Median Arcuate Ligament Syndrome Clinical Presentation, Pathophysiology, and Management: Description of Four Cases
Abstract
1. Introduction
2. Results
3. Discussion
3.1. Clinical Manifestation
3.2. Delayed Gastric Emptying
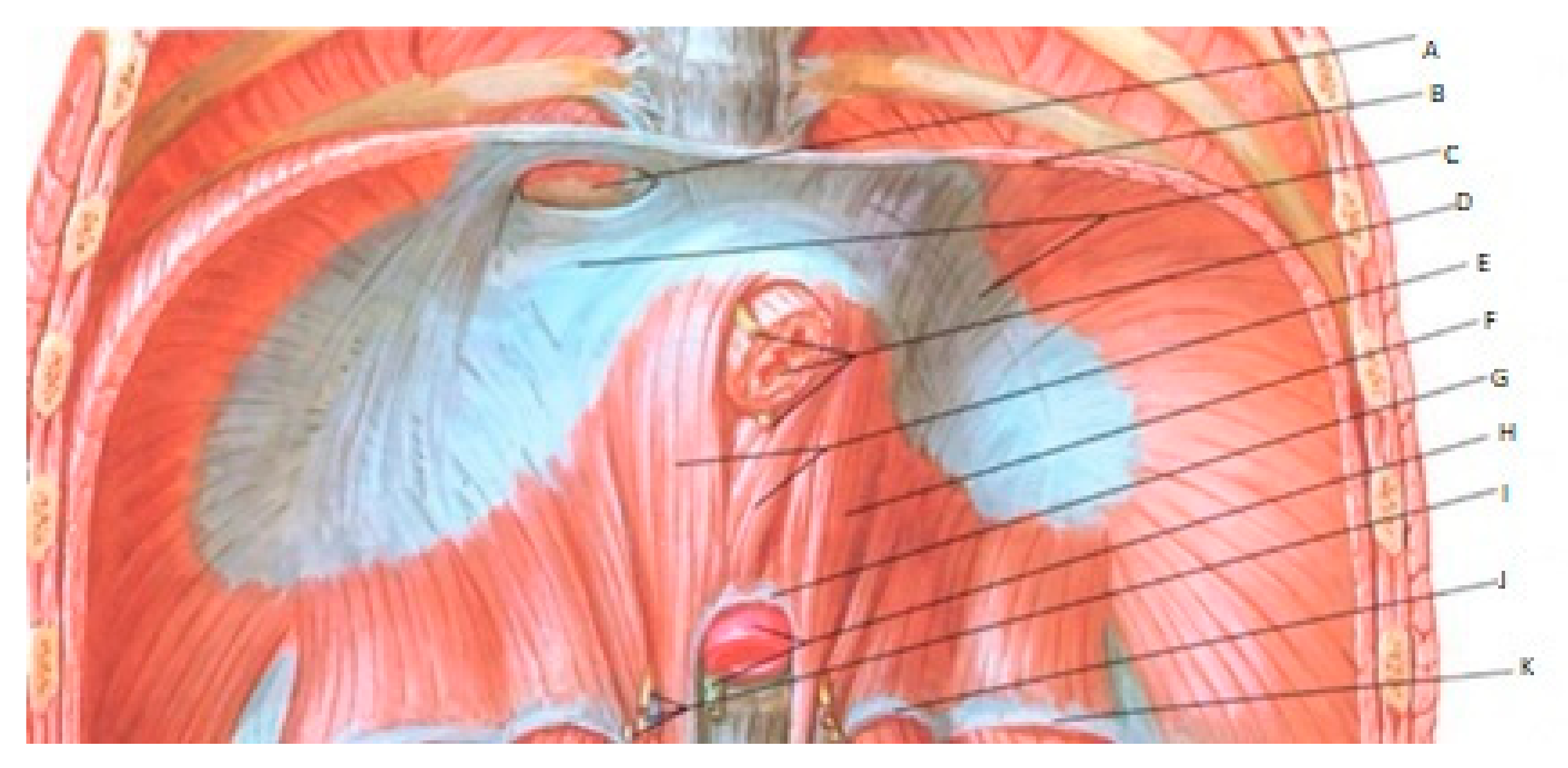
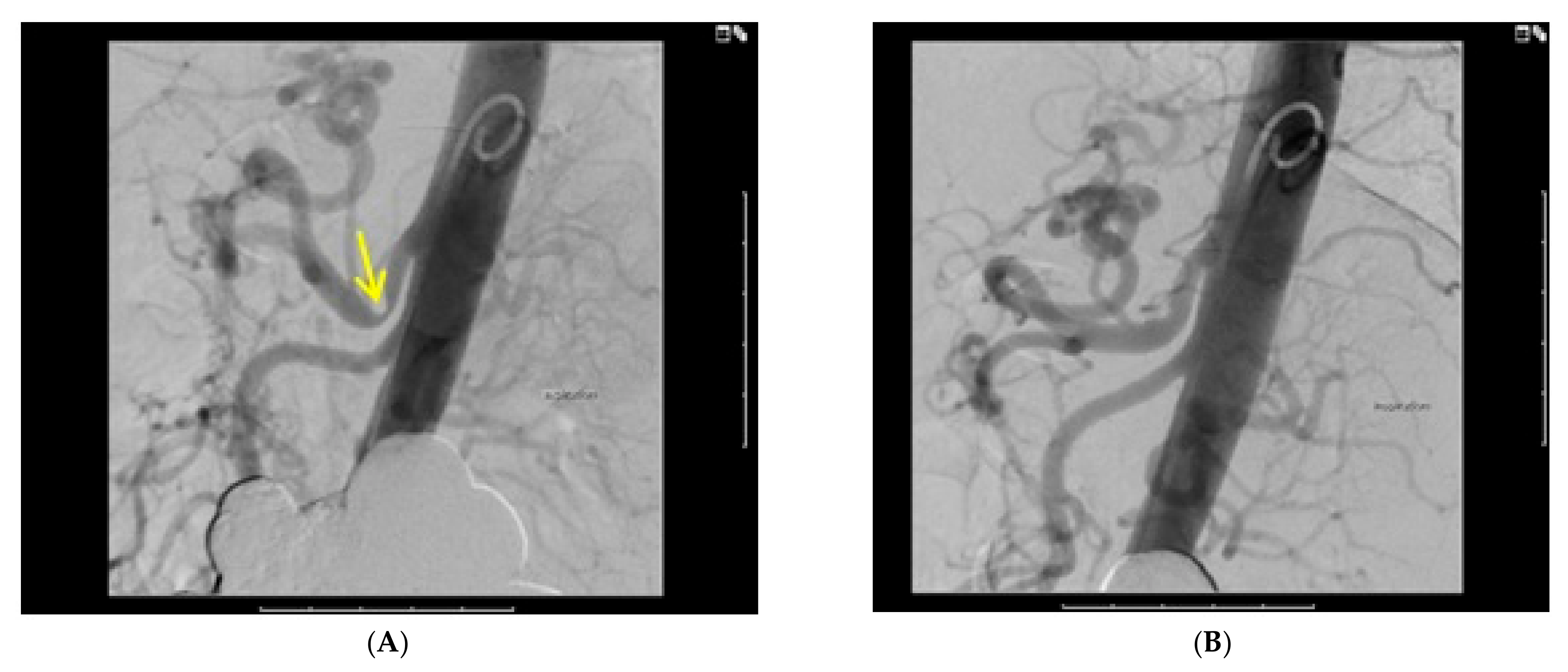
3.3. Diagnostic Studies
3.4. Management
4. Conclusions
5. Materials and Methods
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dunbar, J.D.; Molnar, W.; Beman, F.F.; Marable, S.A. Compression of the celiac trunk and abdominal angina: Preliminary report of 15 cases. Am. J. Roentgenol. 1965, 95, 731–744. [Google Scholar] [CrossRef] [PubMed]
- Rathmell, J.P.; Gallant, J.M.; Brown, D.L. Computed tomography and the anatomy of celiac plexus block. Reg. Anesth. Pain Med. 2000, 25, 411–416. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, J.C.; Harlander-Locke, M.; Dutson, E.P. Open and laparoscopic treatment of median arcuate ligament syndrome. J. Vasc. Surg. 2012, 56, 869–873. [Google Scholar] [CrossRef] [PubMed]
- McCallum, R.W.; Al-Bayati, I. Median Arcuate Ligament Syndrome. Pract. Gastroenterol. 2015, 21. [Google Scholar] [CrossRef]
- Bech, F.; Loesberg, A.; Rosenblum, J.; Glagov, S.; Gewertz, B.L. Median arcuate ligament compression syndrome in monozygotic twins. J. Vasc. Surg. 1994, 19, 934–938. [Google Scholar] [CrossRef]
- Okten, R.S.; Kucukay, F.; Tola, M.; Bostanci, B.; Cumhur, T. Is celiac artery compression syndrome genetically inherited?: A case series from a family and review of the literature. Eur. J. Radiol. 2012, 81, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, J.P.; Angle, J.F.; Matsumoto, A.H.; Young, J.S.; Hagspiel, K.D.; Spinosa, D.J. Acute visceral ischemia occurring subsequent to blunt abdominal trauma: Potential culpability of median arcuate ligament compression. J. Trauma Acute Care Surg. 1998, 45, 404–406. