Montecarlo Simulation and HAXPES Analysis of Organosilane Segregation in Titania Xerogel Films; Towards a Generic Surface Chemofunctionalization Process
Abstract
1. Introduction
2. Materials and Methods
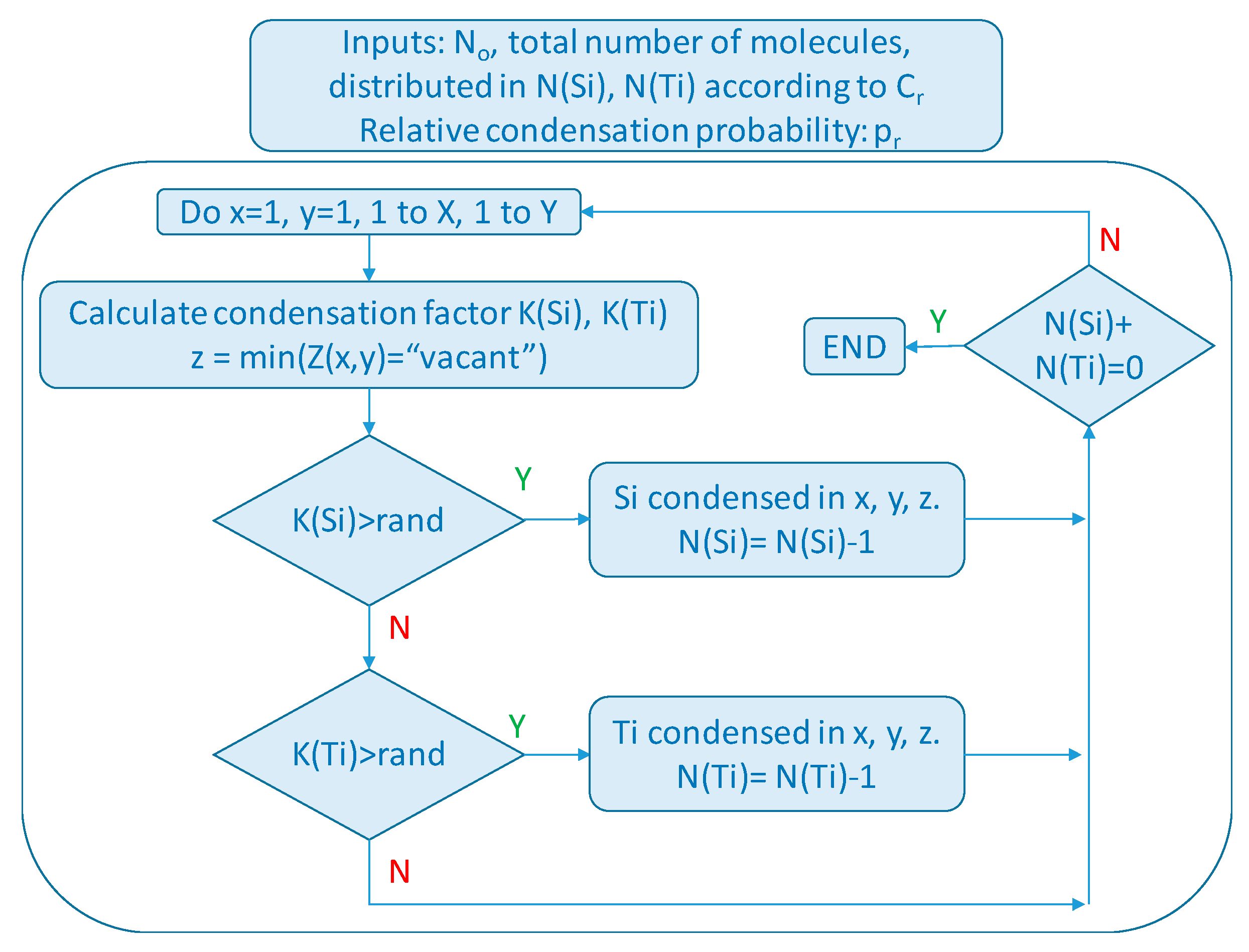
2.1. Simulations and Background
2.2. Preparation of Organosilanized Titanate Films
2.3. Surface and Interface Characterization
3. Results and Discussion
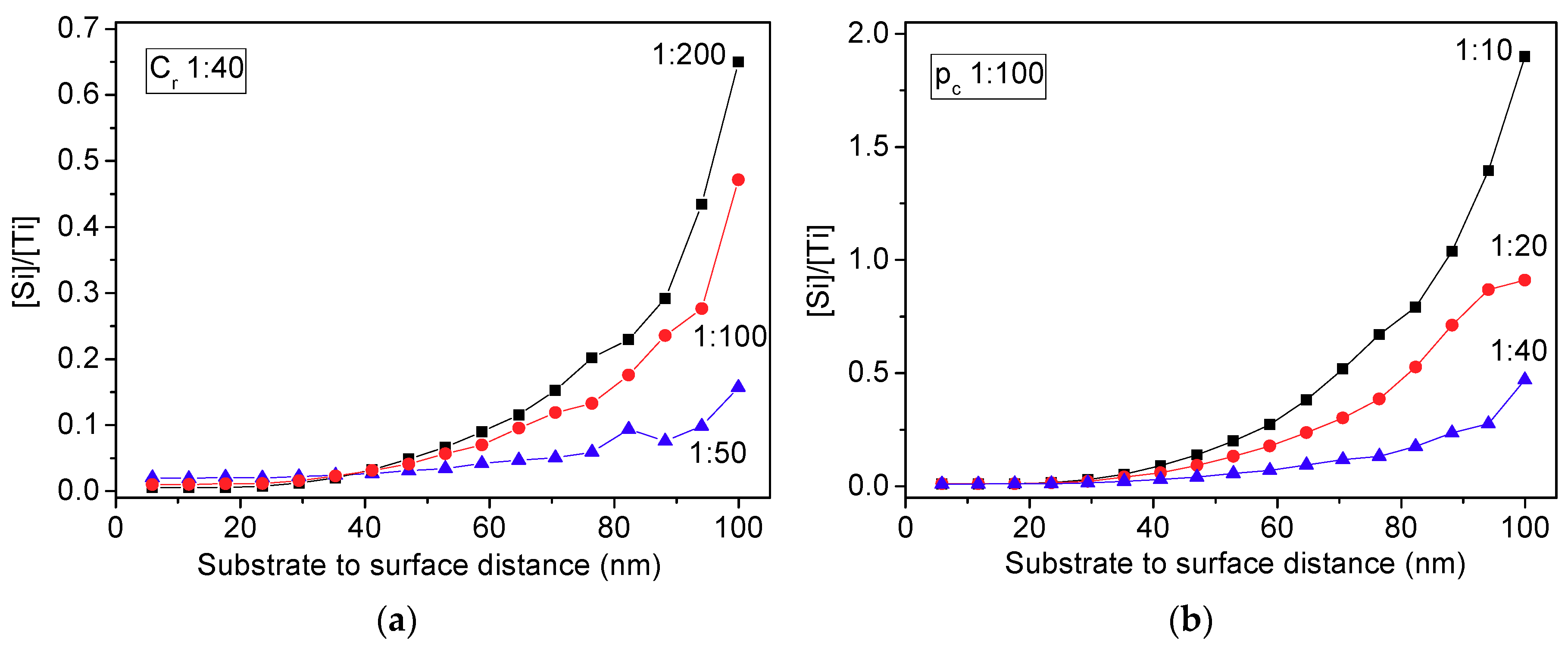
3.1. Montecarlo Simulations
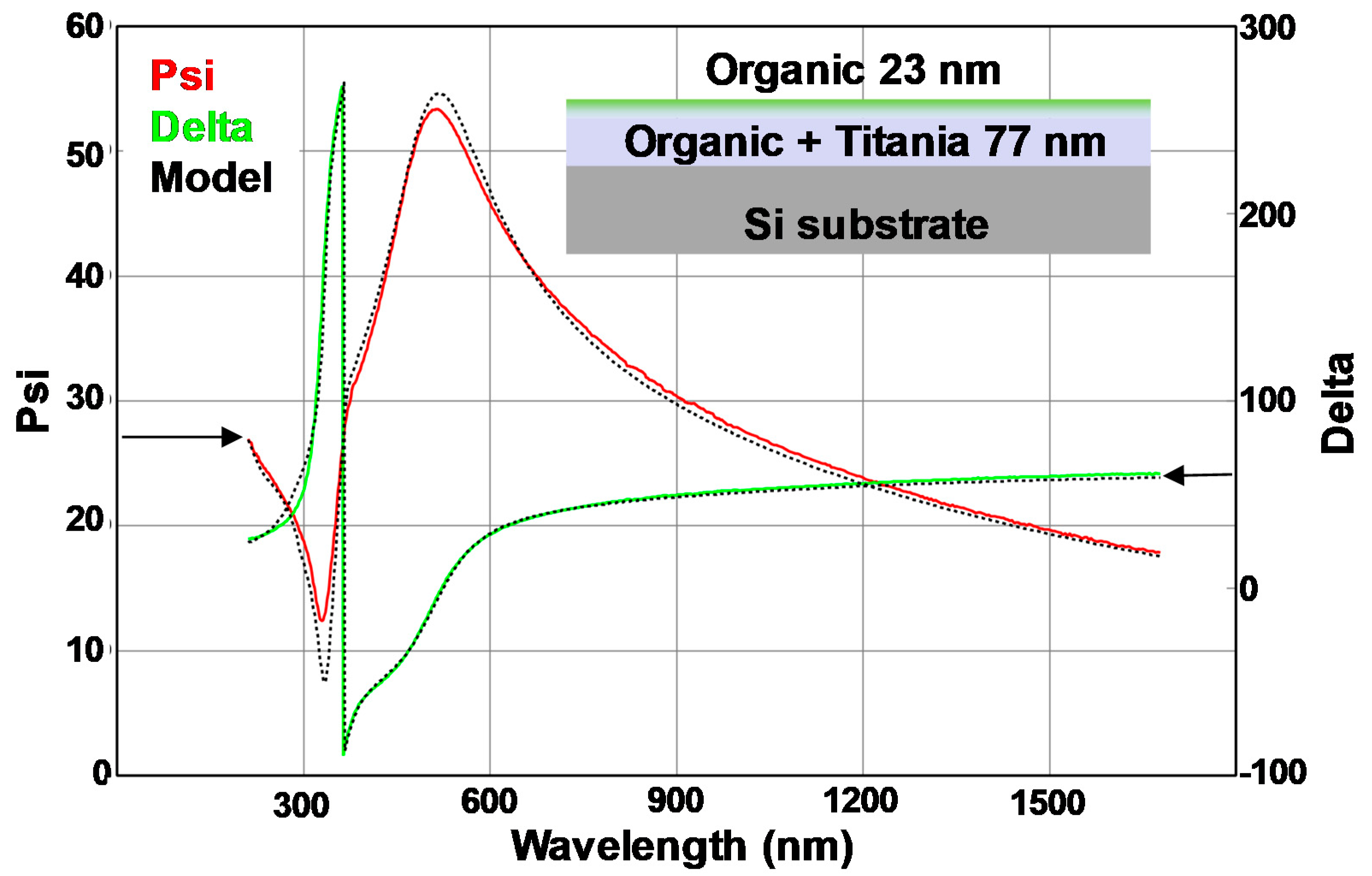
3.2. Optical Analysis
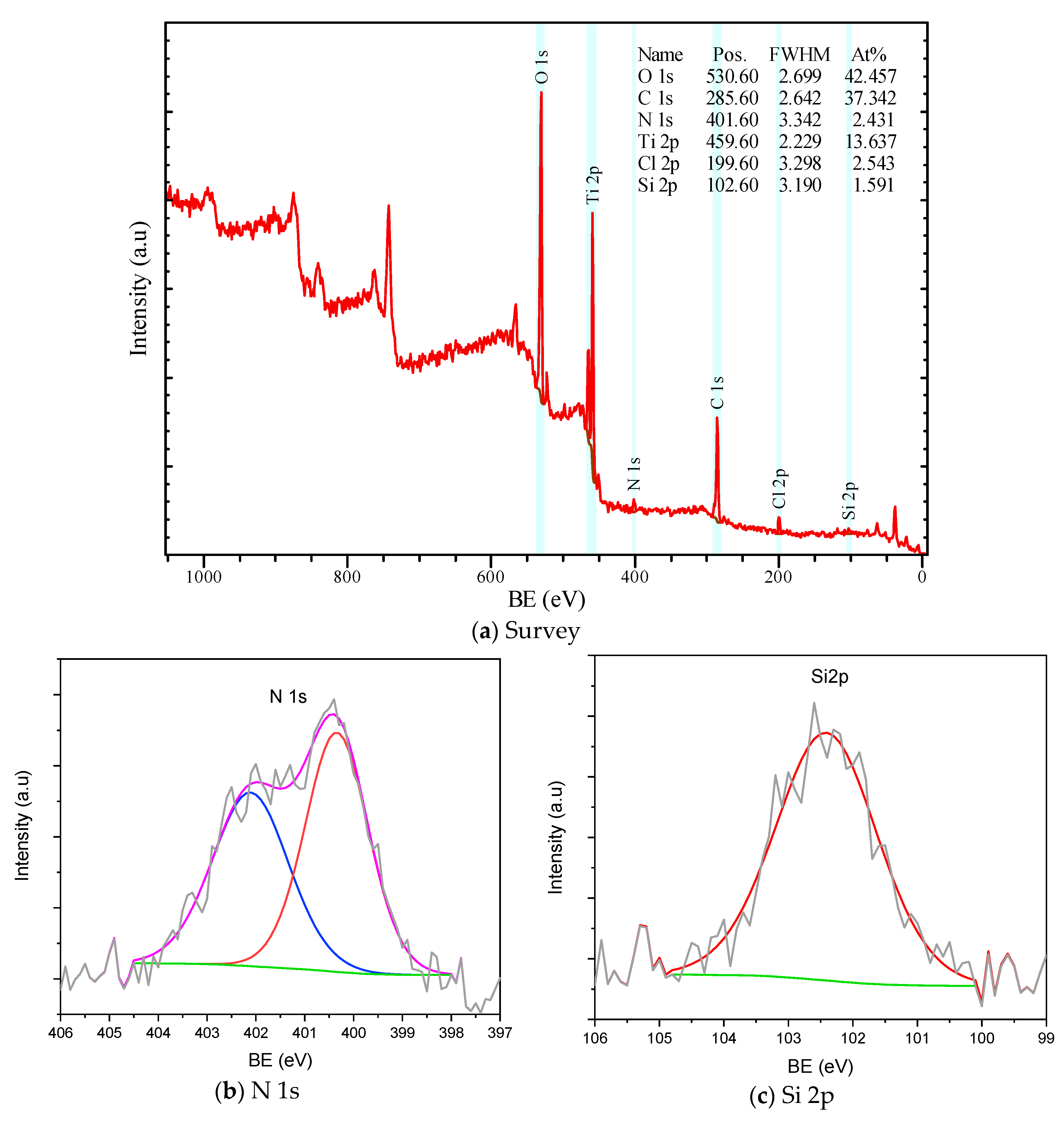
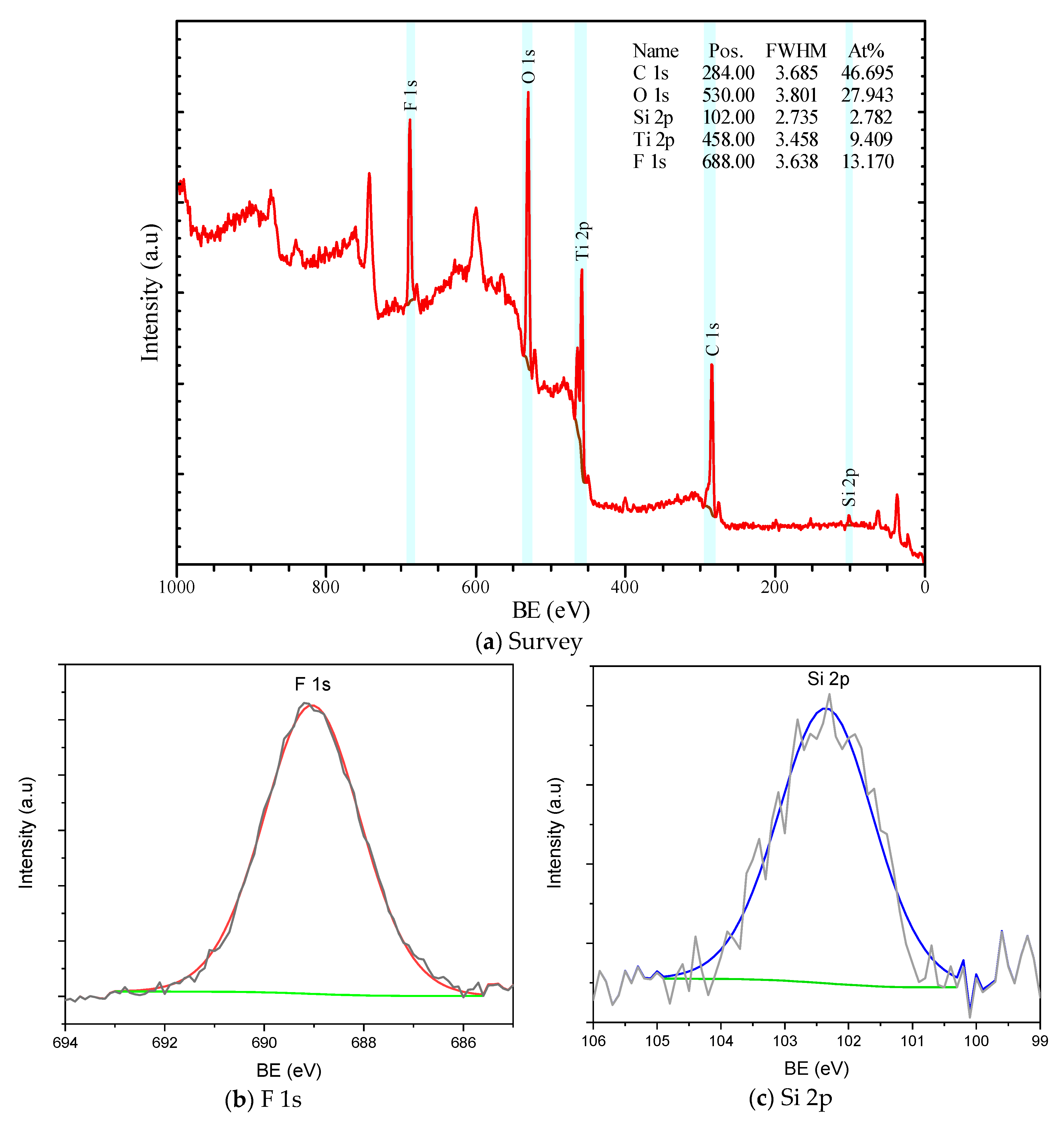
3.3. XPS Analysis
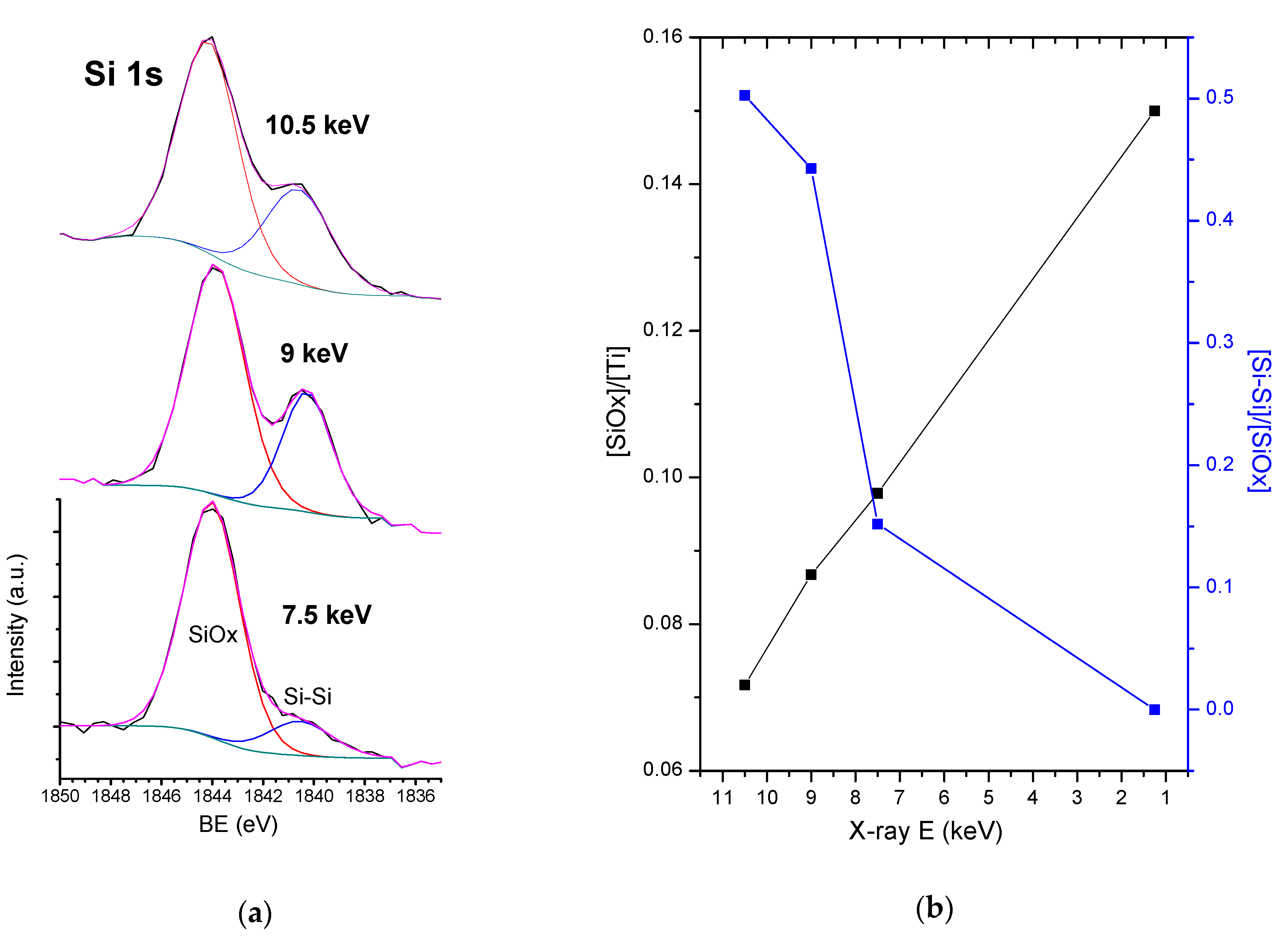
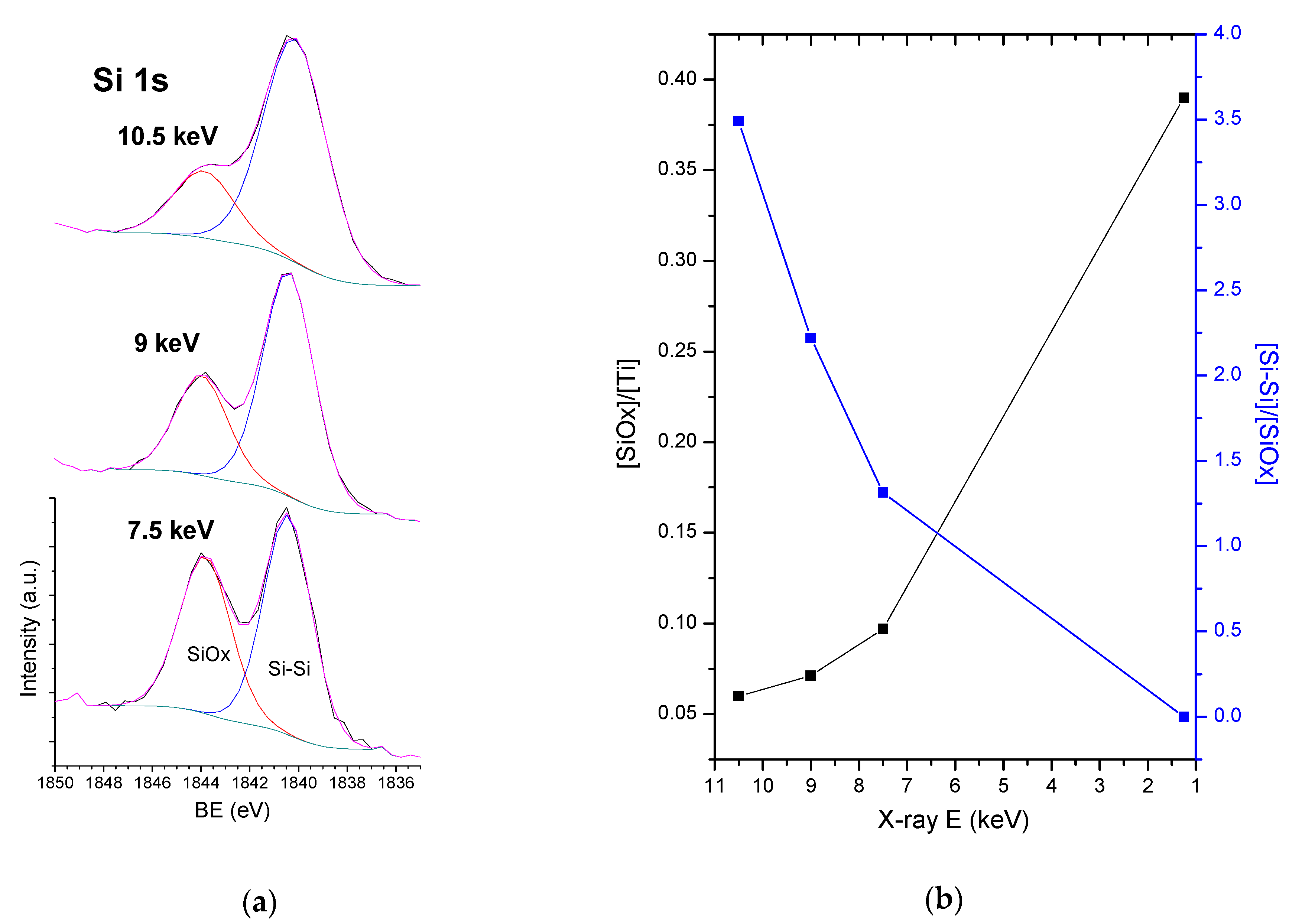
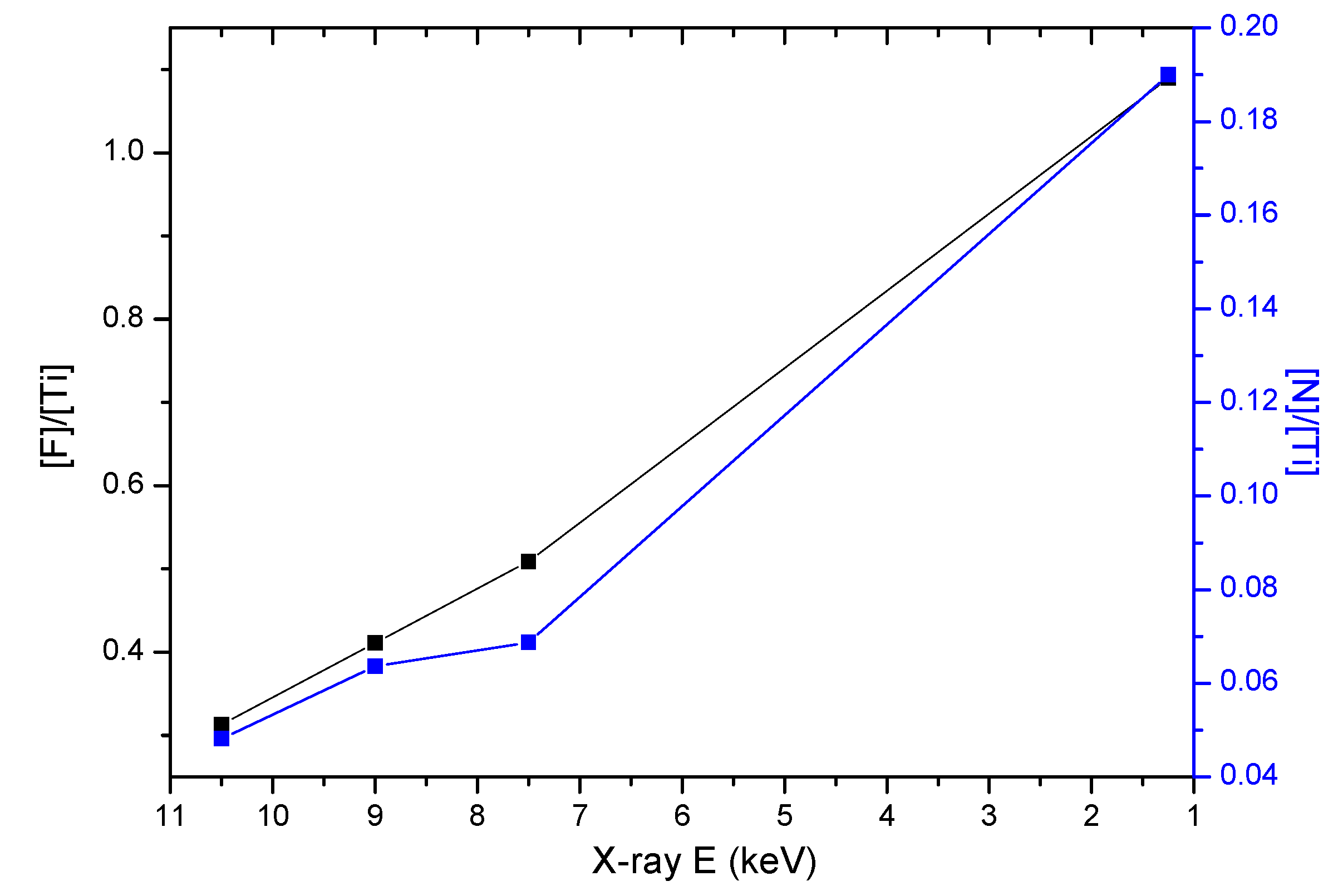
3.4. HAXPES Analysis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Aksay, I.A. Molecular and colloidal engineering of ceramics. Ceram. Int. 1991, 17, 267–274. [Google Scholar] [CrossRef]
- Sakka, S. The current state of sol-gel technology. J. Sol Gel Sci. Technol. 1994, 3, 69–81. [Google Scholar] [CrossRef]
- Messing, G.L.; Minehan, W.T. Synthesis of Ceramic Powders from Metal Alkoxides. J. Ceram. Soc. Jpn. 1991, 99, 1036–1046. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Ishizaka, T.; Kurokawa, Y. Preparation of alumina films by the sol-gel method. J. Mater. Sci. 2005, 40, 263–283. [Google Scholar] [CrossRef]
- Ramadan, R.; Simiz, J.G.; Ynsa, M.D.; Silvan, M.M. Microwave plasma annealing of sol-gel deposited tantalum oxide and zinc oxide films. Vacuum 2018, 149, 336–342. [Google Scholar] [CrossRef]
- Sanchez, C.; Soler-Illia, G.; Ribot, F.; Grosso, D. Design of functional nano-structured materials through the use of controlled hybrid organic-inorganic interfaces. Comptes Rendus Chim. 2003, 6, 1131–1151. [Google Scholar] [CrossRef]
- Niederberger, M.; Garnweitner, G. Organic Reaction Pathways in the Nonaqueous Synthesis of Metal Oxide Nanoparticles. Chem. A Eur. J. 2006, 12, 7282–7302. [Google Scholar] [CrossRef]
