Abstract
Repeated applications of phosphorus (P) fertilizers result in the buildup of P in soil (commonly known as legacy P), a large fraction of which is not immediately available for plant use. Long-term applications and accumulations of soil P is an inefficient use of dwindling P supplies and can result in nutrient runoff, often leading to eutrophication of water bodies. Although soil legacy P is problematic in some regards, it conversely may serve as a source of P for crop use and could potentially decrease dependence on external P fertilizer inputs. This paper reviews the (1) current knowledge on the occurrence and bioaccessibility of different chemical forms of P in soil, (2) legacy P transformations with mineral and organic fertilizer applications in relation to their potential bioaccessibility, and (3) approaches and associated challenges for accessing native soil P that could be used to harness soil legacy P for crop production. We highlight how the occurrence and potential bioaccessibility of different forms of soil inorganic and organic P vary depending on soil properties, such as soil pH and organic matter content. We also found that accumulation of inorganic legacy P forms changes more than organic P species with fertilizer applications and cessations. We also discuss progress and challenges with current approaches for accessing native soil P that could be used for accessing legacy P, including natural and genetically modified plant-based strategies, the use of P-solubilizing microorganisms, and immobilized organic P-hydrolyzing enzymes. It is foreseeable that accessing legacy P will require multidisciplinary approaches to address these limitations.
1. Introduction
Phosphorus (P) is essential to life on Earth. It plays critical roles in core biological systems associated with energy storage, cell replication, and protein synthesis [1]. Among essential macronutrients, it is often the concentration of bioaccessible P, or dissolved inorganic P, in soil that limits plant growth [2]. This fact makes the utilization of P, a finite resource that is mined from specific locations worldwide [3], a critical component of the global food system that aims to feed a growing population [4]. When inorganic fertilizers are added to soil to ameliorate deficiencies, P undergoes sorption, precipitation, and organic matter complexation reactions that render it unavailable for plant uptake [5]. Consequently, large quantities of fertilizers (globally, 16.5 million metric tons P y−1 [4]) are added to maintain soil solution P levels that are optimal for plant growth [6]. With only 10–36% of added P taken up by most crops in the year of application [7,8], long-term P fertilization has led to the buildup of residual P in soil (at a rate of ≈10 million metric tons P y−1 globally [9]) that is not immediately accessible to plants, and is commonly known as legacy P. Legacy P can be categorized as inorganic and organic legacy P, referring to excess, unassimilated inorganic and organic P, respectively, from added inorganic and/or organic fertilizers in the year of application.
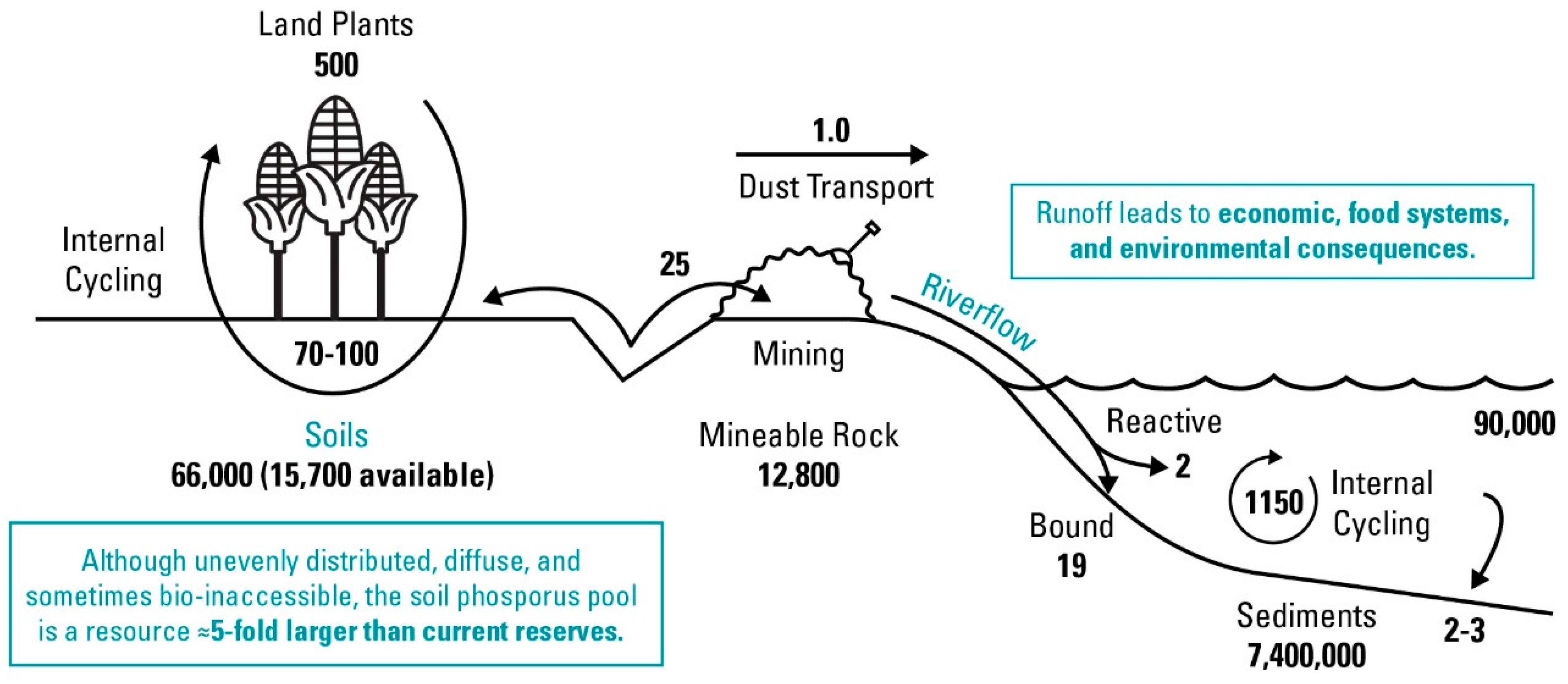
Long-term accumulation of soil P is undesirable from agricultural, economic, and environmental perspectives. Apart from simply being an inefficient use of a finite resource, the buildup of legacy P, such as in European and U.S. soils under long-term P fertilizer applications [10], also presents environmental challenges. Excessive legacy soil P can result in its loss from soil by leaching and erosion to surrounding water bodies. For instance, it is estimated that freshwater ecosystems have had a 75% increase in total P as compared to their pre-industrial revolution state [9]; estuaries and other waterbodies may also be impacted [11]. These increased loadings of P can lead to elevated concentrations and widespread eutrophication of water bodies [12]. For example, 40% of U.S. lakes contain excess P, and ≈80% of states reported the annual occurrence of harmful algal blooms in fresh waterbodies [13,14]. At the same time, legacy P has drawn interest as a potential resource that may be harnessed to reduce use of P fertilizers. Worldwide, soil P is estimated to be five times greater than minable P [15], represented graphically in Figure 1, making the enhancement of legacy P for plant use potentially transformative to the global food system.
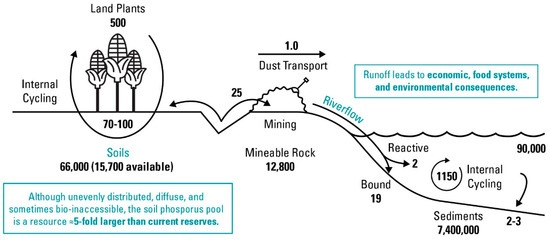
Figure 1.
Global phosphorus flows with flux shown in units of 1012 g P/year. Recreated from [15].
Currently, there are significant technological and societal challenges associated with accessing legacy soil P as a resource for agriculture. First, legacy P naturally becomes available for plant use. For example, highly P-enriched soils in North Carolina, USA, are estimated to support 50–250 years of crop growth without P application [16]. However, actual crop recoveries of this residual P are highly variable across soils, with 4 to >100 years after last fertilizer application needed to recover up to 80% of added P [7,17,18]. Secondly, controlling and increasing soil P solubilization of inorganic legacy P or mineralization of organic legacy P for plant use requires a detailed understanding of the occurrence and reactivity of different soil chemical P forms. Most soil P is bound in bioinaccessible forms with iron, aluminum, or calcium minerals, and organic matter [10,19]. Therefore, with more than 20,000 different soil types mapped across the U.S. alone [20], determining the relative importance of these soil P chemical forms in relation to edaphic factors is a challenge for developing effective management strategies. Third, understanding the dynamic processes associated with P transformation from mineral and animal waste fertilizers into these soil P chemical forms is necessary to maximize the contributions of legacy P as a complement or substitute for externally added P fertilizers. Finally, technologies and strategies for potentially enhancing the capacity to dissolve inorganic legacy P or hydrolyze organic legacy P, herein referred to as bioaccessibility, that are feasible on a large scale are not yet available.
In light of these challenges, this paper aims to review (1) the chemical forms of P in soil and their variation with chemically relevant soil properties (Section 2), (2) the transformation of P into different proportions of various chemical species after mineral and organic P fertilizer applications (Section 3), and (3) different plant- and microbial-based approaches for accessing native soil P and potentially legacy P and associated challenges (Section 4). We conclude with an overall outlook on legacy P as a currently untapped resource that could potentially decrease the dependence on external fertilizer applications in the future as research and technology advances, particularly with the generation of plant cultivars with more efficient P mobilization and utilization processes/capabilities, and with the use of P-solubilizing microorganisms (Section 5).
2. Occurrence and Bioaccessibility of Different Chemical Forms of Soil Legacy P
Inorganic P typically accounts for the bulk of total P in mineral soils [21]. Phosphorus is predominantly taken up by plant roots via dissolved inorganic forms, viz. soluble orthophosphate and its protonated forms. However, concentrations of soluble or bioaccessible phosphate in soils are typically too low for optimal plant growth (ranging from 0.1 to 1 µmol P L−1 soil solution [22]). This low dissolved concentration stems from the strong association of inorganic P with a myriad of minerals. Kizewski et al. [19] compiled a list of the most commonly identified inorganic P species in soils by spectroscopic techniques, which include the calcium phosphate minerals hydroxyapatite and octacalcium phosphate, and phosphate adsorbed on Fe- or Al-oxide minerals.
Unlike inorganic P wherein solubilization equals bioaccessibility, organic P becomes bioaccessible only after mineralization, a biological degradation process catalyzed by specific phosphatase enzymes. Orthophosphate monoesters and diesters are the most common classes of organic P, with monoesters typically comprising 50–70% of organic soil P [23,24]. Among monoester compounds, either the inositol hexakisphosates (IHPs) or humic P constitute the predominant portion of this class [25,26,27,28,29]. These species have differing behaviors in soils, and thus understanding organic speciation of soil P is also critical.
Building off of this knowledge, the following section focuses on the relationship between different P forms and bioaccessibility as influenced by relevant soil properties—a topic that is critical to understanding legacy P accumulation and the potential for legacy P utilization.
2.1. Inorganic P Forms and Bioaccessibility
Among edaphic factors, soil pH largely governs the speciation and bioaccessibility of inorganic P species. In general, acidic pH (pH < 7) favors P association with Al and Fe whereas alkaline pH (pH > 7) favors association with Ca [5]. For instance, in slightly acidic agricultural soils (pH 5.5–6.0), bulk sample P-XANES (X-ray absorption near edge structure) spectroscopy analysis showed 46–56% of total P adsorbed to Fe, 31–42% as Al phosphate mineral, 8–15% as apatite, and 0–12% as organic P [30]. In alkaline, calcareous soils (pH 7.6–7.9), 54–74% of total P existed as hydroxyapatite and/or dicalcium phosphate dihydrate, 25–35% adsorbed to Fe mineral (goethite), and 0–19% organic P [31].
