Green Valorization of Olive Leaves to Produce Polyphenol-Enriched Extracts Using an Environmentally Benign Deep Eutectic Solvent
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Collection of Plant Material and Handling
2.3. Synthesis of DES
2.4. Batch Stirred-Tank Extraction
2.5. Design of Experiment—Response Surface Methodology
2.6. Total Polyphenol Determination
2.7. Total Flavonoid Determination
2.8. Antiradical Activity (AAR) Determination
2.9. Ferric-Reducing Power (PR) Determination
2.10. Liquid Chromatography-Diode Array-Mass Spectrometry (LC-DAD-MS)
2.11. High-Performance Liquid Chromatography (HPLC)
2.12. Statistical Analysis
3. Results and Discussion
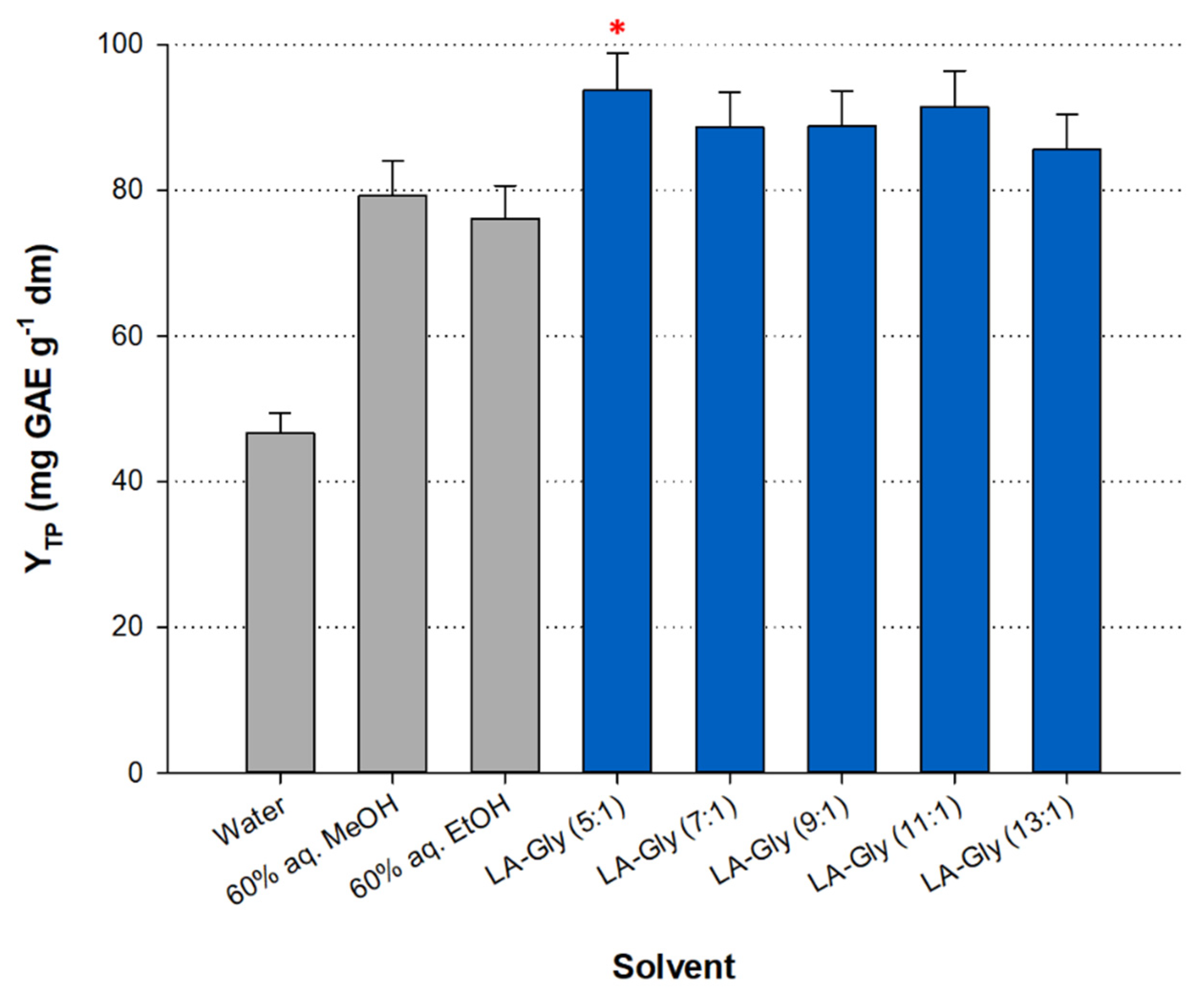
3.1. Effect of DES Composition
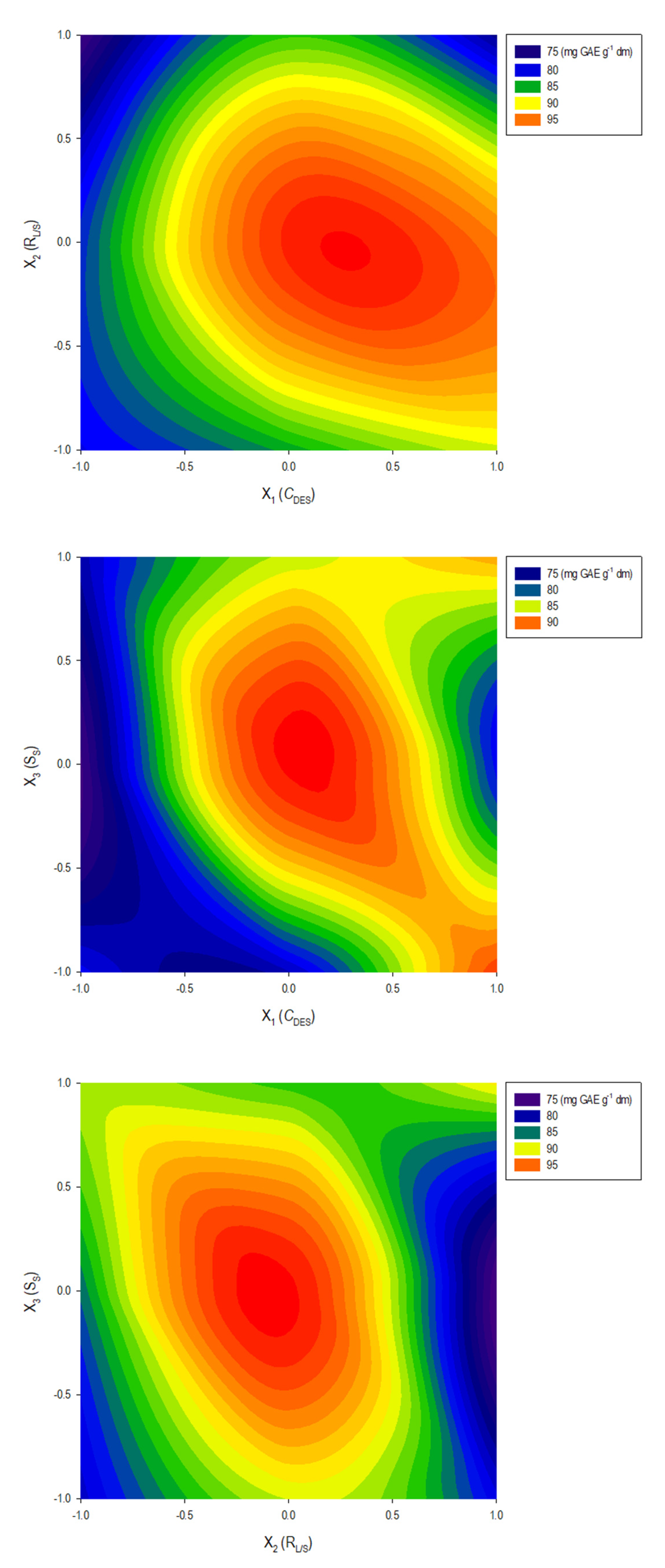
3.2. Extraction Process Optimization
3.3. Temperature Effects
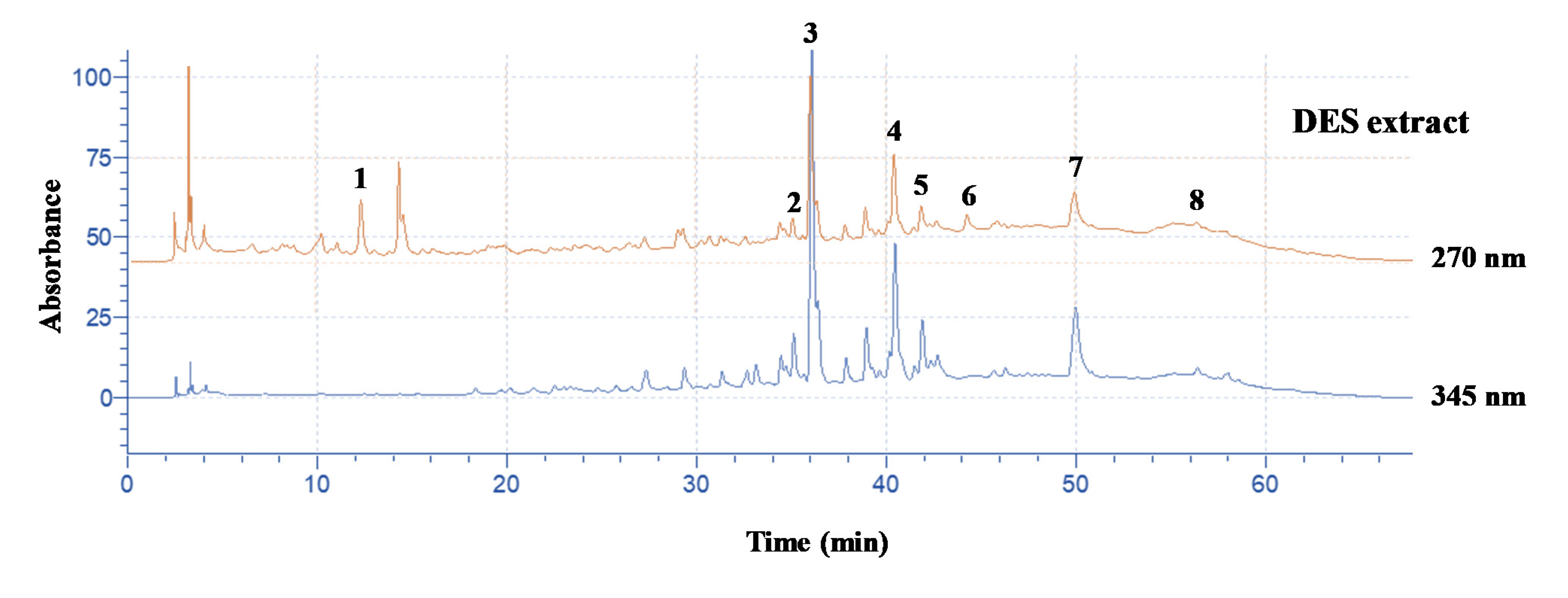
3.4. Polyphenolic Composition
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Nomenclature
| AAR | antiradical activity (μmol DPPH g−1) |
| CDES | proportion of DES/water (%, w/v) |
| PR | reducing power (μmol AAE g−1) |
| RL/S | liquid-to-solid ratio (mL g−1) |
| SS | stirring speed (rpm) |
| t | time (min) |
| T | temperature (°C) |
| YTFn | yield in total flavonoids (mg RtE g−1) |
| YTP | yield in total polyphenols (mg GAE g−1) |
Abbreviations
| AAE | ascorbic acid equivalents |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl radical |
| GAE | gallic acid equivalents |
| Gly | glycine |
| HBA | hydrogen bond acceptor |
| LA | lactic acid |
| OLL | olive leaves |
| TPTZ | 2,4,6-tripyridyl-s-triazine |
References
- Pfaltzgraff, L.A.; Cooper, E.C.; Budarin, V.; Clark, J.H. Food waste biomass: A resource for high-value chemicals. Green Chem. 2013, 15, 307–314. [Google Scholar] [CrossRef]
- Zuin, V.G.; Ramin, L.Z. Green and sustainable separation of natural products from agro-industrial waste: Challenges, potentialities, and perspectives on emerging approaches. In Chemistry and Chemical Technologies in Waste Valorization; Springer: Berlin/Heidelberg, Germany, 2018; pp. 229–282. [Google Scholar]
