Sarcopenia as a Risk Factor of Morbimortality in Colorectal Cancer Surgery
Abstract
1. Introduction
2. Results
2.1. Relationship between Sarcopenia and the Clinical Pathological Data
2.2. Relation between Sarcopenia and Surgical Outcomes
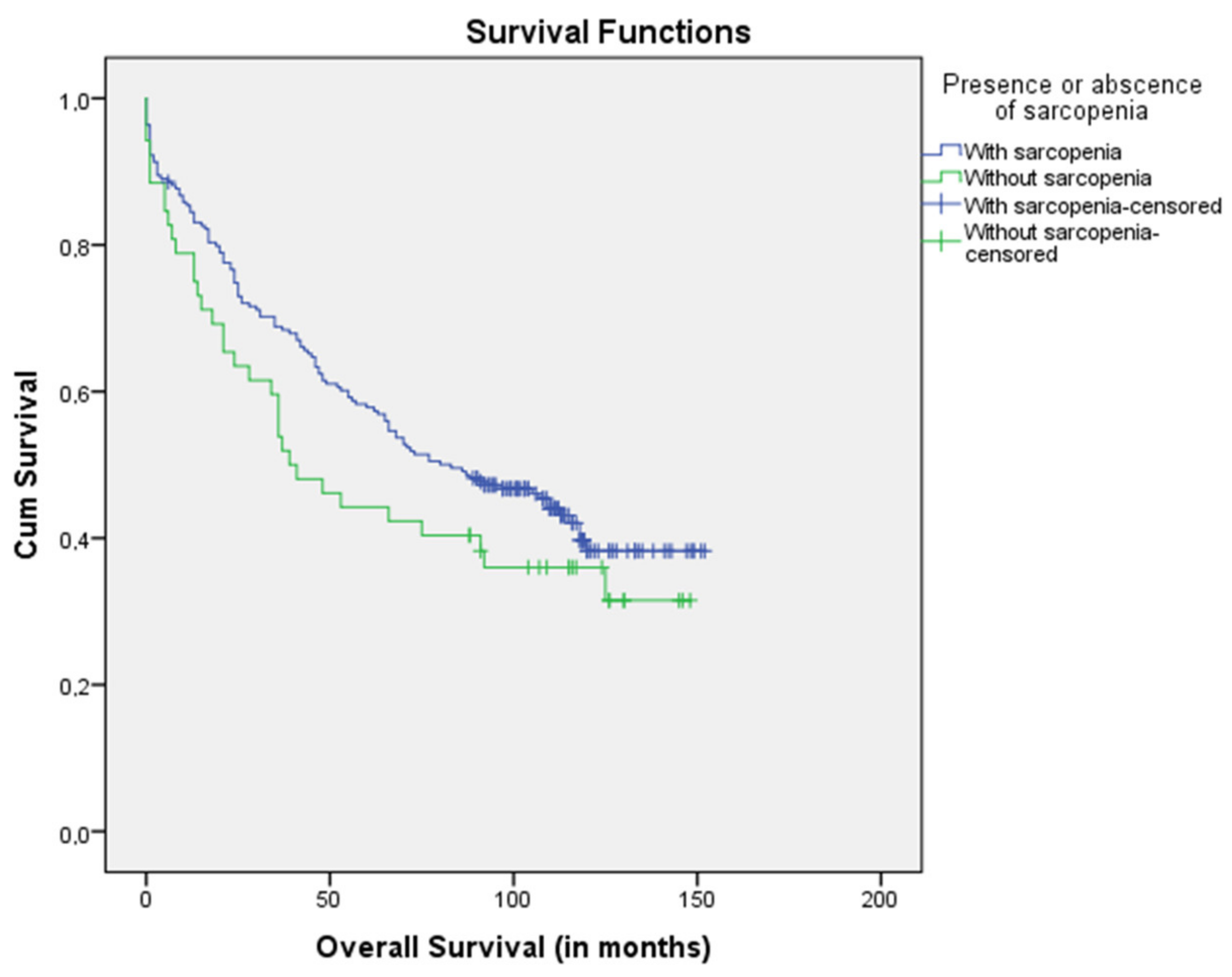
2.3. Relation between Sarcopenia and Overall Survival
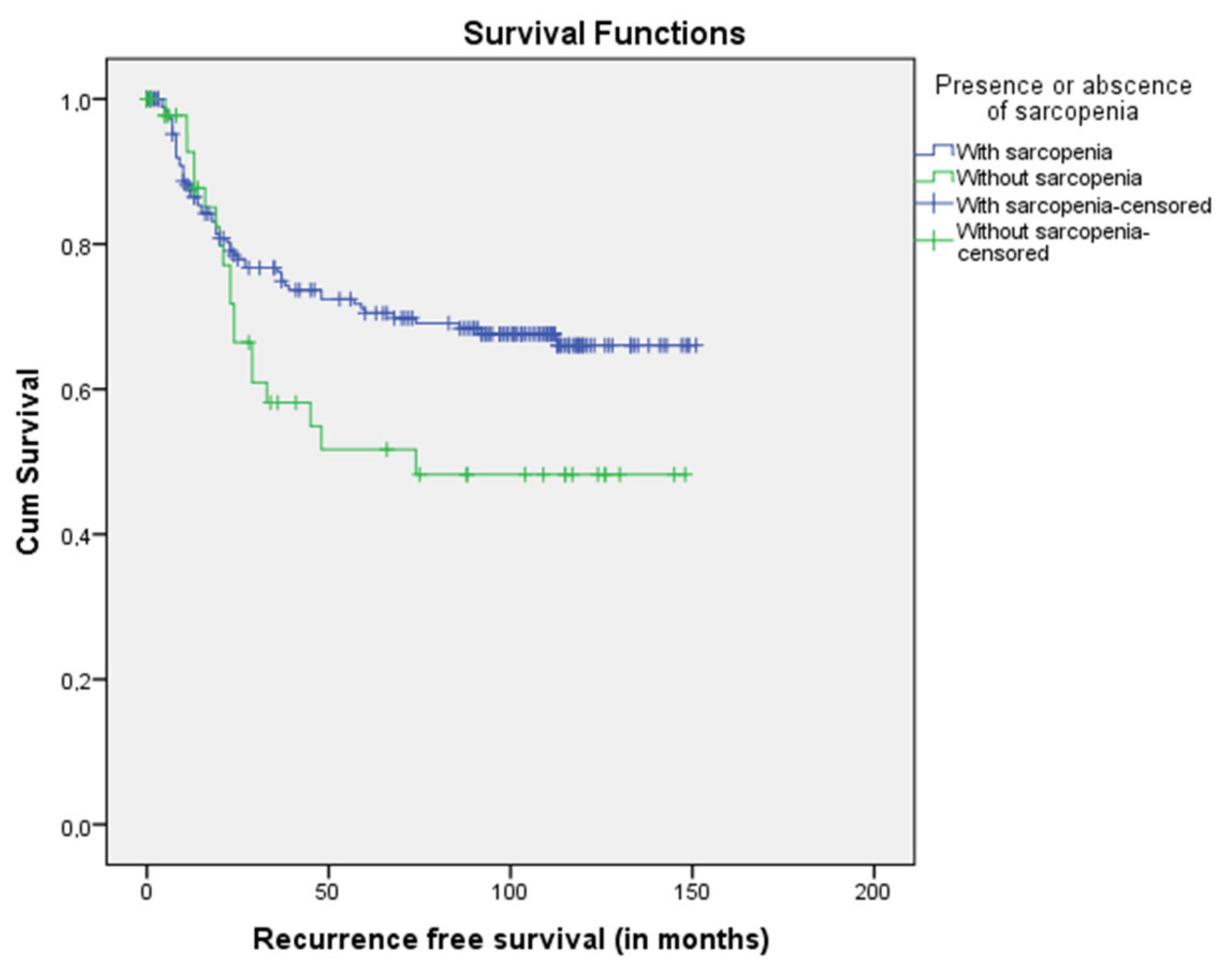
2.4. Relationship between Sarcopenia and Disease-Free Survival
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Data Collection
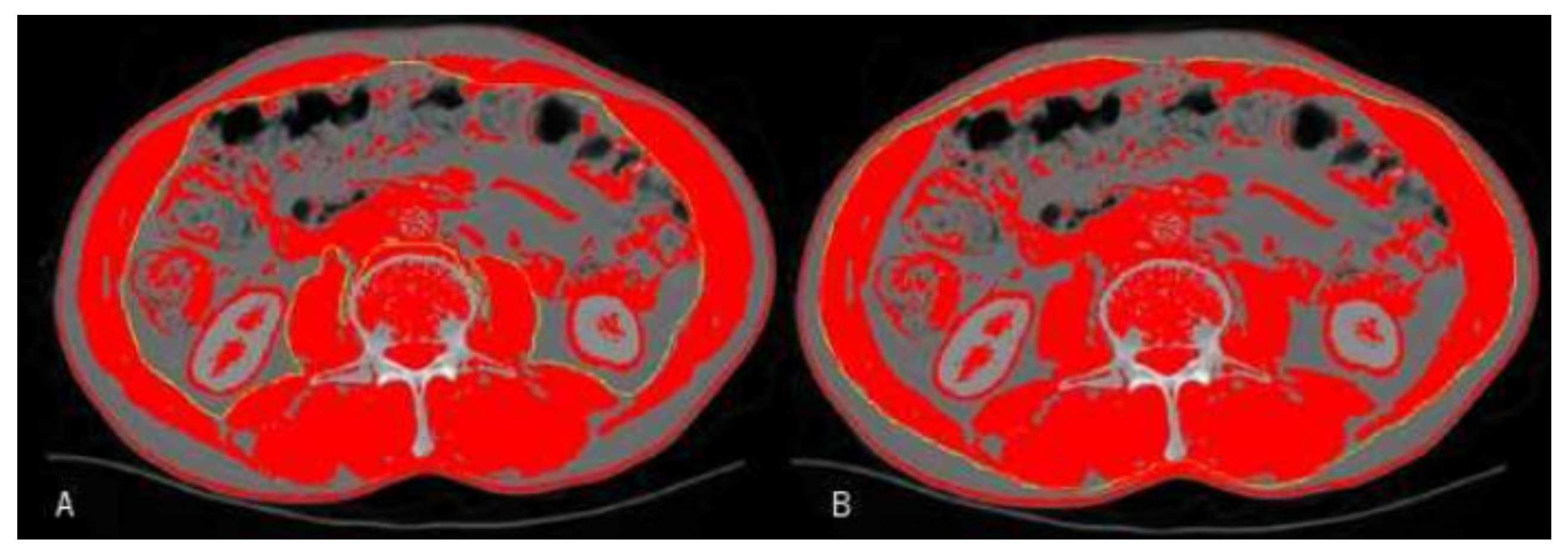
4.3. Image Analysis
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Torre, L.A.; Bray, F.; Siegel, R.L.; Ferlay, J.; Lortet-tieulent, J.; Jemal, A. Global cancer statistics, 2012. CA Cancer J. Clin. 2015, 65, 87–108. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics. CA Cancer J. Clin. 2016, 66, 7–30. [Google Scholar] [CrossRef]
- Granados-Romero, J.J.; Valderrama-Treviño, A.I.; Contreras-Flores, E.H.; Barrera-Mera, B.; Herrera Enríquez, M.; Uriarte-Ruíz, K.; Ceballos-Villalva, J.C.; Estrada-Mata, A.G.; Rodríguez, C.A.; Arauz-Peña, G. Colorectal cancer: A review. Int. J. Res. Med. Sci. 2017, 5, 4667–4676. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.e.; Rolland, Y.; Topinková, E.; et al. Sarcopenia: European consensus on definition and diagnosis. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef] [PubMed]
- Dodds, R.M.; Roberts, H.C.; Cooper, P.C.; Avan, P.; Sayer, A.; Europe PMC Funders Group. The epidemiology of sarcopenia. J. Clin. Densitom. 2016, 18, 461–466. [Google Scholar]
- Schmoll, H.J.; Van Cutsem, E.; Stein, A.; Valentini, V.; Glimelius, B.; Haustermans, K.; Nordlinger, B.; Van de Velde, C.J.; Balmana, J.; Regula, J.; et al. Esmo consensus guidelines for management of patients with colon and rectal cancer. A personalized approach to clinical decision making. Ann. Oncol. 2012, 23, 2479–2516. [Google Scholar] [CrossRef] [PubMed]
