Flood Pulse Irrigation of Meadows Shapes Soil Chemical and Microbial Parameters More Than Mineral Fertilization
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Agricultural Practice
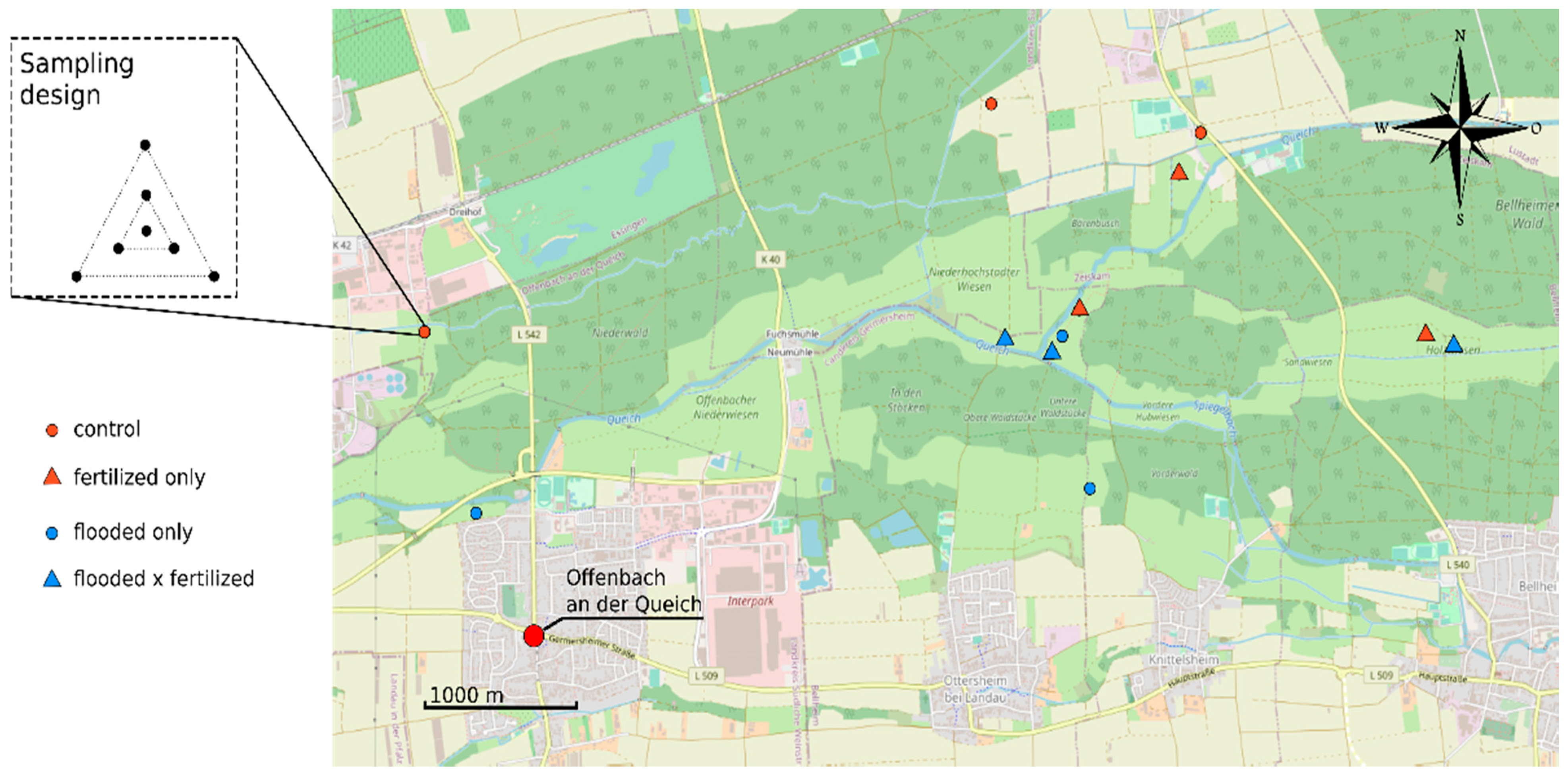
2.2. Experimental Design and Soil Characterization
2.3. Soil Chemical Parameters and Texture
2.4. PLFA Analysis
2.5. Data Analysis
3. Results
3.1. Soil Chemical Properties and Texture
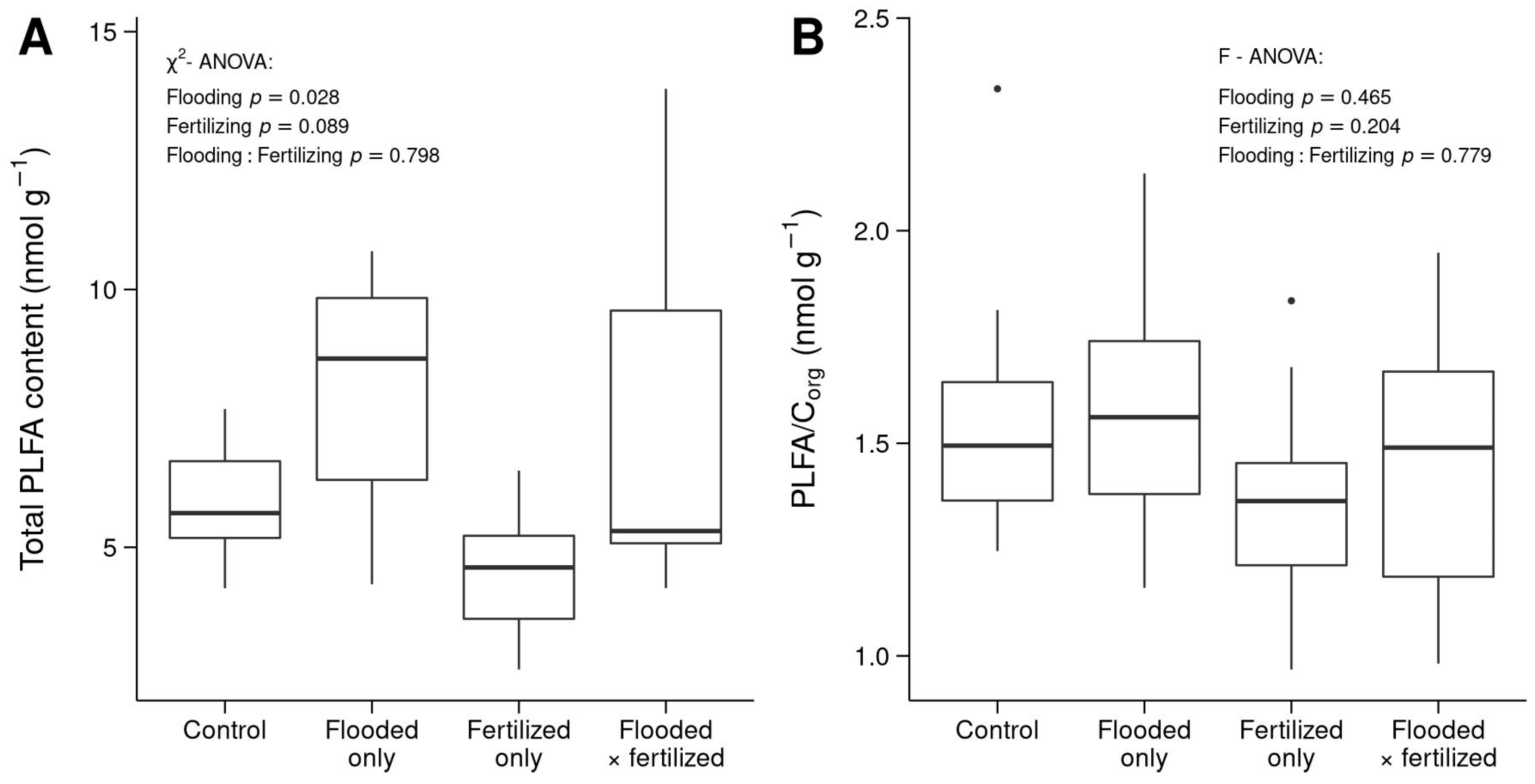
3.2. Total PLFA Content
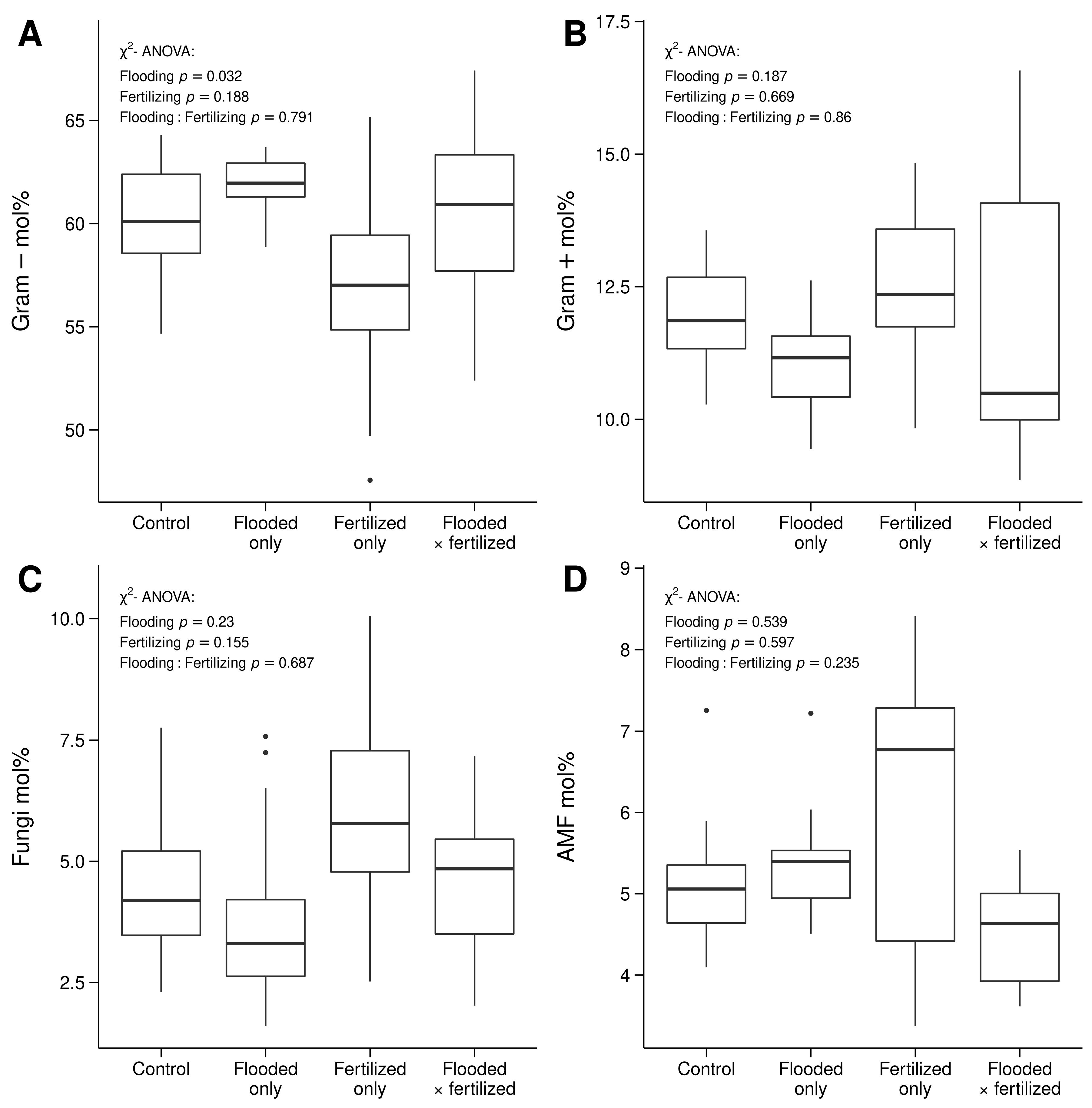
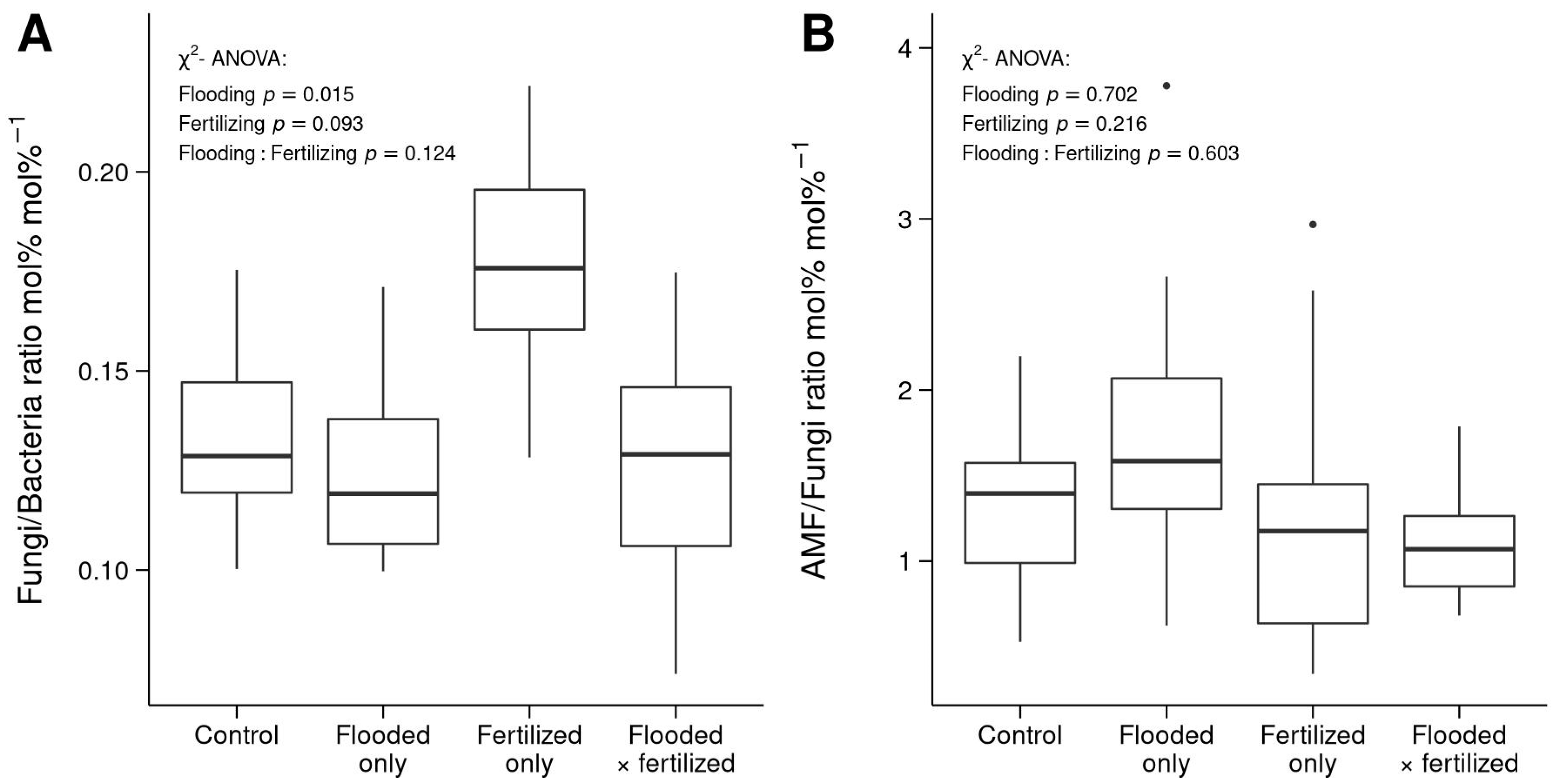
3.3. Differences in Microbial Community Structure
3.4. Effect of Soil Chemistry on PLFA Biomarkers
4. Discussion
4.1. Soil Chemical Parameter under the Influence of Irrigation and Fertilization
4.2. Interactions between Microbial Biomass and Corg
4.3. Differences in Community Structures
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Leibundgut, C.; Kohn, I. European traditional irrigation in transition part I: Irrigation in times past—A historic land use practice across Europe. Irrig. Drain. 2014, 63, 273–293. [Google Scholar] [CrossRef]
- Buhk, C.; Schirmel, J.; Rebekka, G.; Frör, O. Traditional water meadows: A sustainable management type for the future? In Irrigation in Agroecosystems; IntechOpen: London, UK, 2019. [Google Scholar]
- Trost, B.; Ellmer, F.; Baumecker, M.; Meyer-Aurich, A.; Prochnow, A.; Drastig, K. Effects of irrigation and nitrogen fertilizer on yield, carbon inputs from above ground harvest residues and soil organic carbon contents of a sandy soil in Germany. Soil Use Manag. 2014, 30, 209–218. [Google Scholar] [CrossRef]
- Trost, B.; Prochnow, A.; Drastig, K.; Meyer-Aurich, A.; Ellmer, F.; Baumecker, M. Irrigation, soil organic carbon and N2O emissions. A review. Agron. Sustain. Dev. 2013, 33, 733–749. [Google Scholar] [CrossRef]
- Sun, S.; Che, T.; Gentine, P.; Chen, Q.; Wang, L.; Yan, Z.; Chen, B.; Song, Z. Shallow groundwater inhibits soil respiration and favors carbon uptake in a wet alpine meadow ecosystem. Agric. For. Meteorol. 2021, 297, 108254. [Google Scholar] [CrossRef]