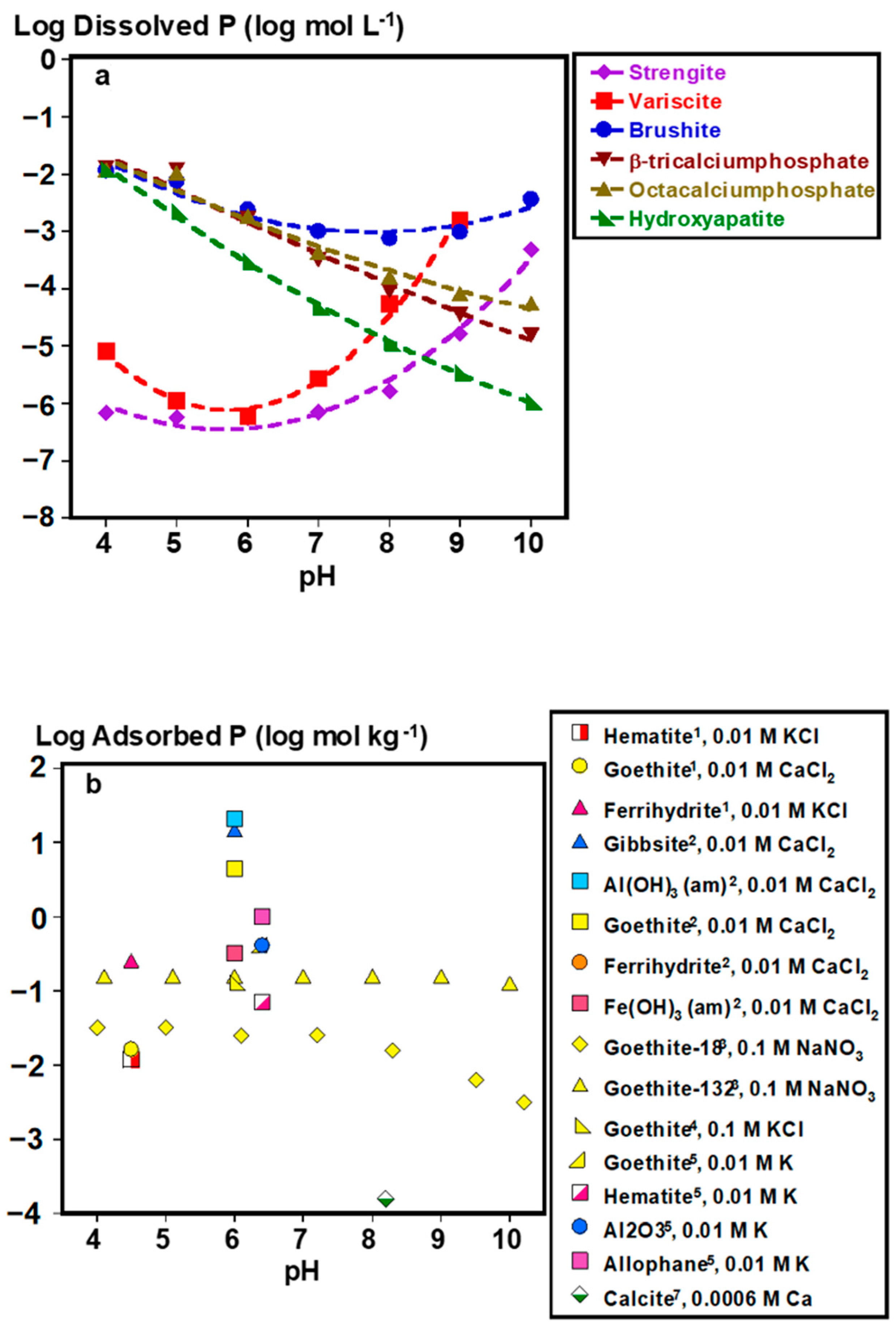
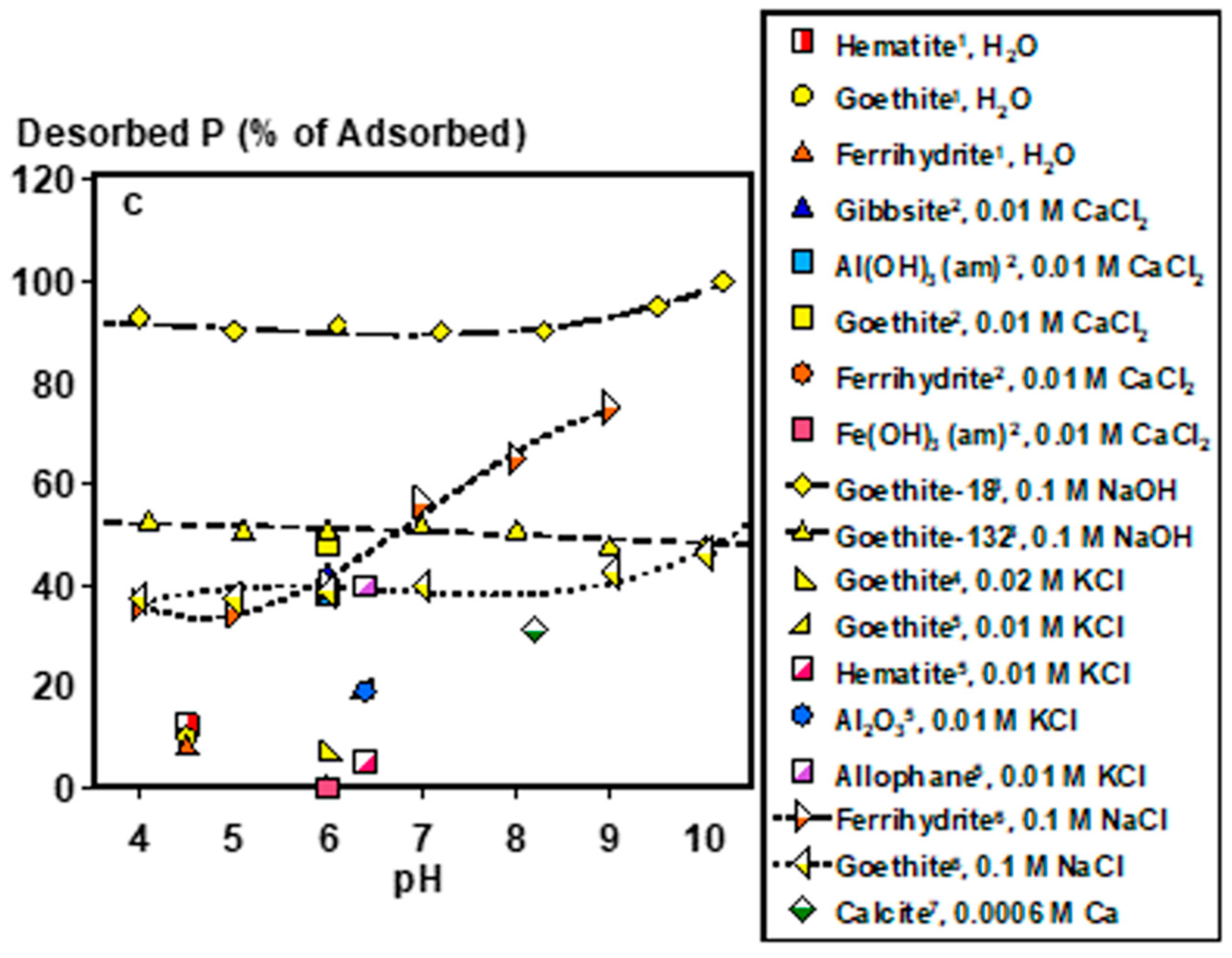
Under acidic soil conditions, Fe- or Al-adsorbed and precipitated P forms control soil solution P (Figure 2a–c). For adsorbed species, composition and phase impact their bioaccessibility. For example, P adsorbed to goethite (α-FeOOH) desorbs similarly to gibbsite (Al(OH)3) and alumina (Al2O3) (within 10% difference) at pH ≈6 under similar experimental conditions [32]. In fact, plant P uptake by ryegrass has been demonstrated with goethite- and poorly crystalline Al(OH)3-adsorbed P at near sorption capacity as lone P sources, demonstrating the potential bioaccessibility of adsorbed P species [33]. However, specific soil mineralogies can strongly impact P extractability and potentially bioaccessibility. Phosphorus adsorbed to amorphous Fe (hydr)oxide phases (i.e., ferrihydrite and Fe(OH)3) is 400–500 times less desorbable than P adsorbed to goethite and amorphous Al(OH)3, with negligible desorption when Fe to Al ratios are increased in amorphous Fe/Al(OH)3 mixtures [34]. It is worth noting that current analytical capabilities, including spectroscopic techniques, such as P K-edge XANES, are limited in their ability to distinguish between P species lacking in distinct spectral features [35], emphasizing that identifying the key adsorbent species in soils is not always straightforward. Thus, functionally defined extractions have been designed to estimate the concentration of poorly crystalline Fe and Al minerals and the P associated with them [36]. Derived from these measurements, the degree of P saturation, defined as the ratio of oxalate-extractable P to the sum of oxalate extractable Fe and Al [37], is an estimate of how much of the P binding capacity of these minerals is occupied, and may serve as a useful tool in assessing the relatively bioaccessible P stocks of legacy P, particularly for acidic soils [38].
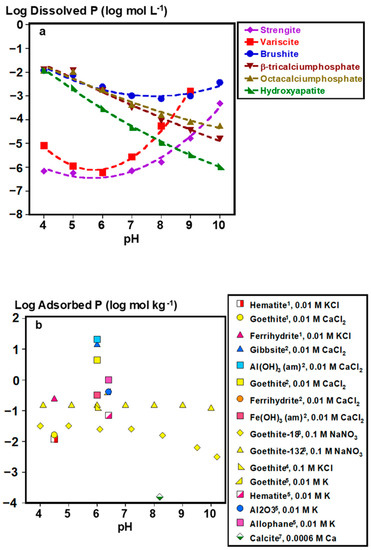
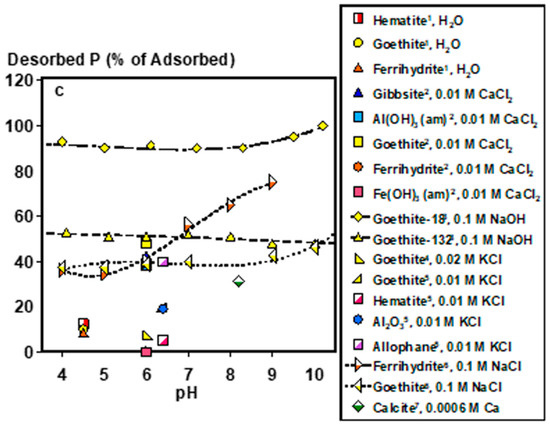
Figure 2.
Soil solution pH controls: (a) dissolved phosphate concentrations in equilibrium with major phosphate minerals, (b) adsorbed P concentrations on to various soil minerals, and (c) desorbed P from different soil minerals as fractions of adsorbed concentrations. *Plotted data points were calculated by Visual MINTEQ Ver. 3.1 at 0.001 KCl and excess concentrations of each mineral (2 g L−1). Adsorption and desorption data were computed from the following works: 1 [39], 2 [34], 3 [40], 4 [41], 5 [32], 6 [42], 7 [43]. Lines are meant to guide the eye and do not represent model fits.
In alkaline soils, bioaccessibility of inorganic precipitated P is controlled by the following Ca phosphate minerals of decreasing solubility: brushite (CaHPO4•2H2O) > β-tricalcium phosphate (Ca3(PO4)2) > octacalcium phosphate (Ca8H2(PO4)6•5H2O) > hydroxyapatite (Ca5(PO4)3(OH)) (Figure 2a). In addition to brushite, calcite-adsorbed P can be another source of labile P, particularly for calcareous alkaline soils (Figure 2). Although P adsorption to Fe (hydr)oxide minerals still occurs under alkaline pH [40], fractions of desorbed P could be similar or greater from these Fe adsorbents than from calcite at pH 8.2 (Figure 2b,c). This suggests that Fe adsorbents could still be controlling more of solution P even in alkaline conditions. However, there is a lack of data on the plant uptake of P from Fe or Al adsorbents under alkaline conditions.
In soils, P is often categorized into labile or non-labile pools, but this concept is rather arbitrary with unclear molecular boundaries [44]. For instance, the use of Hedley sequential extraction method could lead to chemical P redistributions in the course of the extraction process, overestimating Ca-bound P species (i.e., hydroxyapatite), particularly for soils high in exchangeable Ca [45]. Similarly, agronomic soil P tests using various types of chemical solutions (e.g., Bray 1 [46], Mehlich-3 [47], Olsen [48], or Morgan [49] extractants), measure labile P, but the extractable P does not equate to P molecules that plants access in soil. Hence, these tests are agronomically useful only when correlated and calibrated with actual crop response [50]. The use of anion exchange resin membranes may offer a more direct alternative index for bioaccessible fractions of soil legacy P by simulating root surfaces in removing dissolved P at native soil pH, thus providing a better approach for predicting bioavailable P [51,52,53]. However, use of resins can be laborious, time-consuming, and likely incompatible with commercial soil test laboratories where the farmers require quick soil diagnostic tests. Moreover, when well calibrated, soil test extraction methods (e.g., Mehlich-3, Bray, Olsen, and other tests [54]) can determine the fertilizer needs for crops. Consequently, despite being a better method to measure soil bioavailable P, the current use of the anion exchange method is largely limited to research purposes and has low adoption by commercial soil test laboratories. It is worth noting that neither the soil test nor other commonly used labile P extractions, anion resin P measurements, or any field sensing tools can distinguish the individual P species contributing to dissolved inorganic P in soil solution.
2.2. Organic P Forms and Bioaccessibility
Soil pH has been shown to influence occurrence of total organic P and specific organic P species differently. At higher pH in tropical rainforest soils, total organic P concentrations were found to be greater (pH 3.3–7; R2 = 0.89), but this relationship appears to be reversed in temperate agricultural grasslands (pH 5.3–6.7; R2 = −0.60) [55,56]. A more comprehensive regional study by Hou et al. [57] showed generally uniform concentrations of total organic P with pH in tropical, subtropical, and temperate forest soils. This agrees with total organic P concentrations remaining constant across soil pH gradients (pH 3.7–7.8) along the arable Hoosfield strip at Rothamsted Research Station, UK [58]. In terms of specific organic P species, Turner and Blackwell [58] found that non-IHP phosphomonoesters constituted the bulk of soil organic P (51–67%) and did not vary considerably across the Hoosfield pH gradient. However, concentrations of IHPs (myo- and scyllo-isomers), DNA, and phosphonates increased for pH ≤ 5 from relatively uniform concentrations (for IHP) or undetected presence (for DNA and phosphonates) at higher pH (pH 5–8) [58]. The degradation of IHP may be limited by lower enzymatic hydrolysis (breakdown) by phytase enzyme at higher pH, as its activity is mostly optimum between pH 2 and 6.5 with fewer characterized phytases showing optimum activities between pH 7 and 8 [59,60,61,62,63]. In contrast, phosphodiesterase activities decrease at lower soil pH [64]. Additionally, greater accumulation of IHPs and DNAs at highly acidic pH may arise from their stronger associations with clay surfaces at low pH [65,66], with presumably decreased microbial activity under these conditions.
Soil organic P is positively related with soil organic C in natural ecosystems and cultivated grassland soils [57,67,68,69]. No relationships have been drawn between IHPs and organic C, but mixed relationships have been reported between DNA and organic C in non-agricultural and cropland soils [67,70]. In arable soils, humic P has been highly correlated with soil organic C [70]. Positive associations with organic C, however, do not necessitate equal rates of turnover for organic P. In a meta-analysis of 80 topsoils in different countries, Spohn [71] found no changes in organic P concentrations in bulk soils or clay-size fractions in comparison to the significant reductions in organic C concentrations due to land-use change (i.e., native woodlands to croplands). Although different organic P species were not distinguished in the meta-analysis, this analysis suggests that organic P forms are likely more persistent than organic C, or their degradation is limited by factors independent of those of bulk organic C.