- Compton, C.; Fenoglio-Preiser, C.M.; Pettigrew, N.; Fielding, L.P. American Joint Committee on Cancer prognostic factors consensus conference. Cancer 2000, 88, 1739–1757. [Google Scholar] [CrossRef]
- George, F.H.M. Diagnóstico, estadiamento e tratamento do adenocarcinoma do cólon e do reto. Norma Direção Geral Saúde 2013, 1, 9. [Google Scholar]
- Broughman, J.R.; Williams, G.R.; Deal, A.M.; Yu, H.; Nyrop, K.A.; Alston, S.M.; Gordon, B.-B.; Sanoff, H.K.; Muss, H.B. Prevalence of sarcopenia in older patients with colorectal cancer. J. Geriatr. Oncol. 2015, 6, 442–445. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Punyanitya, M.; Wang, Z.; Gallagher, D.; St-Onge, M.P.; Albu, J.; Heymsfield, S.B.; Heshka, S. Total body skeletal muscle and adipose tissue volumes: Estimation from a single abdominal cross-sectional image. J. Appl. Physiol. 2004, 97, 2333–2338. [Google Scholar] [CrossRef]
- Mourtzakis, M.; Prado, C.M.M.; Lieffers, J.R.; Reiman, T.; McCargar, L.J.; Baracos, V.E. A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care. Appl. Physiol. Nutr. Metab. 2008, 33, 997–1006. [Google Scholar] [CrossRef]
- Malietzis, G.; Aziz, O.; Bagnall, N.M.; Johns, N.; Fearon, K.C.; Jenkins, J.T. The role of body composition evaluation by computerized tomography in determining colorectal cancer treatment outcomes: A systematic review. Eur. J. Surg. Oncol. 2015, 41, 186–196. [Google Scholar] [CrossRef] [PubMed]
- Lieffers, J.R.; Bathe, O.F.; Fassbender, K.; Winget, M.; Baracos, V.E. Sarcopenia is associated with postoperative infection and delayed recovery from colorectal cancer resection surgery. Br. J. Cancer 2012, 107, 931–936. [Google Scholar] [CrossRef]
- Jones, K.I.; Doleman, B.; Scott, S.; Lund, J.N.; Williams, J.P. Simple psoas cross-sectional area measurement is a quick and easy method to assess sarcopenia and predicts major surgical complications. Colorectal Dis. 2015, 17, O20–O26. [Google Scholar] [CrossRef] [PubMed]