- Austin, A.T.; Yahdjian, L.; Stark, J.M.; Belnap, J.; Porporato, A.; Norton, U.; Ravetta, D.A.; Schaeffer, S.M. Water pulses and biogeochemical cycles in arid and semiarid ecosystems. Oecologia 2004, 141, 221–235. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Huang, Y.; Qiu, Q.; Xin, G.; Yang, Z.; Shi, S. Flooding greatly affects the diversity of arbuscular mycorrhizal fungi communities in the roots of wetland plants. PLoS ONE 2011, 6, e24512. [Google Scholar] [CrossRef]
- Bossio, D.A.; Scow, K.M. Impacts of carbon and flooding on soil microbial communities: Phospholipid fatty acid profiles and substrate utilization patterns. Microb. Ecol. 1998, 35, 265–278. [Google Scholar] [CrossRef]
- De-Campos, A.B.; Huang, C.; Johnston, C.T. Biogeochemistry of terrestrial soils as influenced by short-term flooding. Biogeochemistry 2012, 111, 239–252. [Google Scholar] [CrossRef]
- Drenovsky, R.E.; Steenwerth, K.L.; Jackson, L.E.; Scow, K.M. Land use and climatic factors structure regional patterns in soil microbial communities. Glob. Ecol. Biogeogr. 2010, 19, 27–39. [Google Scholar] [CrossRef]
- Siebielec, S.; Siebielec, G.; Klimkowicz-Pawlas, A.; Gałązka, A.; Grządziel, J.; Stuczyński, T. Impact of water stress on microbial community and activity in sandy and loamy soils. Agronomy 2020, 10, 1429. [Google Scholar] [CrossRef]
- Hawkes, C.V.; Kivlin, S.N.; Rocca, J.D.; Huguet, V.; Thomsen, M.A.; Suttle, K.B. Fungal community responses to precipitation. Glob. Chang. Biol. 2011, 17, 1637–1645. [Google Scholar] [CrossRef]
- Millard, P.; Singh, B.K. Does grassland vegetation drive soil microbial diversity? Nutr. Cycl. Agroecosyst. 2010, 88, 147–158. [Google Scholar] [CrossRef]
- Geisseler, D.; Scow, K.M. Long-term effects of mineral fertilizers on soil microorganisms—A review. Soil Biol. Biochem. 2014, 75, 54–63. [Google Scholar] [CrossRef]