Among commonly identifiable organic P compounds, simple non-IHP phosphomonesters enzymatically degrade the fastest, followed by phosphodiester, with IHP being most resistant [72]. The relatively rapid degradation of non-IHP monoesters could either be due to greater enzymatic efficiencies of non-IHP phosphomonesterases [73,74] or to the longer persistence of these enzymes in soils relative to phosphodiesterases or phytases [75,76]. Although sustained activities have been reported for adsorbed non-IHP phosphomonoesterase [77] and adsorbed phytase [78], it is not clear how much of these immobilized enzyme activities constitute in their activities in field soils. Moreover, although many laboratory experiments imply limited IHP mineralization, when added in the field, IHP has been reported to degrade rapidly, with 12–18% remaining in calcareous soils after 13 weeks [79,80]. However, it has also been recognized that freshly added IHP may be different from residual IHP that has aged in field soils under much longer periods [78]. Dissolved organic P (<0.2 μm, molybdate-unreactive) in soil water extracts hydrolyzed between 0 and 61% of molybdate-unreactive P in the presence of added enzymes with highly variable fractions of hydrolyzed P class or species [81,82]. Similarly, organic P in soil leachate from soils receiving inorganic fertilizer and dairy effluent hydrolyzed 36–54% of malachite-green unreactive P (another measure for organic P) using enzyme additions [83,84]. However, Toor et al. [84] reported 10–21% of unreactive P inherently hydrolyzed in the leachate in the absence of phosphatase enzymes. These studies suggest that organic P could mineralize during transport in water, but their field hydrolysis may be less than laboratory measurements considering the optimized conditions used in added phosphatase assays [85].
As for humic P, information is currently limited regarding its potential mineralization. Jarosch et al. [86] reported highly significant correlation between enzyme-stable P and high molecular weight unhydrolyzed organic P, consistent with humic P bearing monoester P linkages [87]. Collectively, these studies not only show a gradient chemical stability of organic P but also suggest that humic P might be the most recalcitrant form of organic P, thereby presenting a challenge towards mineralizing this pool of soil organic legacy P for plant use.
3. Legacy P Transformations with Mineral and Organic Fertilizer Applications
As noted above, legacy P accumulates in soil with repeated additions of excess P fertilizers. In the USA, inorganic P fertilizer is applied approximately 10 times more than organic fertilizers (by mass; Table 1). This section reviews particularly how legacy inorganic P transforms into various species with mineral and organic fertilizer applications as it relates to its overall speciation and potential bioaccessibility in soils. We also review studies on impacts of fertilizer application and cessation on implied transformations of specific organic legacy P. This section focuses on soil P species transformation studies before and after fertilizer applications as probed by spectroscopic techniques (i.e., P-XANES for inorganic P and solution P-NMR (nuclear magnetic resonance) for organic P). Considering the analytical limitations of these techniques in distinguishing between fertilizer-derived versus native parent material-derived inorganic P in soil, we consider existing soil inorganic P largely as fertilizer-derived or legacy inorganic P throughout this section.

Table 1.
Types and annual usage of phosphate fertilizers in the USA (the year 2015). Data are from the United States Department of Agriculture Economic Research Service (https://www.ers.usda.gov/data-products/fertilizer-use-and-price.aspx).
3.1. Soil Inorganic Legacy P Transformations
Different types of P fertilizers have been shown to transform inorganic legacy P species of contacted soils depending on soil properties, fertilizer placement, and incubation time. For instance, addition of granular monoammonium phosphate (MAP; (NH4)H2PO4) or liquid ammonium polyphosphate (APP) changes P speciation from predominantly Al-adsorbed P (>90%) to Fe-adsorbed (>70%) within 7.5 mm around the point of application. These shifts towards Fe-adsorbed species also yielded the greatest resin P extractability with MAP in acidic Oxisol (pH 3.9) and APP in Andisol (pH 5.9) [88]. Khatiwada et al. [51] also reported predominantly Fe- and Al-adsorbed P (63%) and greater resin-extractable P from liquid MAP than granular MAP (≈50%) when both fertilizers were deep-banded (at 10 cm below the soil surface) and the soils were sampled at 7.5–10 cm depth. Incorporating single superphosphate (monocalcium phosphate) with or without hog manure in bulk soil (0–20 cm) in an acidic soil (pH 5.87–6.13), increased the proportion of Fe-adsorbed P (at 43 or 47%, respectively), and produced Ca-adsorbed P (31.5%) while consuming 100% of apatite in an unfertilized control after 21 years of fertilization [89]. For the same soils, NPK (nitrogen–phosphorus–potassium) addition more than tripled Olsen-extractable P to 10.4 and 38.2 mg kg−1 for surface and subsurface layers, respectively; however, NPK application coupled with hog manure increased extractable P up to nine times relative to unfertilized control soils (11.4 to 99.4 and 9.6 to 43.4 mg kg−1 for surface and subsurface soils, respectively) [89]. Positive association suggested by these studies between soluble P and adsorbed P forms from additions of synthetic and organic P fertilizers may indicate greater contribution of adsorbed P forms over precipitated P minerals in supplying dissolved P in soils. However, although liquid MAP remained mostly adsorbed (≈80%) even after 6 months of field application, resin-P extractability decreased by as much as two thirds [51]. This highlights the need to couple molecular-scale P speciation studies with macroscale measurements evaluating soluble P (i.e., resin-P extractability), as well as experiments that address in-field kinetics of P solubilization from identified P forms, ideally throughout the growing season and in the long term (i.e., between years of production), when developing P fertility regimes.
Transformations of legacy P have also been shown to differ in alkaline soils upon additions of inorganic P fertilizers. For example, Lou et al. [89] observed a slight increase in hydroxyapatite (71–78% from 67–72%) with concomitant decreases in proportions of P adsorbed to goethite and alumina from NPK additions to a calcareous soil (pH 8.1–8.4). However, to a non-calcareous alkaline soil (pH 7.4–8), the same NPK applications (i.e., P as diammonium phosphate) shifted P speciation from predominantly hydroxyapatite-P in unfertilized soil (52–57%) to brushite-P (44–63%), with a slight increase in goethite- or alumina-bound P [89]. Inorganic P fertilizer applications generally increased Olsen-P from 11 to 19% for calcareous or 9 to 41% for the non-calcareous surface soils, respectively [89]. On the other hand, Kar et al. [90] reported that most soil P (55–90% of total P) precipitated as hydroxyapatite after the addition of MAP coupled with urea, with the rest being adsorbed in a non-calcareous alkaline soil (pH 7.9) from 0–20 cm away from the point of application. However, 27 years of urea-based fertilization with or without P led to >50% reduction of hydroxyapatite species and enrichment of FePO4 for an acidified alkaline Mollisol (originally pH 7.6 to 5.7) [91]. Together, these studies demonstrate how both soil and fertilizer types influence predominant soil legacy P forms in the short and the long term.
Contrary to purely mineral fertilizations, adding manure alone or in combination with inorganic fertilizers tends to show more consistent outcomes in diminishing proportions of more stable legacy P species in alkaline soils. For example, applying hog manure with NPK promoted transformation of hydroxyapatite into brushite (39–50%) on both non-calcareous and calcareous surface soils examined by Lou et al. [89]. This agrees with the findings of Kar et al. [90] who showed that addition of solid cattle manure to a calcareous alkaline soil (pH 7.8) also led to dominant precipitation of brushite (60%), particularly at the point of application (i.e., center of the band). However, for both MAP- and manure-fertilized soils, resin-extractable P decreased with increasing distance away from the point of subsurface band application. Nevertheless, resin-P fractions were 10 times greater in manured compared to MAP-fertilized soils [90]. Under laboratory conditions, Ajiboye et al. [92] also reported decreased hydroxyapatite precipitation and altered distributions among more soluble P species (i.e., β-tricalcium phosphate or calcite-adsorbed P) when different organic amendments were incorporated with calcareous alkaline soils. Increased fractions of soluble P observed in manured alkaline soils has been attributed to organic acids inhibiting precipitation of sparingly soluble Ca phosphate minerals and to the lower Ca to P ratios in manure-amended relative to MAP-amended soils [90]. Together these speciation studies suggest that manure enhances transformation of inorganic legacy P into more soluble chemical forms, confirming their lability in resin P extractions. Management strategies or technological advances that catalyze the solubilization from these pools would be necessary for legacy P to either substitute, if not eliminate, mineral P fertilizers for crop needs.
Overall, results of reviewed literature in this section show that inorganic legacy P could convert to more soluble forms with appropriate fertilizer management (i.e., fertilizer type and placement), but these effects are soil-specific and vary with space and time. For example, formation of insoluble soil P species was decreased in an Andisol (pH 5.9) with a liquid synthetic APP fertilizer, but decreased with a granular fertilizer (MAP) in an Oxisol (pH 3.9) [88]. Additionally, although subsurface banding of some inorganic and manure fertilizers enhanced more soluble inorganic legacy P speciation (e.g., brushite and adsorbed P forms) along with increased resin-extractable P, these effects were diminished farther away from the point and time of application [51]. These results suggest that, depending on long-term P application rates, placement, and soil type, fertilization-enhanced solubilization of inorganic legacy P may be time-sensitive and likely microscopic in scale, which may not necessarily impact the bulk soils in the rooting zone. This observation is consistent with the findings of Weyers et al. [31] and Koch et al. [30], who reported insignificant effects of 3 to 16 years of mineral or manure fertilizer applications on inorganic legacy P speciation of bulk soils. Moreover, the reviewed studies suggest that manure applications may offer a benefit for enhancing the solubilization of soil legacy P (i.e., hydroxyapatite), particularly for alkaline soils, and thus combining inorganic and organic P fertilizer applications may be a simple management strategy for decreasing dependence for mineral P fertilizers. However, it should also be mentioned that hydroxyapatite has also been detected as the main P species in acid soils (pH 4.2–5.9) with a long history of receiving higher rates of poultry litter [93,94]. Therefore, the relationships between application, solubilization, and ultimately crop utilization of manure-driven legacy P requires additional long-term evaluation both for acid and alkaline soils. To further enrich our understanding on legacy P transformations, we review organic legacy P transformations with fertilizer applications and cessations in the succeeding section.
3.2. Soil Organic P Transformations
Existing soil organic P does not seem to be impacted by P fertilizer applications [95], suggesting that organic legacy P may not be actively accumulating in organically P-fertilized soils. However, unlike those of inorganic soil legacy P, many P fertilization studies evaluating organic legacy P transformations are limited in investigating other specific fertilization variables (i.e., changes in organic legacy P speciation as affected by fertilizer application in space and time). For example, in a field study on bulk soils (0–20 cm) by Annaheim et al. [96], no significant changes were observed in concentrations of different organic P forms receiving organic fertilizers relative to unfertilized controls, despite stark differences in contents of organic P species in applied organic fertilizers (i.e., dairy manure, compost, dry sewage sludge). By comparing the expected concentrations of what could have been accumulated after 62 years of additions, they also demonstrated the limited accumulation for all forms of added organic P species, including IHPs, nucleic acids, and unidentified monoesters. Similarly, Dou et al. [29] reported negligible IHP accumulation in farm soils receiving different types of manure for 8–10 years, despite an estimated 30 kg P ha−1 annual additions from manure-derived IHPs. These long-term studies suggested organic P mineralization that may have released and supplied inorganic P for plant uptake, although they did not exclude transport losses (i.e., leaching and runoff). Although the fate of organic P species was not determined, these studies suggest that organic fertilizers are not likely to build up organic legacy P in mineral soils in a matter of years or decades. Moreover, their results indicate that inherent or native soil organic P species in bulk soils are relatively more stable against mineralization (and/or transport) in comparison to newly added organic P forms that appeared to be rapidly transformed to inorganic P in the soil.
Indeed, native organic P has been found stable in soils even after cessation of external P additions. For inorganically P-fertilized soils in Canada without history of manure additions, halting chemical P fertilization for 15 years increased organic P and orthophosphate diesters and decreased most of the IHP isomers including myo-IHP, although total IHP remained unchanged relative to continuously P-fertilized soils [23]. Build-up of organic P when inorganic P decreased has been attributed to drawdown or crop utilization of inorganic legacy P [95] from 28 years of prior inorganic fertilizations before stopping P inputs [23]. Although Liu et al. [23] demonstrated crop utilization of inorganic legacy P from soils, mineralization and consequently crop utilization of organic P appeared to be limited in these soils. In another experiment in Northern Ireland, Cade-Menum et al. [97] reported similar findings when orthophosphate decreased, but neither total organic P nor specific organic P forms changed between various P-fertilized treatments from zero-P controls 5 years after P fertilizations had stopped. Liu et al. [91] also found similar levels of accumulated organic P for both P-fertilized and non-P-fertilized soils in China after 27 years of cropping relative to baseline soils, except that phosphodiesters considerably accumulated more in P-fertilized soils. Liu et al. [91] suggested that P deficits induced degradation of phosphodiesters in soils not receiving P. Taken together, these studies suggest that native organic P forms are not likely to mineralize or contribute to bioaccessible P for soils with prolonged inorganic P fertilization history when soil test P is still sufficient.