- Parashar, M.; Shukla, V.K.; Singh, R. Metal oxides nanoparticles via sol-gel method: A review on synthesis, characterization and applications. J. Mater. Sci. Mater. Electron. 2020, 31, 3729–3749. [Google Scholar] [CrossRef]
- Sakka, S.; Yoko, T. Fibers from gels. J. Non Cryst. Solids 1992, 147, 394–403. [Google Scholar] [CrossRef]
- Kirkbir, F.; Murata, H.; Meyers, D.; Chaudhuri, S.R.; Sarkar, A. Drying and sintering of sol-gel derived large SiO2 monoliths. J. Sol Gel Sci. Technol. 1996, 6, 203–217. [Google Scholar] [CrossRef]
- Barton, T.J.; Bull, L.M.; Klemperer, W.G.; Loy, D.A.; McEnaney, B.; Misono, M.; Monson, P.A.; Pez, G.; Scherer, G.W.; Vartuli, J.C.; et al. Tailored Porous Materials. Chem. Mater. 1999, 11, 2633–2656. [Google Scholar] [CrossRef]
- Brinker, C.J.; Hurd, A.J.; Schunk, P.R.; Frye, G.C.; Ashley, C.S. Review of sol-gel thin-film formation. J. Non Cryst. Solids 1992, 147, 424–436. [Google Scholar] [CrossRef]
- Kim, S.J.; Yoon, S.; Kim, H.J. Review of solution-processed oxide thin-film transistors. Jpn. J. Appl. Phys. 2014, 53, 10. [Google Scholar] [CrossRef]
- Manso-Silvan, A.; Fuentes-Cobas, L.; Martin-Palma, R.J.; Hernandez-Velez, M.; Martinez-Duart, J.M. BaTiO3 thin films obtained by sol-gel spin coating. Surf. Coat. Technol. 2002, 151, 118–121. [Google Scholar] [CrossRef]
- Russellfloyd, R.S.; Harris, B.; Jones, R.W.; Cooke, R.G.; Wang, T.H.; Laurie, J.; Hammett, F.W. Sol-gel processing of fiber-reinforced ceramic shapes. Br. Ceram. Trans. 1993, 92, 8–12. [Google Scholar]
- Langlet, M.; Jenouvrier, P.; Kim, A.; Manso, M.; Valdez, M.T. Functionality of aerosol-gel deposited TiO2 thin films processed at low temperature. J. Sol Gel Sci. Technol. 2003, 26, 759–763. [Google Scholar] [CrossRef]
