Probiotics and Phytochemicals: Role on Gut Microbiota and Efficacy on Irritable Bowel Syndrome, Functional Dyspepsia, and Functional Constipation
Abstract
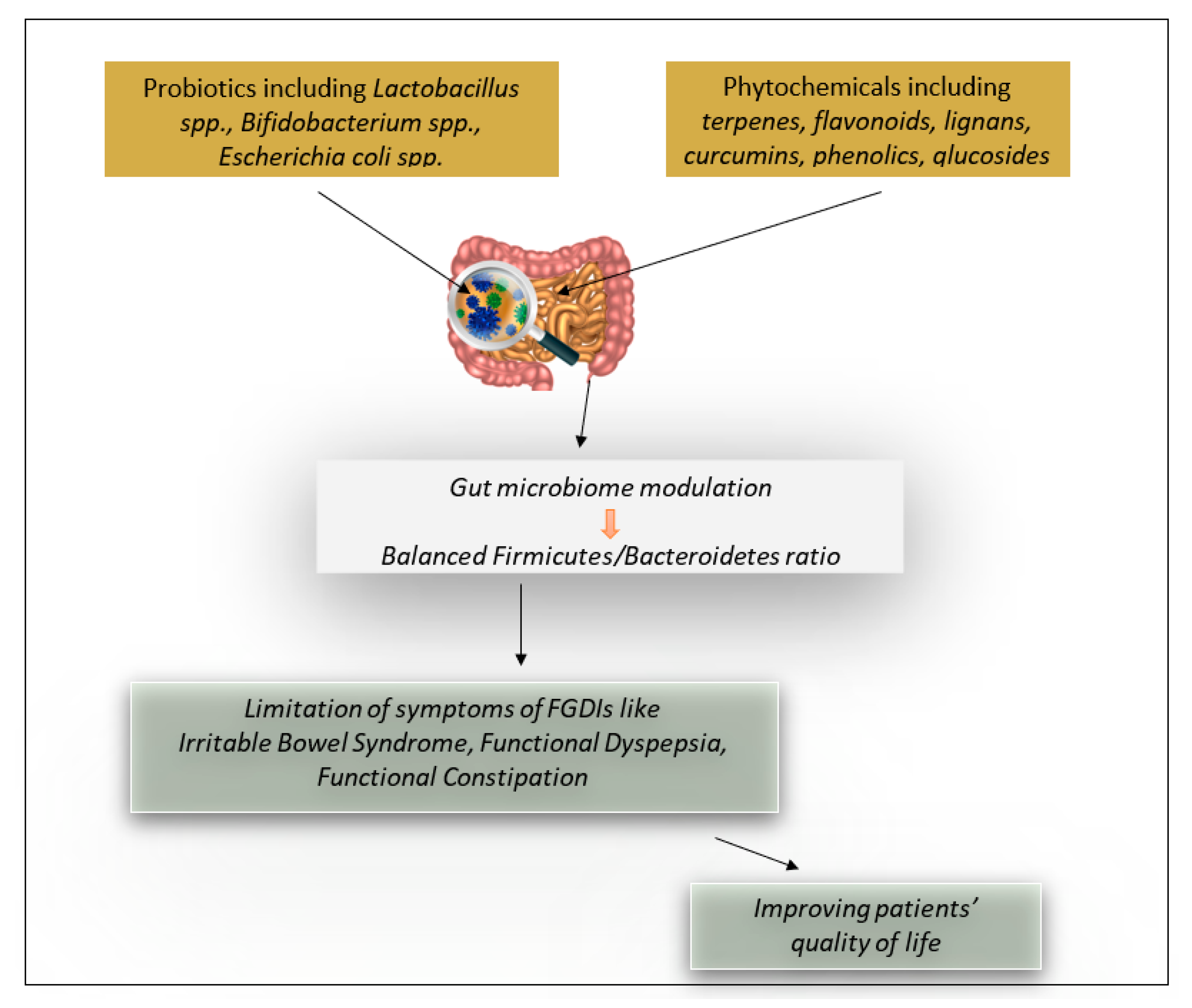
:1. Introduction
2. Results
2.1. Irritable Bowel Syndrome (IBS)
2.1.1. Efficacy of Probiotics in Irritable Bowel Syndrome
2.1.2. Efficacy of Phytochemicals in Treating Irritable Bowel Syndrome
2.2. Functional Dyspepsia (FD)
2.2.1. Efficacy of Probiotics in Functional Dyspepsia
2.2.2. Efficacy of Phytochemicals in Treating Functional Dyspepsia
2.3. Functional Constipation (FC)
2.3.1. Efficacy of Probiotics in Treating Functional Constipation
2.3.2. Efficacy of Phytochemicals in Treating Functional Constipation
3. Discussion
4. Methods
5. Conclusions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Gilbert, J.A.; Blaser, M.J.; Caporaso, J.G.; Jansson, J.K.; Lynch, S.V.; Knight, R. Current Understanding of the Human Microbiome. Nat. Med. 2018, 24, 392–400. [Google Scholar] [CrossRef] [PubMed]
- Young, V.B. The Role of the Microbiome in Human Health and Disease: An Introduction for Clinicians. BMJ 2017, 356, j831. [Google Scholar] [CrossRef] [PubMed]
- Dethlefsen, L.; McFall-Ngai, M.; Relman, D.A. An Ecological and Evolutionary Perspective on Human-Microbe Mutualism and Disease. Nature 2007, 449, 811–818. [Google Scholar] [CrossRef]
- Mondot, S.; de Wouters, T.; Doré, J.; Lepage, P. The Human Gut Microbiome and Its Dysfunctions. Dig. Dis. 2013, 31, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An Obesity-Associated Gut Microbiome with Increased Capacity for Energy Harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Bibbò, S.; Ianiro, G.; Giorgio, V.; Scaldaferri, F.; Masucci, L.; Gasbarrini, A.; Cammarota, G. The Role of Diet on Gut Microbiota Composition. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 4742–4749. [Google Scholar] [PubMed]
- Illiano, P.; Brambilla, R.; Parolini, C. The Mutual Interplay of Gut Microbiota, Diet and Human Disease. FEBS J. 2020, 287, 833–855. [Google Scholar] [CrossRef]
- Functional Gastrointestinal Disorders: Advances in Understanding and Management—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/33049221/ (accessed on 15 December 2021).
- Holtmann, G.; Shah, A.; Morrison, M. Pathophysiology of Functional Gastrointestinal Disorders: A Holistic Overview. Dig. Dis. 2017, 35 (Suppl. 1), 5–13. [Google Scholar] [CrossRef]
- Wan, M.L.Y.; Ling, K.H.; El-Nezami, H.; Wang, M.F. Influence of Functional Food Components on Gut Health. Crit. Rev. Food Sci. Nutr. 2019, 59, 1927–1936. [Google Scholar] [CrossRef]
- Hosseini, A.; Nikfar, S.; Abdollahi, M. Are Probiotics Effective in Management of Irritable Bowel Syndrome? Arch. Med. Sci. 2012, 8, 403–405. [Google Scholar] [CrossRef]
- Cremon, C.; Barbaro, M.R.; Ventura, M.; Barbara, G. Pre- and Probiotic Overview. Curr. Opin. Pharmacol. 2018, 43, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Behnsen, J.; Deriu, E.; Sassone-Corsi, M.; Raffatellu, M. Probiotics: Properties, Examples, and Specific Applications. Cold Spring Harb. Perspect. Med. 2013, 3, a010074. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mizock, B.A. Probiotics. Dis. Mon. 2015, 61, 259–290. [Google Scholar] [CrossRef] [PubMed]
- Islam, S.U. Clinical Uses of Probiotics. Medicine 2016, 95, e2658. [Google Scholar] [CrossRef]