- Kaiser, K.; Wemheuer, B.; Korolkow, V.; Wemheuer, F.; Nacke, H.; Schöning, I.; Schrumpf, M.; Daniel, R. Driving forces of soil bacterial community structure, diversity, and function in temperate grasslands and forests. Sci. Rep. 2016, 6, 33696. [Google Scholar] [CrossRef] [PubMed]
- Bradley, K.; Drijber, R.A.; Knops, J. Increased N availability in grassland soils modifies their microbial communities and decreases the abundance of arbuscular mycorrhizal fungi. Soil Biol. Biochem. 2006, 38, 1583–1595. [Google Scholar] [CrossRef]
- Williams, M.A.; Rice, C.W. Seven years of enhanced water availability influences the physiological, structural, and functional attributes of a soil microbial community. Appl. Soil Ecol. 2007, 35, 535–545. [Google Scholar] [CrossRef]
- Drenovsky, R.E.; Vo, D.; Graham, K.J.; Scow, K.M. Soil water content and organic carbon availability are major determinants of soil microbial community composition. Microb. Ecol. 2004, 48, 424–430. [Google Scholar] [CrossRef]
- Fierer, N.; Schimel, J.P.; Holden, P.A. Influence of drying-rewetting frequency on soil bacterial community structure. Microb. Ecol. 2003, 45, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Bapiri, A.; Bååth, E.; Rousk, J. Drying-rewetting cycles affect fungal and bacterial growth differently in an arable soil. Microb. Ecol. 2010, 60, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.S.; Baldwin, D.S.; Rees, G.N.; Wilson, B.P. The effects of short-term inundation on carbon dynamics, microbial community structure and microbial activity in floodplain soil. River Res. Appl. 2011, 27, 213–225. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Physiol. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Zelles, L. Fatty acid patterns of phospholipids and lipopolysaccharides in the characterisation of microbial communities in soil: A review. Biol. Fertil. Soils 1999, 29, 111–129. [Google Scholar] [CrossRef]
- Müller, I.B.; Buhk, C.; Alt, M.; Entling, M.H.; Schirmel, J. Plant functional shifts in Central European grassland under traditional flood irrigation. Appl. Veg. Sci. 2016, 19, 122–131. [Google Scholar] [CrossRef]