- Kinadjian, N.; Depardieu, M.; Hillard, E.A.; Backov, R. Extrusion-based Integrative Chemistry: Generation and applications of inorganic fibers. Comptes Rendus Chim. 2016, 19, 674–683. [Google Scholar] [CrossRef]
- Rivero, P.J.; Urrutia, A.; Goicoechea, J.; Arregui, F.J. Nanomaterials for Functional Textiles and Fibers. Nanoscale Res. Lett. 2015, 10, 22. [Google Scholar] [CrossRef]
- Levy, D. Sol-gel glasses for optics and electro-optics. J. Non Cryst. Solids 1992, 147, 508–517. [Google Scholar] [CrossRef]
- Reisfeld, R.; Saraidarov, T. Innovative materials based on sol-gel technology. Opt. Mater. 2006, 28, 64–70. [Google Scholar] [CrossRef]
- Hodgson, S.N.B.; Weng, L. Chemical and sol-gel processing of tellurite glasses for optoelectronics. J. Mater. Sci. Mater. Electron. 2006, 17, 723–733. [Google Scholar] [CrossRef]
- Muller-Buschbaum, P.; Thelakkat, M.; Fassler, T.F.; Stutzmann, M. Hybrid Photovoltaics-from Fundamentals towards Application. Adv. Energy Mater. 2017, 7, 1700248. [Google Scholar] [CrossRef]
- Duran, A.; Castro, Y.; Aparicio, M.; Conde, A.; de Damborenea, J.J. Protection and surface modification of metals with sol-gel coatings. Int. Mater. Rev. 2007, 52, 175–192. [Google Scholar] [CrossRef]
- Schottner, G. Hybrid sol-gel-derived polymers: Applications of multifunctional materials. Chem. Mater. 2001, 13, 3422–3435. [Google Scholar] [CrossRef]
- Ward, D.A.; Ho, E.I. Preparing catalytic materials by the sol-gel method. Ind. Eng. Chem. Res. 1995, 34, 421–433. [Google Scholar] [CrossRef]
- Burns, A.; Ow, H.; Wiesner, U. Fluorescent core-shell silica nanoparticles: Towards “Lab on a Particle” architectures for nanobiotechnology. Chem. Soc. Rev. 2006, 35, 1028–1042. [Google Scholar] [CrossRef]
- Manso, M.; Langlet, M.; Fernandez, M.; Vazquez, L.; Martinez-Duart, J.M. Surface and interface analysis of hydroxyapatite/TiO2 biocompatible structures. Mater. Sci. Eng. C-Biomim. Supramol. Syst. 2003, 23, 451–454. [Google Scholar] [CrossRef]