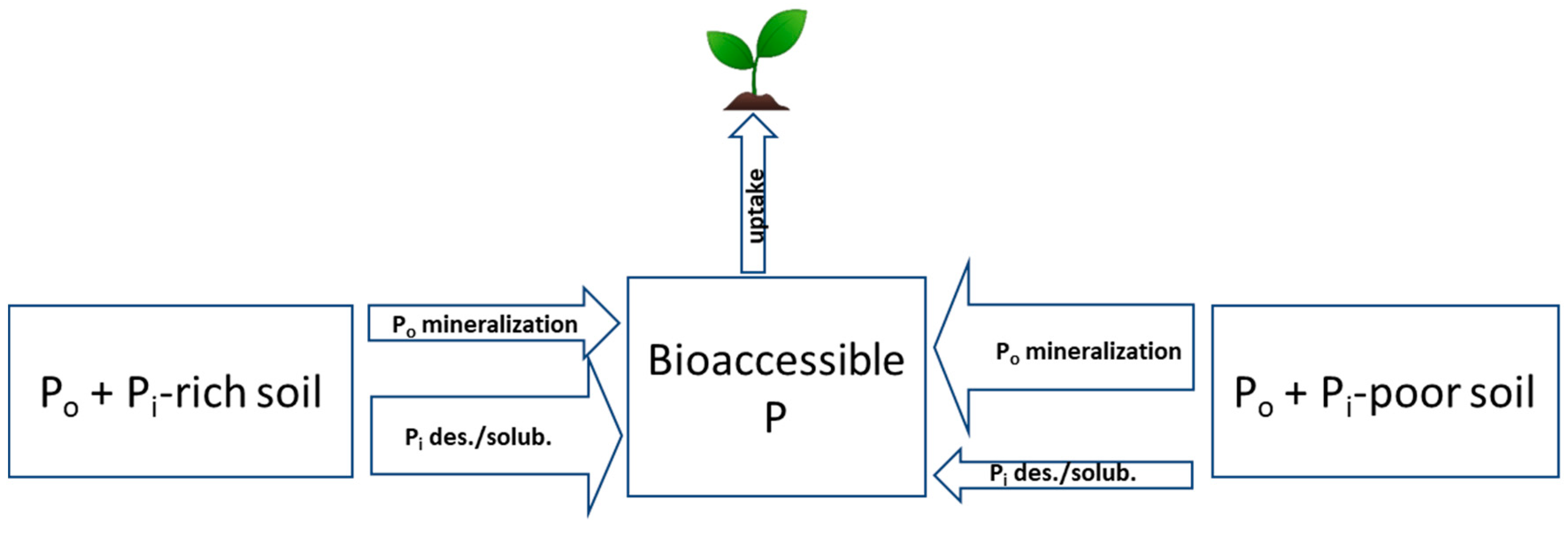
Based on the reviewed studies above, organic P may be constituting a minor portion of legacy P. Moreover, existing non-fertilizer-derived soil organic P may contribute less to plant uptake than inorganic P in soils highly enriched with inorganic legacy P (i.e., soil test P above optimum) (Figure 3). Organic P mineralization by microorganisms or plant roots may not be promoted when sufficient levels of bioaccessible inorganic P exists. Conversely, mineralization of organic P into inorganic P may occur to a greater extent than desorption and solubilization of inorganic P when bioaccessible inorganic P is deficient (i.e., soil test P is below optimum). This is consistent with the work of Recena et al. [98], who observed greater contribution of organic P to actual plant P uptake more in low-P than in high-P soils. Mineralization of organic P forms under deficient soil P conditions could arise from stimulation of microbial growth when C and N levels are not limiting [99,100], meaning when sufficient C supply is present and soil C/N ratios are lower. Hence, balanced nutrient and organic matter management may be required for releasing bound orthophosphates from natural organic P reserves in P-deficient soils. More work is needed to better track the transformations of added organic P compounds from organic P fertilizers under different fertilizer management scenarios and field conditions over different periods of time. Building off this knowledge, the following section reviews the current literature on potential plant, microbial, and biochemical strategies for accessing soil legacy P.
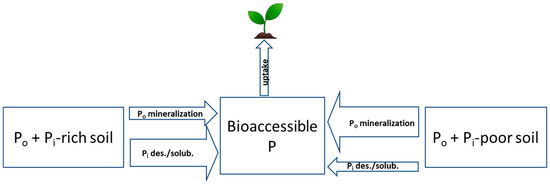
Figure 3.
Conceptual representation of organic and inorganic P transformations towards releasing dissolved P and their expected magnitude of occurrence when legacy P is higher and largely dominated by bioaccessible inorganic P (Po + Pi-rich soil) versus when legacy P is lower and deficient of bioaccessible inorganic P (Po + Pi-poor soil), assuming equal concentrations of organic P. Arrow size indicates the relative magnitude of fluxes. Legend: Po, soil organic P; Pi, soil inorganic P; des/solub, desorption and dissolution.
4. Approaches for Accessing Native Soil P and Associated Challenges
4.1. Plant-Based Strategies
Plants can possibly utilize soil legacy P through naturally evolved strategies, such as induction of root architectural changes and exudation of organic acids and enzymes, as well as possibly through scientific pursuits for improving internal P use efficiency (i.e., plant yield per unit P uptake) [101,102]. Another way that plants can thrive on legacy P is through facilitation of symbiotic and beneficial fungi and other soil microbes. However, the role of microorganisms will be discussed separately in the ensuing section.
Root architectural response is a well-recognized plant mechanism for alleviating soil conditions of low bioaccessible soil P [103], making it likely adaptable for extracting legacy P. Examples of efficient root configurational changes include highly branched root systems, increased production and length of lateral roots, proliferation of root hairs, and formation of cluster roots among various crops [101,104,105,106,107,108,109]. However, these adaptations vary among crop species and genetic varieties [110,111,112,113]. Moreover, most of root architectural studies have been performed on seedlings, which do not necessary correspond to root architecture of mature plants grown in the field [114,115]. With limited field measurements in this area [116], soil factors, particularly those associated with low soil P availability (i.e., soil pH), could potentially restrict efficient root architectural responses in field settings. For instance, foraging traits of maize cultivars characterized by greater root surface correlated well with improved P uptake in neutral field soil, but not in alkaline soil [117]. Furthermore, root architecture has been found to vary depending on the chemical forms and supply patterns of P [118], highlighting the importance for understanding soil legacy P speciation and likely distributions across agricultural soils. Interestingly, genotypes of common bean bearing more adaptive root architectures effectively grew in an unfertilized low-P Ultisol, showing comparable P uptake with P-fertilized commercial varieties [111]. Together these studies emphasize that, while efficient root systems can potentially mine soil legacy P, there is a need for a field-based approach for identifying and matching efficient root systems with soil legacy P and other soil conditions. Moreover, soil properties need to be considered in breeding programs aiming for plants with high P uptake efficiencies.
Another plant adaptive means for potentially exploiting soil legacy P is root exudation of organic acids and phosphatases. Organic acids are known to increase dissolved P by rhizosphere acidification, competition for P adsorption sites, complexation of P-precipitating metals, and provision of C source for P-mobilizing microorganisms [119,120,121,122,123]. However, organic acid production is highly specific to certain plant species [121]. For instance, legumes are known to produce more root exudates than grasses [2,22,124,125], which could be advantageous for P acquisition when legumes are intercropped with cereals. However, increasing evidence exists regarding the minor role of organic acids on actual plant P uptake compared to phosphatase activities or root architectural changes. For instance, legacy P uptake was positively correlated with rhizosphere acid phosphatases, but not with organic anions, with wheat (Triticum aestivum), oat (Avena sativa), potato (Solanum tuberosum), and canola (Brassica napus) in low bioaccessible P soils [126]. Using statistical redundancy analysis, Sun et al. [127] also found that P uptake of field-grown maize and alfalfa was explained more by root architectural changes (58–87%) than total organic anion concentrations (<0.2–24%). This observation emphasizes the importance of root architecture in acquiring P as compared to other mechanisms (e.g., strategic placement of organic acids [128]), although experimental variables also present challenges in understanding organic anions effects [120]. Nevertheless, these studies suggest that enhanced root exudation could effectively allow plants to utilize soil legacy P when combined with highly adaptive root systems.
Apart from natural plant adaptations, genetic engineering can also be used to increase plant uptake capacity and internal use efficiency of soil legacy P. Efforts related to this field have included increasing P uptake by overexpressing P transporters responsible for root P uptake and P transport to shoots, driving crop resilience to P deficiency [129]. For example, overexpression of transporter TaPht1.4 in wheat promoted growth and P accumulation [130]. Similarly, modulating the expression of OsPHT1 transporters in rice has shown to directly impact P uptake [131,132,133,134,135,136]. Phosphorus transporters are regulated by genes and transcription factors, which are altogether induced under P-deficient conditions [129,137]. Phosphorus-related genes have already been identified in major agronomic crops, such as rice (Oryza sativa), maize (Zea mays), and soybean (Glycine max) [138,139,140,141]. Candidate gene overexpression is a powerful approach to enhance nutrient uptake under conditions of low P availability. It is also foreseeable that a combination of both increased uptake capacity and internal P-use efficiency will be equally desirable for crops that have adapted and developed better P uptake and efficiency use strategies.
Whereas natural plant adaptations (i.e., root architectural changes and root exudation) to mobilize legacy P can be highly specific to plant species or varieties as noted above, genetic engineering can increase legacy P acquisition and internal use efficiency for any desired crops. However, this work continues to be a long-term research trajectory. Consequently, much is still unknown as to whether genetically engineered plants may successfully utilize soil legacy P and reduce, if not eliminate, dependence of mineral P fertilizers. As with natural adaptative plant mechanisms, there is an added uncertainty that these plants could address other soil stresses associated with low bioaccessible P (i.e., Al toxicity in highly acidic soils). To this end, material science, microelectronics, and nanotechnology should develop and deploy several sensor platforms (chemical, electromagnetic, optical, and genetic) for detecting real-time below-ground root development and plasticity response to bioaccessible legacy P. Genetic and genomics approaches should complement this sensor approach and will hone in on the identification of the genes regulating P uptake, use efficiency, and tolerance to other soil conditions. For example, the systems level understanding of the many genes, and their regulation, associated with phosphate deficiency once unveiled would allow for predictive and prescriptive interventions. It would be foreseeable to use genome editing technologies, such as CRISPR (clustered regularly interspaced short palindromic repeats) technologies [142,143], to genetically test the functional relevance of known (e.g., OsPSTOL1, AVP1, PHO1 and OsPHT1;6) [141,144,145,146] and unknown genes identified in molecular studies. At the same time, it will be important to consider and incorporate stakeholder perceptions and socio-economic considerations of genetically engineered crops for increased legacy P mobilization and crop utilization to ensure adoption by end-users.
4.2. Phosphate-Solubilizing Microorganisms
A diverse genera of microorganisms including bacteria, fungi, actinomycetes, and cyanobacteria have been reported to solubilize native soil P [147], making them potentially capable of solubilizing legacy P. Phosphate-solubilizing microorganisms (PSM) dissolve or mineralize soil P largely through the production of organic acids and phosphatases [148], and recent reviews have reported generally increased crop growth and yield from the use of PSMs [147,149,150,151]. In general, most studies in this field have focused on effects of PSMs on crop performance, but information is lacking on how PSMs transform legacy P species during or after plant growth, Hence, in the following studies reviewed below, we discuss PSM effects mostly on plant P uptake and growth and suggest research directions for ascertaining the roles of PSMs in impacting legacy P.
Mycorrhizal fungi are perhaps the longest known microorganisms capable of accessing native soil P owing to their capacity for symbiotic associations with 72% of vascular plant roots that began 450 million years ago [152] and could likely be used to access legacy P. Arbuscular mycorrhiza (AM) scavenge greater soil volumes than plant roots for soluble P and rapidly transport acquired P to roots via arbuscules, thereby minimizing the impact of naturally slow replenishment of dissolved P in soil solution [153]. However, AM fungi are diverse; different taxa do not produce the same response from the same plant species, and that the same fungus does not yield the same effects across different plant species or varieties [154]. These effects could range from positive to negative with unknown reasons for these inconsistencies, although elevated C expense by the plant to maintain the symbiotic relationship has been suggested as being detrimental [126,155]. This suggests that compatibility between the plant host and colonizing AM are required for a beneficial outcome of the symbiosis. However, interspecific compatibility between the host plant and the fungi may also vary depending on soil chemical conditions such as soil pH and associated occurrence of different forms of legacy P. Research is therefore needed in understanding t legacy P speciation impacts symbiotic mycorrhizal associations, and vice-versa.
Inoculation of other PSMs to soils has also shown inconsistent effects on plant P uptake and production. For instance, maize increased P uptake by 10% without P addition after 28 d on acidic soil (pH 5.6) with Enterobacter radicincitans DSM 16,656 and Pseudomonas fluorescens DR54 inoculations [156]. However, the observed increase in P uptake had no effect on plant growth. In contrast, Bacillus mucilaginosus inoculation to acidic soil (pH 5.2) did not improve shoot P uptake in maize [157], but Aspergillus FS9 and Bacillus FS-3 increased strawberry (Fragaria x ananasa cv. Fern) yield by 7% and 30%, respectively, in a calcareous soil (pH 7.6) without added P [158]. Although PSM inoculations have been shown to increase water- and resin-extractable P [156], future research needs to focus on how PSMs transform actual soil legacy P species. Direct information on solubilization or mineralization of soil legacy P forms could serve as a tool in matching appropriate PSMs for the a given soil. Moreover, these data are needed both in the short- and the long-term to explore sustainability issues with the use of PSMs in the context of potentially minimizing mineral P fertilizer applications.
