---|
| Model | C0 | C1 | A | Model | C0 | C1 | A | |
| Clay | Sph | 13.59 | 102.60 | 24.10 | Exp | 42.71 | 59.70 | 36.80 |
| OM | Exp | 1.82 | 3.85 | 23.00 | Gau | 0.00 | 10.18 | 21.10 |
| CEC | Exp | 0.00 | 8.50 | 18.50 | Gau | 0.00 | 10.82 | 20.50 |
| pH | Lin | 0.00 | 0.01 | 88.90 | Exp | 0.00 | 0.02 | 16.70 |
| P | Gau | 0.24 | 21.40 | 32.90 | Exp | 0.03 | 0.80 | 37.60 |
| K | Gau | 0.10 | 0.46 | 27.30 | Lin | 0.01 | 0.19 | 35.90 |
| Ca | Sph | 5.62 | 4.50 | 95.60 | Gau | 0.00 | 26.79 | 25.20 |
| Mg | Exp | 0.42 | 0.32 | 46.40 | Exp | 0.02 | 0.26 | 17.50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Canal Filho, R.; Molin, J.P.; Wei, M.C.F.; Silva, E.R.O.d. Soil Attributes Mapping with Online Near-Infrared Spectroscopy Requires Spatio-Temporal Local Calibrations. AgriEngineering 2023, 5, 1163-1177. https://doi.org/10.3390/agriengineering5030074
Canal Filho R, Molin JP, Wei MCF, Silva EROd. Soil Attributes Mapping with Online Near-Infrared Spectroscopy Requires Spatio-Temporal Local Calibrations. AgriEngineering. 2023; 5(3):1163-1177. https://doi.org/10.3390/agriengineering5030074
Chicago/Turabian StyleCanal Filho, Ricardo, José Paulo Molin, Marcelo Chan Fu Wei, and Eudocio Rafael Otavio da Silva. 2023. "Soil Attributes Mapping with Online Near-Infrared Spectroscopy Requires Spatio-Temporal Local Calibrations" AgriEngineering 5, no. 3: 1163-1177. https://doi.org/10.3390/agriengineering5030074
APA StyleCanal Filho, R., Molin, J. P., Wei, M. C. F., & Silva, E. R. O. d. (2023). Soil Attributes Mapping with Online Near-Infrared Spectroscopy Requires Spatio-Temporal Local Calibrations. AgriEngineering, 5(3), 1163-1177. https://doi.org/10.3390/agriengineering5030074







