An Electrochemist Perspective of Microbiologically Influenced Corrosion
Abstract
1. Introduction
2. A Brief History of Microbiologically Influenced Corrosion (MIC)
3. The Biofilm
4. MIC Mechanisms
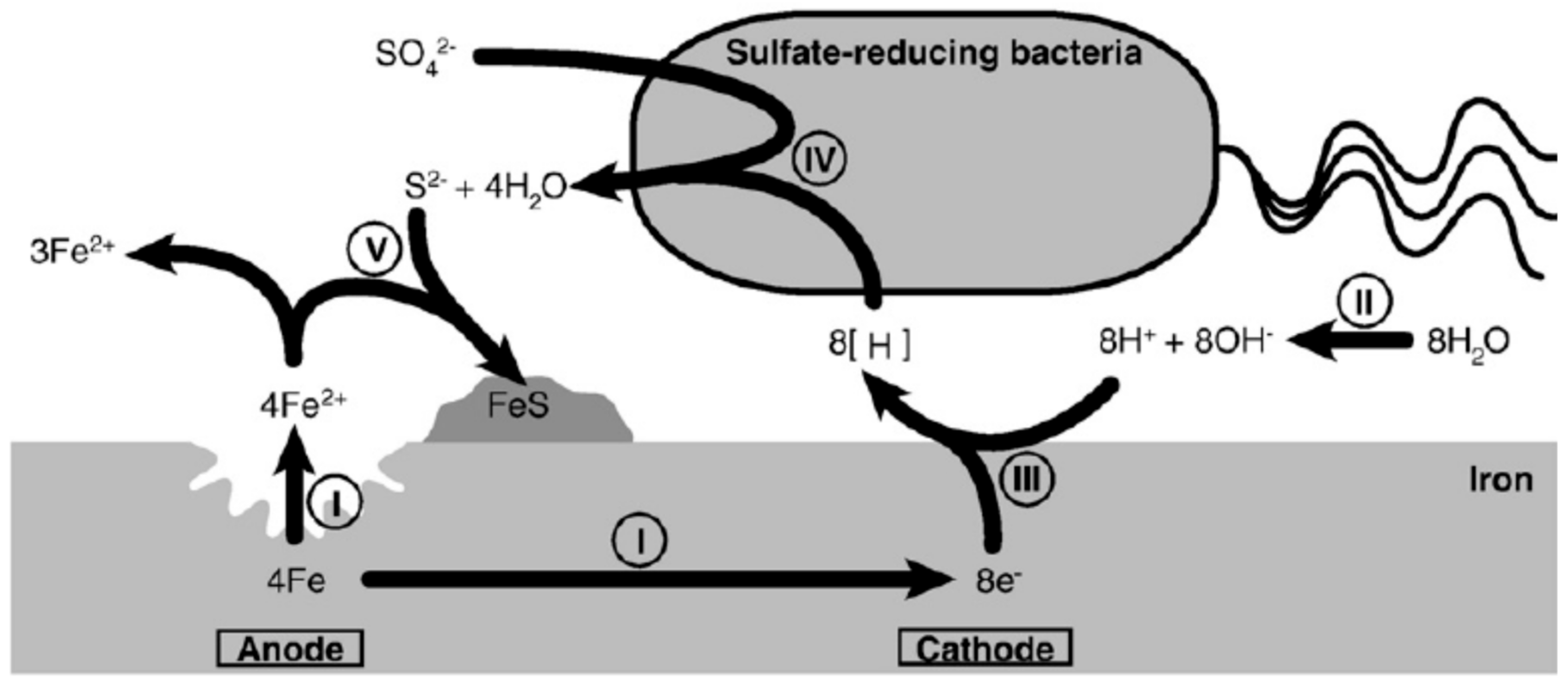
4.1. Cathodic Depolarization Theory (CDP)
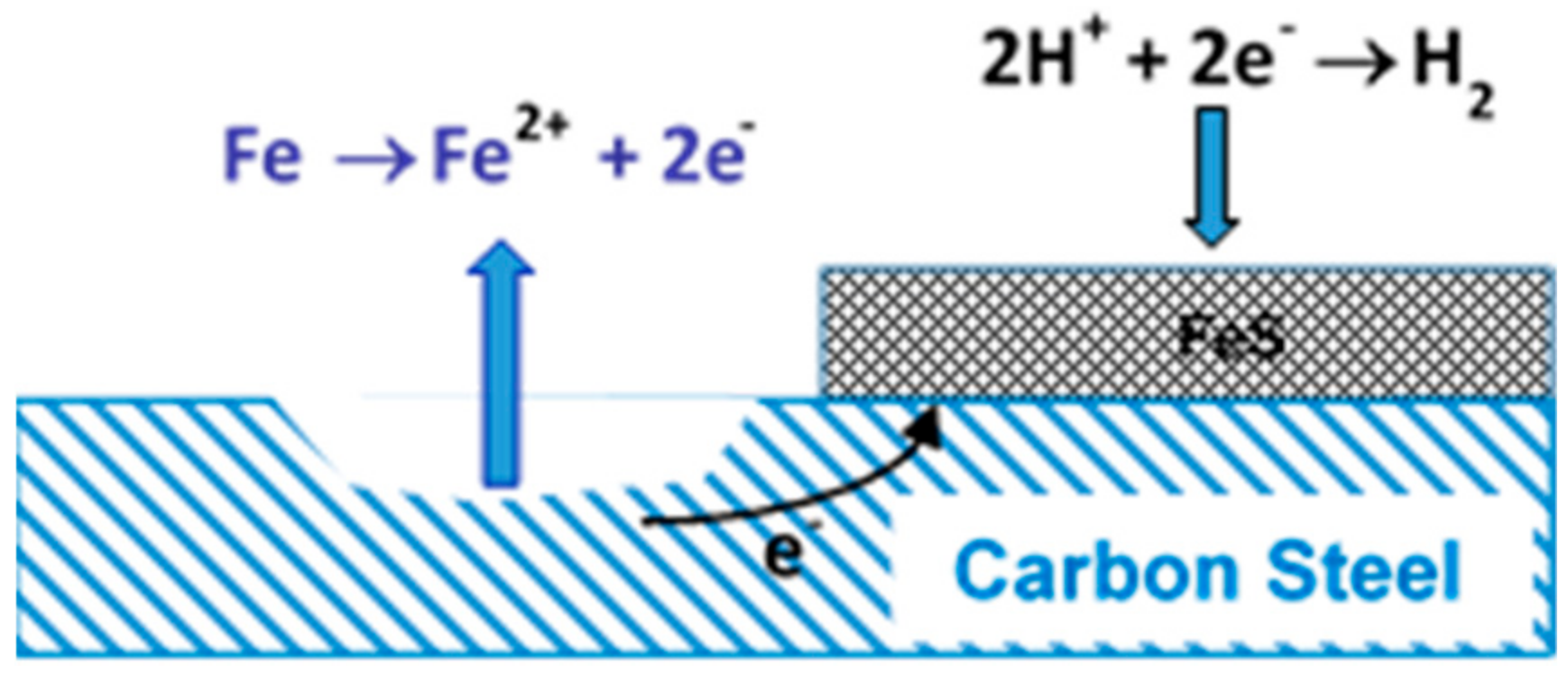
4.2. Attack by Sulphide
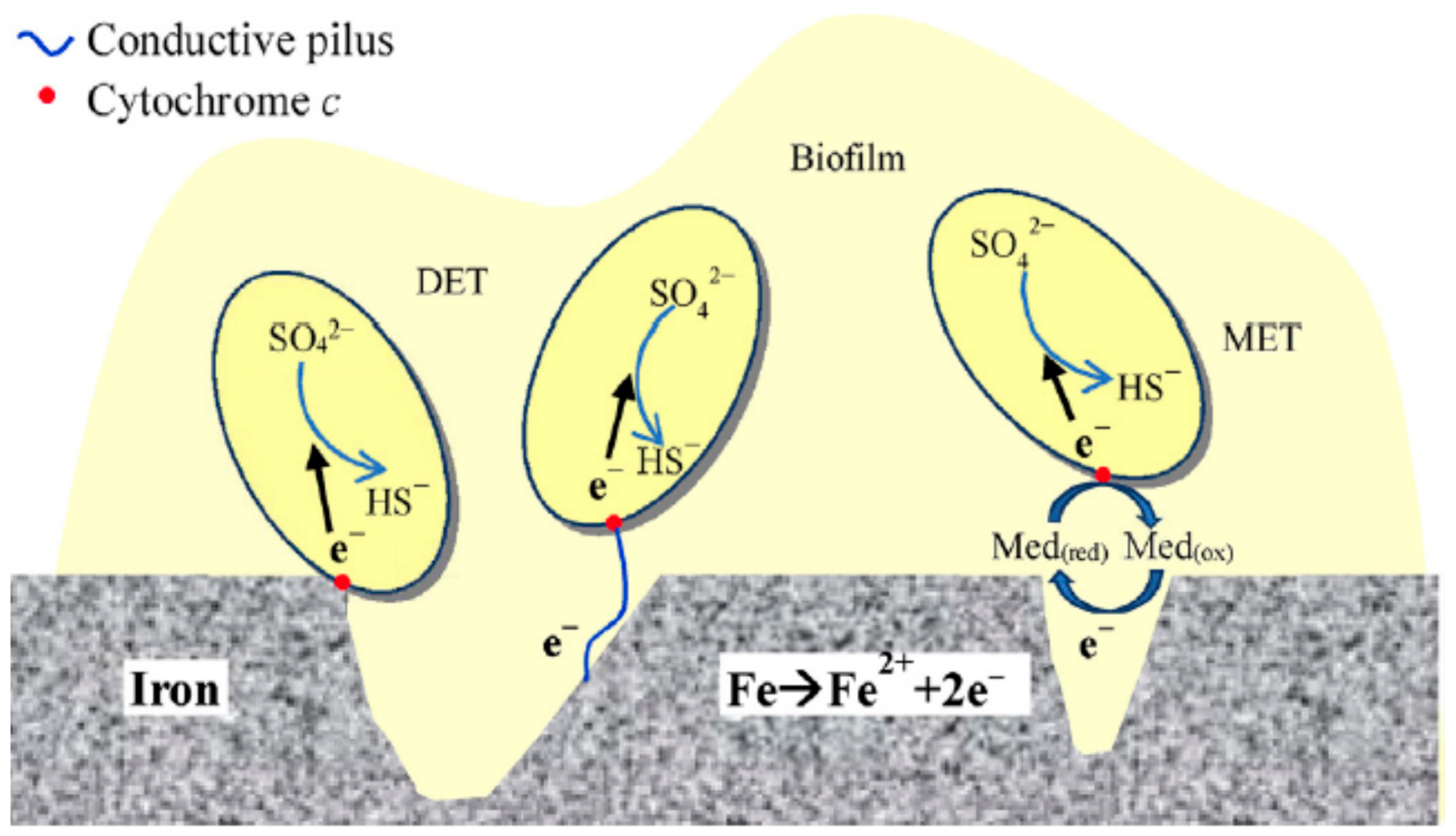
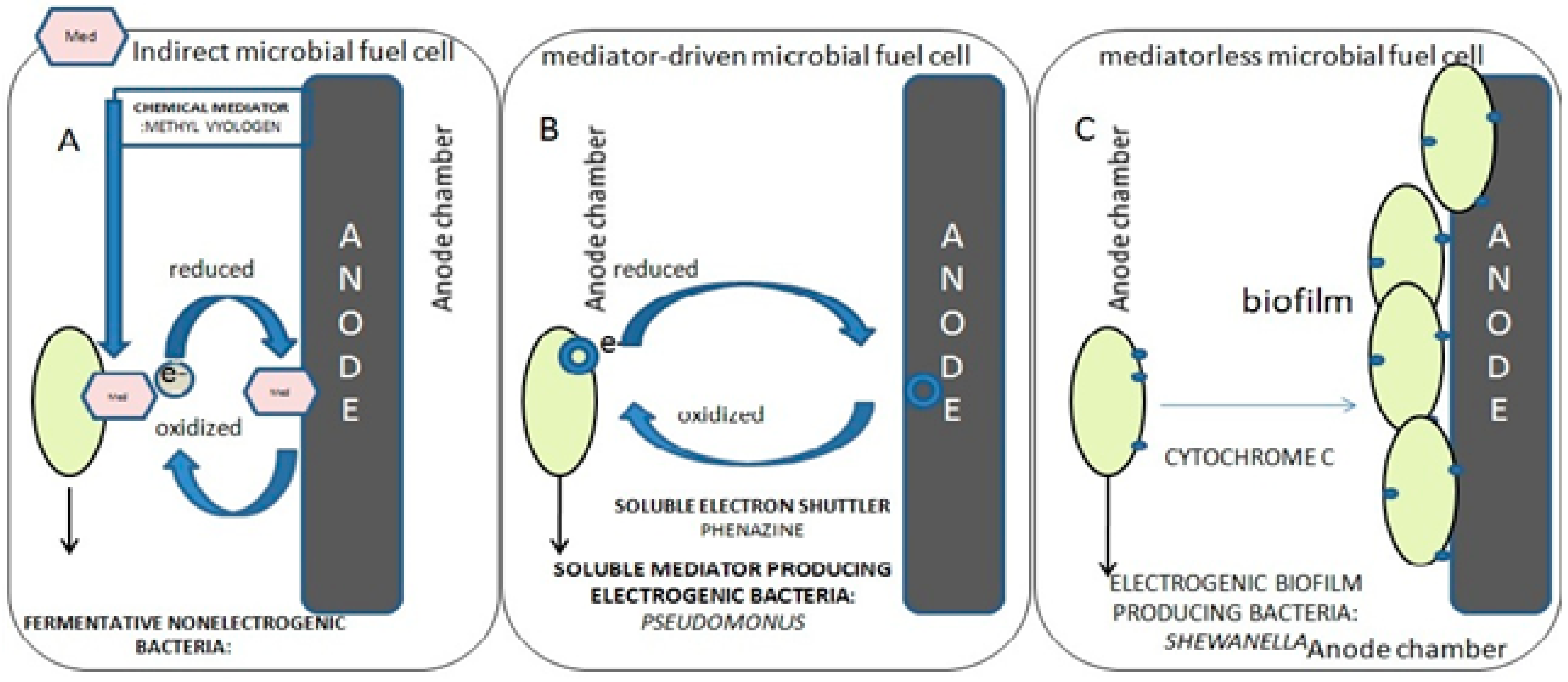
4.3. ‘Direct’ Electron Uptake
- (i)
- direct contact between the cell and the metal substrate;
- (ii)
- conductive pili—microorganisms producing extended flagellum or pili to be in contact with the electrode surface to conduct electrons;
- (iii)
- endogenous mediators—redox molecules being produced by the microorganism itself, for example riboflavin and ferredoxin;
- (iv)
- exogenous mediators—chemicals added externally to the system, for example ferrocyanide.
4.4. Impact on Surface Films
5. Concluding Remarks
Funding
Conflicts of Interest
References
- Kobrin, G. A Practical Manual on Microbiologically Influenced Corrosion; NACE International: Houston, TX, USA, 1993. [Google Scholar]
- Little, B.; Lee, J.; Ray, R. A review of ‘green’ strategies to prevent or mitigate microbiologically influenced corrosion. Biofouling 2007, 23, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Flemming, H.C. Mikrobielle werkstoffzerstörung-grundlagen: Ökonomisch-technischer überblick. Mater. Corros. 1994, 45, 5–9. [Google Scholar] [CrossRef]
- Venkatesan, R.; Muthiah, M.A.; Murugesh, P. Unusual corrosion of instruments deployed in the deep sea for Indian tsunami early warning system. Mar. Technol. Soc. J. 2014, 48, 6–13. [Google Scholar] [CrossRef]
- Little, B.; Wagner, P.; Mansfeld, F. An overview of microbiologically influenced corrosion. Electrochim. Acta 1992, 37, 2185–2194. [Google Scholar] [CrossRef]
- Iofa, Z.A.; Batrakov, V.V.; Cho Ngok, B. Influence of anion adsorption on the action of inhibitors on the acid corrosion of iron and cobalt. Electrochim. Acta 1964, 9, 1645–1653. [Google Scholar] [CrossRef]
- Stott, J.F.D. What progress in the understanding of microbially induced corrosion has been made in the last 25 years? A personal viewpoint. Corros. Sci. 1993, 35, 667–673. [Google Scholar] [CrossRef]
- Videla, H.A.; Herrera, L.K. Microbiologically influenced corrosion: Looking to the future. Int. Microbiol. 2005, 8, 169–180. [Google Scholar] [PubMed]
- Gaines, R.H. Bacterial Activity as a Corrosive Influence in the Soil. Ind. Eng. Chem. 1910, 2, 128–130. [Google Scholar] [CrossRef]
- Von Wolzogen Kühr, C.A.H.; Van der Vlugt, L.S. Graphitization of Cast Iron as an Electrobiochemical Process in Anaerobic Soils; Defense Technical Information Center: Fort Belvoir, VA, USA, 1964. [Google Scholar]
- Iverson, W.P. Direct evidence for the cathodic depolarization theory of bacterial corrosion. Science 1966, 151, 986–988. [Google Scholar] [CrossRef] [PubMed]
- Mara, D.D.; Williams, D.J.A. Polarization studies of pure Fe in the presence of hydrogenase-positive microbes—I. Non-photosynthetic bacteria. Corros. Sci. 1971, 12, 895–900. [Google Scholar] [CrossRef]
- Iverson, W.P. Corrosion of iron and formation of iron phosphide by Desulfovibrio Desulfuricans. Nature 1968, 217, 1265–1267. [Google Scholar] [CrossRef] [PubMed]
- Booth, G.H.; Tiller, A.K. Polarization studies of mild steel in cultures of sulphate-reducing bacteria. Trans. Faraday Soc. 1960, 56, 1689–1696. [Google Scholar] [CrossRef]