- Boer, B.C.; De Graaff, F.; Brusse-Keizer, M.; Bouman, D.E.; Slump, C.H.; Slee-Valentijn, M.; Klaase, J.M. Skeletal muscle mass and quality as risk factors for postoperative outcome after open colon resection for cancer. Int. J. Colorectal Dis. 2016, 31, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Miyamoto, Y.; Baba, Y.; Sakamoto, Y.; Ohuchi, M.; Tokunaga, R.; Kurashige, J.; Hiyoshi, Y.; Iwagami, S.; Yoshida, N.; Watanabe, M.; et al. Sarcopenia is a negative prognostic factor after curative resection of colorectal cancer. Ann. Surg. Oncol. 2015, 22, 2663–2668. [Google Scholar] [CrossRef]
- Kirchhoff, P.; Clavien, P.-A.; Hahnloser, D. Complications in colorectal surgery: Risk factors and preventive strategies. Patient Saf. Surg. 2010, 4, 5. [Google Scholar] [CrossRef]
- Khuri, S.F.; Henderson, W.G.; DePalma, R.G.; Mosca, C.; Healey, N.A.; Kumbhani, D.J. Determinants of long-term survival after major surgery and the adverse effect of postoperative complications. Trans. Meet Am. Surg Assoc. 2005, 123, 32–48. [Google Scholar] [CrossRef]
- Nakanishi, R.; Oki, E.; Sasaki, S.; Hirose, K.; Jogo, T.; Edahiro, K.; Korehisa, S.; Taniguchi, D.; Kudo, K.; Sugiyama, M.; et al. Sarcopenia is an independent predictor of complications after colorectal cancer surgery. Surg. Today 2017, 48, 151–157. [Google Scholar] [CrossRef]
- Black, D.; Mackay, C.; Ramsay, G.; Hamoodi, Z.; Nanthakumaran, S.; Park, K.G.; Loudon, M.A.; Richards, C.H. Prognostic value of computed tomography: Measured parameters of body composition in primary operable gastrointestinal cancers. Ann. Surg. Oncol. 2017, 24, 2241–2251. [Google Scholar] [CrossRef]