- Müller, I.B.; Buhk, C.; Lange, D.; Entling, M.H.; Schirmel, J. Contrasting effects of irrigation and fertilization on plant diversity in hay meadows. Basic Appl. Ecol. 2016, 17, 576–585. [Google Scholar] [CrossRef]
- Bardgett, R.D.; van der Putten, W.H. Belowground biodiversity and ecosystem functioning. Nature 2014, 515, 505. [Google Scholar] [CrossRef] [PubMed]
- Geue, H.; Hock, B. Determination of Acaulospora Longula and Glomus Subgroup Aa in plant roots from grassland using new primers against the large subunit ribosomal DNA. Mycol. Res. 2004, 108, 76–83. [Google Scholar] [CrossRef]
- White, D.C.; Davis, W.M.; Nickels, J.S.; King, J.D.; Bobbie, R.J. Determination of the sedimentary microbial biomass by extractible lipid phosphate. Oecologia 1979, 40, 51–62. [Google Scholar] [CrossRef]
- Butte, W. Rapid method for the determination of fatty acid profiles from fats and oils using trimethylsulphonium hydroxide for transesterification. J. Chromatogr. A 1983, 261, 142–145. [Google Scholar] [CrossRef]
- Gómez-Brandón, M.; Lores, M.; Domínguez, J. A new combination of extraction and derivatization methods that reduces the complexity and preparation time in determining phospholipid fatty acids in solid environmental samples. Bioresour. Technol. 2010, 101, 1348–1354. [Google Scholar] [CrossRef]
- Ratledge, C.; Wilkinson, S.G. Microbial Lipids; Academic Press: London, UK; San Diego, CA, USA, 1988; ISBN 978-0-12-582304-3. [Google Scholar]
- Olsson, P.A.; Bååth, E.; Jakobsen, I.; Söderström, B. The use of phospholipid and neutral lipid fatty acids to estimate biomass of arbuscular mycorrhizal fungi in soil. Mycol. Res. 1995, 99, 623–629. [Google Scholar] [CrossRef]
- Van Aarle, I.M.; Olsson, P.A. Fungal lipid accumulation and development of mycelial structures by two arbuscular mycorrhizal fungi. Appl. Environ. Microbiol. 2003, 69, 6762–6767. [Google Scholar] [CrossRef]