- Metroke, T.L.; Parkhill, R.L.; Knobbe, E.T. Passivation of metal alloys using sol-gel-derived materials-a review. Prog. Org. Coat. 2001, 41, 233–238. [Google Scholar] [CrossRef]
- Yan, W.F.; Mahurin, S.M.; Overbury, S.H.; Dai, S. Nanoengineering catalyst supports via layer-by-layer surface functionalization. Top. Catal. 2006, 39, 199–212. [Google Scholar] [CrossRef]
- Baldan, A. Adhesively-bonded joints and repairs in metallic alloys, polymers and composite materials: Adhesives, adhesion theories and surface pretreatment. J. Mater. Sci. 2004, 39, 1–49. [Google Scholar] [CrossRef]
- Wong, J.Y.; Leach, J.B.; Brown, X.Q. Balance of chemistry, topography, and mechanics at the cell-biomaterial interface: Issues and challenges for assessing the role of substrate mechanics on cell response. Surf. Sci. 2004, 570, 119–133. [Google Scholar] [CrossRef]
- Mrksich, M. A surface chemistry approach to studying cell adhesion. Chem. Soc. Rev. 2000, 29, 267–273. [Google Scholar] [CrossRef]
- Sanchez-Vaquero, V.; Satriano, C.; Tejera-Sanchez, N.; Mendez, L.G.; Ruiz, J.P.G.; Silvan, M.M. Characterization and cytocompatibility of hybrid aminosilane-agarose hydrogel scaffolds. Biointerphases 2010, 5, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Castner, D.G.; Ratner, B.D. Biomedical surface science: Foundations to frontiers. Surf. Sci. 2002, 500, 28–60. [Google Scholar] [CrossRef]
- Silvan, M.M.; Messina, G.M.L.; Montero, I.; Satriano, C.; Ruiz, J.P.G.; Marletta, G. Aminofunctionalization and sub-micrometer patterning on silicon through silane doped agarose hydrogels. J. Mater. Chem. 2009, 19, 5226–5233. [Google Scholar] [CrossRef]
- Zreiqat, H.; Evans, P.; Howlett, C.R. Effect of surface chemical modification of bioceramic on phenotype of human bone-derived cells. J. Biomed. Mater. Res. 1999, 44, 389–396. [Google Scholar] [CrossRef]