- Wilkins, T.; Sequoia, J. Probiotics for Gastrointestinal Conditions: A Summary of the Evidence. Am. Fam. Phys. 2017, 96, 170–178. [Google Scholar]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.E.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (Poly)Phenolics in Human Health: Structures, Bioavailability, and Evidence of Protective Effects against Chronic Diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [Green Version]
- Craig, W.J. Phytochemicals: Guardians of Our Health. J. Am. Diet Assoc. 1997, 97, S199–S204. [Google Scholar] [CrossRef]
- Leitzmann, C. Characteristics and Health Benefits of Phytochemicals. Forsch. Komplementmed. 2016, 23, 69–74. [Google Scholar] [CrossRef]
- Dillard, C.J.; German, J.B. Phytochemicals: Nutraceuticals and Human Health. J. Sci. Food Agric. 2000, 80, 1744–1756. [Google Scholar] [CrossRef]
- Upadhyay, S.; Dixit, M. Role of Polyphenols and Other Phytochemicals on Molecular Signaling. Oxid. Med. Cell. Longev. 2015, 2015, 504253. [Google Scholar] [CrossRef]
- Probst, Y.; Guan, V.; Kent, K. Dietary Phytochemical Intake from Foods and Health Outcomes: A Systematic Review Protocol and Preliminary Scoping. BMJ Open 2017, 7, e013337. [Google Scholar] [CrossRef] [PubMed]
- Clifford, M.N. Diet-Derived Phenols in Plasma and Tissues and Their Implications for Health. Planta Med. 2004, 70, 1103–1114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tomás-Barberán, F.A.; Selma, M.V.; Espín, J.C. Interactions of Gut Microbiota with Dietary Polyphenols and Consequences to Human Health. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Lovell, R.M.; Ford, A.C. Global Prevalence of and Risk Factors for Irritable Bowel Syndrome: A Meta-Analysis. Clin. Gastroenterol. Hepatol. 2012, 10, 712–721.e4. [Google Scholar] [CrossRef]
- Laparra, J.M.; Sanz, Y. Interactions of Gut Microbiota with Functional Food Components and Nutraceuticals. Pharmacol. Res. 2010, 61, 219–225. [Google Scholar] [CrossRef] [Green Version]
- Saha, L. Irritable Bowel Syndrome: Pathogenesis, Diagnosis, Treatment, and Evidence-Based Medicine. World J. Gastroenterol. 2014, 20, 6759–6773. [Google Scholar] [CrossRef]
- Asha, M.Z.; Khalil, S.F.H. Efficacy and Safety of Probiotics, Prebiotics and Synbiotics in the Treatment of Irritable Bowel Syndrome: A Systematic Review and Meta-Analysis. Sultan Qaboos Univ. Med. J. 2020, 20, e13–e24. [Google Scholar] [CrossRef] [Green Version]
- Ford, A.C.; Talley, N.J.; Spiegel, B.M.R.; Foxx-Orenstein, A.E.; Schiller, L.; Quigley, E.M.M.; Moayyedi, P. Effect of Fibre, Antispasmodics, and Peppermint Oil in the Treatment of Irritable Bowel Syndrome: Systematic Review and Meta-Analysis. BMJ 2008, 337, a2313. [Google Scholar] [CrossRef] [Green Version]
- Ford, A.C.; Talley, N.J.; Schoenfeld, P.S.; Quigley, E.M.M.; Moayyedi, P. Efficacy of Antidepressants and Psychological Therapies in Irritable Bowel Syndrome: Systematic Review and Meta-Analysis. Gut 2009, 58, 367–378. [Google Scholar] [CrossRef]
- Ooi, S.L.; Correa, D.; Pak, S.C. Probiotics, Prebiotics, and Low FODMAP Diet for Irritable Bowel Syndrome—What Is the Current Evidence? Complement. Ther. Med. 2019, 43, 73–80. [Google Scholar] [CrossRef]