- Booth, G.H. Sulphur bacteria in relation to corrosion. J. Appl. Microbiol. 1964, 27, 174–181. [Google Scholar] [CrossRef]
- Ferris, F.G.; Schultze, S.; Witten, T.C.; Fyfe, W.S.; Beveridge, T.J. Metal interactions with microbial biofilms in acidic and neutral pH environments. Appl. Environ. Microbiol. 1989, 55, 1249–1257. [Google Scholar] [PubMed]
- Javaherdashti, R. A review of some characteristics of MIC caused by sulfate-reducing bacteria: Past, present and future. Anti-Corros. Methods Mater. 1999, 46, 173–180. [Google Scholar] [CrossRef]
- Watnick, P.; Kolter, R. Biofilm, city of microbes. J. Bacteriol. 2000, 182, 2675–2679. [Google Scholar] [CrossRef] [PubMed]
- Borenstein, S. Microbiologically Influenced Corrosion Handbook; Elsevier: Amsterdam, The Netherlands, 1994. [Google Scholar]
- Videla, H.A.; Characklis, W.G. Biofouling and microbially influenced corrosion. Int. Biodeterior. Biodegrad. 1992, 29, 195–212. [Google Scholar] [CrossRef]
- Mansfeld, F.; Little, B. A technical review of electrochemical techniques applied to microbiologically influenced corrosion. Corros. Sci. 1991, 32, 247–272. [Google Scholar] [CrossRef]
- Walsh, D.; Pope, D.; Danford, M.; Huff, T. The effect of microstructure on microbiologically influenced corrosion. JOM J. Miner. Met. Mater. Soc. 1993, 45, 22–30. [Google Scholar] [CrossRef]
- Magot, M.; Ollivier, B.; Patel, B.K.C. Microbiology of petroleum reservoirs. Antonie Van Leeuwenhoek 2000, 77, 103–116. [Google Scholar] [CrossRef] [PubMed]
- Mitterer, R.M. Methanogenesis and sulfate reduction in marine sediments: A new model. Earth Planet. Sci. Lett. 2010, 295, 358–366. [Google Scholar] [CrossRef]
- Magot, M. Indigenous microbial communities in oil fields. In Petroleum Microbiology; Oliver, B., Magot, B., Eds.; American Society of Microbiology: Washington, DC, USA, 2005; pp. 21–34. [Google Scholar]
- Larsen, J.; Rasmussen, K.; Pedersen, H.; Sørensen, K.; Lundgaard, T.; Skovhus, T.L. Consortia of MIC bacteria and archaea causing pitting corrosion in top side oil production facilities. In Proceedings of the Corrosion 2010, Paper No 10252. San Antonio, TX, USA, 14–18 March 2010. [Google Scholar]
- Vigneron, A.; Alsop, E.B.; Chambers, B.; Lomans, B.P.; Head, I.M.; Tsesmetzis, N. Complementary microorganisms in highly corrosive biofilms from an offshore oil production facility. Appl. Environ. Microbiol. 2016, 82, 2545–2554. [Google Scholar] [CrossRef] [PubMed]
- Javaherdashti, R. Microbiologically Influenced Corrosion: An Engineering Insight; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2008. [Google Scholar]
- Usher, K.M.; Kaksonen, A.H.; Bouquet, D.; Cheng, K.Y.; Geste, Y.; Chapman, P.G.; Johnston, C.D. The role of bacterial communities and carbon dioxide on the corrosion of steel. Corros. Sci. 2015, 98, 354–365. [Google Scholar] [CrossRef]
- Usher, K.M.; Kaksonen, A.H.; Cole, I.; Marney, D. Critical review: Microbially influenced corrosion of buried carbon steel pipes. Int. Biodeterior. Biodegrad. 2014, 93, 84–106. [Google Scholar] [CrossRef]
- Dinh, H.T.; Kuever, J.; Mußmann, M.; Hassel, A.W.; Stratmann, M.; Widdel, F. Iron corrosion by novel anaerobic microorganisms. Nature 2004, 427, 829–832. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Fang, H.H.P.; Ko, B.C.B. Methanogen population in a marine biofilm corrosive to mild steel. Appl. Microbiol. Biotechnol. 2003, 63, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, T.; Ito, K.; Mori, K.; Tsurumaru, H.; Harayama, S. Iron-corroding methanogen isolated from a crude-oil storage tank. Appl. Environ. Microbiol. 2010, 76, 1783–1788. [Google Scholar] [CrossRef] [PubMed]