- Massia, S.P.; Holecko, M.M.; Ehteshami, G.R. In vitro assessment of bioactive coatings for neural implant applications. J. Biomed. Mater. Res. Part A 2004, 68, 177–186. [Google Scholar] [CrossRef]
- Arroyo-Hernandez, M.; Manso-Silvan, M.; Lopez-Elvira, E.; Munoz, A.; Climent, A.; Martinez Duart, J.M. One step processing of aminofunctionalized gate oxides. Biosens. Bioelectron. 2007, 22, 2786–2789. [Google Scholar] [CrossRef]
- Lata, S.; Piehler, J. Stable and functional immobilization of histidine-tagged proteins via multivalent chelator headgroups on a molecular poly(ethylene glycol) brush. Anal. Chem. 2005, 77, 1096–1105. [Google Scholar] [CrossRef]
- Zhang, Z.L.; Crozatier, C.; Le Berre, M.; Chen, Y. In situ bio-functionalization and cell adhesion in microfluidic devices. Microelectron. Eng. 2005, 78–79, 556–562. [Google Scholar] [CrossRef]
- Ahmed, A.; Bonner, C.; Desai, T.A. Bioadhesive Microde vices for Drug Delivery: A Feasibility Study. Biomed. Microdevices 2001, 3, 89–95. [Google Scholar] [CrossRef]
- Feng, S.S.; Mu, L.; Chen, B.H.; Pack, D. Polymeric nanospheres fabricated with natural emulsifiers for clinical administration of an anticancer drug paclitaxel (Taxol((R))). Mater. Sci. Eng. C-Biomim. Supramol. Syst. 2002, 20, 85–92. [Google Scholar] [CrossRef]
- Ndlovu, T.M.; Ward, A.C.; Glassey, J.; Eskildsen, J.; Akay, G. Bioprocess intensification of antibiotic production by Streptomyces coelicolor A3(2) in micro-porous culture. Mater. Sci. Eng. C-Mater. Biol. Appl. 2015, 49, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Luo, Y.F.; Xie, Z.; Xing, J.; Lin, M.P.; Yang, L.; Wang, Y.L.; Huang, K. The optimal combination of substrate chemistry with physiological fluid shear stress. Colloids Surf. B-Biointerfaces 2013, 112, 51–60. [Google Scholar] [CrossRef]
- Maximiano, P.; Duraes, L.; Simoes, P. Overview of Multiscale Molecular Modeling and Simulation of Silica Aerogels. Ind. Eng. Chem. Res. 2019, 58, 18905–18929. [Google Scholar] [CrossRef]