- Reisinger, K.W.; Van Vugt, J.L.; Tegels, J.J.; Snijders, C.; Hulsewé, K.W.; Hoofwijk, A.G.; Stoot, J.H.; Von Meyenfeldt, M.F.; Beets, G.L.; Poeze, M.; et al. Functional compromise reflected by sarcopenia, frailty, and nutritional depletion predicts adverse postoperative outcome after colorectal cancer surgery. Ann. Surg. 2015, 261, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Perez, S.L.; Haus, J.M.; Sheean, P.; Patel, B.; Mar, W.; Chaudhry, V.; McKeever, L.; Braunschweig, C. Measuring Abdominal Circumference and Skeletal Muscle From a Single Cross-Sectional Computed Tomography Image: A Step-by-Step Guide for Clinicians Using National Institutes of Health ImageJ. J. Parenter. Enteral Nutr. 2016, 40, 308–318. [Google Scholar]
- Prado, C.M.M.; Heymsfield, S.B. Lean tissue imaging. J. Parenter Enter. Nutr. 2014, 38, 940–953. [Google Scholar] [CrossRef] [PubMed]
- Prado, C.M.; Lieffers, J.R.; McCargar, L.J.; Reiman, T.; Sawyer, M.B.; Martin, L.; Baracos, V.E. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: A population-based study. Lancet Oncol. 2008, 9, 629–635. [Google Scholar] [CrossRef]
| All Patients (n = 272) | Sarcopenic (n = 52) | Non-Sarcopenic (n = 220) | Statistics Test | |
|---|---|---|---|---|
| Age, Mdn (IQR) | 72.0 (17.0) | 71.0 (18.0) | 72.0 (16.0) | U = 5460, p = 0.609, r = −0.031 |
| Gender, n (%) | ||||
| Male | 167 (61.4) | 46 (88.5) | 121 (55.0) | χ2 (1) = 19.9, p < 0.001 *, Φ = −0.27 |
| Female | 105 (38.6) | 6 (11.5) | 99 (45.0) | |
| CRC family historya, n (%) | ||||
| Absent | 237 (92.6) | 48 (100.0) | 189 (90.9) | Fisher’s test, p = 0.029 *, Φ = −0.14 |
| Present | 19 (7.40) | 0 (0.00) | 19 (9.10) | |
| Clinical presentation, n (%) | ||||
| Asymptomatic | 48 (17.6) | 6 (11.5) | 42 (19.1) | χ2 (1) = 1.65, p = 0.230, Φ = 0.078 |
| Symptomatic | 224 (82.4) | 46 (88.5) | 178 (80.9) | |
| Tumour site, n (%) | ||||
| Colon | 193 (71.9) | 44 (84.6) | 149 (67.7) | χ2 (1) = 5.82, p = 0.017, Φ = −0.15 |
| Rectum | 79 (29.0) | 8 (15.4) | 71 (32.3) | |
| Macroscopic aspecta, n (%) | ||||
| Polypoid | 135 (53.6) | 24 (52.2) | 111 (53.9) | Fisher’s test, p = 0.085, Φc = 0.19 |
| Ulcerative | 66 (26.2) | 9 (19.6) | 57 (27.7) | |