- Boopathy, R.; Daniels, L. Effect of pH on anaerobic mild steel corrosion by methanogenic bacteria. Appl. Environ. Microbiol. 1991, 57, 2104–2108. [Google Scholar] [PubMed]
- Mori, K.; Tsurumaru, H.; Harayama, S. Iron corrosion activity of anaerobic hydrogen-consuming microorganisms isolated from oil facilities. J. Biosci. Bioeng. 2010, 110, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Usher, K.M.; Kaksonen, A.H.; MacLeod, L.D. Marine rust tubercles harbour iron corroding archaea and sulphate reducing bacteria. Corros. Sci. 2014, 83, 189–197. [Google Scholar] [CrossRef]
- Kip, N.; van Veen, J.A. The dual role of microbes in corrosion. ISME J. 2015, 9, 542–551. [Google Scholar] [CrossRef] [PubMed]
- Videla, H.A.; Herrera, L.K. Understanding microbial inhibition of corrosion. A comprehensive overview. Int. Biodeterior. Biodegrad. 2009, 63, 896–900. [Google Scholar] [CrossRef]
- Herrera, L.K.; Videla, H.A. Role of iron-reducing bacteria in corrosion and protection of carbon steel. Int. Biodeterior. Biodegrad. 2009, 63, 891–895. [Google Scholar] [CrossRef]
- Jefferson, K.K. What drives bacteria to produce a biofilm? FEMS Microbiol. Lett. 2004, 236, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Zobell, C.E.; Allen, E.C. The significance of marine bacteria in the fouling of submerged surfaces. J. Bacteriol. 1935, 29, 239–251. [Google Scholar] [PubMed]
- Dempsey, M.J. Marine bacterial fouling: A scanning electron microscope study. Mar. Biol. 1981, 61, 305–315. [Google Scholar] [CrossRef]
- Li, K.; Whitfield, M.; van Vliet, K.J. Beating the bugs: Roles of microbial biofilms in corrosion. Corros. Rev. 2013, 31, 73–84. [Google Scholar] [CrossRef]
- Callow, M.E.; Fletcher, R.L. The influence of low surface energy materials on bioadhesion—A review. Int. Biodeterior. Biodegrad. 1994, 34, 333–334. [Google Scholar] [CrossRef]
- Eashwar, M.; Dexter, S. Relation of Bacterial Settlement Patterns to Anodic Activity on Stainless Steel Weldments. In Proceedings of the Corrosion 1999, San Antonio, TX, USA, Paper No 174. 25–30 April 1999. [Google Scholar]
- Little, B.; Wagner, P.; Jacobus, J. The Impact of Sulfate-Reducing Bacteria on Welded Copper Nickel Seawater Piping Systems; Report No. DTIC ADA204956; Defense Technical Information Center: Fort Belvoir, VA, USA, 1988. [Google Scholar]
- Tiller, A.K. Aspect of microbial corrosion. In Corrosion Processes; Parkins, R.N., Ed.; Applied Science Publication: London, UK, 1982; pp. 115–159. [Google Scholar]
- Little, B.; Wagner, P.; Mansfeld, F. Microbiologically influenced corrosion of metals and alloys. Int. Mater. Rev. 1991, 36, 253–272. [Google Scholar] [CrossRef]
- Characklis, W. Bioengineering report: Fouling biofilm development: A process analysis. Biotechnol. Bioeng. 1981, 23, 1923–1960. [Google Scholar] [CrossRef]
- Pedersen, A.; Hermansson, M. The effects on metal corrosion by Serratia marcescens and a Pseudomonas SP. Biofouling 2009, 313–322. [Google Scholar]
- Pope, D.H.; Pope, R.M. Guide for the Monitoring and Treatment of Microbiologically Influenced Corrosion in the Natural Gas Industry; Gas Research Institute: Des Plaines, IL, USA, 1998. [Google Scholar]
- Little, B.J.; Lee, J.S.; Ray, R.I. The influence of marine biofilms on corrosion: A concise review. Electrochim. Acta 2008, 54, 2–7. [Google Scholar] [CrossRef]