- Gelb, L.D. Modeling Amorphous Porous Materials and Confined Fluids. Mrs Bull. 2009, 34, 592–601. [Google Scholar] [CrossRef]
- Garofalini, S.H.; Martin, G. Molecular simulations of the polymerization of silicic-acid molecules and network formation. J. Phys. Chem. 1994, 98, 1311–1316. [Google Scholar] [CrossRef]
- Morales-Florez, V.; De La Rosa-Fox, N.; Pinero, M.; Esquivias, L. The cluster model: A simulation of the aerogel structure as a hierarchically-ordered arrangement of randomly packed spheres. J. Sol Gel Sci. Technol. 2005, 35, 203–210. [Google Scholar] [CrossRef]
- Tilocca, A. Models of structure, dynamics and reactivity of bioglasses: A review. J. Mater. Chem. 2010, 20, 6848–6858. [Google Scholar] [CrossRef]
- Azenha, M.; Szefczyk, B.; Loureiro, D.; Kathirvel, P.; Cordeiro, M.; Fernando-Silva, A. Computational and Experimental Study of the Effect of PEG in the Preparation of Damascenone-Imprinted Xerogels. Langmuir 2013, 29, 2024–2032. [Google Scholar] [CrossRef]
- Schumacher, C.; Gonzalez, J.; Wright, P.A.; Seaton, N.A. Generation of atomistic models of periodic mesoporous silica by kinetic Monte Carlo simulation of the synthesis of the material. J. Phys. Chem. B 2006, 110, 319–333. [Google Scholar] [CrossRef] [PubMed]
- Majd, M.T.; Bahlakeh, G.; Dehghani, A.; Ramezanzadeh, B.; Ramezanzadeh, M. A green complex film based on the extract of Persian Echium amoenum and zinc nitrate for mild steel protection in saline solution; Electrochemical and surface explorations besides dynamic simulation. J. Mol. Liq. 2019, 291. [Google Scholar] [CrossRef]
- Magdaleno, A.; Gordillo, N.; Pau, J.L.; Manso Silvan, M. A fibrinogen biosensing platform based on plasmonic Ga nanoparticles and aminosilane-titanate antibody trapping. Med. Devices Sens. 2020, 00, 10083. [Google Scholar] [CrossRef]