- Chemat, F.; Abert Vian, M.; Ravi, H.K.; Khadhraoui, B.; Hilali, S.; Perino, S.; Fabiano Tixier, A.-S. Review of alternative solvents for green extraction of food and natural products: Panorama, principles, applications and prospects. Molecules 2019, 24, 3007. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torres-Valenzuela, L.S.; Ballesteros-Gómez, A.; Rubio, S. Green solvents for the extraction of high added-value compounds from agri-food waste. Food Eng. Rev. 2019, 12, 83–100. [Google Scholar] [CrossRef]
- Vanda, H.; Dai, Y.; Wilson, E.G.; Verpoorte, R.; Choi, Y.H. Green solvents from ionic liquids and deep eutectic solvents to natural deep eutectic solvents. Comptes Rendus Chim. 2018, 21, 628–638. [Google Scholar] [CrossRef]
- Skarpalezos, D.; Detsi, A. Deep eutectic solvents as extraction media for valuable flavonoids from natural sources. Appl. Sci. 2019, 9, 4169. [Google Scholar] [CrossRef] [Green Version]
- Peralbo-Molina, Á.; de Castro, M.D.L. Potential of residues from the Mediterranean agriculture and agrifood industry. Trends Food Sci. Technol. 2013, 32, 16–24. [Google Scholar] [CrossRef]
- Lakka, A.; Grigorakis, S.; Karageorgou, I.; Batra, G.; Kaltsa, O.; Bozinou, E.; Lalas, S.; Makris, D.P. Saffron processing wastes as a bioresource of high-value added compounds: Development of a green extraction process for polyphenol recovery using a natural deep eutectic solvent. Antioxidants 2019, 8, 586. [Google Scholar] [CrossRef] [Green Version]
- Mylonaki, S.; Kiassos, E.; Makris, D.P.; Kefalas, P. Optimisation of the extraction of olive (Olea europaea) leaf phenolics using water/ethanol-based solvent systems and response surface methodology. Anal. Bioanal. Chem. 2008, 392, 977. [Google Scholar] [CrossRef]
- Lakka, A.; Karageorgou, I.; Kaltsa, O.; Batra, G.; Bozinou, E.; Lalas, S.; Makris, D. Polyphenol extraction from Humulus lupulus (hop) using a neoteric glycerol/L-alanine deep eutectic solvent: Optimisation, kinetics and the effect of ultrasound-assisted pretreatment. AgriEngineering 2019, 1, 403–417. [Google Scholar] [CrossRef] [Green Version]