- Meyer, B. Approaches to prevention, removal and killing of biofilms. Int. Biodeterior. Biodegrad. 2003, 51, 249–253. [Google Scholar] [CrossRef]
- Duan, J.; Wu, S.; Zhang, X.; Huang, G.; Du, M.; Hou, B. Corrosion of carbon steel influenced by anaerobic biofilm in natural seawater. Electrochim. Acta 2008, 54, 22–28. [Google Scholar] [CrossRef]
- Deutzmann, J.S.; Sahin, M.; Spormann, A.M. Extracellular enzymes facilitate electron uptake in biocorrosion and bioelectrosynthesis. mBio 2015, 6, e00496-15. [Google Scholar] [CrossRef] [PubMed]
- Blackwood, D.J.; Seah, K.H.W.; Teoh, S.H. Corrosion of metallic implants. In Engineering Materials for Biomedical Applications; Teoh, S.H., Ed.; World Scientific: Singapore, 2004; Chapter 3; pp. 3.1–3.56. [Google Scholar]
- Little, B.J.; Lee, J.S. Microbiologically influenced corrosion: An update. J. Int. Mater. Rev. 2014, 59, 384–393. [Google Scholar] [CrossRef]
- Gu, T.; Galicia, B. Can acid producing bacteria be responsible for very fast MIC pitting? In Proceedings of the Corrosion 2012, Salt Lake City, UT, USA, 11–15 March 2012. Paper No 1214. [Google Scholar]
- Parker, C.D. Species of sulphur bacteria associated with the corrosion of concrete. Nature 1947, 159, 439–440. [Google Scholar] [CrossRef] [PubMed]
- Ray, R.I.; Lee, J.S.; Little, B.J. Iron-oxidizing bacteria: A review of corrosion mechanisms in fresh water and marine environments. In Proceedings of the Corrosion 2010, Paper No 10218. San Antonio, TX, USA, 14—18 March 2010. [Google Scholar]
- Little, B.; Lee, J.; Ray, R. Recent Developments in the Identification of Mechanisms for Microbiologically Influenced Corrosion; Report No. DTIC A537292; Defense Technical Information Center: Fort Belvoir, VA, USA, 2010. [Google Scholar]
- Banfield, J.F.; Welch, S.A.; Zhang, H.; Ebert, T.T.; Penn, R.L. Aggregation-based crystal growth and microstructure development in natural iron oxyhydroxide biomineralization products. Science 2000, 289, 751–754. [Google Scholar] [CrossRef] [PubMed]
- Linhardt, P. Microbially influenced corrosion of stainless steel by manganese oxidizing microorganisms. Mater. Corros. 2004, 55, 158–163. [Google Scholar] [CrossRef]
- Linhardt, P. Twenty years of experience with corrosion failures caused by manganese oxidizing microorganisms. Mater. Corros. 2010, 61, 1034–1039. [Google Scholar] [CrossRef]
- Sly, L.I.; Hodgkinson, M.C.; Arunpairojana, V. Deposition of manganese in a drinking water distribution system. Appl. Environ. Microbiol. 1990, 56, 628–639. [Google Scholar] [PubMed]
- Rhoads, A.; Beyenal, H.; Lewandowski, Z. Microbial fuel cell using anaerobic respiration as an anodic reaction and biomineralized manganese as a cathodic reactant. Environ. Sci. Technol. 2005, 39, 4666–4671. [Google Scholar] [CrossRef] [PubMed]
- Iverson, W.P. Direct Evidence for the Cathodic Depolarization Theory of Bacterial Corrosion; Defense Technical Information Center: Fort Belvoir, VA, USA, 1965; Report No. DTIC AD0476409. [Google Scholar]
- Bryant, R.D.; Jansen, W.; Boivin, J.; Laishley, E.J.; Costerton, J.W. Effect of hydrogenase and mixed sulfate-reducing bacterial populations on the corrosion of steel. Appl. Environ. Microbiol. 1991, 57, 2804–2809. [Google Scholar] [PubMed]
- Bryant, R.D.; Laishley, E.J. The effect of inorganic phosphate and hydrogenase on the corrosion of mild steel. Appl. Environ. Microbiol. 1993, 38, 824–827. [Google Scholar] [CrossRef]
- Trasatti, S. Work function, electronegativity, and electrochemical behaviour of metals: III. Electrolytic hydrogen evolution in acid solutions. J. Electroanal. Chem. 1972, 39, 163–184. [Google Scholar] [CrossRef]