- Ford, A.C.; Quigley, E.M.M.; Lacy, B.E.; Lembo, A.J.; Saito, Y.A.; Schiller, L.R.; Soffer, E.E.; Spiegel, B.M.R.; Moayyedi, P. Efficacy of Prebiotics, Probiotics, and Synbiotics in Irritable Bowel Syndrome and Chronic Idiopathic Constipation: Systematic Review and Meta-Analysis. Am. J. Gastroenterol. 2014, 109, 1547–1561. [Google Scholar] [CrossRef] [PubMed]
- Ford, A.C.; Harris, L.A.; Lacy, B.E.; Quigley, E.M.M.; Moayyedi, P. Systematic Review with Meta-Analysis: The Efficacy of Prebiotics, Probiotics, Synbiotics and Antibiotics in Irritable Bowel Syndrome. Aliment. Pharmacol. Ther. 2018, 48, 1044–1060. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Didari, T.; Mozaffari, S.; Nikfar, S.; Abdollahi, M. Effectiveness of Probiotics in Irritable Bowel Syndrome: Updated Systematic Review with Meta-Analysis. World J. Gastroenterol. 2015, 21, 3072–3084. [Google Scholar] [CrossRef] [PubMed]
- Yuan, F.; Ni, H.; Asche, C.V.; Kim, M.; Walayat, S.; Ren, J. Efficacy of Bifidobacterium Infantis 35624 in Patients with Irritable Bowel Syndrome: A Meta-Analysis. Curr. Med. Res. Opin. 2017, 33, 1191–1197. [Google Scholar] [CrossRef] [PubMed]
- Dale, H.F.; Rasmussen, S.H.; Asiller, Ö.Ö.; Lied, G.A. Probiotics in Irritable Bowel Syndrome: An Up-to-Date Systematic Review. Nutrients 2019, 11, 2048. [Google Scholar] [CrossRef] [Green Version]
- Lewis, E.D.; Antony, J.M.; Crowley, D.C.; Piano, A.; Bhardwaj, R.; Tompkins, T.A.; Evans, M. Efficacy of Lactobacillus Paracasei HA-196 and Bifidobacterium Longum R0175 in Alleviating Symptoms of Irritable Bowel Syndrome (IBS): A Randomized, Placebo-Controlled Study. Nutrients 2020, 12, 1159. [Google Scholar] [CrossRef] [Green Version]
- Giang, J.; Lan, X.; Crichton, M.; Marx, W.; Marshall, S. Efficacy and Safety of Biophenol-Rich Nutraceuticals in Adults with Inflammatory Gastrointestinal Diseases or Irritable Bowel Syndrome: A Systematic Literature Review and Meta-Analysis. Nutr. Diet 2021, 1–18. [Google Scholar] [CrossRef]
- Khanna, R.; MacDonald, J.K.; Levesque, B.G. Peppermint Oil for the Treatment of Irritable Bowel Syndrome: A Systematic Review and Meta-Analysis. J. Clin. Gastroenterol. 2014, 48, 505–512. [Google Scholar] [CrossRef]
- Enck, P.; Azpiroz, F.; Boeckxstaens, G.; Elsenbruch, S.; Feinle-Bisset, C.; Holtmann, G.; Lackner, J.M.; Ronkainen, J.; Schemann, M.; Stengel, A.; et al. Functional Dyspepsia. Nat. Rev. Dis. Primers 2017, 3, 17081. [Google Scholar] [CrossRef]
- Ford, A.C.; Mahadeva, S.; Carbone, M.F.; Lacy, B.E.; Talley, N.J. Functional Dyspepsia. Lancet 2020, 396, 1689–1702. [Google Scholar] [CrossRef]
- Duboc, H.; Latrache, S.; Nebunu, N.; Coffin, B. The Role of Diet in Functional Dyspepsia Management. Front. Psychiatry 2020, 11, 23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pesce, M.; Cargiolli, M.; Cassarano, S.; Polese, B.; De Conno, B.; Aurino, L.; Mancino, N.; Sarnelli, G. Diet and Functional Dyspepsia: Clinical Correlates and Therapeutic Perspectives. World J. Gastroenterol. 2020, 26, 456–465. [Google Scholar] [CrossRef] [PubMed]
- Wauters, L.; Slaets, H.; De Paepe, K.; Ceulemans, M.; Wetzels, S.; Geboers, K.; Toth, J.; Thys, W.; Dybajlo, R.; Walgraeve, D.; et al. Efficacy and Safety of Spore-Forming Probiotics in the Treatment of Functional Dyspepsia: A Pilot Randomised, Double-Blind, Placebo-Controlled Trial. Lancet Gastroenterol. Hepatol. 2021, 6, 784–792. [Google Scholar] [CrossRef]