- Lakka, A.; Grigorakis, S.; Kaltsa, O.; Karageorgou, I.; Batra, G.; Bozinou, E.; Lalas, S.; Makris, D.P. The effect of ultrasonication pretreatment on the production of polyphenol-enriched extracts from Moringa oleifera L. (drumstick tree) using a novel bio-based deep eutectic solvent. Appl. Sci. 2020, 10, 220. [Google Scholar] [CrossRef] [Green Version]
- Cicco, N.; Lanorte, M.T.; Paraggio, M.; Viggiano, M.; Lattanzio, V. A reproducible, rapid and inexpensive Folin–Ciocalteu micro-method in determining phenolics of plant methanol extracts. Microchem. J. 2009, 91, 107–110. [Google Scholar] [CrossRef]
- Manousaki, A.; Jancheva, M.; Grigorakis, S.; Makris, D. Extraction of antioxidant phenolics from agri-food waste biomass using a newly designed glycerol-based natural low-transition temperature mixture: A comparison with conventional eco-friendly solvents. Recycling 2016, 1, 194–204. [Google Scholar] [CrossRef]
- Paleologou, I.; Vasiliou, A.; Grigorakis, S.; Makris, D.P. Optimisation of a green ultrasound-assisted extraction process for potato peel (Solanum tuberosum) polyphenols using bio-solvents and response surface methodology. Biomass Convers. Bioref. 2016, 6, 289–299. [Google Scholar] [CrossRef]
- Karakashov, B.; Grigorakis, S.; Loupassaki, S.; Makris, D.P. Optimisation of polyphenol extraction from Hypericum perforatum (St. John’s Wort) using aqueous glycerol and response surface methodology. J. Appl. Res. Med. Aromat. Plants 2015, 2, 1–8. [Google Scholar] [CrossRef]
- Dai, Y.; Witkamp, G.-J.; Verpoorte, R.; Choi, Y.H. Tailoring properties of natural deep eutectic solvents with water to facilitate their applications. Food Chem. 2015, 187, 14–19. [Google Scholar] [CrossRef]
- Chakroun, D.; Grigorakis, S.; Loupassaki, S.; Makris, D.P. Enhanced-performance extraction of olive (Olea europaea) leaf polyphenols using L-lactic acid/ammonium acetate deep eutectic solvent combined with β-cyclodextrin: Screening, optimisation, temperature effects and stability. Biomass Convers. Bioref. 2019. [Google Scholar] [CrossRef]
- Stefou, I.; Grigorakis, S.; Loupassaki, S.; Makris, D.P. Development of sodium propionate-based deep eutectic solvents for polyphenol extraction from onion solid wastes. Clean Technol. Environ. Policy 2019, 21, 1563–1574. [Google Scholar] [CrossRef]