| Infiltrative | 24 (9.50) | 3 (6.50) | 21 (10.2) | |
| Exophytic | 26 (10.30) | 10 (21.7) | 16 (7.80) | |
| Villous | 1 (0.40) | 0 (0.00) | 1 (0.50) | |
| Measurementa, n (%) | ||||
| ≤45 mm | 167 (64.5) | 27 (56.3) | 140 (66.4) | χ2 (1) = 1.74, p = 0.242, Φ = 0.082 |
| >45 mm | 92 (35.5) | 21 (43.8) | 71 (33.6) | |
| Histological type, n (%) | ||||
| Adenocarcinoma | 245 (90.1) | 48 (92.3) | 197 (89.5) | Fisher’s test, p = 0.834, Φc = 0.043 |
| Mucinous | 26 (9.60) | 4 (7.70) | 22 (10.0) | |
| Signet ring cells | 1 (0.40) | 0 (0.00) | 1 (0.50) | |
| Differentiationa, n (%) | ||||
| Well differentiated | 125 (47.9) | 22 (45.8) | 103 (48.4) | Fisher’s test, p = 0.959, Φc = 0.038 |
| Moderately differentiated | 102 (39.1) | 20 (41.7) | 82 (38.5) | |
| Poorly differentiated | 33 (12.6) | 6 (12.5) | 27 (12.7) | |
| Undifferentiated | 1 (0.40) | 0 (0.00) | 1 (0.50) | |
| Venous invasiona, n (%) | ||||
| Absent | 146 (57.5) | 28 (59.6) | 118 (57.0) | χ2 (1) = 0.009, p = 0.870, Φ = -0.020 |
| Present | 108 (42.5) | 19 (40.4) | 89 (43.0) | |
| Lymphatic invasiona, n (%) | ||||
| Absent | 103 (41.0) | 19 (40.4) | 84 (41.2) | χ2 (1) = 0.01, p = 1.000, Φ = 0.006 |
| Present | 148 (59.0) | 28 (59.6) | 120 (58.8) | |
| Stagea, n (%) | ||||
| I | 43 (16.2) | 7 (14.0) | 36 (16.7) | χ2 (3) = 0.93, p = 0.817, Φc = 0.059 |
| II | 87 (32.7) | 17 (34.0) | 70 (32.4) | |
| III | 93 (35.0) | 16 (32.0) | 77 (35.6) | |
| IV | 43 (16.2) | 10 (20.0) | 33 (15.3) | |
| CEAa, n (%) | ||||
| ≤10 ng/mL | 197 (82.4) | 35 (77.8) | 162 (83.5) | χ2 (1) = 0.83, p = 0.386, Φ = 0.059 |
| >10 ng/mL | 42 (17.6) | 10 (22.2) | 32 (16.5) |
| All Patients | Sarcopenic | Non-Sarcopenic | Statistics Test | |
|---|---|---|---|---|
| Total abdominal muscle area, L3 (cm2), Mdn (IQR) | 142 (47.0) | 131 (24.9) | 149 (51.2) | U = 3525, p < 0.001 *, r = −0.26 |
| Skeletal muscle index (SMI) (cm2/m2), Mdn (IQR) | 53.7 (12.5) | 48.2 (8.50) | 56.2 (12.6) | U = 1955, p < 0.001 *, r = −0.45 |