- Frankenthal, R.P.; Milner, P.C. Technical note: Hydrogen evolution kinetics on a high-carbon steel and on tin in seawater. Corrosion 1986, 42, 51–53. [Google Scholar] [CrossRef]
- Smart, N.R.; Blackwood, D.J.; Werme, L. Anaerobic corrosion of carbon steel and cast iron in artificial groundwaters: Part 1—Electrochemical aspects. Corrosion 2002, 58, 547–559. [Google Scholar] [CrossRef]
- Smart, N.R.; Blackwood, D.J.; Werme, L. Anaerobic corrosion of carbon steel and cast iron in artificial groundwaters: Part 2—Gas generation. Corrosion 2002, 58, 627–637. [Google Scholar] [CrossRef]
- Spruit, C.J.P.; Wanklyn, J.N. Iron sulphide ratios in corrosion by sulphate-reducing bacteria. Nature 1951, 168, 951–952. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.A. Utilization of cathodic hydrogen by sulphate-reducing bacteria. Br. Corros. J. 1983, 18, 190–193. [Google Scholar] [CrossRef]
- Costello, J.A. Cathodic depolarization by sulfate-reducing bacteria. S. Afr. J. Sci. 1974, 70, 202–204. [Google Scholar]
- King, R.A.; Miller, J.D.A.; Smith, J.S. Corrosion of mild steel by iron sulphides. Br. Corros. J. 1973, 8, 137–141. [Google Scholar]
- Enning, D.; Venzlaff, H.; Garrelfs, J.; Dinh, H.T.; Meyer, V.; Mayrhofer, K.; Hassel, A.W.; Stratmann, M.; Widdel, F. Marine sulfate-reducing bacteria cause serious corrosion of iron under electroconductive biogenic mineral crust. Environ. Microbiol. 2012, 14, 1772–1787. [Google Scholar]
- Jia, R.; Tan, J.L.; Jin, P.; Blackwood, D.J.; Xu, D.; Gu, T. Effects of biogenic H2S on the microbiologically influenced corrosion of C1018 carbon steel by sulfate reducing Desulfovibrio vulgaris biofilm. Corros. Sci. 2018, 130, 1–11. [Google Scholar] [CrossRef]
- Shoesmith, D.W.; Taylor, P.; Bailey, M.G.; Owen, D.G. The formation of ferrous monosulfide polymorphs during the corrosion of iron by aqueous hydrogen sulfide at 21 °C. J. Electrochem. Soc. 1980, 127, 1007–1015. [Google Scholar] [CrossRef]
- Zheng, Y.; Ning, J.; Brown, B.; Young, D.; Nešić, S. Mechanistic study of the effect of iron sulphide layers on hydrogen sulphide corrosion of carbon steel. In Proceedings of the Corrosion 2015, Paper No 5933. Dallas, TX, USA, 15–19 March 2015. [Google Scholar]
- Gu, T.; Zhao, K.; Nešić, S. A new mechanistic model for MIC based on a biocatalytic cathodic sulfate reduction theory. In Proceedings of the Corrosion 2009, Atlanta, GA, USA, 22–26 March 2009. Paper No 09390. [Google Scholar]
- Elliott, P.; Ragusa, S.; Catcheside, D. Growth of sulfate-reducing bacteria under acidic conditions in an upflow anaerobic bioreactor as a treatment system for acid mine drainage. Water Res. 1998, 32, 3724–3730. [Google Scholar] [CrossRef]
- Morse, J.W.; Millero, F.J.; Cornwell, J.C.; Rickard, D. The chemistry of the hydrogen sulfide and iron sulfide systems in natural waters. Earth-Sci. Rev. 1987, 24, 1–42. [Google Scholar] [CrossRef]
- Sun, W.; Nešić, S.; Young, D.; Woollam, R.C. Equilibrium expressions related to the solubility of the sour corrosion product mackinawite. Ind. Eng. Chem. Res. 2008, 47, 1738–1742. [Google Scholar] [CrossRef]
- Enning, D.; Garrelfs, J. Corrosion of iron by sulfate-reducing bacteria: New views of an old problem. Appl. Environ. Microbiol. 2014, 80, 1226–1236. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Ray, R.I.; Little, B.J.; Lemieux, E.J. Evaluation of deoxygenation as a corrosion control measure for ballast tanks. Corrosion 2005, 61, 1173–1188. [Google Scholar] [CrossRef]