- Cacace, J.; Mazza, G. Mass transfer process during extraction of phenolic compounds from milled berries. J. Food Eng. 2003, 59, 379–389. [Google Scholar] [CrossRef]
- Ho, C.H.; Cacace, J.E.; Mazza, G. Mass transfer during pressurized low polarity water extraction of lignans from flaxseed meal. J. Food Eng. 2008, 89, 64–71. [Google Scholar] [CrossRef]
- Rakotondramasy-Rabesiaka, L.; Havet, J.-L.; Porte, C.; Fauduet, H. Estimation of effective diffusion and transfer rate during the protopine extraction process from Fumaria officinalis L. Sep. Purif. Technol. 2010, 76, 126–131. [Google Scholar] [CrossRef]
- Pinelo, M.; Zornoza, B.; Meyer, A.S. Selective release of phenols from apple skin: Mass transfer kinetics during solvent and enzyme-assisted extraction. Sep. Purif. Technol. 2008, 63, 620–627. [Google Scholar] [CrossRef]
- Shewale, S.; Rathod, V.K. Extraction of total phenolic content from Azadirachta indica or (neem) leaves: Kinetics study. Prep. Biochem. Biotech. 2018, 48, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Alañón, M.E.; Ivanović, M.; Gómez-Caravaca, A.M.; Arráez-Román, D.; Segura-Carretero, A. Choline chloride derivative-based deep eutectic liquids as novel green alternative solvents for extraction of phenolic compounds from olive leaf. Arabian J. Chem. 2018, 13, 1685–1701. [Google Scholar] [CrossRef]
- Athanasiadis, V.; Grigorakis, S.; Lalas, S.; Makris, D.P. Highly efficient extraction of antioxidant polyphenols from Olea europaea leaves using an eco-friendly glycerol/glycine deep eutectic solvent. Waste Biomass Valorif. 2018, 9, 1985–1992. [Google Scholar] [CrossRef]
- Dedousi, M.; Mamoudaki, V.; Grigorakis, S.; Makris, D.P. Ultrasound-assisted extraction of polyphenolic antioxidants from olive (Olea europaea) leaves using a novel glycerol/sodium-potassium tartrate low-transition temperature mixture (LTTM). Environments 2017, 4, 31. [Google Scholar] [CrossRef] [Green Version]
- Slim, Z.; Jancheva, M.; Grigorakis, S.; Makris, D.P. Polyphenol extraction from Origanum dictamnus using low-transition temperature mixtures composed of glycerol and organic salts: Effect of organic anion carbon chain length. Chem. Eng. Commun. 2018, 205, 1494–1506. [Google Scholar] [CrossRef]