| All Patients (n = 272) | Sarcopenic (n = 52) | Non-Sarcopenic (n = 220) | Statistics Test | |
|---|---|---|---|---|
| Morbimortalitya, n (%) | ||||
| Absent | 176 (65.3) | 34 (66.7) | 141 (65.0) | χ2 (1) = 0.052, p = 0.871, Φ = −0.014 |
| Present | 92 (34.7) | 17 (33.3) | 76 (35.0) | |
| Surgical site infectiona, n (%) | ||||
| Absent | 235 (87.7) | 45 (88.2) | 190 (87.6) | χ2 (1) = 0.018, p = 1.000, Φ = −0.008 |
| Present | 33 (12.3) | 6 (11.8) | 27 (12.4) | |
| Anastomosis leakagea, n (%) | ||||
| Absent | 246 (91.8) | 45 (88.2) | 201 (92.6) | Fisher’s test, p = 0.392, Φ = 0.063 |
| Present | 22 (8.20) | 6 (11.8) | 16 (7.40) | |
| Intraabdominal abscessa, n (%) | ||||
| Absent | 255 (95.1) | 48 (94.1) | 207 (95.4) | Fisher’s test, p = 0.718, Φ = 0.023 |
| Present | 13 (4.90) | 3 (5.90) | 10 (4.60) | |
| Other infectionsa,b, n (%) | ||||
| Absent | 261 (97.4) | 51 (100.0) | 210 (96.8) | Fisher’s test, p = 0.353, Φ = −0.079 |
| Present | 7 (2.60) | 0 (0.00) | 7 (3.20) | |
| Pulmonary complicationsa, n (%) | ||||
| Absent | 254 (94.8) | 48 (94.1) | 206 (94.9) | Fisher’s test, p = 0.734, Φ = 0.014 |
| Present | 14 (5.20) | 3 (5.90) | 11 (5.10) | |
| Other complicationsa,c, n (%) | ||||
| Absent | 248 (92.5) | 48 (94.1) | 200 (92.2) | Fisher’s test, p = 0.774, Φ = −0.029 |
| Present | 20 (7.50) | 3 (5.90) | 17 (7.80) | |
| Clavien-Dindo classification, n (%) | ||||
| Grade I | 27 (29.3) | 2 (11.8) | 25 (33.3) | Fisher’s test, p = 0.003 *, Φc = 0.44 |
| Grade II | 28 (30.4) | 2 (11.8) | 26 (34.7) | |
| Grade IIIb | 21 (22.8) | 7 (41.2) | 14 (18.7) | |
| Grade Iva | 1 (1.10) | 1 (5.90) | 0 (0.00) | |
| Grade IVb | 1 (1.10) | 1 (5.90) | 0 (0.00) | |
| Grade V | 14 (15.2) | 4 (23.5) | 10 (13.3) | |
| ≥ Grade II, n (%) | ||||
| Absent | 26 (28.3) | 2 (11.8) | 24 (32.0) | Fisher’s test, p = 0.137, Φ = 0.17 |
| Present | 66 (71.7) | 15 (88.2) | 51 (68.0) | |