- Iino, T.; Ito, K.; Wakai, S.; Tsurumaru, H.; Ohkuma, M.; Harayama, S. Iron Corrosion Induced by Nonhydrogenotrophic Nitrate-Reducing Prolixibacter sp. Strain MIC1-1. Appl. Environ. Microbiol. 2015, 81, 1839–1846. [Google Scholar] [CrossRef] [PubMed]
- Jia, R.; Yang, D.; Xu, D.; Gu, T. Electron transfer mediators accelerated the microbiologically influence corrosion against carbon steel by nitrate reducing Pseudomonas aeruginosa biofilm. Bioelectrochemistry 2017, 118, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.L.; Goh, P.C.; Blackwood, D.J. Influence of H2S-producing chemical species in culture medium and energy source starvation on carbon steel corrosion caused by methanogens. Corros. Sci. 2017, 119, 102–111. [Google Scholar] [CrossRef]
- Venzlaff, H.; Enning, D.; Srinivasan, J.; Mayrhofer, K.J.J.; Hassel, A.W.; Widdel, F.; Stratmann, M. Accelerated cathodic reaction in microbial corrosion of iron due to direct electron uptake by sulfate-reducing bacteria. Corros. Sci. 2013, 66, 88–96. [Google Scholar] [CrossRef]
- Kato, S. Microbial extracellular electron transfer and its relevance to iron corrosion. Microb. Biotechnol. 2016, 9, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Mand, J.; Park, H.S.; Okoro, C.; Lomans, B.P.; Smith, S.; Chiejina, L.; Voordouw, G. Microbial methane production associated with carbon steel corrosion in a Nigerian oil field. Front. Microbiol. 2015, 6, 1538. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xu, D.; Chen, C.; Li, X.; Jia, R.; Zhang, D.; Sand, W.; Wang, F.; Gu, T. Anaerobic microbiologically influenced corrosion mechanisms interpreted using bioenergetics and bioelectrochemistry: A review. J. Mater. Sci. Technol. 2018, 34, 1713–1718. [Google Scholar] [CrossRef]
- Xu, D.; Gu, T. Carbon source starvation triggered more aggressive corrosion against carbon steel by the Desulfovibrio vulgaris biofilm. Int. Biodeterior. Biodegrad. 2014, 91, 74–81. [Google Scholar] [CrossRef]
- Jia, R.; Yang, D.; Xu, J.; Xu, D.; Gu, T. Microbiologically influenced corrosion of C1018 carbon steel by nitrate reducing Pseudomonas aeruginosa biofilm under organic carbon starvation. Corros. Sci. 2017, 127, 1–9. [Google Scholar] [CrossRef]
- Xu, D.; Li, Y.; Gu, T. Mechanistic modeling of biocorrosion caused by biofilms of sulfate reducing bacteria and acid producing bacteria. Bioelectrochemistry 2016, 110, 52–58. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Wang, H.; Hassett, D.J.; Gu, T. Recent advances in microbial fuel cells (MFCs) and microbial electrolysis cells (MECs) for wastewater treatment, bioenergy and bioproducts. J. Chem. Technol. Biotechnol. 2013, 88, 508–518. [Google Scholar] [CrossRef]
- Torres, C.I.; Marcus, A.K.; Lee, H.S.; Parameswaran, P.; Krajmalnik-Brown, R.; Rittmann, B.E. A kinetic perspective on extracellular electron transfer by anode-respiring bacteria. FEMS Microbiol. Rev. 2010, 34, 3–17. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.; Xu, D.; Li, Y.; Yang, K.; Gu, T. Electron mediators accelerate the microbiologically influenced corrosion of 304 stainless steel by the Desulfovibrio vulgaris biofilm. Bioelectrochemistry 2015, 101, 14–21. [Google Scholar] [CrossRef] [PubMed]
- Bockris, J.; Reddy, A.K.N. Modern Electrochemistry; Plenum Press: New York, NY, USA, 1970; Volume 2, Chapter 11; 630p. [Google Scholar]
- Gray, H.B.; Winkler, J.R. Long-Range Electron Transfer. Proc. Natl. Acad. Sci. USA 2005, 102, 3534–3539. [Google Scholar] [CrossRef] [PubMed]