- Khiari, Z.; Makris, D.P.; Kefalas, P. An investigation on the recovery of antioxidant phenolics from onion solid wastes employing water/ethanol-based solvent systems. Food Bioprocess Technol. 2009, 2, 337. [Google Scholar] [CrossRef]
- Shang, X.; Dou, Y.; Zhang, Y.; Tan, J.-N.; Liu, X.; Zhang, Z. Tailor-made natural deep eutectic solvents for green extraction of isoflavones from chickpea (Cicer arietinum L.) sprouts. Ind. Crops Prod. 2019, 140, 111724. [Google Scholar] [CrossRef]
- Shehata, E.; Grigorakis, S.; Loupassaki, S.; Makris, D.P. Extraction optimisation using water/glycerol for the efficient recovery of polyphenolic antioxidants from two Artemisia species. Sep. Purif. Technol. 2015, 149, 462–469. [Google Scholar] [CrossRef]
- Jancheva, M.; Grigorakis, S.; Loupassaki, S.; Makris, D.P. Optimised extraction of antioxidant polyphenols from Satureja thymbra using newly designed glycerol-based natural low-transition temperature mixtures (LTTMs). J. Appl. Res. Med. Aromat. Plants 2017, 6, 31–40. [Google Scholar] [CrossRef]
- Athanasiadis, V.; Grigorakis, S.; Lalas, S.; Makris, D.P. Methyl β-cyclodextrin as a booster for the extraction for Olea europaea leaf polyphenols with a bio-based deep eutectic solvent. Biomass Convers. Bioref. 2018, 8, 345–355. [Google Scholar] [CrossRef]
- Cifá, D.; Skrt, M.; Pittia, P.; Di Mattia, C.; Poklar Ulrih, N. Enhanced yield of oleuropein from olive leaves using ultrasound-assisted extraction. Food Sci. Nutr. 2018, 6, 1128–1137. [Google Scholar] [CrossRef]
- Hassen, I.; Casabianca, H.; Hosni, K. Biological activities of the natural antioxidant oleuropein: Exceeding the expectation–A mini-review. J. Funct. Foods 2015, 18, 926–940. [Google Scholar] [CrossRef]
- Athanasiadis, V.; Grigorakis, S.; Lalas, S.; Makris, D.P. Stability effects of methyl β-cyclodextrin on Olea europaea leaf extracts in a natural deep eutectic solvent. Eur. Food Res. Technol. 2018, 244, 1783–1792. [Google Scholar] [CrossRef]