PSMs may not thrive on soil legacy P alone and thus may not completely eliminate dependence to external P inputs. The need for supplemental P with PSMs is expected, considering that microorganisms also need bioaccessible P and other essential nutrients for their metabolic processes to establish and sustain microbial activity in soil. Supplementing PSMs with P fertilizers has improved shoot production [156], increased P uptake [157], or exceeded yield of plants from uninoculated P-fertilized control soils by 54–71% [158]. However, inoculating PSM with co-added P not improving plant P uptake and yield has also been reported. This has been observed with Pseudomonas fluorescens Pf153 inoculations to soils (pH 4.8 to pH 6.6) after 8 weeks of maize growth compared to uninoculated controls, despite supplemental rock phosphate [159]. In this respect, future research needs to evaluate compatibilities between supplemental P fertilizers and individual or combinations of various PSM species for a given soil. Considering how mineral and organic fertilizers transform soil legacy P (Section 3.1), co-adding mineral and manure with PSM inoculations may enhance utilization of existing P in soils. On one hand, supplementing PSMs with inorganic P may provide for the immediate nutrient need of both the microorganisms and the plants during PSM establishment in soil. On the other hand, supplementing PSMs with organic P fertilizers may alter legacy P speciation into predominantly less sparingly soluble forms. However, given that external P inputs may unavoidably add more to soil legacy P stocks, more work is needed in finding the right balance between minimizing supplemental P additions while maximizing solubilization/mineralization and consequently crop utilization of existing soil legacy P. Moreover, additional research is needed in evaluating any potential combination among PSM species capable of solubilizing P exclusively outside the roots with those capable of solubilizing P within the roots (e.g., endophytic bacteria [160]) with particular application to agricultural crops. Finally, PSM studies involving inoculations to actual test soils require molecular investigation of mechanisms of action to ascertain utilization of legacy P.
4.3. Immobilized Organic P Hydrolyzing Enzymes
Loading organic P-hydrolyzing enzymes onto clay or nanoclay supports has also been explored as a potential biofertilization strategy to mineralize organic P that could likely be applied to hydrolyze organic legacy P. Menezes-Blackburn et al. [161] loaded phytases to either montmorillonite or allophane, found that phytases produced by different microbial species differed in their activities. In their study, phytase activity derived from Escherichia coli showed peak activity at pH 5 in water, representing roughly 85% of free phytase activity at the same pH. However, phytase from Aspergillus niger showed considerably reduced phytase activity at pH below 5.5 with the tested nanoclay supports [161]. Contrary to decreased activities of immobilized enzymes compared to their freely dissolved forms, Calabi-Floody et al. [162] reported increased activities of up to 48% with allophane-immobilized compared to freely dissolved acid phosphatases. Using the Michaelis–Menten equation to describe degradation of p-nitrophenolphosphate as model organic P substrate, maximum rate of substrate degradation by the enzymes reportedly increased from 33 to 38% with the allophanic compared to montmorillonitic support. However, when composted cattle dung was used as the organic P source, none of the immobilized or the free acid phosphatase showed significant differences from the unamended dung in increasing dissolved inorganic P, which they attributed to high inorganic P concentrations contained in the dung. In contrast, Menezes-Blackburn et al. [163] treated cattle manure with phytases stabilized in allophanic nanoclays and found a significant increase in NaOH-EDTA P (commonly used to extract organic P) and Olsen-extractable inorganic P in soil. However, they did not observe significant hydrolysis of freshly added phytate with soil or manure amended with the immobilized phytases. The stability of these nanoclay-supported enzymes in soils is also of concern, as greatly reduced enzyme activity was also observed after merely 2 h of exposure to high temperature and after 1 d of exposure to protein degradation [161]. Overall, while immobilizing enzymes inside in carrier materials are promising under laboratory conditions, this approach appears to be challenged by limited accessibility of substrates and the interference of other dissolved compounds in the soil solution. Moreover, with a short lifespan, the capacity of nanoclay-immobilized enzymes to release organically bound legacy P in soils and sustain crop P needs would be highly challenged. Future research needs to focus on finding new approaches to adding these enzymes using other carrier materials [164] without negatively affecting their activities when added to soil or when combined with organic fertilizers.
5. Conclusions
Repeated P fertilizer applications over time has led to build-up of soil legacy P, which theoretically could be utilized to substitute for, if not eliminate dependence on, mineral P fertilizers. However, crop recovery of legacy P varies considerably across soils and duration. This paper reviewed (1) the occurrence and factors controlling bioaccessibility of different soil P forms, (2) transformation of different legacy P species with P fertilizer additions, and (3) currently studied strategies that could possibly be used for exploiting soil legacy P for crop use. We found that soil legacy P exists predominantly as inorganic P in either adsorbed or precipitated forms, and secondarily as organic P, mainly as monoester or diester forms. Bioaccessibility of different chemical P forms is predominantly controlled by soil pH either directly (e.g., desorption/dissolution of inorganic P species) or indirectly (e.g., enzymatic degradation of organic P). We also found that inorganic legacy P could transform into more bioaccessible forms with fertilizer applications, but this effect is highly specific to soil and fertilizer type and may diminish away from the point and time of fertilizer application. In contrast, transformation of native soil organic P forms does not appear to be impacted by fertilizer applications and cessations, suggesting lesser accumulation of organic legacy P in soils, as well as lower potential for crop utilization of native organic P in soil particularly when bioaccessible inorganic legacy P is sufficient.
Most currently pursued approaches for possibly increasing crop utility of legacy P are biological in nature. These range from natural to genetically engineered plant adaptations for enhanced soil P uptake and use efficiencies to the use of symbiotic or beneficial P-solubilizing microorganisms and immobilized organic P-hydrolyzing enzymes. However, plant- or microorganism-based strategies are limited by the high specificity of acquired adaptation mechanisms capable of solubilizing soil P, whereas immobilized enzyme efficacy is mainly limited by physical and chemical constraints. Moreover, studies investigating these approaches typically do not elucidate the mechanism of actions for utilization of soil legacy P largely due to lack of molecular soil P speciation component.
As scarcity of P resources becomes a more pressing global issue, mining legacy P from soils may emerge as a more pressing societal need. Harnessing this resource will require not only development of new and existing technologies for improved legacy P utilization but also holistic management strategies that incorporate multiple approaches tailored to specific soil environments. Overall, enhancing our ability to access soil legacy P for croplands will require a multidisciplinary approach to establish soil and crop management systems that enhance complementary use of the different chemical forms, spatial variability, and temporal changes of legacy P. A systems approach should integrate many of the strategies identified above, as well as future technologies, for accessing native soil P. As multidisciplinary advances, for example in genetic engineering and material sciences, are combined with molecular soil P speciation tools in addressing each approach’s limitations, it is foreseeable that prospects for widespread utilization of legacy P as a partial or complete substitute for mineral P fertilizer may increase in the future.
Author Contributions
Conceptualization, O.W.D. and S.D.; writing—original draft preparation, Section 1, Section 2 and Section 3 and Section 5, S.D., O.W.D., L.G., E.S.M., K.G., and J.L.J.; Section 4, S.D., R.S., R.P., and L.V.d.B.; writing—review and editing, O.W.D., K.G., D.H., and E.S.M. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the USDA National Institute of Food and Agriculture, Hatch project NC02713. We thank the Game-Changing Research Initiative Program (GRIP) through the NC State Office of Research and Innovation (ORI), RTI International, and the Kenan Institute for Engineering, Science and Technology.
Acknowledgments
We thank Sarah Powers (NC State University) for assistance with graphics.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Fraústo da Silva, J.J.R.; Williams, R.J.P. The Biological Chemistry of the Elements: The Inorganic Chemistry of Life, 2nd ed.; Oxford University Press: Oxford, UK, 2001. [Google Scholar]
- Vance, C.P.; Uhde-Stone, C.; Allan, D.L. Phosphorus acquisition and use: Critical adaptations by plants for securing a nonrenewable resource. New Phytol. 2003, 157, 423–447. [Google Scholar] [CrossRef]
- Stewart, W.M.; Hammond, L.L.; Van Kauwenbergh, S.J. Phosphorus as a Natural Resource. In Phosphorus: Agriculture and the Environment; Pierzynski, G.M., McDowell, R.W., Sims, J.T., Eds.; Soil Science Society of America: Madison, WI, USA, 2005; pp. 1–22. [Google Scholar]
- Cordell, D.; White, S. Life’s Bottleneck: Sustaining the World’s Phosphorus for a Food Secure Future. Ann. Rev. Environ. Resour. 2014, 39, 161–188. [Google Scholar] [CrossRef]
- Pierzynski, G.M.; McDowell, R.W.; Sims, J.T. Chemistry, Cycling, and Potential Movement of Inorganic Phosphorus in Soils. In Phosphorus: Agriculture and the Environment; Sims, J.T., Sharpley, A.N., Westermann, T.D., Eds.; Soil Science Society of America: Madison, WI, USA, 2005; pp. 51–86. [Google Scholar]
- Fox, R.L.; Kamprath, E.J. Phosphate Sorption Isotherms for Evaluating the Phosphate Requirements of Soils. Soil Sci. Soc. Am. J. 1970, 34, 902–907. [Google Scholar] [CrossRef]
- Syers, J.K.; Johnston, A.E.; Curtin, D. Efficiency of Soil Fertilizer Phosphorus Use—Reconciling Changing Concepts of Soil Phosphorus Behavior with Agronomic Information; Food And Agriculture Organization of The United Nations (FAO): Rome, Italy, 2008. [Google Scholar]
- Dhillon, J.; Torres, G.; Driver, E.; Figueiredo, B.; Raun, W.R. World Phosphorus Use Efficiency in Cereal Crops. Agron. J. 2017, 109, 1670–1677. [Google Scholar] [CrossRef]
- Bennett, E.M.; Carpenter, S.R.; Caraco, N.F. Human Impact on Erodable Phosphorus and Eutrophication: A Global Perspective: Increasing accumulation of phosphorus in soil threatens rivers, lakes, and coastal oceans with eutrophication. BioScience 2001, 51, 227–234. [Google Scholar] [CrossRef]
- Menezes-Blackburn, D.; Giles, C.; Darch, T.; George, T.S.; Blackwell, M.; Stutter, M.; Shand, C.; Lumsdon, D.; Cooper, P.; Wendler, R.; et al. Opportunities for mobilizing recalcitrant phosphorus from agricultural soils: A review. Plant Soil 2018, 427, 5–16. [Google Scholar] [CrossRef]
- Yang, Y.-Y.; Asal, S.; Toor, G.S. Residential catchments to coastal waters: Forms, fluxes, and mechanisms of phosphorus transport. Sci. Total Environ. 2020, 142767. [Google Scholar] [CrossRef]
- Jarvie, H.P.; Sharpley, A.N.; Withers, P.J.A.; Scott, J.T.; Haggard, B.E.; Neal, C. Phosphorus Mitigation to Control River Eutrophication: Murky Waters, Inconvenient Truths, and “Postnormal” Science. J. Environ. Qual. 2013, 42, 295–304. [Google Scholar] [CrossRef]
- USEPA. National Lakes Assessment 2012 Results. Available online: https://www.epa.gov/national-aquatic-resource-surveys/national-lakes-assessment-2012-results (accessed on 15 June 2020).
- CDC. Harmful Algal Bloom (HAB)-Associated Illness. Available online: https://www.cdc.gov/habs/general.html#:~:text=Fresh%20water%20%E2%80%94In%202014%2C%20a,or%20other%20fresh%20water%20bodies (accessed on 15 June 2020).