- Hine, R. “Membrane.” The Facts on File Dictionary of Biology, 3rd ed.; Checkmark: New York, NY, USA, 1999; 198p. [Google Scholar]
- Eddowes, M.J.; Hill, H.A.O. Novel method for the investigation of the electrochemistry of metalloproteins: Cytochrome. J. Chem. Soc. Chem. Com. 1977, 45, 771–772. [Google Scholar] [CrossRef]
- Sherar, B.W.A.; Power, I.M.; Keech, P.G.; Mitlin, S.; Southam, G.; Shoesmith, D.W. Characterizing the effect of carbon steel exposure in sulfide containing solutions to microbially induced corrosion. Corros. Sci. 2011, 53, 955–960. [Google Scholar] [CrossRef]
- Xu, D.; Li, Y.; Song, F.; Gu, T. Laboratory investigation of microbiologically influenced corrosion of C1018 carbon steel by nitrate reducing bacterium Bacillus licheniformis. Corros. Sci. 2013, 77, 385–390. [Google Scholar] [CrossRef]
- Reguera, G.; McCarthy, K.D.; Mehta, T.; Nicoll, J.S.; Tuominen, M.T.; Lovley, D.R. Extracellular electron transfer via microbial nanowires. Nature 2005, 435, 1098–1101. [Google Scholar] [CrossRef] [PubMed]
- Gorby, Y.A.; Yanina, S.; McLean, J.S.; Rosso, K.M.; Moyles, D.; Dohnalkova, A.; Beveridge, T.J.; Chang, I.S.; Kim, B.H.; Kim, K.S. Electrically conductive bacterial nanowires produced by Shewanella oneidensis strain MR-1 and other microorganisms. Proc. Natl. Acad. Sci. USA 2006, 103, 11358–11363. [Google Scholar] [CrossRef] [PubMed]
- Lovley, D.R. Happy together: Microbial communities that hook up to swap electrons. ISME J. 2017, 11, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Amdursky, N.; Marchak, D.; Sepunaru, L.; Pecht, I.; Sheves, M.; Cahen, D. Electronic Transport via Proteins. Adv. Mater. 2014, 26, 7142–7161. [Google Scholar] [CrossRef] [PubMed]
- Fink, H.W.; Schönenberger, C. Electrical conduction through DNA molecules. Nature 1999, 398, 407–410. [Google Scholar] [CrossRef] [PubMed]
- Buck, R.P. Electron hopping in one dimension: Mixed conductor membranes. J. Phys. Chem. 1988, 92, 4196–4200. [Google Scholar] [CrossRef]
- Witt, H.T.; Müller, A.; Rumberg, B. Oxidized cytochrome and chlorophyll in photosynthesis. Nature 1961, 192, 967–969. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.B.; Midya, A.; Loh, K.P.; Adams, S.; Blackwood, D.J.; Wang, J.; Zhang, X. Highly efficient dye-sensitized solar cells using phenothiazine derivative organic dyes. Prog. Photovoltaics 2010, 18, 573–581. [Google Scholar] [CrossRef]
- Harper, A.; Anderson, M.R. Electrochemical glucose sensors—Developments using electrostatic assembly and carbon nanotubes for biosensor construction. Sensors 2010, 10, 8248–8274. [Google Scholar] [CrossRef] [PubMed]
- Eschbach, M.; Schreiber, K.; Trunk, K.; Buer, J.; Jahn, D.; Schobert, M. Long-term anaerobic survival of the opportunistic pathogen Pseudomonas aeruginosa via pyruvate fermentation. J. Bacteriol. 2004, 186, 4596–4604. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, W.; Holliger, C.; Zehnder, A.J.B.; Hagen, W.R. Redox chemistry of cobalamin and iron-sulfur cofactors in the tetrachloroethene reductase of Dehalobacter restrictus. FEBS Lett. 1997, 409, 421–425. [Google Scholar] [CrossRef]
- Müller, V.; Blaut, M.; Gottschalk, G. Bioenergetics of methanogenesis. In Methanogenesis: Ecology, Physiology, Biochemistry & Genetics; Ferry, J.G., Ed.; Springer: Dordrecht, The Netherlands, 1993; pp. 360–406. [Google Scholar]