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.N.; Lamb, K.; Relles, D.; Moudgill, N.; DiMuzio, P.J.; Eisenberg, J.A. Median Arcuate Ligament Syndrome—Review of This Rare Disease. JAMA Surg. 2016, 151, 471–477. [Google Scholar] [CrossRef] [PubMed]
- Balaban, D.H.; Chen, J.; Lin, Z.; Tribble, C.G.; McCallum, R.W. Median arcuate ligament syndrome: A possible cause of idiopathic gastroparesis. Am. J. Gastroenterol. 1997, 92, 519–523. [Google Scholar] [PubMed]
- Tack, J.; Talley, N.J.; Camilleri, M.; Holtmann, G.; Hu, P.; Malagelada, J.-R.; Stanghellini, V. Functional Gastroduodenal Disorders. Gastroenterology 2006, 130, 1466–1479. [Google Scholar] [CrossRef] [PubMed]
- Watson, W.; Sadikali, F. Celiac axis compression: Experience with 20 patients and a critical appraisal of the syndrome. Ann. Intern. Med. 1977, 86, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Aschenbach, R.; Basche, S.; Vogl, T.J. Compression of the celiac trunk caused by median arcuate ligament in children and adolescent subjects: Evaluation with contrast-enhanced MR angiography and comparison with Doppler US evaluation. J. Vasc. Interv. Radiol. 2011, 22, 556–561. [Google Scholar] [CrossRef] [PubMed]
- Delis, K.T.; Gloviczki, P.; Altuwaijri, M.; McKusick, M.A. Median arcuate ligament syndrome: Open celiac artery reconstruction and ligament division after endovascular failure. J. Vasc. Surg. 2007, 46, 799–802. [Google Scholar] [CrossRef] [PubMed]
- Carbonell, A.; Kercher, K.; Heniford, B.; Matthews, B. Laparoscopic management of median arcuate ligament syndrome. Surg. Endosc. 2005, 19, 729. [Google Scholar] [CrossRef] [PubMed]