| ≥ Grade III, n (%) | ||||
| Absent | 54 (58.7) | 4 (23.5) | 50 (66.7) | χ2 (1) = 10.6, p = 0.002 *, Φ = 0.34 |
| Present | 38 (41.3) | 13 (76.5) | 25 (33.3) |
| Univariate Analysis | Multivariate Analysis * | |||
|---|---|---|---|---|
| Unadjusted OR (CI 95%) | p Value | Adjusted OR (CI 95%) | p Value | |
| Age | 1.02 (0.98, 1.07) | 0.328 | 1.04 (0.98, 1.11) | 0.155 |
| Gender (male vs. female) | 0.66 (0.26, 1.64) | 0.369 | 1.30 (0.35, 4.90) | 0.685 |
| Tumour site (colon vs. rectum) | 0.85 (0.36, 1.99) | 0.706 | ||
| Stage (I, II vs. III, IV) | 0.87 (0.38, 2.01) | 0.747 | ||
| Total abdominal muscle area | 0.99 (0.98, 1.01) | 0.272 | ||
| Sarcopenia (absent vs. present) | 6.50 (1.92, 22.0) | 0.003 | 13.6 (2.42, 76.7) | 0.003 * |
| Anastomosis leakage (absent vs. present) | 28.9 (6.14, 136) | <0.001 | 42.9 (6.75, 272) | <0.001 * |
| Intraabdominal abscess (absent vs. present) | 2.61 (0.78, 8.73) | 0.119 | 5.42 (1.16, 25.3) | 0.031 * |
| Surgical site infection (absent vs. present) | 0.39 (0.16, 0.97) | 0.044 | 0.76 (0.19, 3.04) | 0.700 |
| Other infections (absent vs. present) | 0.27 (0.030, 2.36) | 0.234 | ||
| Pulmonary complications (absent vs. present) | 0.34 (0.087, 1.30) | 0.113 | 0.44 (0.071, 2.69) | 0.371 |
| All Patients (n = 272) | Sarcopenic (n = 52) | Non-Sarcopenic (n = 220) | Statistics Test | |
|---|---|---|---|---|
| Postoperative hospital stay (days), Mdn (IQR) | 7.00 (4.00) | 7.00 (4.00) | 7.00 (4.00) | U = 5589, p = 0.915, r = 0.007 |
| Readmission, n (%) | ||||
| Absent | 265 (97.4) | 50 (96.2) | 215 (97.7) | Fisher’s test, p = 0.622, Φ = 0.039 |
| Present | 7 (2.60) | 2 (3.80) | 5 (2.30) | |
| 30-day mortalitya, n (%) | ||||
| Absent | 251 (92.6) | 46 (88.5) | 205 (93.6) | Fisher’s test, p = 0.235, Φ = 0.078 |
| Present | 20 (7.40) | 6 (11.5) | 14 (6.40) |
| Univariate Analysis | Multivariate Analysis | |||
|---|---|---|---|---|
| Unadjusted HR (CI 95%) | p Value | Adjusted HR (CI 95%) | p | |