- Loach, P.A. Oxidation-reduction potentials, absorbance bands and molar absorbance of compounds used in biochemical studies. In Handbook of Biochemistry and Molecular Biology, 4th ed.; Lundlad, R.L., MacDonald, F.M., Eds.; CRC Press: Boca Raton, FL, USA, 2010; pp. 557–563. [Google Scholar]
- Pandit, S.; Chandrasekhar, K.; Kakarla, R.; Kadier, A.; Jeevitha, V. Basic Principles of Microbial Fuel Cell: Technical Challenges and Economic Feasibility. In Microbial Applications; Kalia, V., Kumar, P., Eds.; Springer: Cham, Switzerland, 2017; Volume 1, pp. 165–188. [Google Scholar]
- Eckert, R.; Skovhus, T.L. Advances in the application of molecular microbiological methods in the oil and gas industry and links to microbiologically influenced corrosion. Int. Biodeterior. Biodegrad. 2018, 126, 169–176. [Google Scholar] [CrossRef]
- Pourbaix, M. Atlas of Electrochemical Equilibria in Aqueous Solutions; National Association of Corrosion Engineers: Houston, TX, USA, 1974. [Google Scholar]
- Fontana, M.G. Corrosion Engineering; McGraw Hill: New York, NY, USA, 1986; 94p. [Google Scholar]
- Obuekwe, C.O.; Westlake, D.W.S.; Plambeck, J.A.; Cook, F.D. Corrosion of mild steel in cultures of ferric iron reducing bacterium isolated from crude oil I. polarization characteristics. Corrosion 1981, 37, 461–467. [Google Scholar] [CrossRef]
- Beech, I.B.; Sunner, J. Biocorrosion: Towards understanding interactions between biofilms and metals. Curr. Opin. Biotechnol. 2004, 15, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Lovley, D.R.; Stolz, J.F.; Nord, G.L.; Philips, E.J.P. Anaerobic production of magnetite by a dissumlatory iron-reducing microorganism. Nature 1987, 330, 252–254. [Google Scholar] [CrossRef]
- Valencia-Cantero, E.; Peña-Cabriales, J.J. Effects of iron-reducing bacteria on carbon steel corrosion Induced by Thermophilic sulfate-reducing consortia. J. Microbiol. Biotechnol. 2014, 24, 280–286. [Google Scholar] [CrossRef] [PubMed]

| Gas | Pressure | Temp | Ecorr | Icorr | βc | βa |
|---|---|---|---|---|---|---|
| (Atms) | (°C) | (mV vs. SHE) | (µA.cm−2) | (mV dec−1) | (mV dec−1) | |
| Argon | 1 | 24 | −575 | 1.7 | −156 | 71 |
| Hydrogen | 1 | 24 | −607 | 1.1 | −132 | 71 |
| “ | 10 | 24 | −567 | 3.5 | −199 | 75 |
| “ | 85 | 24 | −527 | 1.4 | −194 | 59 |
| Argon | 1 | 50 | −593 | 1.2 | −126 | 74 |
| Hydrogen | 1 | 50 | −613 | 0.98 | −144 | 66 |
| “ | 10 | 50 | −589 | 3.1 | −158 | 67 |
| “ | 85 | 50 | −567 | 4.3 | −204 | 54 |
| Argon | 1 | 80 | −596 | 2.7 | −168 | 46 |
| Hydrogen | 1 | 80 | −591 | 2.8 | −161 | 47 |
| “ | 10 | 80 | −588 | 7.2 | −183 | 52 |
| “ | 85 | 80 | −559 | 12 | −149 | 50 |
| Redox Reaction | Potential V vs. SHE at pH 7 |
|---|---|
| −0.4137 V | |
| −0.35 V | |
| −0.34 V | |
| −0.32 V | |
| −0.22 V | |
| −0.19 V |
© 2018 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blackwood, D.J. An Electrochemist Perspective of Microbiologically Influenced Corrosion. Corros. Mater. Degrad. 2020, 1, 59-76. https://doi.org/10.3390/cmd1010005
Blackwood DJ. An Electrochemist Perspective of Microbiologically Influenced Corrosion. Corrosion and Materials Degradation. 2020; 1(1):59-76. https://doi.org/10.3390/cmd1010005
Chicago/Turabian StyleBlackwood, Daniel J. 2020. "An Electrochemist Perspective of Microbiologically Influenced Corrosion" Corrosion and Materials Degradation 1, no. 1: 59-76. https://doi.org/10.3390/cmd1010005
APA StyleBlackwood, D. J. (2020). An Electrochemist Perspective of Microbiologically Influenced Corrosion. Corrosion and Materials Degradation, 1(1), 59-76. https://doi.org/10.3390/cmd1010005