- Schlesinger, W.H.; Bernhardt, E.S. Chapter 12—The Global Cycles of Nitrogen and Phosphorus. In Biogeochemistry, 3rd ed.; Schlesinger, W.H., Bernhardt, E.S., Eds.; Academic Press: Boston, MA, USA, 2013; pp. 445–467. [Google Scholar]
- Kamprath, E.J. Changes in phosphate availability of ultisols with long-term cropping. Commun. Soil Sci. Plant Anal. 1999, 30, 909–919. [Google Scholar] [CrossRef]
- Roberts, T.; Johnston, A. Phosphorus use efficiency and management in agriculture. Resour. Conserv. Recycl. 2015, 105. [Google Scholar] [CrossRef]
- Johnston, A.E.; Poulton, P.R. Phosphorus in Agriculture: A Review of Results from 175 Years of Research at Rothamsted, UK. J. Environ. Qual. 2019, 48, 1133–1144. [Google Scholar] [CrossRef] [PubMed]
- Kizewski, F.; Liu, Y.-T.; Morris, A.; Hesterberg, D. Spectroscopic Approaches for Phosphorus Speciation in Soils and Other Environmental Systems. J. Environ. Qual. 2011, 40, 751–766. [Google Scholar] [CrossRef] [PubMed]
- Hou, E.; Tan, X.; Heenan, M.; Wen, D. A global dataset of plant available and unavailable phosphorus in natural soils derived by Hedley method. Sci. Data 2018, 5, 180166. [Google Scholar] [CrossRef] [PubMed]
- Sims, J.T.; Pierzynski, G.M. Chemistry of Phosphorus in Soils. In Chemical Processes in Soils; Tabatabai, M.A., Sparks, D.L., Eds.; Soil Science Society of America: Madison, WI, USA, 2005; pp. 151–192. [Google Scholar]
- Hinsinger, P. Bioavailability of soil inorganic P in the rhizosphere as affected by root-induced chemical changes: A review. Plant Soil 2001, 237, 173–195. [Google Scholar] [CrossRef]
- Liu, J.; Hu, Y.; Yang, J.; Abdi, D.; Cade-Menun, B.J. Investigation of Soil Legacy Phosphorus Transformation in Long-Term Agricultural Fields Using Sequential Fractionation, P K-edge XANES and Solution P NMR Spectroscopy. Environ. Sci. Technol. 2015, 49, 168–176. [Google Scholar] [CrossRef]
- Cade-Menun, B.J.; He, Z.; Zhang, H.; Endale, D.M.; Schomberg, H.H.; Liu, C.W. Stratification of Phosphorus Forms from Long-Term Conservation Tillage and Poultry Litter Application. Soil Sci. Soc. Am. J. 2015, 79, 504–516. [Google Scholar] [CrossRef]
- Vincent, A.G.; Vestergren, J.; Gröbner, G.; Persson, P.; Schleucher, J.; Giesler, R. Soil organic phosphorus transformations in a boreal forest chronosequence. Plant Soil 2013, 367, 149–162. [Google Scholar] [CrossRef]
- Turner, B.L.; Cheesman, A.W.; Godage, H.Y.; Riley, A.M.; Potter, B.V.L. Determination of neo- and d-chiro-Inositol Hexakisphosphate in Soils by Solution 31P NMR Spectroscopy. Environ. Sci. Technol. 2012, 46, 4994–5002. [Google Scholar] [CrossRef]
- Ahlgren, J.; Djodjic, F.; Börjesson, G.; Mattsson, L. Identification and quantification of organic phosphorus forms in soils from fertility experiments. Soil Use Manag. 2013, 29, 24–35. [Google Scholar] [CrossRef]
- Cade-Menun, B.J.; Carter, M.R.; James, D.C.; Liu, C.W. Phosphorus Forms and Chemistry in the Soil Profile under Long-Term Conservation Tillage: A Phosphorus-31 Nuclear Magnetic Resonance Study. J. Environ. Qual. 2010, 39, 1647–1656. [Google Scholar] [CrossRef]
- Dou, Z.; Ramberg, C.F.; Toth, J.D.; Wang, Y.; Sharpley, A.N.; Boyd, S.E.; Chen, C.R.; Williams, D.; Xu, Z.H. Phosphorus Speciation and Sorption-Desorption Characteristics in Heavily Manured Soils. Soil Sci. Soc. Am. J. 2009, 73, 93–101. [Google Scholar] [CrossRef]
- Koch, M.; Kruse, J.; Eichler-Löbermann, B.; Zimmer, D.; Willbold, S.; Leinweber, P.; Siebers, N. Phosphorus stocks and speciation in soil profiles of a long-term fertilizer experiment: Evidence from sequential fractionation, P K-edge XANES, and 31P NMR spectroscopy. Geoderma 2018, 316, 115–126. [Google Scholar] [CrossRef]
- Weyers, E.; Strawn, D.G.; Peak, D.; Moore, A.D.; Baker, L.L.; Cade-Menun, B. Phosphorus Speciation in Calcareous Soils Following Annual Dairy Manure Amendments. Soil Sci. Soc. Am. J. 2016, 80, 1531–1542. [Google Scholar] [CrossRef]
- Oh, Y.-M.; Hesterberg, D.L.; Nelson, P.V.; Niedziela, C.E. Desorption Characteristics of Three Mineral Oxides and a Non-crystalline Aluminosilicate for Supplying Phosphate in Soilless Root Media. Commun. Soil Sci. Plant Anal. 2016, 47, 753–760. [Google Scholar] [CrossRef]
- Parfitt, R.L. The availability of P from phosphate-goethite bridging complexes. Desorption and uptake by ryegrass. Plant Soil 1979, 53, 55–65. [Google Scholar] [CrossRef]
- Gypser, S.; Hirsch, F.; Schleicher, A.M.; Freese, D. Impact of crystalline and amorphous iron- and aluminum hydroxides on mechanisms of phosphate adsorption and desorption. J. Environ. Sci. 2018, 70, 175–189. [Google Scholar] [CrossRef]
- Kruse, J.; Abraham, M.; Amelung, W.; Baum, C.; Bol, R.; Kühn, O.; Lewandowski, H.; Niederberger, J.; Oelmann, Y.; Rüger, C.; et al. Innovative methods in soil phosphorus research: A review. J. Plant Nutr. Soil Sci. 2015, 178, 43–88. [Google Scholar] [CrossRef]
- Mehra, O.P.; Jackson, M.L. Iron oxide removal from soils and clays by a dithionite–citrate system buffered with sodium bicarbonate. In Clays and Clay Minerals; Ingerson, E., Ed.; Pergamon: Oxford, UK, 2013; pp. 317–327. [Google Scholar]
- Nair, V.D. Soil phosphorus saturation ratio for risk assessment in land use systems. Front. Environ. Sci. 2014, 2. [Google Scholar] [CrossRef]
- Beauchemin, S.; Simard, R.R. Soil phosphorus saturation degree: Review of some indices and their suitability for P management in Québec, Canada. Can. J. Soil Sci. 1999, 79, 615–625. [Google Scholar] [CrossRef]
- Wang, X.M.; Liu, F.; Tan, W.F.; Li, W.; Feng, X.H.; Sparks, D.L. Characteristics of Phosphate Adsorption-Desorption Onto Ferrihydrite: Comparison With Well-Crystalline Fe (Hydr)Oxides. Soil Sci. 2013, 178, 1–11. [Google Scholar] [CrossRef]
- Strauss, R.; Brümmer, G.W.; Barrow, N.J. Effects of crystallinity of goethite: II. Rates of sorption and desorption of phosphate. Eur. J. Soil Sci. 1997, 48, 101–114. [Google Scholar] [CrossRef]
- Yan, Y.; Koopal, L.K.; Liu, F.; Huang, Q.; Feng, X. Desorption of myo-inositol hexakisphosphate and phosphate from goethite by different reagents. J. Plant Nutr. Soil Sci. 2015, 178, 878–887. [Google Scholar] [CrossRef]
- Krumina, L.; Kenney, J.P.L.; Loring, J.S.; Persson, P. Desorption mechanisms of phosphate from ferrihydrite and goethite surfaces. Chem. Geol. 2016, 427, 54–64. [Google Scholar] [CrossRef]
- Sø, H.U.; Postma, D.; Jakobsen, R.; Larsen, F. Sorption of phosphate onto calcite; results from batch experiments and surface complexation modeling. Geochim. Cosmochim. Acta 2011, 75, 2911–2923. [Google Scholar]
- Hesterberg, D. Macroscale Chemical Properties and X-Ray Absorption Spectroscopy of Soil Phosphorus. In Developments in Soil Science; Singh, B., Gräfe, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2010; Volume 34, pp. 313–356. [Google Scholar]
- Gu, C.; Dam, T.; Hart, S.C.; Turner, B.L.; Chadwick, O.A.; Berhe, A.A.; Hu, Y.; Zhu, M. Quantifying Uncertainties in Sequential Chemical Extraction of Soil Phosphorus Using XANES Spectroscopy. Environ. Sci. Technol. 2020, 54, 2257–2267. [Google Scholar] [CrossRef]
- Bray, R.H.; Kurtz, L.T. Determination of Total, Organic, and Available Forms of Phosphorus in Soils. Soil Sci. 1945, 59, 39–46. [Google Scholar] [CrossRef]
- Mehlich, A. Mehlich 3 soil test extractant: A modification of Mehlich 2 extractant. Commun. Soil Sci. Plant Anal. 1984, 15, 1409–1416. [Google Scholar] [CrossRef]
- Watanabe, F.S.; Olsen, S.R. Test of an Ascorbic Acid Method for Determining Phosphorus in Water and NaHCO3 Extracts from Soil. Soil Sci. Soc. Am. J. 1965, 29, 677–678. [Google Scholar] [CrossRef]
- McIntosh, J.L. Bray and Morgan Soil Extractants Modified for Testing Acid Soils from Different Parent Materials1. Agron. J. 1969, 61, 259–265. [Google Scholar] [CrossRef]
- Hochmuth, G.J.; Mylavarapu, R.S.; Hanlon, E.A. Developing a Soil Test Extractant: The Correlation and Calibration Processes; University of Florida: Gainesville, FL, USA, 2017. [Google Scholar]
- Khatiwada, R.; Hettiarachchi, G.M.; Mengel, D.B.; Fei, M. Speciation of Phosphorus in a Fertilized, Reduced-Till Soil System: In-Field Treatment Incubation Study. Soil Sci. Soc. Am. J. 2012, 76, 2006–2018. [Google Scholar] [CrossRef]
- Sims, J.T.; Edwards, A.C.; Schoumans, O.F.; Simard, R.R. Integrating Soil Phosphorus Testing into Environmentally Based Agricultural Management Practices. J. Environ. Qual. 2000, 29, 60–71. [Google Scholar] [CrossRef]
- Mallarino, A.P.; Atia, A.M. Correlation of a Resin Membrane Soil Phosphorus Test with Corn Yield and Routine Soil Tests. Soil Sci. Soc. Am. J. 2005, 69, 266–272. [Google Scholar] [CrossRef]
- Sims, J.T. Soil Test Phosphorus: Principles and Methods In Methods of Phosphorus Analysis for Soils, Sediments, Residuals, and Waters, 2nd ed.; Kovar, J., Pierzynski, G., Eds.; Southern Cooperative Series Bulletin 408; Southern Extension and Research Activity (SERA): Blacksburg, VA, USA, 2009; Volume 408. [Google Scholar]
- Turner, B.L.; Engelbrecht, B.M.J. Soil organic phosphorus in lowland tropical rain forests. Biogeochemistry 2011, 103, 297–315. [Google Scholar] [CrossRef]
- Murphy, P.N.C.; Bell, A.; Turner, B.L. Phosphorus speciation in temperate basaltic grassland soils by solution 31P NMR spectroscopy. Eur. J. Soil Sci. 2009, 60, 638–651. [Google Scholar] [CrossRef]
- Hou, E.; Wen, D.; Kuang, Y.; Cong, J.; Chen, C.; He, X.; Heenan, M.; Lu, H.; Zhang, Y. Soil pH predominantly controls the forms of organic phosphorus in topsoils under natural broadleaved forests along a 2500km latitudinal gradient. Geoderma 2018, 315, 65–74. [Google Scholar] [CrossRef]
- Turner, B.L.; Blackwell, M.S.A. Isolating the influence of pH on the amounts and forms of soil organic phosphorus. Eur. J. Soil Sci. 2013, 64, 249–259. [Google Scholar] [CrossRef]
- Li, R.; Zhao, J.; Sun, C.; Lu, W.; Guo, C.; Xiao, K. Biochemical properties, molecular characterizations, functions, and application perspectives of phytases. Front. Agric. China 2010, 4, 195–209. [Google Scholar] [CrossRef]
- Vohra, A.; Satyanarayana, T. Phytases: Microbial Sources, Production, Purification, and Potential Biotechnological Applications. Crit. Rev. Biotechnol. 2003, 23, 29–60. [Google Scholar] [CrossRef]
- McKelvie, I.D.; Hart, B.T.; Cardwell, T.J.; Cattrall, R.W. Use of immobilized 3-phytase and flow injection for the determination of phosphorus species in natural waters. Anal. Chim. Acta 1995, 316, 277–289. [Google Scholar] [CrossRef]
- Ullah, A.H.J.; Gibson, D.M. Extracellular Phytase (E. C. 3.1.3.8) from Aspergillus Ficuum NRRL 3135: Purification and Characterization. Prep. Biochem. 1987, 17, 63–91. [Google Scholar]
- Konietzny, U.; Greiner, R. Molecular and catalytic properties of phytate-degrading enzymes (phytases). Int. J. Food Sci. Technol. 2002, 37, 791–812. [Google Scholar] [CrossRef]
- Turner, B.L.; Haygarth, P.M. Phosphatase activity in temperate pasture soils: Potential regulation of labile organic phosphorus turnover by phosphodiesterase activity. Sci. Total Environ. 2005, 344, 27–36. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Wan, B.; Liu, F.; Tan, W.; Liu, M.; Feng, X. Adsorption-Desorption of Myo-Inositol Hexakisphosphate on Hematite. Soil Sci. 2014, 179, 476–485. [Google Scholar] [CrossRef]
- Saeki, K.; Kunito, T.; Sakai, M. Effects of pH, ionic strength, and solutes on DNA adsorption by andosols. Biol. Fertil. Soils 2010, 46, 531–535. [Google Scholar] [CrossRef]
- Deiss, L.; de Moraes, A.; Maire, V. Environmental drivers of soil phosphorus composition in natural ecosystems. Biogeosciences 2018, 15, 4575–4592. [Google Scholar] [CrossRef]