- Zhang, J.; Wu, H.M.; Wang, X.; Xie, J.; Li, X.; Ma, J.; Wang, F.; Tang, X. Efficacy of Prebiotics and Probiotics for Functional Dyspepsia: A Systematic Review and Meta-Analysis. Medicine 2020, 99, e19107. [Google Scholar] [CrossRef]
- Ianiro, G.; Pizzoferrato, M.; Franceschi, F.; Tarullo, A.; Luisi, T.; Gasbarrini, G. Effect of an Extra-Virgin Olive Oil Enriched with Probiotics or Antioxidants on Functional Dyspepsia: A Pilot Study. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 2085–2090. [Google Scholar]
- Kupcinskas, L.; Lafolie, P.; Lignell, A.; Kiudelis, G.; Jonaitis, L.; Adamonis, K.; Andersen, L.P.; Wadström, T. Efficacy of the Natural Antioxidant Astaxanthin in the Treatment of Functional Dyspepsia in Patients with or without Helicobacter Pylori Infection: A Prospective, Randomized, Double Blind, and Placebo-Controlled Study. Phytomedicine 2008, 15, 391–399. [Google Scholar] [CrossRef]
- Rao, S.S.C.; Rattanakovit, K.; Patcharatrakul, T. Diagnosis and Management of Chronic Constipation in Adults. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 295–305. [Google Scholar] [CrossRef]
- Scarpato, E.; Coppola, V.; Staiano, A. Probiotics and Applications to Constipation; Academic Press: Cambridge, MA, USA, 2019; pp. 193–196. ISBN 978-0-12-814468-8. [Google Scholar]
- Riezzo, G.; Orlando, A.; D’Attoma, B.; Guerra, V.; Valerio, F.; Lavermicocca, P.; De Candia, S.; Russo, F. Randomised Clinical Trial: Efficacy of Lactobacillus Paracasei-Enriched Artichokes in the Treatment of Patients with Functional Constipation--a Double-Blind, Controlled, Crossover Study. Aliment. Pharmacol. Ther. 2012, 35, 441–450. [Google Scholar] [CrossRef]
- Yoon, J.Y.; Cha, J.M.; Oh, J.K.; Tan, P.L.; Kim, S.H.; Kwak, M.S.; Jeon, J.W.; Shin, H.P. Probiotics Ameliorate Stool Consistency in Patients with Chronic Constipation: A Randomized, Double-Blind, Placebo-Controlled Study. Dig. Dis. Sci. 2018, 63, 2754–2764. [Google Scholar] [CrossRef]
- Martoni, C.J.; Evans, M.; Chow, C.-E.T.; Chan, L.S.; Leyer, G. Impact of a Probiotic Product on Bowel Habits and Microbial Profile in Participants with Functional Constipation: A Randomized Controlled Trial. J. Dig. Dis. 2019, 20, 435–446. [Google Scholar] [CrossRef]
- Dimidi, E.; Christodoulides, S.; Fragkos, K.C.; Scott, S.M.; Whelan, K. The Effect of Probiotics on Functional Constipation in Adults: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Am. J. Clin. Nutr. 2014, 100, 1075–1084. [Google Scholar] [CrossRef] [Green Version]
- Mazlyn, M.M.; Nagarajah, L.H.-L.; Fatimah, A.; Norimah, A.K.; Goh, K.-L. Effects of a Probiotic Fermented Milk on Functional Constipation: A Randomized, Double-Blind, Placebo-Controlled Study. J. Gastroenterol. Hepatol. 2013, 28, 1141–1147. [Google Scholar] [CrossRef]
- Ibarra, A.; Latreille-Barbier, M.; Donazzolo, Y.; Pelletier, X.; Ouwehand, A.C. Effects of 28-Day Bifidobacterium Animalis Subsp. Lactis HN019 Supplementation on Colonic Transit Time and Gastrointestinal Symptoms in Adults with Functional Constipation: A Double-Blind, Randomized, Placebo-Controlled, and Dose-Ranging Trial. Gut Microbes 2018, 9, 236–251. [Google Scholar] [CrossRef] [Green Version]