- Frostegård, A.; Bååth, E. The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol. Fertil. Soils 1996, 22, 59–65. [Google Scholar] [CrossRef]
- Kaiser, C.; Frank, A.; Wild, B.; Koranda, M.; Richter, A. Negligible contribution from roots to soil-borne phospholipid fatty acid fungal biomarkers 18:2ω6,9 and 18:1ω9. Soil Biol. Biochem. 2010, 42, 1650–1652. [Google Scholar] [CrossRef] [PubMed]
- R Team. Core. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- Bates, D.; Mächler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using Lme4. J. Stat. Softw. 2015, 67, 48. [Google Scholar] [CrossRef]
- Körschens, M.; Albert, E.; Armbruster, M.; Barkusky, D.; Baumecker, M.; Behle-Schalk, L.; Bischoff, R.; Čergan, Z.; Ellmer, F.; Herbst, F.; et al. Effect of mineral and organic fertilization on crop yield, nitrogen uptake, carbon and nitrogen balances, as well as soil organic carbon content and dynamics: Results from 20 European long-term field experiments of the twenty-first century. Arch. Agron. Soil Sci. 2013, 59, 1017–1040. [Google Scholar] [CrossRef]
- Francioli, D.; Schulz, E.; Lentendu, G.; Wubet, T.; Buscot, F.; Reitz, T. Mineral vs. organic amendments: Microbial community structure, activity and abundance of agriculturally relevant microbes are driven by long-term fertilization strategies. Front. Microbiol. 2016, 7, 1446. [Google Scholar] [CrossRef]
- Menšík, L.; Hlisnikovský, L.; Pospíšilová, L.; Kunzová, E. The effect of application of organic manures and mineral fertilizers on the state of soil organic matter and nutrients in the long-term field experiment. J. Soils Sediments 2018, 1–10. [Google Scholar] [CrossRef]
- Nardi, S.; Morari, F.; Berti, A.; Tosoni, M.; Giardini, L. Soil organic matter properties after 40 years of different use of organic and mineral fertilisers. Eur. J. Agron. 2004, 21, 357–367. [Google Scholar] [CrossRef]
- Zechmeister, H.G.; Schmitzberger, I.; Steurer, B.; Peterseil, J.; Wrbka, T. The influence of land-use practices and economics on plant species richness in meadows. Biol. Conserv. 2003, 114, 165–177. [Google Scholar] [CrossRef]