- Siebers, N.; Sumann, M.; Kaiser, K.; Amelung, W. Climatic Effects on Phosphorus Fractions of Native and Cultivated North American Grassland Soils. Soil Sci. Soc. Am. J. 2017, 81, 299–309. [Google Scholar] [CrossRef]
- Dalai, R.C. Soil Organic Phosphorus. In Advances in Agronomy; Brady, N.C., Ed.; Academic Press: Cambridge, MA, USA, 1977; Volume 29, pp. 83–117. [Google Scholar]
- Moata, M.R.S.; Doolette, A.L.; Smernik, R.J.; McNeill, A.M.; Macdonald, L.M. Organic phosphorus speciation in Australian Red Chromosols: Stoichiometric control. Soil Res. 2016, 54, 11–19. [Google Scholar] [CrossRef]
- Spohn, M. Phosphorus and carbon in soil particle size fractions: A synthesis. Biogeochemistry 2020, 147, 225–242. [Google Scholar] [CrossRef]
- Bowman, R.A.; Cole, C.V. Transformations of organic phosphorus substrates in soils as evaluated by NaHCO3 extraction. Soil Sci. 1978, 125, 49–54. [Google Scholar] [CrossRef]
- Allison, V.J.; Condron, L.M.; Peltzer, D.A.; Richardson, S.J.; Turner, B.L. Changes in enzyme activities and soil microbial community composition along carbon and nutrient gradients at the Franz Josef chronosequence, New Zealand. Soil Biol. Biochem. 2007, 39, 1770–1781. [Google Scholar] [CrossRef]
- Yadav, R.S.; Tarafdar, J.C. Phytase and phosphatase producing fungi in arid and semi-arid soils and their efficiency in hydrolyzing different organic P compounds. Soil Biol. Biochem. 2003, 35, 745–751. [Google Scholar] [CrossRef]
- George, T.S.; Simpson, R.J.; Gregory, P.J.; Richardson, A.E. Differential interaction of Aspergillus niger and Peniophora lycii phytases with soil particles affects the hydrolysis of inositol phosphates. Soil Biol. Biochem. 2007, 39, 793–803. [Google Scholar] [CrossRef]
- Renella, G.; Szukics, U.; Landi, L.; Nannipieri, P. Quantitative assessment of hydrolase production and persistence in soil. Biol. Fertil. Soils 2007, 44, 321–329. [Google Scholar] [CrossRef]
- Skujiņš, J.; Burns, R.G. Extracellular Enzymes in Soil. CRC Crit. Rev. Microbiol. 1976, 4, 383–421. [Google Scholar]
- Gerke, J. Phytate (Inositol Hexakisphosphate) in Soil and Phosphate Acquisition from Inositol Phosphates by Higher Plants. A Review. Plants 2015, 4, 253–266. [Google Scholar] [CrossRef]
- Doolette, A.L.; Smernik, R.J.; Dougherty, W.J. Rapid decomposition of phytate applied to a calcareous soil demonstrated by a solution 31P NMR study. Eur. J. Soil Sci. 2010, 61, 563–575. [Google Scholar] [CrossRef]
- Leytem, A.B.; Smith, D.R.; Applegate, T.J.; Thacker, P.A. The Influence of Manure Phytic Acid on Phosphorus Solubility in Calcareous Soils. Soil Sci. Soc. Am. J. 2006, 70, 1629–1638. [Google Scholar] [CrossRef]
- Annaheim, K.E.; Rufener, C.B.; Frossard, E.; Bünemann, E.K. Hydrolysis of organic phosphorus in soil water suspensions after addition of phosphatase enzymes. Biol. Fertil. Soils 2013, 49, 1203–1213. [Google Scholar] [CrossRef][Green Version]
- Turner, B.L.; McKelvie, I.D.; Haygarth, P.M. Characterisation of water-extractable soil organic phosphorus by phosphatase hydrolysis. Soil Biol. Biochem. 2002, 34, 27–35. [Google Scholar] [CrossRef]
- Toor, G.S.; Condron, L.M.; Cade-Menun, B.J.; Di, H.J.; Cameron, K.C. Preferential phosphorus leaching from an irrigated grassland soil. Eur. J. Soil Sci. 2005, 56, 155–168. [Google Scholar] [CrossRef]
- Toor, G.S.; Condron, L.M.; Di, H.J.; Cameron, K.C.; Cade-Menun, B.J. Characterization of organic phosphorus in leachate from a grassland soil. Soil Biol. Biochem. 2003, 35, 1317–1323. [Google Scholar] [CrossRef]
- Darch, T.; Blackwell, M.S.A.; Hawkins, J.M.B.; Haygarth, P.M.; Chadwick, D. A Meta-Analysis of Organic and Inorganic Phosphorus in Organic Fertilizers, Soils, and Water: Implications for Water Quality. Crit. Rev. Environ. Sci. Technol. 2014, 44, 2172–2202. [Google Scholar] [CrossRef]
- Jarosch, K.A.; Doolette, A.L.; Smernik, R.J.; Tamburini, F.; Frossard, E.; Bünemann, E.K. Characterisation of soil organic phosphorus in NaOH-EDTA extracts: A comparison of 31P NMR spectroscopy and enzyme addition assays. Soil Biol. Biochem. 2015, 91, 298–309. [Google Scholar] [CrossRef]
- McLaren, T.I.; Smernik, R.J.; McLaughlin, M.J.; McBeath, T.M.; Kirby, J.K.; Simpson, R.J.; Guppy, C.N.; Doolette, A.L.; Richardson, A.E. Complex Forms of Soil Organic Phosphorus–A Major Component of Soil Phosphorus. Environ. Sci. Technol. 2015, 49, 13238–13245. [Google Scholar] [CrossRef] [PubMed]
- Pierzynski, J.; Hettiarachchi, G.M. Reactions of Phosphorus Fertilizers with and without a Fertilizer Enhancer in Three Acidic Soils with High Phosphorus-Fixing Capacity. Soil Sci. Soc. Am. J. 2018, 82, 1124–1139. [Google Scholar] [CrossRef]
- Luo, L.; Ma, Y.; Sanders, R.L.; Xu, C.; Li, J.; Myneni, S.C.B. Phosphorus speciation and transformation in long-term fertilized soil: Evidence from chemical fractionation and P K-edge XANES spectroscopy. Nutr. Cycl. Agroecosystems 2017, 107, 215–226. [Google Scholar] [CrossRef]
- Kar, G.; Peak, D.; Schoenau, J.J. Spatial Distribution and Chemical Speciation of Soil Phosphorus in a Band Application. Soil Sci. Soc. Am. J. 2012, 76, 2297–2306. [Google Scholar] [CrossRef]
- Liu, J.; Yang, J.; Cade-Menun, B.J.; Hu, Y.; Li, J.; Peng, C.; Ma, Y. Molecular speciation and transformation of soil legacy phosphorus with and without long-term phosphorus fertilization: Insights from bulk and microprobe spectroscopy. Sci. Rep. 2017, 7, 15354. [Google Scholar] [CrossRef] [PubMed]
- Ajiboye, B.; Akinremi, O.O.; Hu, Y.; Jürgensen, A. XANES Speciation of Phosphorus in Organically Amended and Fertilized Vertisol and Mollisol. Soil Sci. Soc. Am. J. 2008, 72, 1256–1262. [Google Scholar] [CrossRef]
- Gamble, A.V.; Northrup, P.A.; Sparks, D.L. Elucidation of soil phosphorus speciation in mid-Atlantic soils using synchrotron-based microspectroscopic techniques. J. Environ. Qual. 2020, 49, 184–193. [Google Scholar] [CrossRef]
- Abdala, D.B.; Moore, P.A.; Rodrigues, M.; Herrera, W.F.; Pavinato, P.S. Long-term effects of alum-treated litter, untreated litter and NH4NO3 application on phosphorus speciation, distribution and reactivity in soils using K-edge XANES and chemical fractionation. J. Environ. Manag. 2018, 213, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Cade-Menun, B.J. Characterizing phosphorus forms in cropland soils with solution 31P-NMR: Past studies and future research needs. Chem. Biol. Technol. Agric. 2017, 4, 19. [Google Scholar] [CrossRef]
- Annaheim, K.E.; Doolette, A.L.; Smernik, R.J.; Mayer, J.; Oberson, A.; Frossard, E.; Bünemann, E.K. Long-term addition of organic fertilizers has little effect on soil organic phosphorus as characterized by 31P NMR spectroscopy and enzyme additions. Geoderma 2015, 257-258, 67–77. [Google Scholar] [CrossRef]
- Cade-Menun, B.J.; Doody, D.G.; Liu, C.W.; Watson, C.J. Long-term Changes in Grassland Soil Phosphorus with Fertilizer Application and Withdrawal. J. Environ. Qual. 2017, 46, 537–545. [Google Scholar] [CrossRef]
- Recena, R.; Torrent, J.; del Campillo, M.C.; Delgado, A. Accuracy of Olsen P to assess plant P uptake in relation to soil properties and P forms. Agron. Sustain. Dev. 2015, 35, 1571–1579. [Google Scholar] [CrossRef]
- Mander, C.; Wakelin, S.; Young, S.; Condron, L.; O’Callaghan, M. Incidence and diversity of phosphate-solubilising bacteria are linked to phosphorus status in grassland soils. Soil Biol. Biochem. 2012, 44, 93–101. [Google Scholar] [CrossRef]
- Richardson, A.E.; Simpson, R.J. Soil Microorganisms Mediating Phosphorus Availability Update on Microbial Phosphorus. Plant Physiol. 2011, 156, 989–996. [Google Scholar] [CrossRef]
- Lynch, J. Root Architecture and Plant Productivity. Plant Physiol. 1995, 109, 7–13. [Google Scholar] [CrossRef] [PubMed]
- Richardson, A.E.; Lynch, J.P.; Ryan, P.R.; Delhaize, E.; Smith, F.A.; Smith, S.E.; Harvey, P.R.; Ryan, M.H.; Veneklaas, E.J.; Lambers, H.; et al. Plant and microbial strategies to improve the phosphorus efficiency of agriculture. Plant Soil 2011, 349, 121–156. [Google Scholar] [CrossRef]
- Niu, Y.F.; Chai, R.S.; Jin, G.L.; Wang, H.; Tang, C.X.; Zhang, Y.S. Responses of root architecture development to low phosphorus availability: A review. Ann. Bot. 2012, 112, 391–408. [Google Scholar] [CrossRef] [PubMed]
- Lambers, H.; Shane, M.W.; Cramer, M.D.; Pearse, S.J.; Veneklaas, E.J. Root Structure and Functioning for Efficient Acquisition of Phosphorus: Matching Morphological and Physiological Traits. Ann. Bot. 2006, 98, 693–713. [Google Scholar] [CrossRef] [PubMed]
- Lambers, H.; Finnegan, P.M.; Laliberté, E.; Pearse, S.J.; Ryan, M.H.; Shane, M.W.; Veneklaas, E.J. Phosphorus Nutrition of Proteaceae in Severely Phosphorus-Impoverished Soils: Are There Lessons To Be Learned for Future Crops? Plant Physiol. 2011, 156, 1058–1066. [Google Scholar] [CrossRef] [PubMed]
- Williamson, L.C.; Ribrioux, S.P.C.P.; Fitter, A.H.; Leyser, H.M.O. Phosphate Availability Regulates Root System Architecture in Arabidopsis. Plant Physiol. 2001, 126, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Tang, C.; Armstrong, R.; Sale, P. Phosphorus supply enhances the response of legumes to elevated CO2 (FACE) in a phosphorus-deficient vertisol. Plant Soil 2012, 358, 91–104. [Google Scholar] [CrossRef]
- Rose, T.J.; Impa, S.M.; Rose, M.T.; Pariasca-Tanaka, J.; Mori, A.; Heuer, S.; Johnson-Beebout, S.E.; Wissuwa, M. Enhancing phosphorus and zinc acquisition efficiency in rice: A critical review of root traits and their potential utility in rice breeding. Ann. Bot. 2013, 112, 331–345. [Google Scholar] [CrossRef]
- Lynch, J.; van Beem, J.J. Growth and Architecture of Seedling Roots of Common Bean Genotypes. Crop Sci. 1993, 33, cropsci1993.0011183X003300060028x. [Google Scholar] [CrossRef]
- Wang, Y.L.; Almvik, M.; Clarke, N.; Eich-Greatorex, S.; Øgaard, A.F.; Krogstad, T.; Lambers, H.; Clarke, J.L. Contrasting responses of root morphology and root-exuded organic acids to low phosphorus availability in three important food crops with divergent root traits. AoB Plants 2015, 7. [Google Scholar] [CrossRef]
- Henry, A.; Chaves, N.F.; Kleinman, P.J.A.; Lynch, J.P. Will nutrient-efficient genotypes mine the soil? Effects of genetic differences in root architecture in common bean (Phaseolus vulgaris L.) on soil phosphorus depletion in a low-input agro-ecosystem in Central America. Field Crops Res. 2010, 115, 67–78. [Google Scholar] [CrossRef]
- da Silva, A.; Bruno, I.P.; Franzini, V.I.; Marcante, N.C.; Benitiz, L.; Muraoka, T. Phosphorus uptake efficiency, root morphology and architecture in Brazilian wheat cultivars. J. Radioanal. Nucl. Chem. 2016, 307, 1055–1063. [Google Scholar] [CrossRef]
- Salvi, S. An evo-devo perspective on root genetic variation in cereals. J. Exp. Bot. 2017, 68, 351–354. [Google Scholar] [CrossRef]
- Rich, S.M.; Watt, M. Soil conditions and cereal root system architecture: Review and considerations for linking Darwin and Weaver. J. Exp. Bot. 2013, 64, 1193–1208. [Google Scholar] [CrossRef] [PubMed]
- Watt, M.; Moosavi, S.; Cunningham, S.C.; Kirkegaard, J.A.; Rebetzke, G.J.; Richards, R.A. A rapid, controlled-environment seedling root screen for wheat correlates well with rooting depths at vegetative, but not reproductive, stages at two field sites. Ann. Bot. 2013, 112, 447–455. [Google Scholar] [CrossRef] [PubMed]
- White, P.J.; George, T.S.; Gregory, P.J.; Bengough, A.G.; Hallett, P.D.; McKenzie, B.M. Matching roots to their environment. Ann. Bot. 2013, 112, 207–222. [Google Scholar] [CrossRef] [PubMed]
- Erel, R.; Bérard, A.; Capowiez, L.; Doussan, C.; Arnal, D.; Souche, G.; Gavaland, A.; Fritz, C.; Visser, E.J.W.; Salvi, S.; et al. Soil type determines how root and rhizosphere traits relate to phosphorus acquisition in field-grown maize genotypes. Plant Soil 2017, 412, 115–132. [Google Scholar] [CrossRef]