- Airaksinen, K.; Yeung, N.; Lyra, A.; Lahtinen, S.J.; Huttunen, T.; Shanahan, F.; Ouwehand, A.C. The Effect of a Probiotic Blend on Gastrointestinal Symptoms in Constipated Patients: A Double Blind, Randomised, Placebo Controlled 2-Week Trial. Benef. Microbes 2019, 10, 617–627. [Google Scholar] [CrossRef] [PubMed]
- Venancio, V.P.; Kim, H.; Sirven, M.A.; Tekwe, C.D.; Honvoh, G.; Talcott, S.T.; Mertens-Talcott, S.U. Polyphenol-Rich Mango (Mangifera indica L.) Ameliorate Functional Constipation Symptoms in Humans beyond Equivalent Amount of Fiber. Mol. Nutr. Food Res. 2018, 62, e1701034. [Google Scholar] [CrossRef] [PubMed]
- Relationship between the Gut Microbiome and Brain Function—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/29701810/ (accessed on 15 December 2021).
- De Palma, G.; Collins, S.M.; Bercik, P. The Microbiota-Gut-Brain Axis in Functional Gastrointestinal Disorders. Gut Microbes 2014, 5, 419–429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mukhtar, K.; Nawaz, H.; Abid, S. Functional Gastrointestinal Disorders and Gut-Brain Axis: What Does the Future Hold? World J. Gastroenterol. 2019, 25, 552–566. [Google Scholar] [CrossRef]
- Jeffery, I.B.; O’Toole, P.W.; Öhman, L.; Claesson, M.J.; Deane, J.; Quigley, E.M.M.; Simrén, M. An Irritable Bowel Syndrome Subtype Defined by Species-Specific Alterations in Faecal Microbiota. Gut 2012, 61, 997–1006. [Google Scholar] [CrossRef]
- Chassard, C.; Dapoigny, M.; Scott, K.P.; Crouzet, L.; Del’homme, C.; Marquet, P.; Martin, J.C.; Pickering, G.; Ardid, D.; Eschalier, A.; et al. Functional Dysbiosis within the Gut Microbiota of Patients with Constipated-Irritable Bowel Syndrome. Aliment. Pharmacol. Ther. 2012, 35, 828–838. [Google Scholar] [CrossRef]
- Shukla, R.; Ghoshal, U.; Dhole, T.N.; Ghoshal, U.C. Fecal Microbiota in Patients with Irritable Bowel Syndrome Compared with Healthy Controls Using Real-Time Polymerase Chain Reaction: An Evidence of Dysbiosis. Dig. Dis. Sci. 2015, 60, 2953–2962. [Google Scholar] [CrossRef]
- Durbán, A.; Abellán, J.J.; Jiménez-Hernández, N.; Artacho, A.; Garrigues, V.; Ortiz, V.; Ponce, J.; Latorre, A.; Moya, A. Instability of the Faecal Microbiota in Diarrhoea-Predominant Irritable Bowel Syndrome. FEMS Microbiol. Ecol. 2013, 86, 581–589. [Google Scholar] [CrossRef] [Green Version]
- Durbán, A.; Abellán, J.J.; Jiménez-Hernández, N.; Salgado, P.; Ponce, M.; Ponce, J.; Garrigues, V.; Latorre, A.; Moya, A. Structural Alterations of Faecal and Mucosa-Associated Bacterial Communities in Irritable Bowel Syndrome. Environ. Microbiol. Rep. 2012, 4, 242–247. [Google Scholar] [CrossRef]
- El-Salhy, M.; Hatlebakk, J.G.; Gilja, O.H.; Bråthen Kristoffersen, A.; Hausken, T. Efficacy of Faecal Microbiota Transplantation for Patients with Irritable Bowel Syndrome in a Randomised, Double-Blind, Placebo-Controlled Study. Gut 2020, 69, 859–867. [Google Scholar] [CrossRef] [Green Version]
- Harvie, R.M.; Chisholm, A.W.; Bisanz, J.E.; Burton, J.P.; Herbison, P.; Schultz, K.; Schultz, M. Long-Term Irritable Bowel Syndrome Symptom Control with Reintroduction of Selected FODMAPs. World J. Gastroenterol. 2017, 23, 4632–4643. [Google Scholar] [CrossRef]
- Physical Activity Improves Symptoms in Irritable Bowel Syndrome: A Randomized Controlled Trial—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/21206488/ (accessed on 15 December 2021).