- Briante, R.; La Cara, F.; Tonziello, M.P.; Febbraio, F.; Nucci, R. Antioxidant activity of the main bioactive derivatives from oleuropein hydrolysis by hyperthermophilic β-glycosidase. J. Agric. Food Chem. 2001, 49, 3198–3203. [Google Scholar] [CrossRef]
- Yuan, J.-J.; Wang, C.-Z.; Ye, J.-Z.; Tao, R.; Zhang, Y.-S. Enzymatic hydrolysis of oleuropein from Olea europea (olive) leaf extract and antioxidant activities. Molecules 2015, 20, 2903–2921. [Google Scholar] [CrossRef] [Green Version]

| Independent Variables | Code Units | Coded Variable Level | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| CDES (%, w/v) | X1 | 55 | 70 | 85 |
| RL/S (mL g−1) | X2 | 20 | 40 | 60 |
| SS (rpm) | X3 | 200 | 500 | 800 |
| Solvent | YTP (mg GAE g−1 dm) | YTFn (mg RtE g−1 dm) | AAR (μmol DPPH g−1 dm) | PR (μmol AAE g−1 dm) |
|---|---|---|---|---|
| Water | 46.64 ± 2.80 | 7.81 ± 0.47 | 265.93 ± 5.32 | 213.15 ± 3.20 |
| 60% MeOH | 79.23 ± 4.75 | 18.33 ± 1.10 | 381.17 ± 7.62 | 276.26 ± 4.14 |
| 60% EtOH | 76.07 ± 4.56 | 22.12 ± 1.33 | 347.93 ± 6.96 | 265.43 ± 3.98 |
| LA-Gly (5:1) a | 93.73 ± 5.16 * | 17.28 ± 0.95 | 508.60 ± 10.17 * | 400.61 ± 6.01 * |
| Term | Standard Error | t Ratio | Probability > t | Sum of Squares | F Ratio |
|---|---|---|---|---|---|
| CDES | 0.901165 | 5.44 | 0.0029* | 192.17801 | 29.5805 |
| RL/S | 0.901165 | −4.85 | 0.0047* | 152.68781 | 23.5020 |
| SS | 0.901165 | −0.78 | 0.4694 | 3.97620 | 0.6120 |
| CDES RL/S | 1.27444 | −0.78 | 0.4682 | 4.00000 | 0.6157 |
| CDES SS | 1.27444 | −0.24 | 0.8218 | 0.36602 | 0.0563 |
| RL/S SS | 1.27444 | −3.13 | 0.0259 * | 63.76023 | 9.8141 |
| CDESCDES | 1.326479 | −3.61 | 0.0154 * | 84.74616 | 13.0443 |
| RL/S RL/S | 1.326479 | −8.90 | 0.0003 * | 514.62536 | 79.2123 |
| SS SS | 1.326479 | −2.41 | 0.0611 | 37.65186 | 5.7955 |
| Lack-of-fit | 0.3241 | 32.483942 | 2.2340 |
| Design Point | Independent Variables | Response (YTP, mg GAE g−1 dm) | |||
|---|---|---|---|---|---|
| X1 (CDES, % w/v) | X2 (RL/S, mL g−1) | X3 (SS, rpm) | Measured | Predicted | |
| 1 | −1 (55) | −1 (20) | 0 (500) | 79.88 | 77.64 |
| 2 | −1 (55) | 1 (60) | 0 (500) | 71.67 | 70.90 |
| 3 | 1 (85) | −1 (20) | 0 (500) | 88.67 | 89.44 |
| 4 | 1 (85) | 1 (60) | 0 (500) | 76.46 | 78.70 |