- Federal Ministry of Food and Agriculture (Germany). German Fertilizer Enactment Verordnung Über Das Inverkehrbringen von Düngemitteln, Bodenhilfsstoffen, Kultursubstraten Und Pflanzenhilfsmitteln. Bundesgesetzblatt 2012, 58, 2482–2544. [Google Scholar]
- Zak, D.R.; Holmes, W.E.; White, D.C.; Peacock, A.D.; Tilman, D. Plant diversity, soil microbial communities, and ecosystem function: Are there any links? Ecology 2003, 84, 2042–2050. [Google Scholar] [CrossRef]
- Lange, M.; Eisenhauer, N.; Sierra, C.A.; Bessler, H.; Engels, C.; Griffiths, R.I.; Mellado-Vázquez, P.G.; Malik, A.A.; Roy, J.; Scheu, S.; et al. Plant diversity increases soil microbial activity and soil carbon storage. Nat. Commun. 2015, 6, 6707. [Google Scholar] [CrossRef] [PubMed]
- Xue, R.; Shen, Y.; Marschner, P. Soil water content during and after plant growth influence nutrient availability and microbial biomass. J. Soil Sci. Plant. Nutr. 2017, 17, 702–715. [Google Scholar] [CrossRef]
- Treseder, K.K. Nitrogen additions and microbial biomass: A meta-analysis of ecosystem studies. Ecol. Lett. 2008, 11, 1111–1120. [Google Scholar] [CrossRef]
- Yao, H.; He, Z.; Wilson, M.J.; Campbell, C.D. Microbial biomass and community structure in a sequence of soils with increasing fertility and changing land use. Microb. Ecol. 2000, 40, 223–237. [Google Scholar] [CrossRef]
- Stagnari, F.; Perpetuini, G.; Tofalo, R.; Campanelli, G.; Leteo, F.; della Vella, U.; Schirone, M.; Suzzi, G.; Pisante, M. Long-term impact of farm management and crops on soil microorganisms assessed by combined DGGE and PLFA analyses. Front. Microbiol. 2014, 5, 644. [Google Scholar] [CrossRef]
- Tian, J.; Dippold, M.; Pausch, J.; Blagodatskaya, E.; Fan, M.; Li, X.; Kuzyakov, Y. Microbial response to rhizodeposition depending on water regimes in paddy soils. Soil Biol. Biochem. 2013, 65, 195–203. [Google Scholar] [CrossRef]