- Natural Polyphenols for the Prevention of Irritable Bowel Syndrome: Molecular Mechanisms and Targets; a Comprehensive Review—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/31273572/ (accessed on 15 December 2021).
- Masuy, I.; Van Oudenhove, L.; Tack, J. Review Article: Treatment Options for Functional Dyspepsia. Aliment. Pharmacol. Ther. 2019, 49, 1134–1172. [Google Scholar] [CrossRef] [Green Version]
- Moayyedi, P. Helicobacter Pylori Eradication for Functional Dyspepsia: What Are We Treating?: Comment on “Helicobacter Pylori Eradication in Functional Dyspepsia”. Arch. Intern. Med. 2011, 171, 1936–1937. [Google Scholar] [CrossRef]
- Tomita, T.; Oshima, T.; Miwa, H. New Approaches to Diagnosis and Treatment of Functional Dyspepsia. Curr. Gastroenterol. Rep. 2018, 20, 55. [Google Scholar] [CrossRef]
- Zheng, H.; Xu, J.; Sun, X.; Zeng, F.; Li, Y.; Wu, X.; Li, J.; Zhao, L.; Chang, X.-R.; Liu, M.; et al. Electroacupuncture for Patients with Refractory Functional Dyspepsia: A Randomized Controlled Trial. Neurogastroenterol. Motil. 2018, 30, e13316. [Google Scholar] [CrossRef]
- Davis, B.R.; Sarosiek, I.; Bashashati, M.; Alvarado, B.; McCallum, R.W. The Long-Term Efficacy and Safety of Pyloroplasty Combined with Gastric Electrical Stimulation Therapy in Gastroparesis. J. Gastrointest. Surg. 2017, 21, 222–227. [Google Scholar] [CrossRef]
- Malik, Z.; Kataria, R.; Modayil, R.; Ehrlich, A.C.; Schey, R.; Parkman, H.P.; Stavropoulos, S.N. Gastric Per Oral Endoscopic Myotomy (G-POEM) for the Treatment of Refractory Gastroparesis: Early Experience. Dig. Dis. Sci. 2018, 63, 2405–2412. [Google Scholar] [CrossRef]
- Xue, H.B.; Fan, H.Z.; Meng, X.M.; Cristofaro, S.; Mekaroonkamol, P.; Dacha, S.; Li, L.Y.; Fu, X.L.; Zhan, S.H.; Cai, Q. Fluoroscopy-Guided Gastric Peroral Endoscopic Pyloromyotomy (G-POEM): A More Reliable and Efficient Method for Treatment of Refractory Gastroparesis. Surg. Endosc. 2017, 31, 4617–4624. [Google Scholar] [CrossRef] [PubMed]
- Mekaroonkamol, P.; Dacha, S.; Wang, L.; Li, X.; Jiang, Y.; Li, L.; Li, T.; Shahnavaz, N.; Sakaria, S.; LeVert, F.E.; et al. Gastric Peroral Endoscopic Pyloromyotomy Reduces Symptoms, Increases Quality of Life, and Reduces Health Care Use For Patients With Gastroparesis. Clin. Gastroenterol. Hepatol. 2019, 17, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Duncanson, K.R.; Talley, N.J.; Walker, M.M.; Burrows, T.L. Food and Functional Dyspepsia: A Systematic Review. J. Hum. Nutr. Diet 2018, 31, 390–407. [Google Scholar] [CrossRef] [PubMed]
- Feinle-Bisset, C.; Vozzo, R.; Horowitz, M.; Talley, N.J. Diet, Food Intake, and Disturbed Physiology in the Pathogenesis of Symptoms in Functional Dyspepsia. Am. J. Gastroenterol. 2004, 99, 170–181. [Google Scholar] [CrossRef] [PubMed]
- Tziatzios, G.; Gkolfakis, P.; Papanikolaou, I.S.; Mathur, R.; Pimentel, M.; Giamarellos-Bourboulis, E.J.; Triantafyllou, K. Gut Microbiota Dysbiosis in Functional Dyspepsia. Microorganisms 2020, 8, 691. [Google Scholar] [CrossRef]
- Shin, J.E.; Park, K.S.; Nam, K. Chronic Functional Constipation. Korean J. Gastroenterol. 2019, 73, 92–98. [Google Scholar] [CrossRef] [Green Version]
- Huang, L.; Zhu, Q.; Qu, X.; Qin, H. Microbial Treatment in Chronic Constipation. Sci. China Life Sci. 2018, 61, 744–752. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. PRISMA Group Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [Green Version]

| Study Type | Study Sample/Duration | Participants | Diagnosis of IBS | Protocol | Summary of Results | Study Reference |
|---|---|---|---|---|---|---|
| Systematic review and meta-analysis | 23 randomized controlled trials | Adults/both male and female | Clinician’s opinion, or meeting specific diagnostic criteria (Manning, Kruis score, or Rome I, II, or III), supplemented by negative investigations where trials deemed this necessary | Probiotics were administered in the form of a capsule or included in functional foods | Probiotics combination or single strains of Lactobacillus, Bifidobacterium, Escherichia, Saccharomyces, or Streptococcus | [32] |
| ↓ | ||||||
| Improvement in global symptoms, abdominal pain, bloating, and flatulence scores | ||||||