- Li, H.; Zhang, D.; Wang, X.; Li, H.; Rengel, Z.; Shen, J. Competition between Zea mays genotypes with different root morphological and physiological traits is dependent on phosphorus forms and supply patterns. Plant Soil 2019, 434, 125–137. [Google Scholar] [CrossRef]
- Guppy, C.N.; Menzies, N.W.; Moody, P.W.; Blamey, F.P.C. Competitive sorption reactions between phosphorus and organic matter in soil: A review. Soil Res. 2005, 43, 189–202. [Google Scholar] [CrossRef]
- Wang, Y.; Lambers, H. Root-released organic anions in response to low phosphorus availability: Recent progress, challenges and future perspectives. Plant Soil 2020, 447, 135–156. [Google Scholar] [CrossRef]
- Jones, D.L. Organic acids in the rhizosphere—A critical review. Plant Soil 1998, 205, 25–44. [Google Scholar] [CrossRef]
- Oburger, E.; Leitner, D.; Jones, D.L.; Zygalakis, K.C.; Schnepf, A.; Roose, T. Adsorption and desorption dynamics of citric acid anions in soil. Eur. J. Soil Sci. 2011, 62, 733–742. [Google Scholar] [CrossRef]
- Oburger, E.; Jones, D.L.; Wenzel, W.W. Phosphorus saturation and pH differentially regulate the efficiency of organic acid anion-mediated P solubilization mechanisms in soil. Plant Soil 2011, 341, 363–382. [Google Scholar] [CrossRef]
- Touhami, D.; McDowell, R.W.; Condron, L.M. Role of Organic Anions and Phosphatase Enzymes in Phosphorus Acquisition in the Rhizospheres of Legumes and Grasses Grown in a Low Phosphorus Pasture Soil. Plants 2020, 9, 1185. [Google Scholar] [CrossRef]
- Pearse, S.J.; Veneklaas, E.J.; Cawthray, G.R.; Bolland, M.D.A.; Lambers, H. Carboxylate release of wheat, canola and 11 grain legume species as affected by phosphorus status. Plant Soil 2006, 288, 127–139. [Google Scholar] [CrossRef]
- Wang, Y.; Krogstad, T.; Clarke, J.L.; Hallama, M.; Øgaard, A.F.; Eich-Greatorex, S.; Kandeler, E.; Clarke, N. Rhizosphere Organic Anions Play a Minor Role in Improving Crop Species’ Ability to Take Up Residual Phosphorus (P) in Agricultural Soils Low in P Availability. Front. Plant Sci. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Gao, Y.; Wu, X.; Ma, H.; Zheng, C.; Wang, X.; Zhang, H.; Li, Z.; Yang, H. The relative contributions of pH, organic anions, and phosphatase to rhizosphere soil phosphorus mobilization and crop phosphorus uptake in maize/alfalfa polyculture. Plant Soil 2020, 447, 117–133. [Google Scholar] [CrossRef]
- Lynch, J.P. Root Phenes for Enhanced Soil Exploration and Phosphorus Acquisition: Tools for Future Crops. Plant Physiol. 2011, 156, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
- Hasan, M.M.; Hasan, M.M.; Teixeira da Silva, J.A.; Li, X. Regulation of phosphorus uptake and utilization: Transitioning from current knowledge to practical strategies. Cell. Mol. Biol. Lett. 2016, 21, 7. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhao, X.; Zhang, L.; Lu, W.; Li, X.; Xiao, K. TaPht1;4, a high-affinity phosphate transporter gene in wheat (Triticum aestivum), plays an important role in plant phosphate acquisition under phosphorus deprivation. Funct. Plant Biol. 2013, 40, 329–341. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Gu, M.; Cao, Y.; Huang, X.; Zhang, X.; Ai, P.; Zhao, J.; Fan, X.; Xu, G. A Constitutive Expressed Phosphate Transporter, OsPht1;1, Modulates Phosphate Uptake and Translocation in Phosphate-Replete Rice. Plant Physiol. 2012, 159, 1571–1581. [Google Scholar] [CrossRef]
- Liu, T.-Y.; Huang, T.-K.; Tseng, C.-Y.; Lai, Y.-S.; Lin, S.-I.; Lin, W.-Y.; Chen, J.-W.; Chiou, T.-J. PHO2-Dependent Degradation of PHO1 Modulates Phosphate Homeostasis in Arabidopsis. Plant Cell 2012, 24, 2168–2183. [Google Scholar] [CrossRef]
- Ye, Y.; Yuan, J.; Chang, X.; Yang, M.; Zhang, L.; Lu, K.; Lian, X. The Phosphate Transporter Gene OsPht1;4 Is Involved in Phosphate Homeostasis in Rice. PLoS ONE 2015, 10, e0126186. [Google Scholar] [CrossRef]
- Zhang, F.; Wu, X.-N.; Zhou, H.-M.; Wang, D.-F.; Jiang, T.-T.; Sun, Y.-F.; Cao, Y.; Pei, W.-X.; Sun, S.-B.; Xu, G.-H. Overexpression of rice phosphate transporter gene OsPT6 enhances phosphate uptake and accumulation in transgenic rice plants. Plant Soil 2014, 384, 259–270. [Google Scholar] [CrossRef]
- Wang, Z.; Ruan, W.; Shi, J.; Zhang, L.; Xiang, D.; Yang, C.; Li, C.; Wu, Z.; Liu, Y.; Yu, Y.; et al. Rice SPX1 and SPX2 inhibit phosphate starvation responses through interacting with PHR2 in a phosphate-dependent manner. Proc. Natl. Acad. Sci. USA 2014, 111, 14953–14958. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.; Ren, H.; Gu, M.; Zhao, J.; Sun, S.; Zhang, X.; Chen, J.; Wu, P.; Xu, G. The phosphate transporter gene OsPht1;8 is involved in phosphate homeostasis in rice. Plant Physiol. 2011, 156, 1164–1175. [Google Scholar] [CrossRef] [PubMed]
- Heuer, S.; Gaxiola, R.; Schilling, R.; Herrera-Estrella, L.; López-Arredondo, D.; Wissuwa, M.; Delhaize, E.; Rouached, H. Improving phosphorus use efficiency: A complex trait with emerging opportunities. Plant J. 2017, 90, 868–885. [Google Scholar] [CrossRef]
- Shimizu, A.; Kato, K.; Komatsu, A.; Motomura, K.; Ikehashi, H. Genetic analysis of root elongation induced by phosphorus deficiency in rice (Oryza sativa L.): Fine QTL mapping and multivariate analysis of related traits. Theor. Appl. Genet. 2008, 117, 987–996. [Google Scholar] [CrossRef]
- Zhu, J.; Kaeppler, S.M.; Lynch, J.P. Mapping of QTL controlling root hair length in maize (Zea mays L.) under phosphorus deficiency. Plant Soil 2005, 270, 299–310. [Google Scholar] [CrossRef]
- Liang, Q.; Cheng, X.; Mei, M.; Yan, X.; Liao, H. QTL analysis of root traits as related to phosphorus efficiency in soybean. Ann. Bot. 2010, 106, 223–234. [Google Scholar] [CrossRef]
- Wissuwa, M.; Ae, N. Genotypic variation for tolerance to phosphorus deficiency in rice and the potential for its exploitation in rice improvement. Plant Breed. 2001, 120, 43–48. [Google Scholar] [CrossRef]
- Li, Q.; Sapkota, M.; van der Knaap, E. Perspectives of CRISPR/Cas-mediated cis-engineering in horticulture: Unlocking the neglected potential for crop improvement. Hortic. Res. 2020, 7, 36. [Google Scholar] [CrossRef]
- Doudna, J.A.; Charpentier, E. Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science 2014, 346, 1258096. [Google Scholar] [CrossRef]
- Gaxiola, R.A.; Regmi, K.; Hirschi, K.D. Moving On Up: H(+)-PPase Mediated Crop Improvement. Trends Biotechnol. 2016, 34, 347–349. [Google Scholar] [CrossRef] [PubMed]
- Rouached, H.; Stefanovic, A.; Secco, D.; Bulak Arpat, A.; Gout, E.; Bligny, R.; Poirier, Y. Uncoupling phosphate deficiency from its major effects on growth and transcriptome via PHO1 expression in Arabidopsis. Plant J. 2011, 65, 557–570. [Google Scholar] [CrossRef] [PubMed]
- Ai, P.; Sun, S.; Zhao, J.; Fan, X.; Xin, W.; Guo, Q.; Yu, L.; Shen, Q.; Wu, P.; Miller, A.J.; et al. Two rice phosphate transporters, OsPht1;2 and OsPht1;6, have different functions and kinetic properties in uptake and translocation. Plant J. 2009, 57, 798–809. [Google Scholar] [CrossRef] [PubMed]
- Alori, E.T.; Glick, B.R.; Babalola, O.O. Microbial Phosphorus Solubilization and Its Potential for Use in Sustainable Agriculture. Front. Microbiol. 2017, 8, 971. [Google Scholar] [CrossRef] [PubMed]
- Calvo, P.; Nelson, L.; Kloepper, J.W. Agricultural uses of plant biostimulants. Plant Soil 2014, 383, 3–41. [Google Scholar] [CrossRef]
- Pii, Y.; Mimmo, T.; Tomasi, N.; Terzano, R.; Cesco, S.; Crecchio, C. Microbial interactions in the rhizosphere: Beneficial influences of plant growth-promoting rhizobacteria on nutrient acquisition process. A review. Biol. Fertil. Soils 2015, 51, 403–415. [Google Scholar] [CrossRef]
- Bargaz, A.; Lyamlouli, K.; Chtouki, M.; Zeroual, Y.; Dhiba, D. Soil Microbial Resources for Improving Fertilizers Efficiency in an Integrated Plant Nutrient Management System. Front. Microbiol. 2018, 9, 1606. [Google Scholar] [CrossRef]
- Kafle, A.; Cope, K.R.; Raths, R.; Krishna Yakha, J.; Subramanian, S.; Bücking, H.; Garcia, K. Harnessing Soil Microbes to Improve Plant Phosphate Efficiency in Cropping Systems. Agronomy 2019, 9, 127. [Google Scholar] [CrossRef]
- Brundrett, M.C.; Tedersoo, L. Evolutionary history of mycorrhizal symbioses and global host plant diversity. New Phytol. 2018, 220, 1108–1115. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.E.; Smith, F.A. Roles of Arbuscular Mycorrhizas in Plant Nutrition and Growth: New Paradigms from Cellular to Ecosystem Scales. Ann. Rev. Plant Biol. 2011, 62, 227–250. [Google Scholar] [CrossRef]
- Smith, S.E.; Jakobsen, I.; Grønlund, M.; Smith, F.A. Roles of Arbuscular Mycorrhizas in Plant Phosphorus Nutrition: Interactions between Pathways of Phosphorus Uptake in Arbuscular Mycorrhizal Roots Have Important Implications for Understanding and Manipulating Plant Phosphorus Acquisition. Plant Physiol. 2011, 156, 1050–1057. [Google Scholar] [CrossRef] [PubMed]
- Peng, S.; Eissenstat, D.M.; Graham, J.H.; Williams, K.; Hodge, N.C. Growth Depression in Mycorrhizal Citrus at High-Phosphorus Supply (Analysis of Carbon Costs). Plant Physiol. 1993, 101, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Krey, T.; Baum, C.; Ruppel, S.; Seydel, M.; Eichler-Löbermann, B. Organic and Inorganic P Sources Interacting with Applied Rhizosphere Bacteria and Their Effects on Growth and P Supply of Maize. Commun. Soil Sci. Plant Anal. 2013, 44, 3205–3215. [Google Scholar] [CrossRef]
- Mercl, F.; Tejnecký, V.; Ságová-Marečková, M.; Dietel, K.; Kopecký, J.; Břendová, K.; Kulhánek, M.; Košnář, Z.; Száková, J.; Tlustoš, P. Co-application of wood ash and Paenibacillus mucilaginosus to soil: The effect on maize nutritional status, root exudation and composition of soil solution. Plant Soil 2018, 428, 105–122. [Google Scholar] [CrossRef]
- Güneş, A.; Ataoğlu, N.; Turan, M.; Eşitken, A.; Ketterings, Q.M. Effects of phosphate-solubilizing microorganisms on strawberry yield and nutrient concentrations. J. Plant Nutr. Soil Sci. 2009, 172, 385–392. [Google Scholar]
- Mosimann, C.; Oberhänsli, T.; Ziegler, D.; Nassal, D.; Kandeler, E.; Boller, T.; Mäder, P.; Thonar, C. Tracing of Two Pseudomonas Strains in the Root and Rhizoplane of Maize, as Related to Their Plant Growth-Promoting Effect in Contrasting Soils. Front. Microbiol. 2017, 7, 2150. [Google Scholar] [CrossRef]
- Varga, T.; Hixson, K.K.; Ahkami, A.H.; Sher, A.W.; Barnes, M.E.; Chu, R.K.; Battu, A.K.; Nicora, C.D.; Winkler, T.E.; Reno, L.R.; et al. Endophyte-Promoted Phosphorus Solubilization in Populus. Front. Plant Sci. 2020, 11. [Google Scholar] [CrossRef]
- Menezes-Blackburn, D.; Jorquera, M.; Gianfreda, L.; Rao, M.; Greiner, R.; Garrido, E.; de la Luz Mora, M. Activity stabilization of Aspergillus niger and Escherichia coli phytases immobilized on allophanic synthetic compounds and montmorillonite nanoclays. Bioresour. Technol. 2011, 102, 9360–9367. [Google Scholar] [CrossRef] [PubMed]
- Calabi-Floody, M.; Velásquez, G.; Gianfreda, L.; Saggar, S.; Bolan, N.; Rumpel, C.; Mora, M.L. Improving bioavailability of phosphorous from cattle dung by using phosphatase immobilized on natural clay and nanoclay. Chemosphere 2012, 89, 648–655. [Google Scholar] [CrossRef]
- Menezes-Blackburn, D.; Jorquera, M.A.; Gianfreda, L.; Greiner, R.; de la Luz Mora, M. A novel phosphorus biofertilization strategy using cattle manure treated with phytase–nanoclay complexes. Biol. Fertil. Soils 2014, 50, 583–592. [Google Scholar] [CrossRef]
- Jones, J.L.; Yingling, Y.G.; Reaney, I.M.; Westerhoff, P. Materials matter in phosphorus sustainability. MRS Bull. 2020, 45, 7–10. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).