| Age | 1.00 (0.98, 1.02) | 0.941 | 1.02 (1.00. 1.05) | 0.118 |
| Gender (male vs. female) | 0.94 (0.59, 1.52) | 0.813 | 1.11 (0.61. 2.02) | 0.733 |
| CRC family history (absent vs. present) | 0.48 (0.15, 1.52) | 0.211 | ||
| Tumour site (colon vs. rectum) | 1.03 (0.63, 1.70) | 0.899 | ||
| Stage (I, II vs. III, IV) | 2.33 (1.45, 3.24) | <0.001 | 1.21 (0.60. 2.45) | 0.599 |
| Total abdominal muscle area | 1.00 (1.00, 1.00) | 0.357 | ||
| Sarcopenia (absent vs. present) | 1.65 (0.98, 2.78) | 0.058 | 1.83 (0.96. 3.49) | 0.066 |
| Differentiation (G1, G2 vs. G3, G4) | 1.53 (0.78, 2.97) | 0.215 | ||
| Venous invasion (absent vs. present) | 2.81 (1.75, 4.50) | <0.001 | 3.05 (1.64. 5.66) | <0.001 * |
| Lymphatic invasion (absent vs. present) | 1.69 (1.03, 2.77) | 0.036 | 0.95 (0.46. 1.97) | 0.887 |
| CEA (≤10 versus >10 ng/mL) | 2.86 (1.63, 5.03) | <0.001 | 3.21 (1.69, 6.09) | <0.001 * |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pereira, M.; Pereira, A.; Silva, P.; Costa, C.; Martins, S.F. Sarcopenia as a Risk Factor of Morbimortality in Colorectal Cancer Surgery. Gastrointest. Disord. 2020, 2, 107-117. https://doi.org/10.3390/gidisord2020010
Pereira M, Pereira A, Silva P, Costa C, Martins SF. Sarcopenia as a Risk Factor of Morbimortality in Colorectal Cancer Surgery. Gastrointestinal Disorders. 2020; 2(2):107-117. https://doi.org/10.3390/gidisord2020010
Chicago/Turabian StylePereira, Mariana, Ana Pereira, Patrícia Silva, Catarina Costa, and Sandra F. Martins. 2020. "Sarcopenia as a Risk Factor of Morbimortality in Colorectal Cancer Surgery" Gastrointestinal Disorders 2, no. 2: 107-117. https://doi.org/10.3390/gidisord2020010
APA StylePereira, M., Pereira, A., Silva, P., Costa, C., & Martins, S. F. (2020). Sarcopenia as a Risk Factor of Morbimortality in Colorectal Cancer Surgery. Gastrointestinal Disorders, 2(2), 107-117. https://doi.org/10.3390/gidisord2020010