| Systematic review and meta-analysis | 37rRandomized controlled trials | Adults/both male and female | Clinician’s opinion, or meeting specific diagnostic criteria (Manning, Kruis score, Rome I, II, or III), supplemented by negative investigations where trials deemed this necessary | Probiotics were administered in the form of a capsule or included in functional foods | Probiotics combination or single strains of Lactobacillus, Bifidobacterium, Escherichia, Saccharomyces, or Streptococcus | [33] |
| ↓ | ||||||
| Improvement in global symptoms and abdominal pain | ||||||
| Systematic review and meta-analysis | 15 randomized controlled trials | Adults/both male and female | Rome II, Rome III, and International Classification of Health Problems in Primary Care and World Organization of Family Doctors criteria | Probiotics were administered in the form of a capsule | Probiotics combination or single strains of Lactobacillus plantarum, Escherichia coli | [34] |
| ↓ | ||||||
| Improvement in global symptoms and abdominal pain, bloating, flatulence scores and stability of intestinal microbiota | ||||||
| Systematic review and meta-analysis | 5 randomized controlled trials | Adults/both male and female | Rome I, II, or Rome III criteria | Probiotics were administered in the form of a capsule | Comparison of the efficacy of the single probiotic Bifidobacterium infantis 35624 to a probiotic blend | [35] |
| ↓ | ||||||
| Composite probiotics containing B. infantis had significantly reduced abdominal pain and bloating/distention in patients | ||||||
| Systematic review | 11 randomized controlled trials | Adults/both male and female | Rome III or Rome IV criteria | Probiotics were administered in the form of a capsule, tablet, liquid or power mixed water | Probiotics combination or single strains | [36] |
| ↓ | ||||||
| Improvement in global symptoms | ||||||
| Randomized placebo-controlled study | 251 adults/12 weeks | Adults/both male and female | Rome III | Probiotics were administered in the form of a capsule | Single strains of Lactobacillus paracasei HA-196 and Bifidobacterium longum R0175 examined separately | [37] |
| ↓ | ||||||
| L. paracasei and B. longum has a beneficial impact on GI symptom severity |
| Study Type | Study Sample/Duration | Participants | Diagnosis of FD | Protocol | Summary of Results | Study Reference |
|---|---|---|---|---|---|---|
| Randomized, double-blind, placebo-controlled trial | 68 patients/8 weeks | Adults/both male and female | Rome IV criteria | Probiotics were administered in the form of a capsule | Spore-forming probiotics Bacillus coagulans MY01 and Bacillus subtilis MY02 as treatment for two subtypes of functional dyspepsia | [44] |
| ↓ | ||||||
| Beneficial immune and microbial changes as potential treatment targets | ||||||
| Systematic review and meta-analysis | 5 Randomized Controlled Trials | Adults/both male and female | Physician’s opinion or symptom-based diagnostic criteria, with a negative upper GI endoscopy excluding an organic cause of dyspepsia | Probiotics were administered in the form of a capsule | Probiotics combination or single strains of Saccharomyces, Lactobacillus, Bifidobacterium, Escherichia coli | [45] |
| ↓ | ||||||
| Probiotics seemed to be effective treatments for FD | ||||||
| Pilot study | 8 patients/7 days | Adults/both male and female | Rome III criteria | Probiotics were administered via extra-virgin oil enriched with probiotics | Extra-virgin oil enriched with probiotics included to patients’ diet | [46] |
| ↓ | ||||||
| Significant improvement of dyspeptic symptoms was observed in subjects receiving |
| Study Type | Study Sample/Duration | Participants | Diagnosis of FC | Protocol | Summary of Results | Study Reference |
|---|---|---|---|---|---|---|
| Randomized controlled trial | 20 patients/15 days | Adults/both male and female | Rome criteria III for constipation and constipation scoring system (CSS) | 180 g probiotic-enriched artichokes | Efficacy of Lactobacillus paracasei-enriched artichokes in the treatment of patients with functional constipation | [50] |
| ↓ | ||||||
| Positive effect on symptoms in constipated patients after intake of probiotic-enriched artichokes | ||||||
| Randomized controlled trial | 180 patients/4 weeks | Adults/both male and female | Rome IV criteria | 26 gr chocolate enriched with probiotics | Chocolate enriched with probiotics Streptococcus thermophilus and Lactobacillus plantarum | [51] |
| ↓ | ||||||
| Probiotics significantly ameliorated stool consistency in patients with chronic constipation | ||||||