- Jaik, N.P.; Stawicki, S.P.; Weger, N.S.; Lukaszczyk, J.J. Celiac artery compression syndrome: Successful utilization of robotic-assisted laparoscopic approach. J. Gastrointest. Liver Dis. 2007, 16, 93. [Google Scholar]
- Roddy, S.P. Laparoscopic versus open celiac ganglionectomy in patients with median arcuate ligament syndrome. J. Vasc. Surg. 2010, 52, 1419. [Google Scholar] [CrossRef]
- Kohn, G.P.; Bitar, R.S.; Farber, M.A.; Marston, W.A.; Overby, D.W.; Farrell, T.M. Treatment options and outcomes for celiac artery compression syndrome. Surg. Innov. 2011, 18, 338–343. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Patients N = 4 (%) |
|---|---|
| Median Age (Interquartile Range [IQR]) in years | 34.5 (28.5, 42.25) |
| Female Gender | 4 (100) |
| Symptoms | |
| ● Abdominal Pain | 4 (100) |
| ● Nausea or Vomiting | 3 (75) |
| ● Weight Loss | 2 (50) |
| Duration of symptoms in years | 3 (2, 4.75) |
| Delayed Gastric Emptying Study | 4 (100) |
| Increased Doppler Ultrasound Peak Systolic Velocity | 4 (100) |
| Abdominal aortogram during expiration | 4 (100) |
| Celiac Block | 3 (75) |
| Celiac Artery Stent | 1 (25) |
| Surgical MAL release | 4 (100) |
| Resolution of Abd Pain Post-op | 4 (100) |
| Patients | Patient 1 | Patient 2 | Patient 3 | Patient 4 |
|---|---|---|---|---|
| Age in Years | 55 | 38 | 21 | 31 |
| Gender | Female | Female | Female | Female |
| History of previous abdominal surgery | Cholecystectomy | None | None | None |
| Presenting Symptoms | AP, N, V | AP, N, V, Wt Loss (5% over 2 years ) | AP | AP, N, V, Wt loss (8% over 2 years) |
| Duration of Symptoms (years) | 7 | 2 | 4 | 2 |
| GES at 4 hours | 19% | 15% | 31% | 10% |
| DU PSV of Celiac Artery, cm/sec | 362 | 323 | 339 | 440 |