- Bååth, E.; Anderson, T.-H. Comparison of soil fungal/bacterial ratios in a PH gradient using physiological and PLFA-based techniques. Soil Biol. Biochem. 2003, 35, 955–963. [Google Scholar] [CrossRef]
- Egerton-Warburton, L.M.; Johnson, N.C.; Allen, E.B. Mycorrhizal community dynamics following nitrogen fertilization: A cross-site test in five grasslands. Ecol. Monogr. 2007, 77, 527–544. [Google Scholar] [CrossRef]
- Gryndler, M.; Larsen, J.; Hršelová, H.; Řezáčová, V.; Gryndlerová, H.; Kubát, J. Organic and mineral fertilization, respectively, increase and decrease the development of external mycelium of arbuscular mycorrhizal fungi in a long-term field experiment. Mycorrhiza 2006, 16, 159–166. [Google Scholar] [CrossRef]
- Mentzer, J.L.; Goodman, R.M.; Balser, T.C. Microbial response over time to hydrologic and fertilization treatments in a simulated wet prairie. Plant Soil 2006, 284, 85–100. [Google Scholar] [CrossRef]
- Burrows, R.L.; Pfleger, F.L. Arbuscular mycorrhizal fungi respond to increasing plant diversity. Can. J. Bot. 2002, 80, 120–130. [Google Scholar] [CrossRef]
- Morris, E.K.; Buscot, F.; Herbst, C.; Meiners, T.; Obermaier, E.; Wäschke, N.W.; Wubet, T.; Rillig, M.C. Land use and host neighbor identity effects on arbuscular mycorrhizal fungal community composition in focal plant rhizosphere. Biodivers. Conserv. 2013, 22, 2193–2205. [Google Scholar] [CrossRef]
- Chen, Y.-L.; Zhang, X.; Ye, J.-S.; Han, H.-Y.; Wan, S.-Q.; Chen, B.-D. Six-year fertilization modifies the biodiversity of arbuscular mycorrhizal fungi in a temperate steppe in inner mongolia. Soil Biol. Biochem. 2014, 69, 371–381. [Google Scholar] [CrossRef]
- Dumbrell, A.J.; Nelson, M.; Helgason, T.; Dytham, C.; Fitter, A.H. Relative roles of niche and neutral processes in structuring a soil microbial community. ISME J. 2010, 4, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Alt, M.; Buhk, C.; Diehl, D.; Gerlach, R.; Rudolph, I.; Schirmel, J.; Theissinger, K.; Schaumann, G.E. Historical irrigated meadows at the river Queich, Rhineland-Palatinate. In Jahrestagung der DBG 2013: Böden-Lebensgrundlage und Verantwortung; Jahrestagung der DBG: Rostock, Germany, 2013. [Google Scholar]