- Zhang, T.; Tian, B.Z.; Kong, J.L.; Yang, P.Y.; Liu, B.H. A sensitive mediator-free tyrosinase biosensor based on an inorganic-organic hybrid titania sol-gel matrix. Anal. Chim. Acta 2003, 489, 199–206. [Google Scholar] [CrossRef]
- Manso-Silvan, M.; Valsesia, A.; Hasiwa, M.; Rodriguez-Navas, C.; Gilliland, D.; Ceccone, G.; Garcia Ruiz, J.P.; Rossi, F. Micro-spot, UV and wetting patterning pathways for applications of biofunctional aminosilane-titanate coatings. Biomed. Microdevices 2007, 9, 287–294. [Google Scholar] [CrossRef]
- Moreno-Cerrada, D.; Rodriguez, C.; Moreno-Madrid, F.; Selivanovitch, E.; Douglas, T.; de Pablo, P.J.; Manso Silvan, M. Loading the dice: The orientation of virus-like particles adsorbed on titanate assisted organosilanized surfaces. Biointerphases 2019, 14. [Google Scholar] [CrossRef]
- Manso-Silvan, M.; Rodriguez-Navas, C.; Hernandez, M.A.; Lopez-Elvira, E.; Gago, R.; Vazquez, L.; Agullo-Rueda, F.; Climent, A.; Martinez-Duart, J.M.; Garcia-Ruiz, J.P. Hybrid titania-aminosilane platforms evaluated with human mesenchymal stem cells. J. Biomed. Mater. Res. Part B-Appl. Biomater. 2007, 83B, 232–239. [Google Scholar] [CrossRef]
- Benito, N.; Galindo, R.E.; Rubio-Zuazo, J.; Castro, G.R.; Palacio, C. High-and low-energy x-ray photoelectron techniques for compositional depth profiles: Destructive versus non-destructive methods. J. Phys. D Appl. Phys. 2013, 46. [Google Scholar] [CrossRef]
- Rubio-Zuazo, J.R.; Castro, G.R. Hard X-ray photoelectron spectroscopy (HAXPES) (<= 15 keV) at SpLine, the Spanish CRG beamline at the ESRF. Nucl. Instrum. Methods Phys. Res. Sect. A Accel. Spectrometers Detect. Assoc. Equip. 2005, 547, 64–72. [Google Scholar] [CrossRef]
- Bartlett, J.R.; Woolfrey, J.L. Preparation of multicomponent powders by alkoxide hydrolysis 1. Chemical processing. Chem. Mater. 1996, 8, 1167–1174. [Google Scholar] [CrossRef]
- Livage, J.; Henry, M.; Sanchez, C. Sol-gel chemistry of transition-metal oxides. Prog. Solid State Chem. 1988, 18, 259–341. [Google Scholar] [CrossRef]