| Narrowing of Celiac Artery on Abdominal Aortogram during expiration | 54% | 66% | 58% | 60% |
| Underwent Celiac Block | Yes | Yes | No | Yes |
| Underwent Celiac artery Stent | No | Yes | No | No |
| Surgical Treatment | Yes | Yes | Yes | Yes |
| Pyloroplasty and GES | No | No | Yes | No |
| Post-op PSV of Celiac Artery, cm/sec | 130 | 178 | 207 | 151 |
| Post-op Aortogram | normal | normal | normal | Not available |
| Resolution of Abd Pain Post-op | Yes | Yes | Yes | Yes |
| Time of abdominal pain relief (based on follow up time) | 6 years | 4 years | 1 year | 3 months |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bayati, I.A.; Gajendran, M.; Davis, B.R.; Diaz, J.R.; McCallum, R.W. Median Arcuate Ligament Syndrome Clinical Presentation, Pathophysiology, and Management: Description of Four Cases. Gastrointest. Disord. 2021, 3, 44-50. https://doi.org/10.3390/gidisord3010005
Bayati IA, Gajendran M, Davis BR, Diaz JR, McCallum RW. Median Arcuate Ligament Syndrome Clinical Presentation, Pathophysiology, and Management: Description of Four Cases. Gastrointestinal Disorders. 2021; 3(1):44-50. https://doi.org/10.3390/gidisord3010005
Chicago/Turabian StyleBayati, Ihsan Al, Mahesh Gajendran, Brian R. Davis, Jesus R. Diaz, and Richard W. McCallum. 2021. "Median Arcuate Ligament Syndrome Clinical Presentation, Pathophysiology, and Management: Description of Four Cases" Gastrointestinal Disorders 3, no. 1: 44-50. https://doi.org/10.3390/gidisord3010005
APA StyleBayati, I. A., Gajendran, M., Davis, B. R., Diaz, J. R., & McCallum, R. W. (2021). Median Arcuate Ligament Syndrome Clinical Presentation, Pathophysiology, and Management: Description of Four Cases. Gastrointestinal Disorders, 3(1), 44-50. https://doi.org/10.3390/gidisord3010005