| 5 | 0 (70) | −1 (20) | −1 (200) | 80.55 | 81.85 |
| 6 | 0 (70) | −1 (20) | 1 (800) | 88.25 | 88.42 |
| 7 | 0 (70) | 1 (60) | −1 (200) | 81.27 | 81.10 |
| 8 | 0 (70) | 1 (60) | 1 (800) | 73.00 | 71.70 |
| 9 | −1 (55) | 0 (40) | −1 (200) | 82.34 | 83.28 |
| 10 | 1 (85) | 0 (40) | −1 (200) | 95.76 | 93.69 |
| 11 | −1 (55) | 0 (40) | 1 (800) | 80.41 | 82.48 |
| 12 | 1 (85) | 0 (40) | 1 (800) | 92.62 | 91.68 |
| 13 | 0 (70) | 0 (40) | 0 (500) | 93.54 | 95.77 |
| 14 | 0 (70) | 0 (40) | 0 (500) | 97.00 | 95.77 |
| 15 | 0 (70) | 0 (40) | 0 (500) | 96.76 | 95.77 |
| T (° C) | YTP (mg GAE g−1 dm) | YTFn (mg RtE g−1 dm) | AAR (μmol DPPH g−1 dm) | PR (μmol AAE g−1 dm) |
|---|---|---|---|---|
| 50 | 96.41 ± 2.27 | 19.47 ± 0.68 | 637.14 ± 11.57 | 407.00 ± 7.00 |
| 80 | 98.77 ± 2.00 | 26.44 ± 0.98 | 773.21 ± 13.89 | 461.18 ± 8.11 |
| Polyphenol | Content (mg g−1 dm) ± sd |
|---|---|
| Hydroxytyrosol | 8.20 ± 0.12 |
| Rutin | 0.28 ± 0.00 |
| Luteolin 7-O-glucoside | 2.59 ± 0.04 |
| Apigenin 7-O-rutinoside | 0.36 ± 0.01 |
| Luteolin 3’-O-glucoside | 0.36 ± 0.01 |
| Oleuropein | 2.88 ± 0.04 |
| Quercetin | 0.44 ± 0.01 |
| Apigenin | 0.01 ± 0.00 |
| Sum | 15.13 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaltsa, O.; Grigorakis, S.; Lakka, A.; Bozinou, E.; Lalas, S.; Makris, D.P. Green Valorization of Olive Leaves to Produce Polyphenol-Enriched Extracts Using an Environmentally Benign Deep Eutectic Solvent. AgriEngineering 2020, 2, 226-239. https://doi.org/10.3390/agriengineering2020014
Kaltsa O, Grigorakis S, Lakka A, Bozinou E, Lalas S, Makris DP. Green Valorization of Olive Leaves to Produce Polyphenol-Enriched Extracts Using an Environmentally Benign Deep Eutectic Solvent. AgriEngineering. 2020; 2(2):226-239. https://doi.org/10.3390/agriengineering2020014
Chicago/Turabian StyleKaltsa, Olga, Spyros Grigorakis, Achillia Lakka, Eleni Bozinou, Stavros Lalas, and Dimitris P. Makris. 2020. "Green Valorization of Olive Leaves to Produce Polyphenol-Enriched Extracts Using an Environmentally Benign Deep Eutectic Solvent" AgriEngineering 2, no. 2: 226-239. https://doi.org/10.3390/agriengineering2020014