- Rubio-Zuazo, J.; Escher, M.; Merkel, M.; Castro, G.R. High Voltage-Cylinder Sector Analyzer 300/15: A cylindrical sector analyzer for electron kinetic energies up to 15 keV. Rev. Sci. Instrum. 2010, 81. [Google Scholar] [CrossRef] [PubMed]
- Kitayama, S.; Fujiwara, H.; Gloskovsky, A.; Gorgoi, M.; Schaefers, F.; Felser, C.; Funabashi, G.; Yamaguchi, J.; Kimura, M.; Kuwahara, G.; et al. Photon Energy dependent Hard X-Ray Photoemission Spectroscopy of YbCu2Si2. J. Phys. Soc. Jpn. 2012, 81, 4. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mateo Moreno, J.; Calvo Membibre, R.; Pinilla Yanguas, S.; Rubio Zuazo, J.; Manso Siván, M. Montecarlo Simulation and HAXPES Analysis of Organosilane Segregation in Titania Xerogel Films; Towards a Generic Surface Chemofunctionalization Process. Surfaces 2020, 3, 352-365. https://doi.org/10.3390/surfaces3030026
Mateo Moreno J, Calvo Membibre R, Pinilla Yanguas S, Rubio Zuazo J, Manso Siván M. Montecarlo Simulation and HAXPES Analysis of Organosilane Segregation in Titania Xerogel Films; Towards a Generic Surface Chemofunctionalization Process. Surfaces. 2020; 3(3):352-365. https://doi.org/10.3390/surfaces3030026
Chicago/Turabian StyleMateo Moreno, Javier, Rodrigo Calvo Membibre, Sergio Pinilla Yanguas, Juan Rubio Zuazo, and Miguel Manso Siván. 2020. "Montecarlo Simulation and HAXPES Analysis of Organosilane Segregation in Titania Xerogel Films; Towards a Generic Surface Chemofunctionalization Process" Surfaces 3, no. 3: 352-365. https://doi.org/10.3390/surfaces3030026
APA StyleMateo Moreno, J., Calvo Membibre, R., Pinilla Yanguas, S., Rubio Zuazo, J., & Manso Siván, M. (2020). Montecarlo Simulation and HAXPES Analysis of Organosilane Segregation in Titania Xerogel Films; Towards a Generic Surface Chemofunctionalization Process. Surfaces, 3(3), 352-365. https://doi.org/10.3390/surfaces3030026