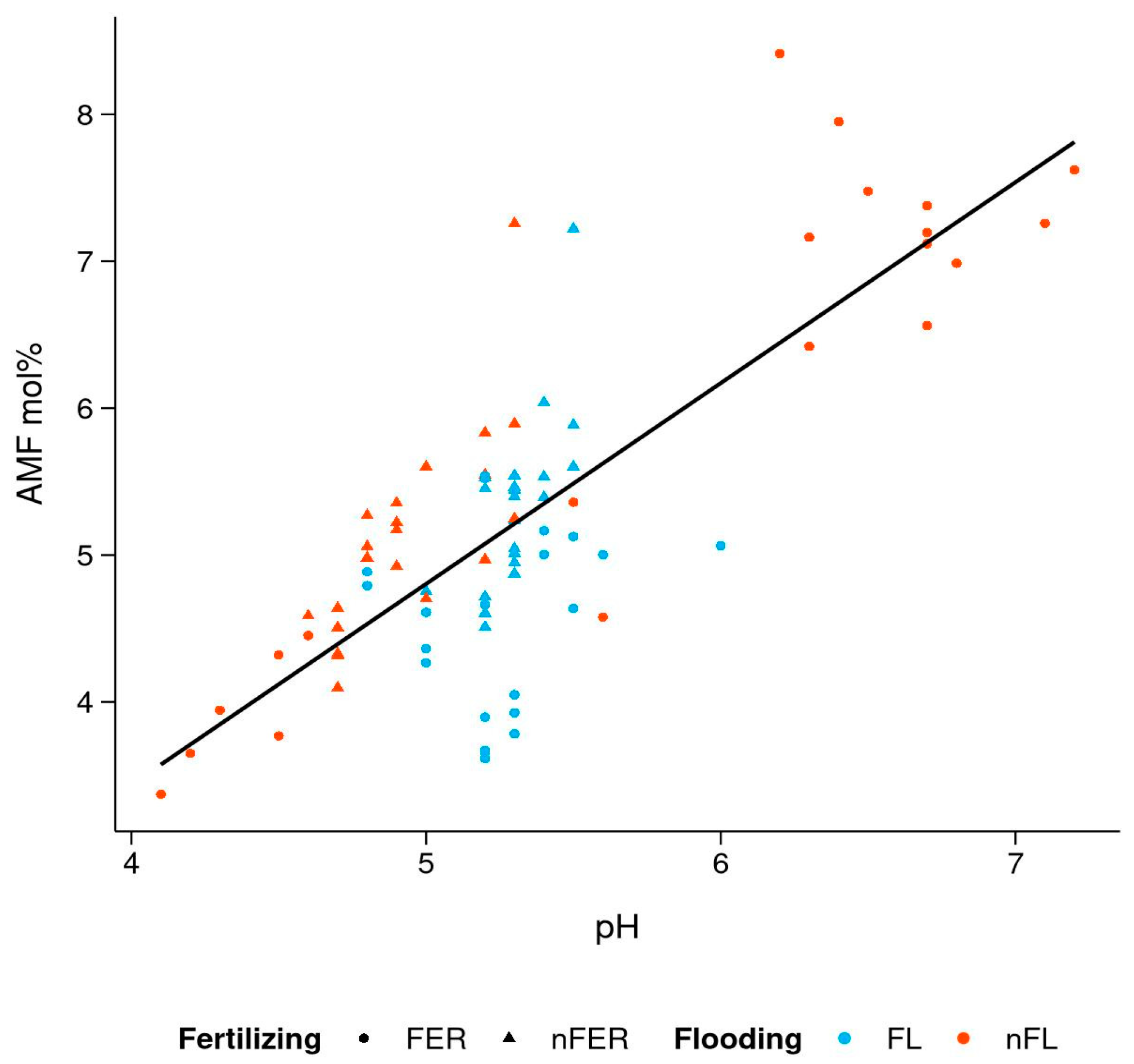
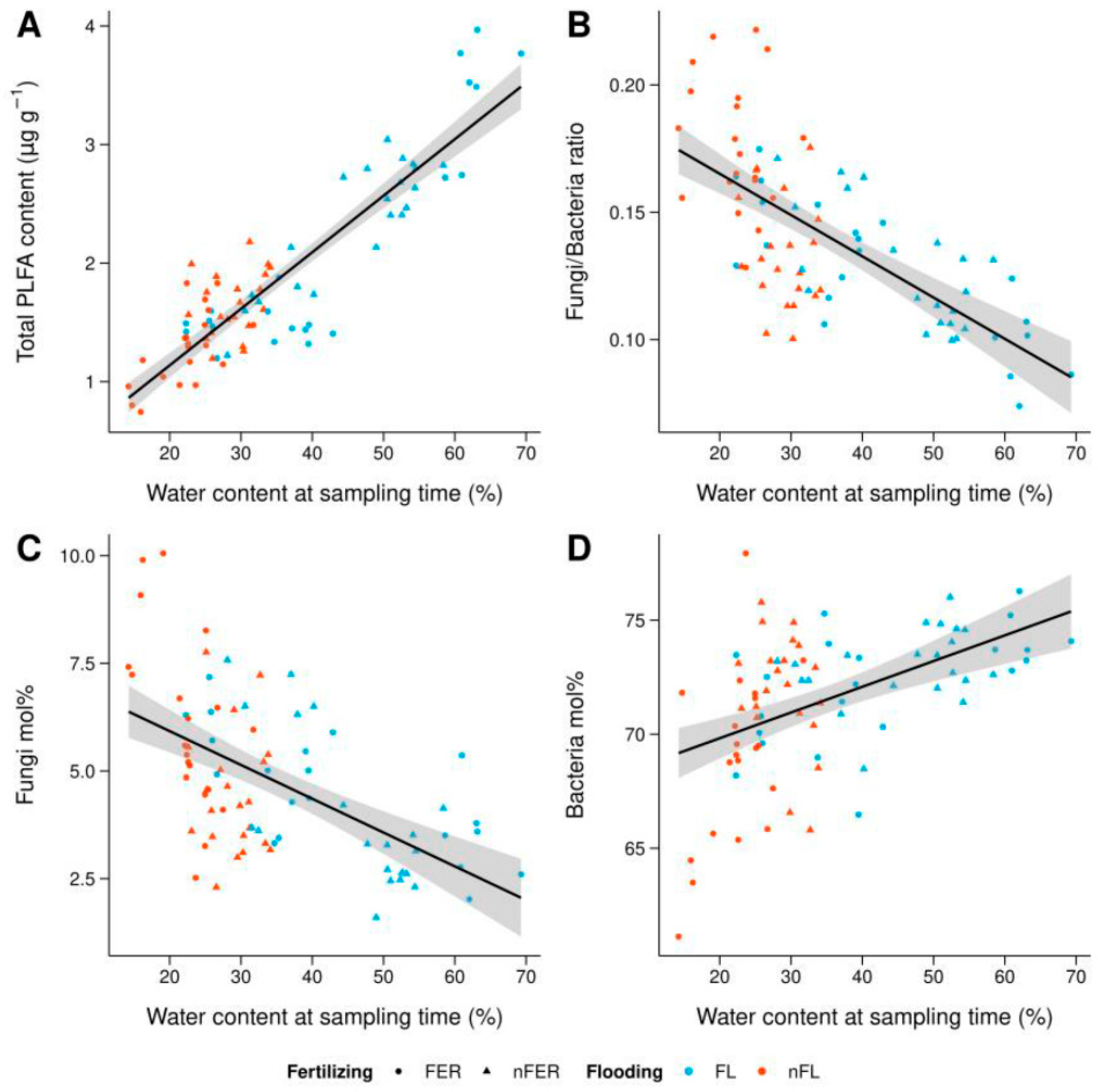
| Parameter | Control | Flooded Only | Fertilized Only | Flooded × Fertilized |
|---|---|---|---|---|
| Corg (%) | 3.8 ± 0.6 | 5 ± 1 | 3 ± 1 | 5 ± 2 |
| Ntot (%) | 0.35 ± 0.05 | 0.5 ± 0.1 | 0.30 ± 0.09 | 0.5 ± 0.2 |
| pH | 4.9 ± 0.2 | 5.3 ± 0.1 | 6 ± 1 | 5.3 ± 0.3 |
| Water extractable NO3− (µg g−1) | 90 ± 30 | 120 ± 90 | 90 ± 60 | 110 ± 80 |
| Water content (g g−1 soil) | 0.29 ± 0.04 | 0.46 ± 0.09 | 0.22 ± 0.04 | 0.4 ± 0.2 |
| Sand (%) | 50 ± 10 | 40 ± 20 | 57 ± 6 | 50 ± 10 |
| Silt (%) | 27 ± 5 | 30 ± 8 | 28 ± 4 | 29 ± 5 |
| Clay (%) | 21 ± 7 | 30 ± 10 | 16 ± 3 | 23 ± 7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kenngott, K.G.J.; Riess, K.; Muñoz, K.; Schaumann, G.E.; Buhk, C.; Diehl, D. Flood Pulse Irrigation of Meadows Shapes Soil Chemical and Microbial Parameters More Than Mineral Fertilization. Soil Syst. 2021, 5, 24. https://doi.org/10.3390/soilsystems5020024
Kenngott KGJ, Riess K, Muñoz K, Schaumann GE, Buhk C, Diehl D. Flood Pulse Irrigation of Meadows Shapes Soil Chemical and Microbial Parameters More Than Mineral Fertilization. Soil Systems. 2021; 5(2):24. https://doi.org/10.3390/soilsystems5020024
Chicago/Turabian StyleKenngott, Kilian G. J., Kai Riess, Katherine Muñoz, Gabriele E. Schaumann, Constanze Buhk, and Dörte Diehl. 2021. "Flood Pulse Irrigation of Meadows Shapes Soil Chemical and Microbial Parameters More Than Mineral Fertilization" Soil Systems 5, no. 2: 24. https://doi.org/10.3390/soilsystems5020024
APA StyleKenngott, K. G. J., Riess, K., Muñoz, K., Schaumann, G. E., Buhk, C., & Diehl, D. (2021). Flood Pulse Irrigation of Meadows Shapes Soil Chemical and Microbial Parameters More Than Mineral Fertilization. Soil Systems, 5(2), 24. https://doi.org/10.3390/soilsystems5020024