| Randomized controlled trial | 94 patients/4 weeks | Adults 18–65 y/both male and female | Rome III | Probiotics were administered in the form of a capsule | Clinical efficacy of a multi-strain probiotic product consisting of Lactobacillus acidophilus, Bifidobacterium animalis, Bifidobacterium longum and Bifidobacterium bifidum | [52] |
| ↓ | ||||||
| Normalization of stool frequency and consistency, with most participants achieving a normalized profile after 1 week | ||||||
| Systematic review and meta-analysis | 14 Randomized controlled trials/1182 patients | Adults/both male and female | Clinical symptoms, a physician’s opinion, or the Rome I, II, or III criteria | Probiotics were administered in tablet, powder, capsule, softgel, or fortified food forms | Probiotics combination or single strains | [53] |
| ↓ | ||||||
| Probiotics improved whole gut transit time, stool frequency, and stool consistency, with subgroup analysis indicating beneficial effects of B. lactis in particular |
| Study Type | Study Sample/Duration | Participants | FGID | Protocol | Summary of Results | Study Reference |
|---|---|---|---|---|---|---|
| Systematic review and meta-analysis | 23 RCTs/ 2–5 weeks | Adults/ both male and female | IBS | Phytochemicals were administered in the form of a capsule, tablet or powder | Biophenol-rich nutraceuticals may be an effective and safe adjuvant treatment for the management of IBS; with higher certainty of evidence for peppermint oil for IBS. | [38] |
| Systematic review and meta-analysis | 9 RCTs/ 726 patients | Adults/ both male and female | IBS/ Rome I or Rome II criteria or clinical symptoms with the exclusion of organic disease | Peppermint oil was administered in the form of a capsule or tablet | Improvement in global symptoms and abdominal pain of patients with IBS. | [39] |
| Pilot study | 8 patients/ 7 days | Adults/ both male and female | FD/ Rome III criteria | Phytochemicals were administered via extra-virgin oil enriched with antioxidants | A significant improvement of dyspeptic symptoms was observed in subjects receiving the antioxidant enriched oil diet. | [46] |
| ↓ | ||||||
| Normalization of stool frequency and consistency, with most participants achieving a normalized profile after 1 week | ||||||
| Randomized, double-blind, and placebo-controlled study | 132 patients/ 12 weeks | Males and females (non-lactating and non-pregnant) aged 18–70 years | FD/ Rome II criteria | 16 mg, or 40 mg astaxanthin in the form of a capsule | Reduction of reflux symptoms was detected in patients treated with the highest dose of astaxanthin while the response was more pronounced in H. pylori-infected patients. | [47] |
| Randomized control trial | 48 participants/ 4 weeks | Adults 18–65 yo/ both male and female | Self-reported | 300 gr of mango fruit daily | Mango consumption improved constipation status (stool frequency, consistency, and shape) and increased gastrin levels and fecal concentrations of short chain fatty acid (valeric acid), while lowering endotoxin and interleukin concentrations in plasma | [57] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bousdouni, P.; Kandyliari, A.; Koutelidakis, A.E. Probiotics and Phytochemicals: Role on Gut Microbiota and Efficacy on Irritable Bowel Syndrome, Functional Dyspepsia, and Functional Constipation. Gastrointest. Disord. 2022, 4, 30-48. https://doi.org/10.3390/gidisord4010005
Bousdouni P, Kandyliari A, Koutelidakis AE. Probiotics and Phytochemicals: Role on Gut Microbiota and Efficacy on Irritable Bowel Syndrome, Functional Dyspepsia, and Functional Constipation. Gastrointestinal Disorders. 2022; 4(1):30-48. https://doi.org/10.3390/gidisord4010005
Chicago/Turabian StyleBousdouni, Panoraia, Aikaterini Kandyliari, and Antonios E. Koutelidakis. 2022. "Probiotics and Phytochemicals: Role on Gut Microbiota and Efficacy on Irritable Bowel Syndrome, Functional Dyspepsia, and Functional Constipation" Gastrointestinal Disorders 4, no. 1: 30-48. https://doi.org/10.3390/gidisord4010005
APA StyleBousdouni, P., Kandyliari, A., & Koutelidakis, A. E. (2022). Probiotics and Phytochemicals: Role on Gut Microbiota and Efficacy on Irritable Bowel Syndrome, Functional Dyspepsia, and Functional Constipation. Gastrointestinal Disorders, 4(1), 30-48. https://doi.org/10.3390/gidisord4010005







